Abstract
Objective
Cytokines are implicated in the pathogenesis of osteoarthritis (OA), and this study aims to assess the therapeutic potential of an IL-8 neutralizing monoclonal antibody (mAb) for OA intervention.
Design
The study employed a rabbit model of OA induced by anterior cruciate ligament transection (ACLT) surgery to investigate the effects of an interleukin (IL)-8 neutralizing mAb, with hyaluronic acid (HA) used as a positive control. Primary outcomes assessed in the rabbits included cartilage repair, synovitis, joint effusion, changes in footprints, and lower limb loading conditions.
Results
Compared to HA, intra-articular injection of the IL-8 neutralizing mAb demonstrated a more pronounced attenuation of OA progression and enhancement of cartilage repair. We observed a reduction in synovitis and joint effusion, indications of bone marrow edema, as well as improvements in lower limb function. In knees treated with the neutralizing IL-8 mAb, there was a significant decrease in IL-8 levels within the synovial tissues.
Conclusions
The IL-8 neutralizing mAb exhibits promising therapeutic potential in the management of OA by attenuating inflammation and facilitating cartilage repair. However, further investigations are warranted to comprehensively elucidate the underlying mechanisms, optimize treatment protocols, and ensure the long-term safety and efficacy of this innovative therapeutic approach.
Introduction
Osteoarthritis (OA) is a prevalent chronic degenerative joint disorder characterized by the degradation of articular cartilage, bone remodeling, and inflammation. 1 Cytokines play a pivotal role in this process, mediating inflammatory responses and contributing to the pathogenesis of OA. 2 Joint inflammation often accompanies OA progression, affecting the metabolism, repair, and regeneration of joint tissues. This inflammation, driven by a surge in pro-inflammatory cytokines such as interleukins (IL-1β, IL-6, and IL-8) and tumor necrosis factor-alpha (TNF-α), can lead to an imbalance in cartilage metabolism, which can disrupt cartilage metabolism leading to its degradation. 3 Moreover, joint inflammation can induce swelling of the joint capsule, pain sensation, and enhanced osteoclast activation; processes intricately regulated by cytokine signaling. 4 Furthermore, inflammation in OA is closely associated with subchondral bone sclerosis that involves alterations in bone structure and metabolism possibly influenced by an imbalanced ratio between osteoprotegerin (OPG) and receptor activator of nuclear factor kappa-B ligand (RANKL), critical mediators for osteoclastogenesis. 5 Inflammatory cytokines act as key regulators within the inflamed joint microenvironment offering significant targets for intervention in conservative treatment approaches for OA.
Neutralizing antibodies against various cytokines have been developed in recent years to treat infections, autoimmune disorders, and inflammatory diseases. These conditions include COVID-19, 6 rheumatoid arthritis, 7 and inflammatory bowel disease. 8 In the field of OA research, significant advancements have been made in cytokine-targeting antibody therapies. 9 For instance, anti-TNF-α, 10 anti-IL-1β, 11 anti-nerve growth factor (NGF), 12 anti-ADAMTS5, 13 and anti-IL-6 14 antibodies have been demonstrated potential therapeutic effects in alleviating joint inflammation and improving joint function in clinical trials. However, the long-term efficacy and complete mechanisms of action for these cytokine-targeting therapies remain uncertain. Given these uncertainties, it becomes imperative to identify and investigate novel cytokine targets as a promising avenue for developing antibody-based treatments for OA. By validating their effectiveness specifically in the context of OA, this approach could potentially offer new hope for patients by introducing an innovative therapeutic strategy.
Interleukin-8 (IL-8) is a pivotal pro-inflammatory cytokine that plays a central role in the recruitment, activation, and proliferation of neutrophils. 15 In addition, IL-8 is known to attract macrophages, another essential immune cell type involved in the inflammatory response, thereby amplifying the cascade of inflammation. Concerning OA, IL-8 has been implicated in joint pain and cartilage damage. The primary mechanism through which IL-8 exerts its effects involves binding to its receptors CXCR1 and CXCR2, 16 inducing chemotaxis of neutrophils and contributing to the initiation and maintenance of joint inflammation. Furthermore, by activating macrophages, IL-8 can promote the release of inflammatory mediators, intensifying the inflammatory environment within the joint. 17 Moreover, IL-8 influences chondrocyte metabolism and facilitates degradation of cartilage matrix components, thus accelerating joint degeneration. 18 Studies indicate that levels of IL-8 are frequently elevated in arthritis patients and correlate with both the extent and severity of joint inflammation. 19 Given its critical role in OA pathogenesis involving recruitment and activation of neutrophils as well as macrophages, targeting IL-8 holds promise as a potential therapeutic approach.
Despite the potential role of IL-8 in the pathogenesis of OA, research into its involvement has been challenging and relatively limited. The lack of understanding stems from multiple factors. Firstly, the intricate cytokine network and the overlapping roles of various cytokines in OA pathogenesis pose challenges in isolating the specific influence exerted by IL-8. 20 And the potential pleiotropic effects of IL-8 and the redundancy in cytokine signaling pose additional challenges. Moreover, the role of IL-8 in OA may vary depending on the stage and severity of the disease, adding another layer of complexity. It is worth noting that developing and validating high-quality neutralizing monoclonal antibodies (mAb) against IL-8 is a complex and costly process, with relatively limited research compared to other cytokines. In addition, there is no direct equivalent for human IL-8 in commonly used model organisms such as rats and mice, which complicates testing and validation of these antibodies in animal models. 21 However, despite these challenges, research on neutralizing antibodies targeting IL-8 for OA is still at an early stage. Preliminary studies have suggested that IL-8 plays a crucial role in neutrophil-mediated acute gouty arthritis, as demonstrated in a model induced by monosodium urate crystals. 22 Neutralizing IL-8 significantly reduces joint swelling induced by MSU crystals and neutrophil infiltration. 23 These findings emphasize the need for further investigation into the therapeutic potential of targeting IL-8 in clinical practice.
By investigating the potential therapeutic effects, mechanisms of action, and clinical application prospects of IL-8 neutralizing mAb in the treatment of OA, this study aims to provide novel insights and therapeutic targets for addressing this degenerative condition. In future research endeavors, our team will focus on deepening the understanding of the molecular mechanisms underlying the effects of IL-8 neutralizing mAbs, optimizing their design and delivery strategies, as well as conducting comprehensive clinical trials to assess their safety and efficacy in managing OA.
Materials and Methods
Isolation and Culture of Bone Marrow–Derived Macrophages (BMDMs)
All animal experiments were conducted in accordance with the animal research committee regulations of the Army Medical University (Third Military Medical University), under approval number AMUWEC20227021. Rabbits were euthanized using a high dose of concentrated sodium pentobarbital, and bilateral femur/tibia were separated. The medullary cavity of the femur/tibia was carefully rinsed with DMEM/F12. The resulting suspension was centrifuged at 500 g for 5 min. The supernatant was removed, and red blood cell lysis buffer (Beyotime) was added. Cells were centrifuged again (500 g for 5 min) and resuspended. For BMDMs, cells were cultured in RMPI 1640 (ThermoFisher) supplemented with 10% fetal bovine serum (FBS) (Gibco), 20 ng/mL macrophage colony-stimulating factor (M-CSF) (Peprotech), and 1% penicillin and streptomycin (P/S) (Gibco). To avoid phenotypic changes, BMDMs were used at passage 0.
Evaluation of Chemotactic Effect of IL-8 on BMDMs and the Interference of IL-8 Neutralizing mAb
To investigate the chemotactic effect of IL-8 on BMDMs and to assess whether the IL-8 neutralizing mAb could significantly interfere with this process, a Transwell migration assay was performed. BMDMs were cultured and adjusted to a concentration of 50,000 cells per well for the migration assay. The experiment was divided into four groups: (1) control group (Control) with no IL-8 or IL-8 neutralizing mAb; (2) IL-8 group (IL-8); (3) IL-8 + antibody group (IL-8 + Ab) with added IL-8 and IL-8 neutralizing mAb; and (4) antibody group (Ab) with only IL-8 neutralizing mAb. The concentration for IL-8 applied is 50 ng/ml, and IL-8 neutralizing mAb is used at 100 μg/ml. 24 Transwell inserts with 8.0 μm pore size were placed in a 24-well plate, and the respective treatments were added to the lower chamber culture medium for each group. The BMDMs suspension was then added to the upper chamber of the Transwell inserts, and the plate was incubated at 37°C in a 5% CO2 incubator for 6 h. After incubation, nonmigrated cells on the upper surface of the membrane were removed with a cotton swab, and the migrated cells on the lower surface were washed with phosphate-buffered saline (PBS), fixed with methanol, and stained with 0.1% crystal violet. The number of migrated cells was counted under a microscope, and the cell migration in the four groups was compared to evaluate the impact of IL-8 on BMDMs chemotaxis and the potential interference of the IL-8 neutralizing antibody on this process.
Assessing the Efficacy of IL-8 Neutralizing mAb in Alleviating OA Progression in Rabbits
In this study, we aimed to investigate the potential therapeutic effects of IL-8 neutralizing mAb on the progression of OA in rabbits, using hyaluronic acid (HA) as a positive control. A total of 48 healthy rabbits (New Zealand white rabbit/female/2500 g) were randomly assigned into four groups (n = 12 per group): control group, ACLT group, HA group, and IL-8 neutralizing mAb group. OA was induced in all rabbits by anterior cruciate ligament transection (ACLT) surgery. Four weeks post-ACLT, the development of OA was confirmed, and the rabbits were further divided into two subgroups (n = 6 per subgroup) for each treatment group, with interventions lasting for 4 weeks and 8 weeks, respectively. Intra-articular injections of IL-8 neutralizing mAb, HA, or saline were administered to the corresponding groups once every 2 weeks. Assessments were conducted 1 week after the completion of each intervention period. The evaluation of OA progression included footprint analysis, magnetic resonance imaging (MRI), and histological examination of joint tissues. We assessed the functional status of the hindlimbs in rabbits through footprint analysis, and used MRI to evaluate the structure of joints and the integrity of cartilage. Histological examination including Safranin O/Fast green staining and immunohistochemistry was used to assess cartilage degeneration and other OA-related changes. By comparing the results across the three groups at the fifth week and ninth week, we aimed to determine the efficacy of IL-8 neutralizing mAb in alleviating OA progression in rabbits and to compare its effects with those of HA treatment.
Evaluation of Hindlimb Function in Rabbit OA Model
To evaluate the hindlimb functional status in rabbits, we constructed a custom-made runway with wooden boards, designed to accommodate the rabbit size. 25 The surface of the runway was covered with a thick layer of clean, fine-grained sand to enable the capture of distinct footprints as the rabbits ambulated across the track. Individual rabbits were placed onto the runway, and their movements were carefully monitored to analyze the footprints left by their hindlimbs in the sand. The surface area of the left and right hindlimb footprints was measured and compared to determine any limb preference for weight-bearing. A discernible preference for one limb over the other could indicate a more significant OA severity in the non-weight-bearing limb, as rabbits may avoid bearing weight on the affected limb due to pain or discomfort.
MRI of Rabbit Knee Joints
To assess structural changes and cartilage integrity in rabbit knee joints, 3.0T MRI was performed. Rabbits were anesthetized with sodium pentobarbital to ensure comfort and minimize movement during the procedure. The anesthetized rabbits were positioned in a prone position with their hindlimbs extended for optimal imaging of the knee joints. The imaging protocol included T1-weighted, T2-weighted, and proton density-weighted sequences to visualize joint structures and cartilage. Imaging parameters were adjusted to optimize image quality and minimize acquisition time. Two experienced, blinded radiologists assessed the images for cartilage degeneration, joint space narrowing, subchondral bone changes, and other OA-related alterations.
Gross and Histologic Evaluation
For histological analysis, rabbits were humanely euthanized, and their knee joints were harvested and fixed in 4% paraformaldehyde. The samples were subsequently decalcified in 10% EDTA solution and embedded in paraffin. Serial sagittal sections of 5 μm thickness were obtained at 50 μm intervals. Paraffin sections were then subjected to Safranin O/Fast green staining, as well as immunohistochemical staining, following the protocols established in our previous studies. 26
Statistical Analysis
Data were presented as means with 95% confidence intervals (CIs) and analyzed using GraphPad Prism 9.0 software. Each in vitro experiment was conducted in triplicate. The independent-sample t-test and one-way analysis of variance (ANOVA) with Tukey’s post hoc test, which were used for two different groups comparisons and multiple group comparisons, respectively, assume that the data are normally distributed and have homogeneity of variances. Normal distribution is a common type of continuous probability distribution with a bell-shaped, symmetrical curve. Homogeneity of variances indicates that the variances are equal across all groups. When dealing with ranked data or data that do not meet the assumptions of normal distribution and homogeneity of variance, non-parametric methods like the Kruskal-Wallis test and Nemenyi test were used. The Kruskal–Wallis test is a non-parametric method for comparing three or more independent samples, which does not require the assumption of normal distribution or equal variances. The Nemenyi test is used for pairwise comparisons following a significant Kruskal–Wallis result. In all cases, results were considered statistically significant when P < 0.05.
Results
IL-8 Neutralizing mAb Effectively Antagonized the Chemotactic Effect of IL-8 on BMDMs
As shown in
Figure 1A
, rabbit BMDMs were isolated according to the established protocol.
27
These BMDMs typically exhibit a distinct morphology, with an irregular shape, a large, round nucleus, and a granular cytoplasm indicative of a healthy state (
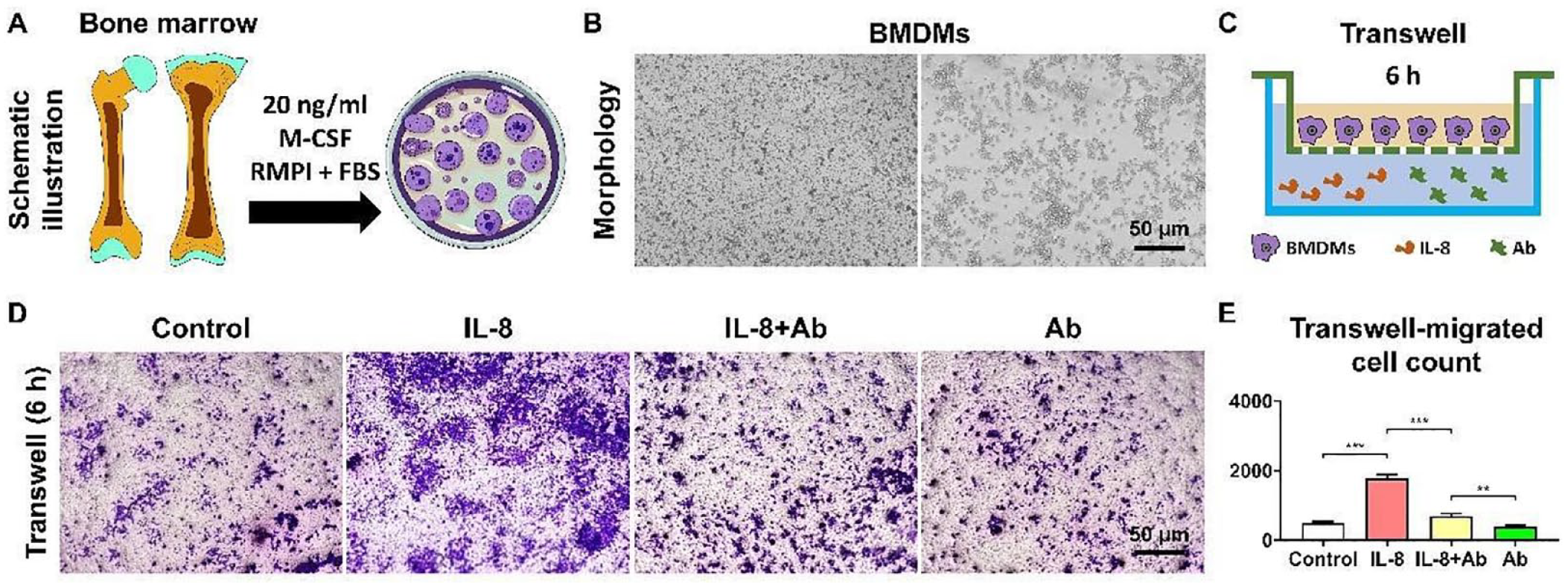
IL-8 neutralizing mAb effectively antagonized the chemotactic effect of IL-8 on BMDMs. A: Isolation of BMDMs; B: Morphology of BMDMs; C: Schematic of Transwell experiment; D: Detection of BMDMs migration via crystal violet staining; E: Quantitative Analysis of BMDMs migration. **P < 0.01; ***P < 0.001.
Intra-Articular Injection of IL-8 Neutralizing mAb Effectively Improved Lower Limb Function in OA Rabbit
Compared with control group, at fifth and ninth weeks posttreatments with IL-8 neutralizing mAb and HA, rabbits from the ACLT group demonstrated a significant reduction in both paw prints and reluctance to bear weight on the affected limb. However, compared to the ACLT group, rabbits in both HA and anti-IL-8 group showed little difference in footprint between the two sides, suggesting a significant improvement in lower limb weight-bearing conditions (
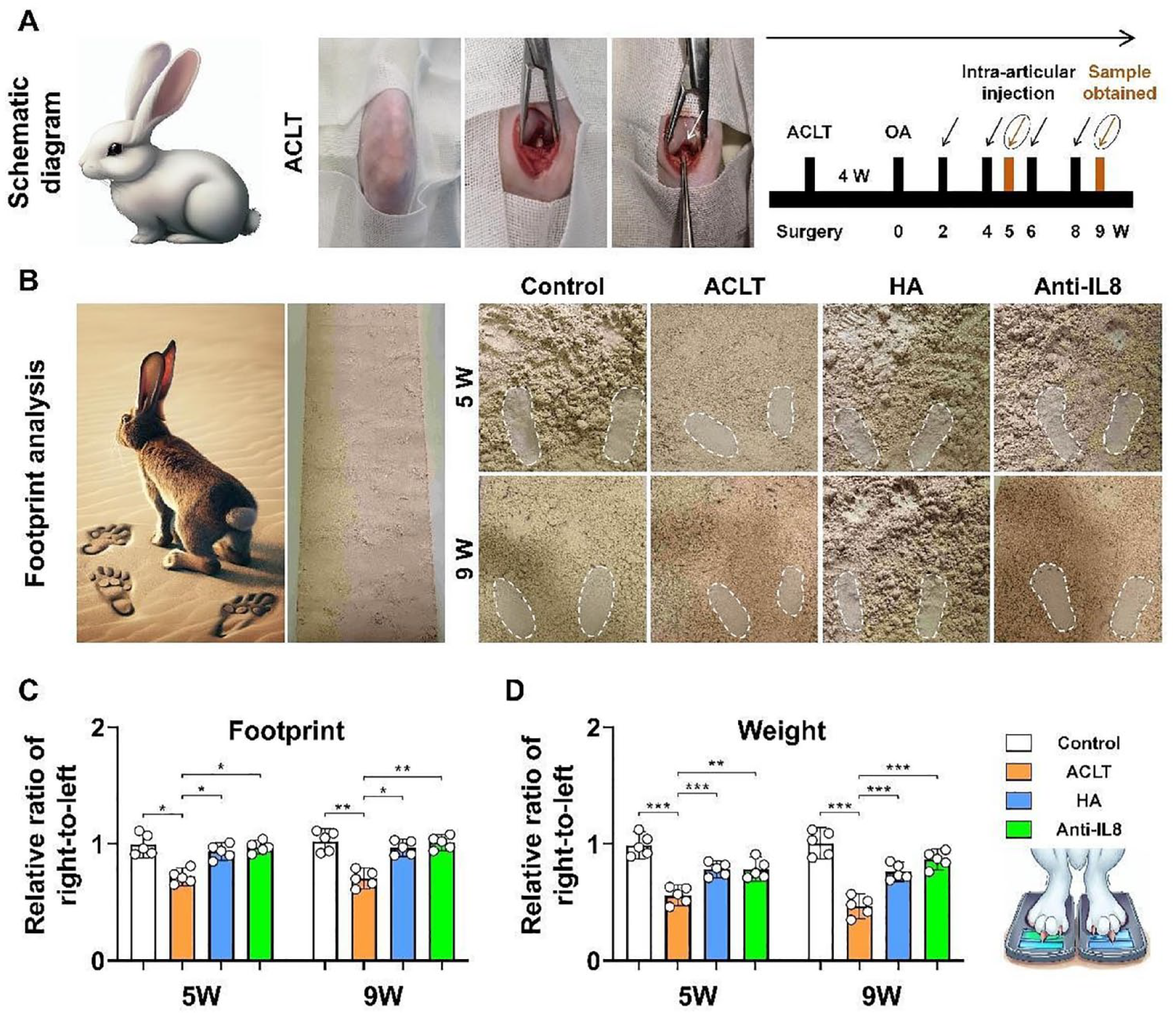
Intra-articular injection of IL-8 neutralizing mAb effectively improved lower limb function in OA rabbit. A: Process of the ACLT Surgery in Rabbits (the black arrows indicate the injection of hyaluronic acid or IL-8, while the orange arrows represent sample collection); B: Footprint analysis of OA rabbits on the sand (the white dashed boxes represent the footprint area of the rabbits); C: Ratio of footprint area between right and left feet of OA rabbits; D: Ratio of weight bearing between right and left feet of OA rabbits. **P < 0.01; ***P < 0.001.
Intra-Articular Injection of IL-8 Neutralizing mAb Effectively Reduced Joint Effusion and Alleviates Bone Marrow Edema
Following intra-articular injection of IL-8 neutralizing mAb and HA, MRI results at the fifth week showed that, compared to the control group, the ACLT group exhibited significant increase in joint effusion in the rabbit knee, presenting with “meniscal extrusion.” In comparison to the ACLT group, both the HA and the anti-IL-8 groups showed significantly reduced joint effusion, except some areas still displaying signs of bone marrow edema. MRI results at the ninth week indicated that, compared to the control group, the ACLT group had more effusion in the joint and showed clear femoral condyle cartilage defects along with significant bone marrow edema. Compared to the ACLT group, both the HA and the anti-IL-8 group showed significantly reduced joint effusion and minimal signs of bone marrow edema (
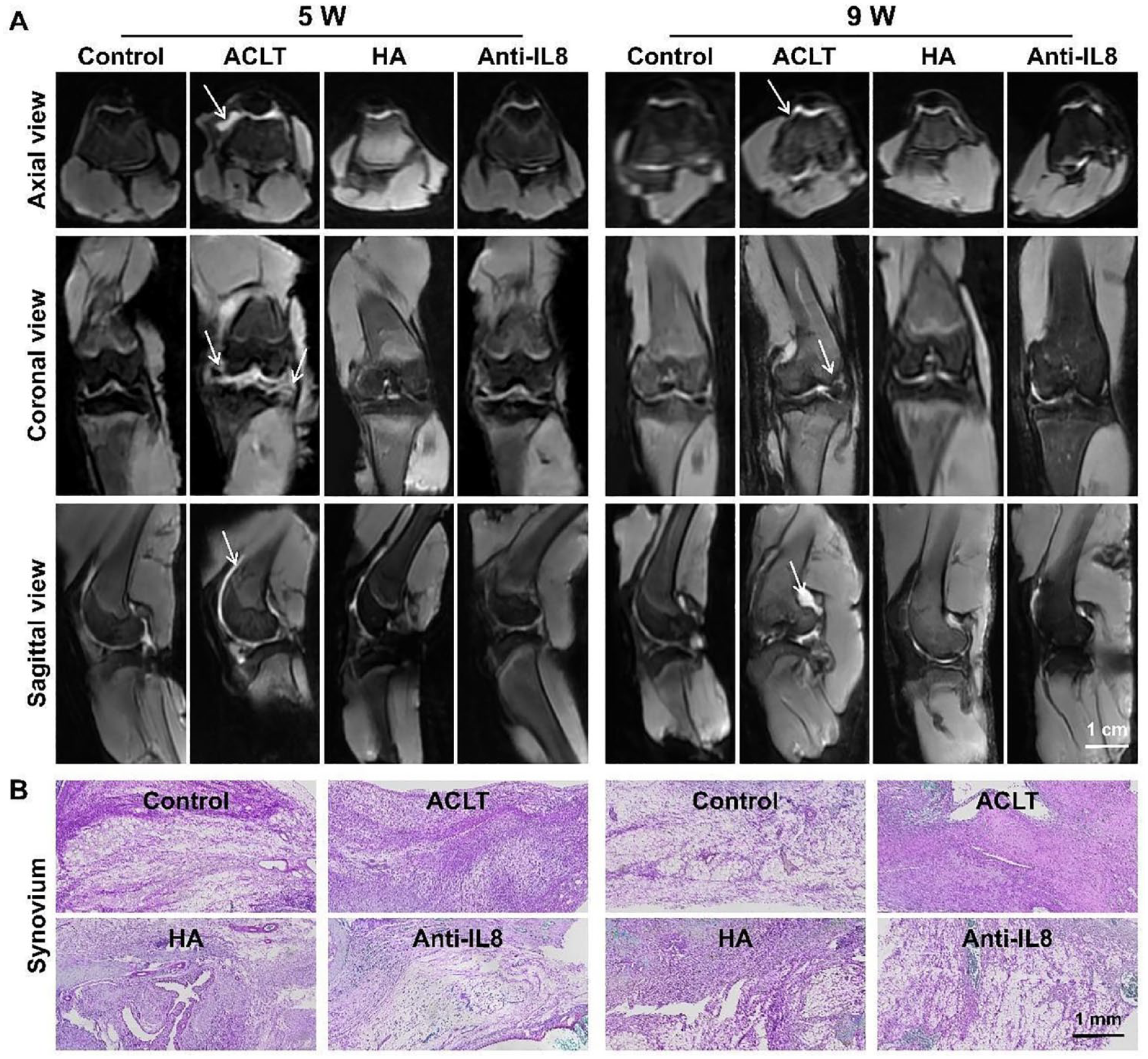
Intra-articular injection of IL-8 neutralizing mAb effectively reduced joint effusion, mitigated bone marrow edema, and alleviated synovitis. A: MRI of rabbit knee joints, with white arrows indicating joint effusion; B: Histological staining of synovial tissue in the knee joint.
Intra-Articular Injection of IL-8 Neutralizing mAb Effectively Alleviates Synovitis
By the fifth week, histological staining of the synovium revealed noticeable signs of synovitis in the ACLT rabbit group, characterized by dense infiltration of inflammatory cells, synovial hyperplasia, and thickening of the synovial lining layer. In contrast, the synovium in both the HA and the anti-IL-8 groups displayed milder inflammation, evidenced by fewer inflammatory cells, reduced synovial hyperplasia, and a thinner synovial lining layer. In addition, the anti-IL-8 treatment with neutralizing mAb resulted in a greater alleviation of synovitis. By the ninth week, the overall trend in histological staining was consistent across all groups as compared to the fifth week. Moreover, the ACLT group showed aggravated synovitis at the ninth week, while the level of inflammation in the HA and anti-IL-8 groups was slightly mitigated relative to the fifth week. In the group treated with IL-8 neutralizing mAb, the remission of synovitis was maintained. Nine weeks posttreatment, the histologic morphology and synovitis score in this group were comparable to those of the control group (
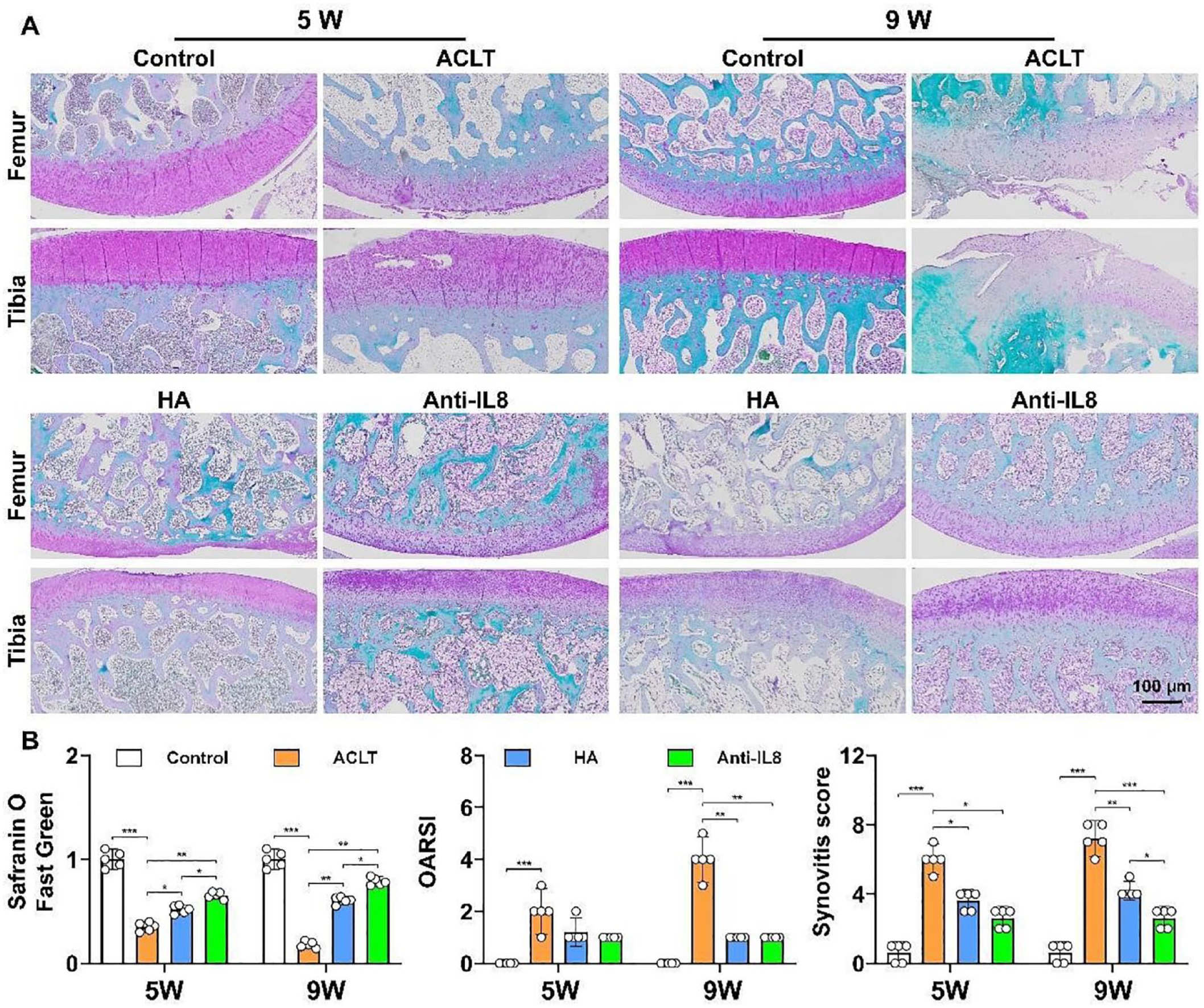
Intra-articular injection of IL-8 neutralizing mAb effectively enhanced cartilage matrix expression in OA rabbits. A: Safranin O/Fast Green staining of rabbit knee joints; B: Semi-quantitative analysis of Safranin O/Fast Green Staining, OARSI scoring, and synovial tissue inflammation scoring. *P < 0.05; **P < 0.01; ***P < 0.001.
Intra-Articular Injection of IL-8 Neutralizing mAb Effectively Enhances Cartilage Matrix Expression in OA Rabbits
Compared to the control group, at the fifth week, Safranin O-Fast Green staining revealed that the articular surface in the ACLT group was unsmooth, with local detachment and damage. Notably, the chondrocytes were proliferating in certain areas, displaying a disorganized arrangement. The expression of the cartilage matrix was reduced, and vacuolation was observed in some areas. Compared to the ACLT group, the knee joint surface in HA group was relatively smoother, with more abundant cartilage matrix expression, though the thickness of the cartilage was reduced in some areas. The anti-IL-8 group showed significant increase in cartilage matrix expression, a slightly more normal cell arrangement, a smooth surface, and no prominent defects. At the ninth week, Safranin O-Fast Green staining revealed that the joint surface in the ACLT group exhibited noticeable peeling, compensatory thickening of the cartilage due to proliferation, local cell morphological disarray, and cartilage surface rupture that reached the subchondral bone, when compared with the control group. Compared to the ACLT group, both the HA group and the anti-IL-8 group effectively improved cartilage matrix expression. The cartilage surface was smooth, the cell arrangement was orderly, and the structure was clear and rational. The anti-IL-8 group showed even higher cartilage matrix content expression than the HA group. In addition, IL-8 neutralizing mAb showed more rapid restoration in articular cartilage and subchondral bone morphology, evidenced by higher cartilage matrix content expression than the HA group at both the fifth and ninth week post treatment (
Intra-Articular Injection of IL-8 Neutralizing mAb Effectively Reduced IL-8 Expression in Joint Tissue
The immunohistochemical staining against IL-8 in the knee joint tissue revealed that the expression levels of IL-8 varied among groups and at each time point. At the fifth week, the knee joint in the ACLT group exhibited the highest IL-8 expression level, followed by the HA group, the anti-IL-8 group, and the control group, in that order. Interestingly, at the ninth week, the expression level of IL-8 in the ACLT group was even higher than at the fifth week. This suggested not only an ongoing inflammatory response but also a potential decline in cartilage repair activity due to the persistent inflammation. In contrast, in both the HA group and the anti-IL-8 group, the IL-8 expression levels were reduced. Among groups, the anti-IL-8 group showed a particularly notable decrease in IL-8 expression levels at the ninth week, which was even more substantial than the reduction observed in the HA group at the fifth week. Despite these observations, it is important to note that IL-8 expression levels in both the ACLT and HA groups were still higher than in the normal group at the ninth week, indicating the persistence of the inflammatory reaction, albeit at a reduced level (
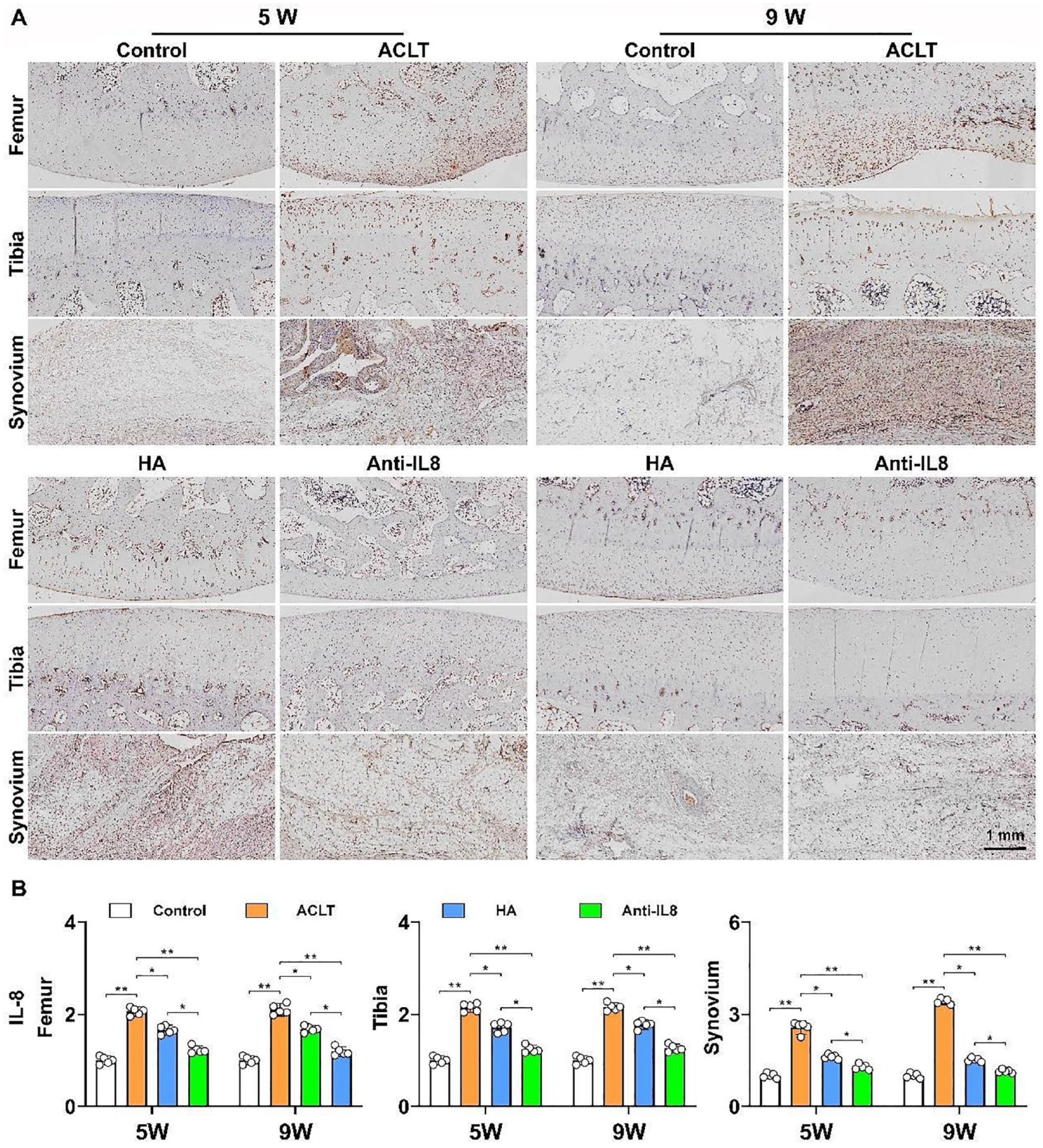
Intra-articular injection of IL-8 neutralizing mAb effectively reduced IL-8 expression in joint tissue. A: Immunohistochemical staining of IL-8 in rabbit knee joints; B: Semi-quantitative analysis of IL-8 immunohistochemical staining. *P < 0.05; **P < 0.01.
Discussion
The pro-inflammatory cytokine IL-8 plays a significant role in the pathogenesis of OA, being involved in the recruitment and activation of neutrophils, thereby promoting inflammation and degradation of the extracellular matrix, ultimately leading to pathological changes in OA. 28 In this study, we investigated the therapeutic potential of an IL-8 neutralizing antibody using a rabbit model of OA. Our findings demonstrated that administration of the IL-8 neutralizing mAb not only mitigated disease progression but also facilitated cartilage repair.
Neutralizing antibodies, which exert their effects through targeted inhibition of specific proteins, have been increasingly employed in the treatment of various diseases, including cystic fibrosis, 29 inflammatory bowel disease, 30 and cancer. 31 Among them, IL-8 neutralizing antibodies possess a distinctive mechanism of action with the potential to mitigate inflammatory responses, 24 thereby alleviating conditions such as synovitis and reducing joint effusion, which are commonly observed in OA. Moreover, by neutralizing IL-8, they may restore the balance between cartilage synthesis and degradation, thus promoting cartilage repair.
Concurrently, an expanding body of research has been investigating the role of joint effusion and bone marrow edema in OA. Joint effusion, characterized by excessive fluid accumulation within the joint cavity, is a prevalent symptom of OA that can contribute to pain and functional impairment. 32 Bone marrow edema, identified by increased fluid in the bone marrow as observed in MRI scans of OA patients, has been associated with higher levels of pain. 33 These two conditions signify the presence of inflammation and may potentially be alleviated through interventions targeting inflammatory pathways such as IL-8 neutralizing antibodies. In our study, we observed a reduction in both joint effusion and signs of bone marrow edema among treated rabbits, providing support for the potential efficacy of IL-8 neutralization in managing these common symptoms associated with OA. In addition, an increasing number of studies are exploring gait analysis as a valuable tool in OA research. 34 Gait analysis evaluates locomotion patterns in animals or humans often by examining footprints and has been used non-invasively to assess pain levels and functional impairment related to OA. Alterations in gait parameters like stride length or weight-bearing can indicate disease severity and treatment effectiveness. Consistent with these findings, our study also noted significant improvements both in footprint characteristics and weight-bearing capacity among treated rabbits further reinforcing the potential benefits offered by IL-8 neutralizing antibodies for treating OA.
In the design of our experiments, we selected HA as the positive control due to its extensive clinical usage, proven efficacy, and established safety profile. Our findings from knee joint MRI and gait analysis demonstrate that the IL-8 neutralizing mAb exhibits comparable effects to HA in terms of joint effusion, bone marrow edema, and weight-bearing stress distribution in the lower limbs. Notably, the IL-8 neutralizing mAb exhibits an enhanced capacity for alleviating synovial inflammation and promoting cartilage tissue repair. This can be attributed to the antibody’s precise targeting of the inflammatory cytokine IL-8 within the joint space. By inhibiting IL-8’s chemotactic activity, this antibody may reduce macrophage infiltration and quantity, which is crucial in OA’s inflammatory cascade. Despite its potential in OA treatment, HA currently remains indispensable due to its convenience in clinical application and cost-effectiveness. Further enhancements in manufacturing processes for IL-8 neutralizing mAbs are essential along with a deeper understanding of their mechanisms in treating OA. Moreover, conducting subsequent clinical trials to evaluate these aspects will be pivotal for translating these IL-8 antibodies into clinical practice.
While our study provided support for the therapeutic potential of IL-8 neutralizing mAb in OA, acknowledging its limitations will guide further research. The persistent presence of IL-8 in the joint implies that future antibody therapies may necessitate enhanced neutralization capacity, potentially through controlled-release or responsive delivery systems to achieve sustained therapeutic effects. 35 Further investigations should scrutinize the underlying mechanisms responsible for the observed effects, as well as assess the biodistribution and pharmacokinetics of these antibodies while optimizing dosage regimens. Addressing these aspects will ensure both safety and efficacy of this therapeutic strategy, thereby facilitating its clinical translation. With a deeper understanding of IL-8’s role in OA, development of more refined antibody therapies could emerge as potent tools to combat this debilitating disease.
Conclusion
Our study has demonstrated the potential of IL-8 neutralizing mAb as a novel therapeutic strategy for treating OA. The antibody has the ability to mitigate inflammatory responses and promote cartilage repair, offering an alternative or adjunct to traditional treatments such as HA. Despite promising results, further research is necessary to fully understand the underlying mechanisms, optimize dosing regimens, and ensure long-term safety and efficacy. This study provides new insights into OA treatment and could pave the way for improved patient outcomes in this debilitating disease.
Footnotes
Author Contributions
Conception and design: Xiaoyuan Gong, Guangxing Chen, and Junjun Yang.
Analysis and interpretation of the data: Junjun Yang, Xin Wang, Zhenlan Fu, Rong Wang, Yanming Ma, and Wang Cai.
Drafting of the article: Junjun Yang, Xin Wang, Yizhao Zhou, and Xiaoyuan Gong.
Critical revision of the article for important intellectual content: Guangxing Chen and Cheng Chen.
Final approval of the article: Xiaoyuan Gong, Guangxing Chen, and Junjun Yang.
Statistical expertise: Junjun Yang, Dejie Fu, and Shuo Meng.
Obtaining of funding: Xiaoyuan Gong, Guangxing Chen, Cheng Chen, and Junjun Yang.
Acknowledgments and Funding
The authors extend their gratitude to Yes Biotech Laboratories Ltd. for their generous donation of IL-8 Neutralizing mAb for our research. They also thank Kunming Baker Norton Pharmaceutical for providing Hyaluronic Acid (HA, ARTZ® Dispo, Imported Drug License: H20140533). Besides, they express their appreciation to Chongqing Yinglian Medical Imaging Diagnosis Center Co., Ltd. for their invaluable provision of MRI scanning services. In addition, they are grateful to Knorigene Technologies for their technical support in histologic staining. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chongqing Natural Science Foundation General Project (grant nos. cstc2021jcyj-msxmX0135 and CSTB2022NSCQ-MSX0139), Chongqing Natural Science Foundation Innovation Development Joint Fund (Municipal Education Commission) Project (grant no. 2022NSCQ-LZX0256), Natural Science Foundation of Hunan Province (grant no. 2021JJ304060) and Chongqing Postdoctoral Research Project Special Fund (grant no. 2021XM3033).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
