Abstract
Narcissism is a personality trait with far-reaching individual, social, and societal consequences. Thus, it is important to understand the sources of individual differences in this trait. Existing hypotheses on the development of narcissism have focused on familial and parental environments that act to make siblings in a family more alike. However, the relative importance of shared environments as opposed to other environmental and genetic sources is still unclear. Using a large extended twin family design, we found that parents’ and children’s narcissism scores were correlated, but this association was entirely genetically driven. Across age and measures, genetics and individual-specific environmental factors each explained 50% of the variance in narcissism, and there was no evidence of environmental sources shared within families. This finding calls for a fundamental shift in the search for the origins of narcissism, including extra-familial environmental factors (e.g., educational and occupational pathways, experiences with peers, and romantic partners).
Introduction
Narcissism is a personality trait that fascinates laypeople and scientists alike. Research on narcissism has made substantial progress in the last two decades in refining the definition and motivational core of narcissism and in exploring its intrapsychic, interpersonal, and institutional consequences (Miller et al., 2021). According to the current state of research, narcissism is centered on differences in grandiosity, sense of entitlement, and striving for social status (Grapsas et al., 2020), and it can have both adaptive (e.g., confidence, initial popularity, dating success, and leadership attainment) and maladaptive (e.g., overly risky decisions, interpersonal conflict, relationship dissatisfaction, and organizational disruption) consequences (Grijalva et al., 2015; Härtel et al., 2023; Leckelt et al., 2015, 2019; Richter et al., 2023; Wurst et al., 2017). Moreover, with regard to development, narcissism has been shown to be observable even in childhood (Grapsas et al., 2021) and to decline with age on average, besides showing strong stability in individual differences in adulthood (Orth et al., 2024; Weidmann et al., 2023). That is, once formed, differences in narcissism are very stable over time.
However, the sources of these individual differences in narcissism are still underexamined and unclear. In this article, we provide a comprehensive analysis of the sources of individual differences in narcissism by applying an extended twin family design across different birth cohorts of twins. This approach provides a well-powered and nuanced test of how much variance in narcissism is driven by genetic factors, environmental factors shared by siblings reared together (shared environmental influences; e.g., familial socioeconomic background, common parental or sibling-specific influences), and environmental factors that are not shared by family members and are, thus, individual-specific (nonshared environmental influences; for example, individual experiences in school, with peers, with romantic partners, or in the workplace).
Traditionally, research on the origins of narcissism has focused strongly on familial environmental factors, particularly parenting styles (Green et al., 2020; Horton, 2011; Horton et al., 2006; Kılıçkaya et al., 2023; Mechanic & Barry, 2015; Thomaes et al., 2013; Watson et al., 1992; Wetzel & Robins, 2016). Psychoanalytic accounts assume that narcissism develops as a compensatory reaction to cold parenting and a lack of parental warmth (Kernberg, 1975; Kohut, 1971). This idea is also fundamental to the prominent mask model (Kuchynka & Bosson, 2018; Morf et al., 2011; Morf & Rhodewalt, 1993), which assumes that narcissistic behavior is a compensatory mask to protect an insecure, fragile self, acquired in early childhood. Learning theories, by contrast, assume that narcissism develops as a learned self-concept in response to parental overvaluation (putting children on a pedestal; Brummelman, 2018; Brummelman et al., 2015, 2022; Brummelman & Sedikides, 2020; Millon, 1981). These different sorts of parenting approaches to the development of narcissism are not only the most salient ones scientifically, but they also dominate lay beliefs and the public discourse about narcissism (Koepernik et al., 2022). Importantly, they rest on the untested assumption that parenting style (in one way or another) is the major source of narcissism development. However, it is far from clear whether this link—if there is one—is actually an environmental link.
Parents and offspring are genetically related, and behavior genetic research in recent decades has shown that individual personality traits are in general substantially heritable (about 50%; Polderman et al., 2015). Importantly, familial environments shared by siblings reared together (e.g., shared parental influences) were found to play a minor role across a broad spectrum of personality characteristics (e.g., broad trait dimensions, motives, values, and interests), with some exceptions, such as religiousness or social attitudes (Kandler, 2021; Kandler et al., 2024; Zapko-Willmes et al., 2018). Genetic and environmental influences, however, may be complexly interwoven and interdependent. Development of individual differences may be driven by dynamic transactions and synergistic interactions between genetic and environmental factors (Briley et al., 2018; Kandler, Zapko-Willmes, et al., 2021). Previous research has shown that genetic differences in personality traits increase from childhood to young adulthood as a consequence of gene–environment interplay (Kandler et al., 2019; Mõttus et al., 2019). As children grow, they become more and more autonomous and self-directed. They have increasing opportunities to pave their own way, evoke, select, and create environments that match their heritable tendencies. These environments in turn can provide experiences that have the potential to reinforce the pre-existing tendencies and stabilize individual differences. Moreover, individual-specific, nonshared environmental factors, such as experiences with peer groups, with romantic partners, or in the workplace, seem to play more prominent roles in personality differences and development across the lifespan (Briley & Tucker-Drob, 2017; Kandler, Bratko, et al., 2021). If parents treat their offspring differently to some extent, then parental influences can act as such individual-specific factors making siblings in a family less alike (Plomin & Daniels, 2011). In other words, objectively shared environmental influences, such as parenting styles of the same parent, can represent environmental influences that are effectively not shared by individuals raised in the same family.
The few existing studies that have investigated the genetic and environmental sources of individual differences in narcissism converge with those on other personality traits and are generally at odds with the predominant explanation of the development of narcissism that focus on parenting: They found 40%–60% genetic and environmental components of variance each, with marginal influences from shared environmental factors (Livesley et al., 1993; Luo et al., 2014; Vernon et al., 2008). However, these studies were limited, as they included relatively small sample sizes and did not consider potential age differences in genetic and environmental contributions. Furthermore, earlier studies were based only on the classical twin study design that relies on several assumptions, such as the absence of assortative mating, the restriction to only three different genetic and environmental sources of variance, and the absence of correlations between these factors (Instinske & Kandler, 2024). These limitations make it difficult to assess the robustness of previous results and their generalizability across different designs. Hence, there is demand for more fine-grained estimates of genetic and environmental factors.
We address the outlined challenges by making use of data from the German TwinLife project, a large extended twin family study. These unique data were used to (1) provide the first robust test of genetic and environmental sources of individual differences in narcissism across age cohorts of twins (about 15, 21, and 27 years of age). By taking the information from twin siblings, their parents, and the nontwin siblings of twins, if available, into account, we (2) could use the advantages of extended twin family modeling. Such a design overcomes several limitations of the classical twin design as it allows the disentanglement of more specific sources of individual differences in narcissism. It distinguishes between (a) genetic and environmental transmission from one generation to the next, (b) different kinds of genetic sources (including additive genetic influences and nonadditive genetic factors due to interactions of genetic variants within or between gene loci), (c) different kinds of environmental sources shared by family members (including those shared by siblings only, shared by twins only, or shared by the entire core family), and (d) nonshared (individual-specific) environmental sources, taking (e) effects of assortative mating into account, and allowing for (f) passive gene–environment covariance. Assortative mating is estimated on the basis of the covariance in parents’ narcissism as a function of nonrandom assortment. Passive gene–environment covariance represents a nonrandom link between genetic and environmental factors transmitted from parents to their offspring. This acts as a function of parental characteristics, which are heritable to some extent and linked with twins’ family environments. Both assortative mating and gene–environment covariance should be taken into account, because otherwise estimates of genetic and environmental variance components can be biased (Bleidorn et al., 2018; Kandler et al., 2024).
Confirmatory hypotheses pertain to the broader estimations of overall genetic variance and variance due to environmental factors shared or not shared by siblings reared together. First, we expected (Hypothesis 1) a substantial amount of genetic variance (including additive and nonadditive genetic components), (Hypothesis 1a) similar to what has been observed in previous studies on personality characteristics (i.e., between 40% and 60%) and (Hypothesis 1b) significantly larger in adults compared with adolescents. Second, we expected (Hypothesis 2) a substantial amount of variance attributable to environmental factors not shared by siblings reared together, similar to what has been observed in previous studies (i.e., between 30% and 60%). Third, we expected (Hypothesis 3) a small amount of environmental variance attributable to environmental factors shared within families, (Hypothesis 3a) similar to what has been observed in previous studies (i.e., between 0% and 20%), but (Hypothesis 3b) significant in adolescents or significantly larger in adolescents compared with adults. These hypotheses and the study analysis plan were preregistered at the Open Science Framework (see https://osf.io/za9gd).
Given that previous research on the behavioral genetics of narcissism was not particularly well-powered and did not include an extended twin family design, our study allowed first and direct estimates of the amount of passive gene–environment covariance as well as the examination and consideration of assortative mating in narcissism. In this regard, our study is exploratory.
Method
Transparency and Openness
The hypotheses and methods, including the analysis plan, were preregistered (https://osf.io/za9gd) with the title “Nature and Nurture of Narcissism: An Extended Twin Family Analysis.” before data analyses. There were minor deviations from the preregistration (for details see “Document Description” on p. 1 of the Supplement A). Specific codebooks, technical reports, and information on measures and data releases are available for download here: https://www.twin-life.de/documentation/downloads. An overview of the data and full data documentation are provided at the TwinLife homepage https://www.twin-life.de/documentation/. Data are available as scientific use files for download from the GESIS homepage: https://search.gesis.org/research_data/ZA6701. For the current investigation, we used the F2F 3a and 3b data releases (third wave of data collection via face-to-face household interviews and self-report measures), which include measures of narcissism for different twin family members. All analysis scripts are publicly available at https://osf.io/za9gd. See also Supplement A for detailed information on the used statistical software programs and specific packages. In addition, we conducted several supplementary and robustness analyses (see Supplement A for more details on the measures of narcissism and the sample and Supplement B for preparatory analyses). Supplementary details on all main analyses and results are described and presented in Supplements C and D.
Data and Sampling
The TwinLife project is an extended twin family study that includes not only three large age cohorts of monozygotic and same-sex dizygotic twins but also their parents and nontwin siblings (Diewald et al., 2022; Hahn et al., 2016; Rohm et al., 2023). Ethical approval was received from the German Psychological Society (protocol numbers RR 11.2009 and RR 09.2013). Participants received information about the study’s aims, the voluntary nature of their participation, and information on data protection before their participation in writing. Informed verbal consent was obtained from each participant (or in the case of underaged children, from participants’ legal guardians) at the beginning of each face-to-face interview. Figure 1 presents an illustration of the TwinLife design and its data on narcissism.
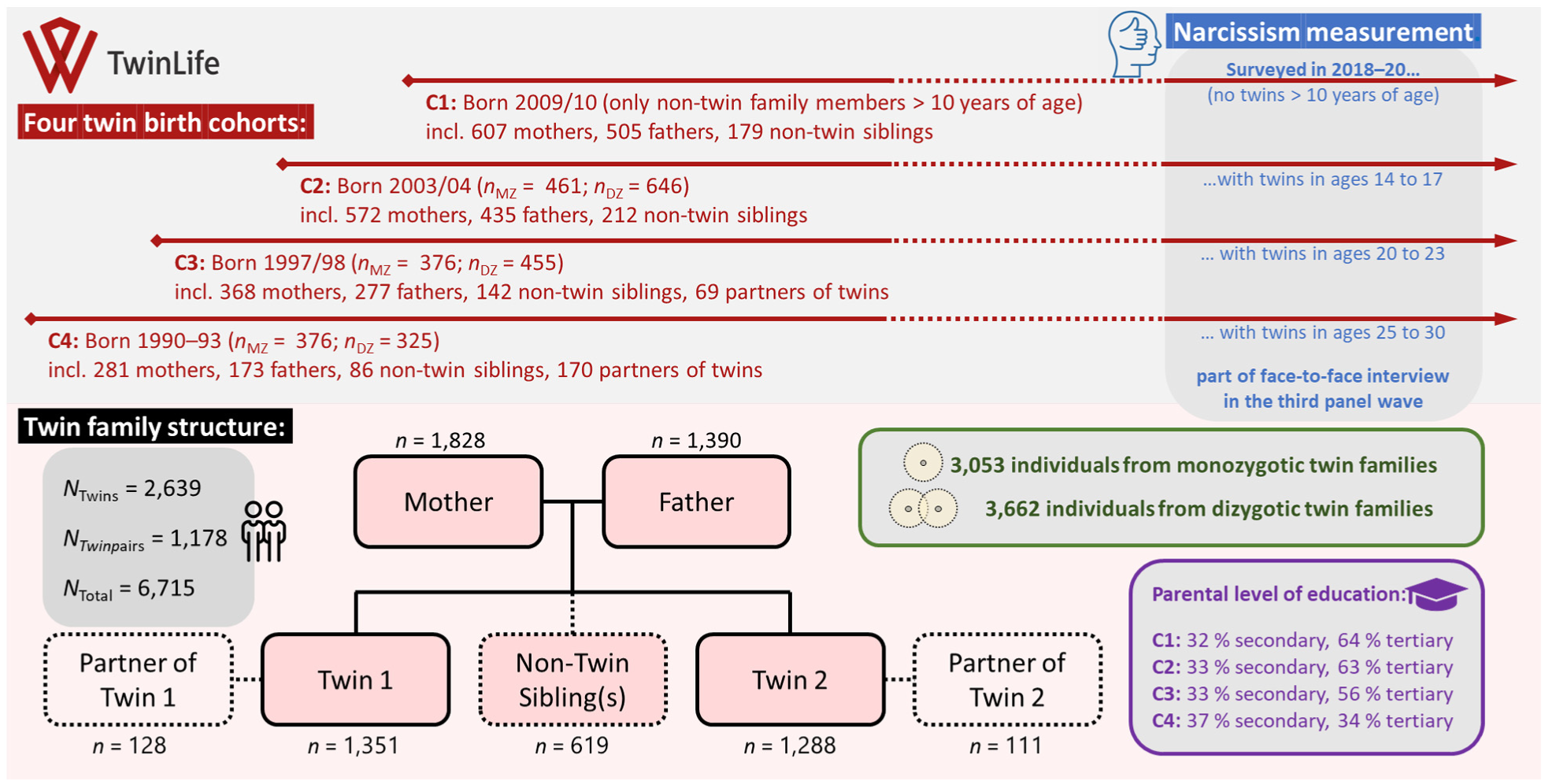
Overview of the TwinLife Extended Twin Family Design and Included Data
The initial round of data was collected by TNS Infratest/Kantar TNS (first face-to-face household survey, parts of the first telephone survey). Since the first telephone survey and second wave of data collection, infas (Institut für angewandte Sozialwissenschaft) carried out data collection via face-to-face and telephone interviews by trained interviewers. Data were cleaned and screened by infas and the TwinLife data management team (Krell et al., 2024).
We analyzed all available data on narcissism collected from twin family members (data collection during Wave 3, which was the only measurement occasion where narcissism was captured within the panel). The preregistered sample size still included observations with invalid age and sex information, as well as a small number of nonbiologically related parents (i.e., adoptive parents, foster parents, and step-parents) or half- and step-siblings. With these individuals excluded, the final sample size used for analysis was N = 6,715, including data from 2,639 twins, 619 nontwin siblings of twins, 1,828 mothers of twins, 1,390 fathers of twins, and 239 spouses/partners of twins. In the preregistration, we stated that “individuals with missing values on relevant items will be excluded from the analyses,” but for our final analyses, we excluded only those individuals who did not have a minimum of two valid items on the analyzed narcissism scales. We used full information maximum likelihood estimation procedures to account for any additional missingness in the data.
Narcissism Measures
The narcissism items from the abbreviated version of the Dirty Dozen Scale (the Naughty Nine Scale, NNS; Küfner et al., 2015) were applied when participants were 17 years of age or older (1,534 twins, 400 nontwin siblings, 1,828 mothers, and 1,390 fathers). The three items (“I tend to want others to admire me”; “I tend to want others to pay attention to me”; and “I tend to seek prestige and status”) were answered on a scale ranging from 1 (does not apply at all) to 9 (applies completely). Adjusted items selected from the Narcissistic Personality Inventory, NPI; Raskin & Terry, 1988) were applied for individuals between 11 and 16 years of age (1,105 twins and 219 nontwin siblings). The four items (“I was born a good leader”; “I am really a special person”; “I am good at getting people to do things my way”; and “It is easy for me to control other people”) were answered on a scale ranging from 1 (does not apply at all) to 5 (applies completely). The NNS and NPI narcissism scores were calculated by factor score extraction based on confirmatory factor analyses. In addition, latent-variable modeling was used for all main analyses (see Supplemental Figures D1, D2, and D3).
Data Preprocessing
All data preprocessing steps, variable and sample descriptions, details on narcissism measures, and reliability analyses thereof are summarized in Supplement A. The population-based sample can be seen as heterogeneous regarding educational attainment (Table A5). The internal consistencies of the NNS and NPI were evaluated as good, ranging from ω = .77 to ω = .79 for the NPI and ω = .83 to ω = .84 for the NNS across different cohorts and groups of family members (Tables A3 and A4).
More detailed analyses on the psychometric quality of the measures are outlined in Supplement B. These include factor analyses of the measures. All analyses yielded homogeneity of the measured constructs, supporting factorial validity (Tables B1, B2, and B4). Furthermore, metric measurement invariance for the NNS across family member types and scalar measurement invariance for the NPI across ages 11 to 16 were supported (Table B3). Regarding the expected increase in genetic variance from adolescence to adulthood (as outlined in the preregistration), the analyses suggested this trend across age cohorts (see Figure 3 and D4), but the model tests indicated that these trends were not statistically significant.
To check for possible confounding effects, we tested for sex and linear as well as nonlinear age differences in narcissism (Table B6). We saw small but statistically significant linear age and sex effects on the NNS factor score, and small but statistically significant sex effects on the NPI factor score. We computed adjusted scores accordingly with residualization (standardized residuals derived from regressions with age and sex as predictors). There were no substantial differences between nonadjusted and adjusted scores regarding family resemblance (see supplemental Table C1). Therefore, we used the nonadjusted factor and item scores for further analyses based on latent-variable modeling.
Statistical Main Analyses
First, we used the NPI and NNS factor scores (adjusted and nonadjusted for age and sex differences) for each family member to estimate dyadic family correlations. Then, we ran the family correlations corrected for random measurement error by using latent-variable modeling for each group of family dyads. We also estimated cohort-specific twin and sibling correlations.
After calculations of dyadic family correlations for a rough inspection of familial similarity or dissimilarity in narcissism, we ran different extended twin family model analyses for examining the sources of variance in narcissism. As we used latent-variable modeling in all twin family analyses, individual differences in narcissism were corrected for variance due to random error of measurement. The model estimates were based on full information maximum likelihood estimation.
We started with a classical model of twins reared together. This twins-only model allows for estimates of additive genetic effects due to genetic factors that are shared between relatives to the degree they are genetically related. Additive genetic effects act to make monozygotic twins more similar to each other than dizygotic twins. Furthermore, two kinds of environmental effects can be estimated, those due to environmental factors shared by both monozygotic and dizygotic twins and those due to factors not shared by twins. Shared environmental effects are indicated when monozygotic and dizygotic twin correlations do not differ substantially. Instead of shared environmental effects, nonadditive genetic effects can be estimated, taking significant interactions of genetic variants either between gene loci (epistatic gene-by-gene interactions) or within gene loci (genetic dominance) into account. Nonadditive genetic influences because of epistasis are more plausible for polygenic characteristics, such as personality traits (Kandler et al., 2024). They are completely shared by genetically identical monozygotic twins and do not correlate among other relatives. Those effects are thus indicated when monozygotic twin correlations are more than twice as high as the dizygotic twin correlations. Shared environmental effects and nonadditive genetic effects cannot be estimated in presence of each other in a twins-only model (see Bleidorn et al., 2018, for more details on the classical twin model and its basic assumptions). Therefore, we started with two alternative models: A model allowing for shared environmental effects and a model allowing for nonadditive genetic effects, both in addition to additive genetic and nonshared environmental effects. We also tested a more parsimonious model only allowing for additive genetic and nonshared environmental effects and compared this model to the other models with the use of a likelihood ratio test.
In the next step, we added the nontwin sibling data. This twin+sibling model allows for estimates of additive genetic effects and environmental effects due to environmental factors shared by twins only and those due to factors not shared by twins. Furthermore, the model allows estimates of either environmental effects shared by all siblings or nonadditive genetic effects because of epistasis. Both effects cannot be estimated in the presence of each other. Therefore, we started with two alternative models: A model allowing for nonadditive genetic effects and a model allowing for environmental effects shared by siblings, both in addition to environmental effects shared by twins only as well as additive genetic and nonshared environmental effects. We again tested a more parsimonious model only allowing for additive genetic and nonshared environmental effects and compared this model to the more complex models with the use of a likelihood ratio test.
Then, we ran nuclear twin family model analyses based on data from twins, their nontwin siblings, and parents. This model allows for estimates of additive genetic effects and nonadditive genetic effects due to gene-by-gene interactions. Both types of genetic effects can be estimated in the presence of several shared and nonshared environmental influences. This prevents the overestimation of additive genetic effects on one hand and the underestimation of nonadditive genetic and shared environmental sources of variance on the other. In a nuclear twin family model, shared environmental influences can be partitioned into shared family environmental effects provided through environmental transmission from parents to their offspring and into estimates of shared environmental influences that go beyond the direct parental transmission to the offspring generation. Because of the inclusion of a nontwin sibling, these within-generational shared environmental influences can be further disentangled into an environmental effect shared by all siblings and an environmental contribution only shared by the same-aged twin siblings, which might exist because of age-related effects. As in the other models, nonshared environmental influences act to make all family members less alike. All these genetic and environmental factors can account for variance in narcissism. A special benefit of the nuclear twin family model is that it allows for the consideration of assortative mating between twins’ parents. Taking assortative mating into account prevents overestimating shared environmental influences and underestimating additive genetic effects. A further value of the used model is that it also allows for an estimation of passive gene–environment correlation, if both genetic and environmental transmission from parents to the offspring matter. One limitation, however, is that environmental effects shared by all siblings cannot be estimated in the presence of nonadditive genetic effects. Therefore, we again started with two alternative models: A model allowing for nonadditive genetic effects in addition to other effects and a model allowing for environmental effects shared by siblings instead. Then, we also ran more parsimonious models and compared them to more complex models.
All twin family model analyses are described in Supplement D with more technical details. In a final step, the twin model analyses were run for the three twin cohorts separately in a six-group twin model (two zygosity groups × three twin birth cohorts) allowing for different parameter estimates across cohorts. We tested for significant differences in genetic and environmental variance components across cohorts.
Results
Familial Similarities in Narcissism
We computed average correlations of narcissism scores with 95% confidence intervals for each group of family dyads using all available information on narcissism and correcting for random measurement error by using latent-variable modeling. As Figure 2 shows, and speaking for a strong genetic component of narcissism, monozygotic twins were much more similar in narcissism than either dizygotic twins or nontwin siblings. The similarity of parents and their offspring was comparable to the similarity of the partners (i.e., mother–father and twin-partner similarity), with mothers being slightly more similar to their offspring than fathers. Analyses separated for different narcissism measures and age cohorts yielded very similar findings and can be found in Supplement C.
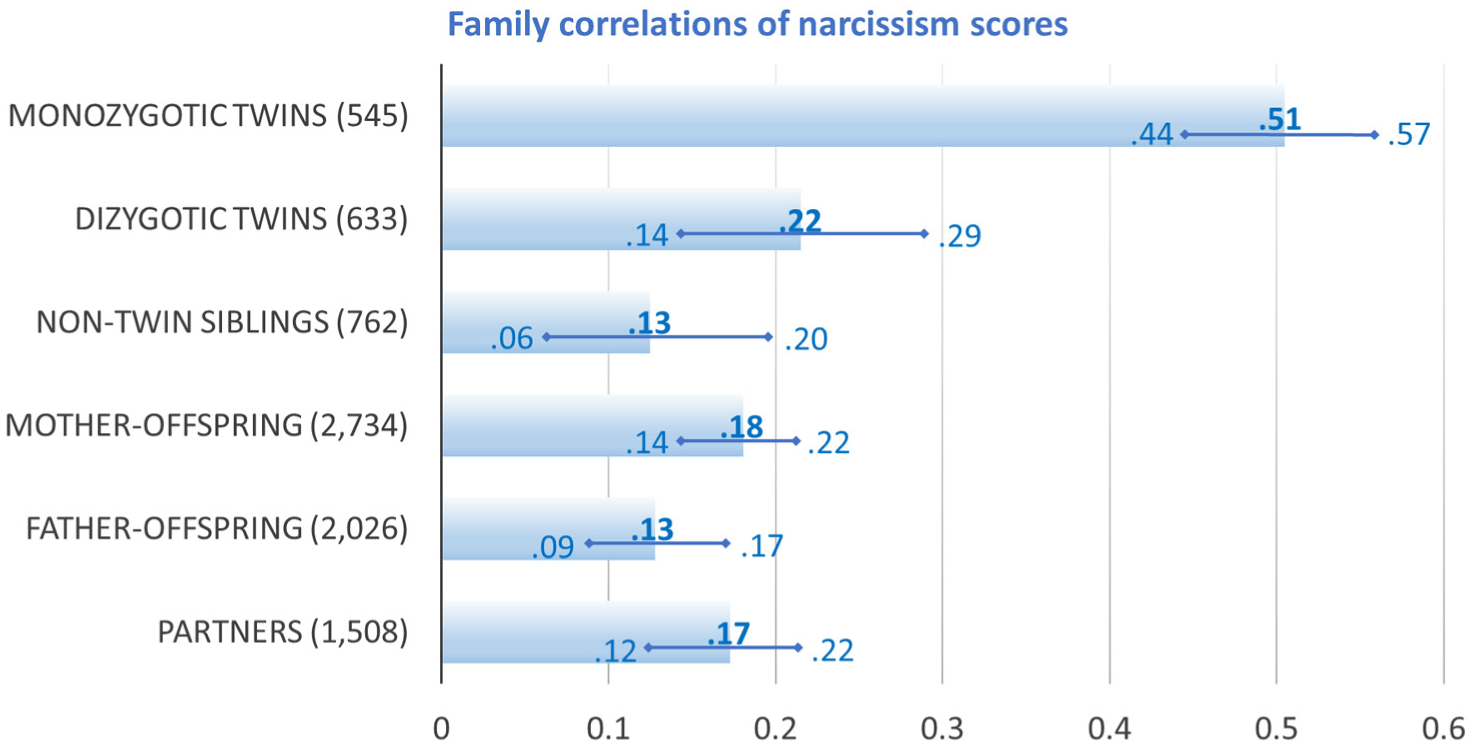
Visualization of Family Correlations
Sources of Variance in Narcissism
To investigate the relative importance of genetic and environmental sources of variance in narcissism, we conducted a series of twin (family) model analyses based on structural equation modeling, starting with classical twins-only models and progressing to increasingly complex twin family models that, in addition, included narcissism information on nontwin siblings and parents of twins (see Supplement D). Figure 3 summarizes the results of the model-based estimates of genetic and environmental variance components, using consistent application of latent-variable modeling of narcissism measures across age cohorts and individuals (i.e., twins, nontwin siblings, and parents).
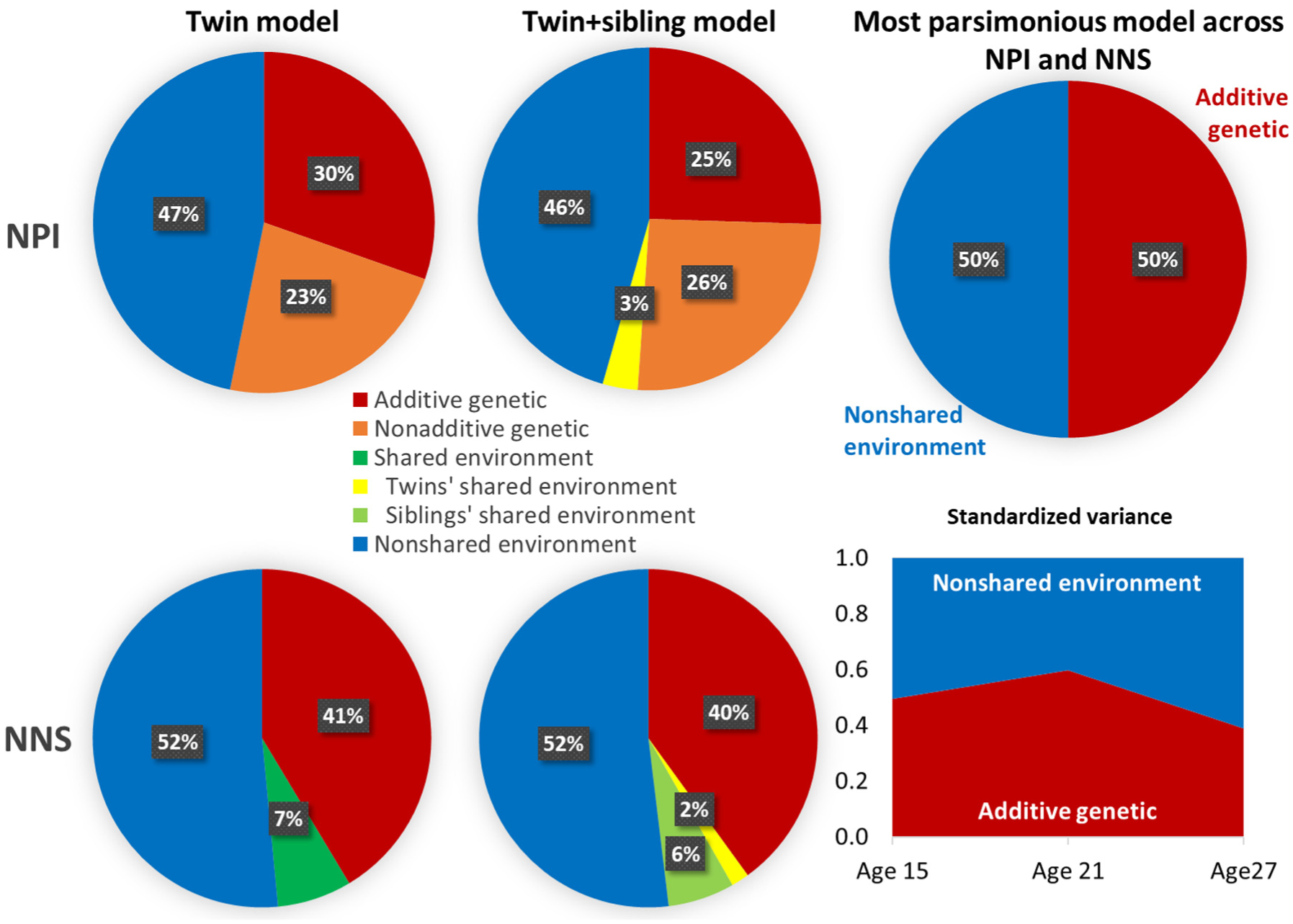
Genetic and Environmental Sources of Variance in Narcissism
Across age cohorts and applied narcissism measures, about 50% of the variance could be attributed to genetic factors (consistent with Hypothesis 1a) and 50% to environmental sources not shared by twins (in line with Hypothesis 2). In line with our Hypothesis 3a, the contribution of environmental influences shared by siblings reared together to the variance in narcissism was generally negligible. In addition, we also tested for the presence of nonadditive genetic effects that take interactions of alleles between gene loci into account. Evidence of such epistatic gene-by-gene interactions was mixed and inconsistent across narcissism measures (see Figure 3 and supplemental Tables D3, D6, D9, and D13 for full model parameter estimates including exact p-values and 95% confidence intervals). Only additive genetic and nonshared environmental effects proved to be robust across all measurements and model variants. The a- and e-effect estimates had a p-value less than .05 in >90%, while the other parameters had a p-value less than .05 in ≤50% of combinations of narcissism measures and model variants.
The more complex extended twin family models corroborated the finding of equally strong influences of genetic and nonshared environmental factors (e.g., for the model based on NNS: a = 0.64, 95% CI = [.38, .90], p < .001, std. a2 = .39; e = 0.74, 95% CI = [.67, .82], p < .001, std. e2 = .53). They also allowed us to better understand (the lack of) familial environmental influences. There was clear evidence that parents tended to be similar in their narcissism, and thus, for the presence of assortative mating (μ = 0.19, 95% CI = [0.13, 0.25], p < .001). Neither the mother’s narcissism nor the father’s narcissism, however, had a direct positive environmentally transmitted effect on their child’s narcissism. These effects of parental environmental transmission were even estimated to be negative (see Table D9). That is, more narcissistic parents influenced their children to be less narcissistic via the parents’ behavioral traits. This finding, in combination with the fact that parents positively affected their children’s narcissism via genetic transmission, resulted in a rarely seen but small negative gene–environment covariation. While these results need to be interpreted cautiously, they clearly speak against a strong positive influence of parents on their children’s narcissism via environmental transmission.
The only effects on narcissism from the family environment that were significant in some of the analyses were effects shared by all siblings in a family: s = .39, 95% CI = [0.11, 0.66], p = .006, std. s2 = .14 (see Table D9). These aspects include shared environmental influences such as generational effects or those from the broader social environment that are not due to direct parental transmission and not specific to same-aged twins. However, these effect estimates were inconsistent across the two narcissism measures and the applied models, and should, therefore, be viewed with caution.
Although, descriptively, genetic variance tended to be larger in the emerging adult cohort (age 21: a = 1.41, 95% CI = [1.22, 1.59], p < .001, a2 = 1.98, std. a2 = .60; see Figure 3 and Table D11) compared with the adolescent cohort (age 15: a = 1.27, 95% CI = [1.09, 1.44], p < .001, a2 = 1.61, std. a2 = .50) and the adult cohort (age 27: a = 1.14, 95% CI = [0.89, 1.38], p < .001, a2 = 1.29, std. a2 = .39), the model comparisons from a six-group twin model (two zygosity groups × three twin birth cohorts) indicated that the differences were not statistically significant. The same was true for the environmental variance (see Table D11). Thus, we found no substantial differences between age cohorts in the general extent of individual differences or in genetic or environmental differences in narcissism across different age cohorts, contradicting Hypotheses 1b and 3b. All analyses—separated by different intermediate models, age cohorts, and narcissism measures—can be found in Supplement D. Across analytical variants and consistent with Hypotheses 1a, 2, and 3a, genetic factors accounted for 39% to 63% of the variance in narcissism. Nonshared environmental contributions ranged from 48% to 59%, whereas shared environmental components were mostly negligible (0%–16%).
We also conducted robustness checks on our model results by using a different statistical software and a different extended twin family model specification. Overall, the results turned out to be similar across both modeling approaches. The deviations are minimal and are reported in Supplement D. These do not modify our main results in any way.
Discussion
In this study, we provided a large-scale and differentiated analysis of the genetic and environmental sources of individual differences in narcissism across age cohorts, extended twin family models, and narcissism measures. The results provide robust evidence of equally large proportions of variance from additive genetic and nonshared environmental sources but low to no variance from shared environmental sources. Parents and children were similar in their narcissism scores, but this association was entirely genetically driven. These results demonstrate that shared environmental sources alone (e.g., common parental influences or family socioeconomic status) play a minor role in the development of narcissism. Although our latent-variable modeling approach controlled for random measurement errors, the narcissism measures were based on self-reports, which are subject to nonrandom rater biases. These biases can artificially reduce the actual family resemblance in personality traits, leading to an underestimation of heritability estimates and an overestimation of nonshared environmental factors (Mõttus et al., 2025). Even though the actual family similarity in terms of narcissism may be higher, this limitation does not affect our main conclusion: Narcissism runs in families, but mainly due to genetics.
Interestingly, we also found initial evidence of another reason why narcissism runs in families: positive assortative mating. Parents tended to be similar to each other in their narcissism levels. This finding corroborates previous findings on the role of narcissism-matching in romantic relationships (Lamkin et al., 2015) and contradicts widespread ideas of complementary matching (i.e., that opposites attract when it comes to narcissism) and victim selection (i.e., narcissists select submissive and weak “victims” they can exploit). Not only does assortative mating make the partners within families more alike in narcissism and via genetic transmission also their offspring, but it also contributes to increase in between-family differences, and, thus, increased genetic variance in narcissism across generations. However, given the rather moderate partner correlation, it can be assumed that this effect will be minor.
Our findings might seem less surprising given similar behavioral genetic results found for other domains of personality traits. Nevertheless, they challenge existing theoretical accounts in the realm of narcissism research that place a strong and almost exclusive focus on the idea that the driving force behind narcissism is parenting that is shared by siblings in a family. They call for a fundamental shift in the search for the sources of narcissism—with regard to genetics, relevant environmental factors, and the interplay of genes and environments. First, we need to better understand the biological differences these genetic influences trigger and how they exert their influence on narcissistic behaviors and experiences. To date and to our knowledge, no GWAS studies have ever included narcissism measures, and thus, there are currently no empirical insights into potentially relevant genetic variants and no existing genetic risk scores for narcissism. These more fine-grained genetic analyses could be combined with a well-powered search for mediating neurophysiological systems. Existing neuroscientific studies on narcissism have revealed an unclear pattern of associations and are typically severely underpowered (Jauk & Kanske, 2021). One particularly promising candidate is, for example, the dopaminergic system, given its strong link to approach-oriented behavior and sensitivity to novel rewards (DeYoung, 2013), a pattern that shows a strong resemblance to narcissistic functioning (Foster & Trimm, 2008). Another candidate is the hormone testosterone, which is connected to dominance and status-seeking behavior (Eisenegger et al., 2011), both hallmark features of narcissism (Grapsas et al., 2020; Zeigler-Hill et al., 2018). Indeed, a recent study found an association between narcissism and basal levels of testosterone (Zajenkowski et al., 2023).
Second, regarding environmental influences, future research should focus more strongly on individual-specific environmental factors. The degree of narcissism can itself drive social relationships that are not shared between siblings but can also develop within such relationships (e.g., an individual’s affiliation with and position in social networks, experiences with peers and romantic partners; Leckelt et al., 2020; Wurst et al., 2017). Narcissism also influences and is potentially influenced by individuals’ pathways and experiences in educational and occupational contexts (Härtel et al., 2023; Heyde et al., 2024; Wille et al., 2019). Closely analyzing repeated status- versus communality-related rewards in social contexts might be a key to understanding individual differences in narcissism trajectories. This calls for an environmentally oriented methodological approach that is similarly ambitious as GWAS in the genetic domain, such as collaborative efforts to realize longitudinal experience-wide association studies (LEWAS; Bleidorn et al., 2020).
Third, these two approaches, GWAS and LEWAS, will need to be linked to achieve a closer understanding of the potential dynamic and synergistic interplay between genetic and environmental factors (Briley & Tucker-Drob, 2017; Kandler, Zapko-Willmes, et al., 2021). The combination and interplay of a genetically based disposition to develop narcissistic features (e.g., a strong reactivity to novel rewards) and certain repeated experiences (e.g., status boosts in peer, romantic, and job contexts) might be particularly strong drivers of narcissism trajectories. The search for such more complex underpinnings of narcissism will be most efficient when top-down theoretical approaches (e.g., combinations of neuroscientific and social-interactive models of narcissism; Grapsas et al., 2020; Jauk & Kanske, 2021) are combined with bottom-up, data-driven approaches (e.g., with interpretable machine-learning approaches that include a wide range of genetic and environmental factors; Gao et al., 2023; Libbrecht & Noble, 2015).
More detailed knowledge of genetic and environmental factors and their interplay that drive individual differences in narcissism will further transform our understanding of narcissism. This will be essential for developing more effective means for dealing with narcissistic individuals in applied contexts, such as in psychotherapy, the workplace, and everyday life.
Supplemental Material
sj-docx-1-spp-10.1177_19485506261429556 – Supplemental material for Narcissism Runs in Families Due to Genetics: An Extended Twin Family Analysis
Supplemental material, sj-docx-1-spp-10.1177_19485506261429556 for Narcissism Runs in Families Due to Genetics: An Extended Twin Family Analysis by Mitja D. Back, Jana Instinske, Theresa Rohm, Marco Deppe and Christian Kandler in Social Psychological and Personality Science
Footnotes
Acknowledgements
The authors thank the Twin Life staff for collecting and providing the data used in this study.
Handling Editor: Rodica Damian
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The TwinLife study was funded by the German Research Foundation (DFG) (Grant No. 220286500, ![]() ). The funder had no role in the study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
). The funder had no role in the study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
The supplemental material is available in the online version of the article.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
