Abstract
Background
Complex meniscal lesions often require meniscectomy with favorable results in the short term but a high risk of early osteoarthritis subsequently. Partial meniscectomy treated with meniscal substitutes may delay articular cartilage degeneration.
Purpose
To evaluate the status of articular cartilage by T2 mapping after meniscal substitution with polyurethane scaffolds enriched with mesenchymal stem cells (MSC) and comparison with acellular scaffolds at 12 months.
Methods
Seventeen patients (18-50 years) with past meniscectomies were enrolled in 2 groups: (1) acellular polyurethane scaffold (APS) or (2) polyurethane scaffold enriched with MSC (MPS). Patients in the MPS group received filgrastim to stimulate MSC production, and CD90+ cells were obtained and cultured in the polyurethane scaffold. The scaffolds were implanted arthroscopically into partial meniscus defects. Concomitant injuries (articular cartilage lesions or cartilage lesions) were treated during the same procedure. Changes in the quality of articular cartilage were evaluated with T2 mapping in femur and tibia at 12 months.
Results
In tibial T2 mapping, values for the MPS group increased slightly at 9 months but returned to initial values at 12 months (P > 0.05). In the APS group, a clear decrease from 3 months to 12 months was observed (P > 0.05). This difference tended to be significantly lower in the APS group compared with the MPS group at the final time point (P = 0.18). In the femur, a slight increase in the MPS group (47.8 ± 3.4) compared with the APS group (45.3 ± 4.9) was observed (P > 0.05).
Conclusion
Meniscal substitution with polyurethane scaffold maintains normal T2 mapping values in adjacent cartilage at 12 months. The addition of MSC did not show any advantage in the protection of articular cartilage over acellular scaffolds (P > 0.05).
Keywords
Introduction
The knee is one of the most frequently injured joints in the human body. In 15% of all knee injuries either one of the menisci is involved. 1 Previously reported incidence rates for hospital admission after meniscal injury vary between 0.35 and 0.7 per 1000 person-years. 2 Knee osteoarthritis (OA) shares many risk factors and biological processes with degenerative meniscal tears. OA affects 27% of women and 21% of men, with an increase in incidence for both groups from the age of 45 years. According to an arthroscopic study, there is a 40% coincidence rate of meniscal injuries in those with knee OA. Furthermore, people who had previously partial or total meniscectomies are at risk of developing OA subsequently. 3
The meniscus plays a pivotal role in load bearing and load transmission to the cartilage and the subchondral bone. 4 After removal of the meniscus, the contact area decreases by 40% medially and by as much as 52% laterally.5,6 Meniscus injuries occur either traumatically and are often associated with an articular cartilage lesion (ACL) rupture, or chronically as a result of structural weakening due to aging and wear.7,8 Repair strategies allow preservation of the meniscal tissue when the rupture is not complex and is located in the vascular area. 9 However, a high number of complex lesions cannot be repaired and usually require partial or total meniscectomy as the current standard of care, and favorable results are observed in the short term following a partial meniscectomy. However, the risk of early OA has been reported due to the decreased contact area when the menisci are missing and subsequent mechanical overload of the underlying articular cartilage. Dhollander et al. reported a decrease in intraarticular contact area of approximately 75% and an increase in peak contact pressure by approximately 235% after a total meniscectomy. 10 Comparable results were found in a study by Ahmed and Burke.4,11 Roos et al. reported a long-term clinical study with a follow-up of 21 years of patients after total meniscectomy compared with matched controls. They confirmed that the increased pressure seen in biomechanical studies led to radiographic evidence of OA with a relative risk factor of 14. 12
Several long-term studies have shown that partial meniscectomy treated with meniscal substitutes may delay degeneration but not prevent it. 13 A study of 136 patients who underwent partial meniscectomy for isolated meniscal tears showed a re-operation rate of 22.8% at 8.5 years, and 53% of patients had osteoarthritic radiographic changes compared with only 22% in the unaffected control knee. 14 Meniscal substitutes have been used to relieve symptoms in patients who underwent partial meniscectomies.15,16 The objective of these implants is to provide a better functionality of the meniscus, reduce the level of pain in young patients, and delay the onset of OA. Meniscal substitution with a polyurethane scaffold (Actifit, Orteq Sports Medicine, London, UK) has been shown to be an effective treatment by providing significant pain relief, and functional improvement in the short and medium term. 17 Using a different material, a polycarbonate urethane, Shriram et al. demonstrated that the contact pressure on the articular cartilage and implant displacement were sensitive to the stiffness of the polycarbonate urethane meniscal implant. According to their study, increasing the material stiffness of the meniscal implant led to elevated contact pressures on the articular cartilage, which increased the risk of physiological damage to the articular cartilage. 14 It is believed that the size of the implant is correlated with these findings. 18
To the best of our knowledge, the evaluation of cartilage protection or delay in deterioration by T2 mapping in patients following meniscectomy and treatment with a polyurethane scaffold has not been described before. Furthermore, the clinical use of mesenchymal stem cell (MSC) with a polyurethane scaffold has not been reported. Therefore, we decided to develop the present study to evaluate the changes in articular cartilage quality after meniscectomy treated with acellular polyurethane scaffolds and compare them to MSC-enriched scaffolds at 12 months.
Methods
The study was performed in accordance with Good Clinical Practice and the Declaration of Helsinki. It was approved by the ethics committee of the National Institute of Rehabilitation “Luis Guillermo Ibarra Ibarra,” Mexico City, Mexico. Written informed consent was obtained from all patients for this prospective, longitudinal, comparative, and analytic study. Patients with a history of previous partial irreparable meniscal injury or partial (or subtotal) meniscectomy were included.
In the original design, only 11 patients were included to be treated with the MSC-enriched polyurethane scaffold. However, 6 young patients diagnosed with pathologies (ACL reconstruction or cartilage repair) treatment were found to have complex meniscal lesions that required meniscectomy in the index procedure and were therefore treated with acellular polyurethane scaffolds.
The patients were between 18 and 50 years old, had a history of chronic symptomatic meniscectomy (>25% to 90%) or acute meniscectomy performed during ACL reconstruction or any cartilage repair techniques, clinical and radiographic alignment, and body mass index (BMI) <30. Patients with OA grade IV evaluated by X-ray, history of septic arthritis, hypersensitivity to any implant component (Actifit) or to filgrastim, personal or familiar history of neoplasia, and presence of a hematopoietic system pathology were excluded. Patients were divided in 2 groups: (1) polyurethane scaffold enriched with mobilized MSCs (MPS) and (2) acellular polyurethane scaffold (APS).
Mesenchymal Stem Cell Mobilization
Patients enrolled in the MPS group received a daily subcutaneous injection of 300 µg of G-CSF (Filgrastim, Amgen, Thousand Oaks, CA) in the deltoid area for 3 consecutive days. The objective of G-CSF was to increase the pool of MSC in the peripheral blood stream. In order to observe the behavior of leukocytes, granulocytes, monocytes, and lymphocytes, a blood count control and hematic cell count were performed from day 1 to day 4. A daily questionnaire was performed to detect side effects that could be caused by the filgrastim application as headache, arthralgia, sternal pain, iliac crest pain, and fatigue.
Mobilized Mesenchymal Stem Cells Harvesting
At day 4, 400 mL of peripheral blood was obtained from a brachial vein. The blood unit was separated to obtain an average of 50 mL of the leukocyte fraction (“buffy coat”) ( Fig. 1A ).

Peripheral blood sample with mobilized mesenchymal stem cells after granulocyte colony stimulating factor application. (
Isolation of Mononuclear Cells from the Buffy Coat
In the Biotechnology Unit of our institution, the buffy coat was processed under sterile conditions inside a class 100 laminar flow hood with a cell separation kit (CS-900) to separate the mononuclear cells from the white fraction in a Biosafe Sepax 2 cell separation system (GE Life Sciences, Marlborough, MA). This system consists of a fully automatic, closed-loop, sterile mononuclear cell separator that allows a mononuclear isolation of the blood components through optical sensors. The content is transferred from the collector bag to a 50 mL tube and then centrifuged for 10 minutes at 300g to begin the process of mononuclear cell count ( Fig. 1B ).
Isolation of CD90+ Cells
CD90+ cells were isolated from the mononuclear cells after centrifugation ( Fig. 1C ). Ten microliters of mononuclear cells were taken, mixed in a 1-mL tube with 990 µL of marking solution, and read in an automated cell counter (Scepter, Millipore Sigma, Burlington, MA). Once the number of mononuclear cells had been established, the content was divided into three 15-mL tubes to which 300 µL of anti-CD90+ antibody (Miltenyi Biotec, Bergisch Gladbach, Germany) was added. The 15-mL tubes were incubated during 20 minutes at 4°C using an orbital agitator (MACSmix Tube Rotator, Miltenyi). After incubation, the manual cell separation kit was assembled inside a laminar flow hood and the LS cell separation columns (Miltenyi) were placed. The cell/antibody sample was passed through the column with magnetic beads. At the end of passing the sample through the column, this column was removed from the separation system and placed in a 15-mL tube ( Fig. 1D ). The tube was centrifuged at 300g for 10 minutes and an aliquot of 10 µL was taken and then processed. The cell count was then repeated with an automated counter (Scepter) ( Fig. 1E ).
Characterization by Flow Cytometry
To establish their immunophenotype, an aliquot of 2.5 × 104 cells in 100 µL of PBS were incubated with 5 µL of the antibody suspension for 30 minutes at 4°C. The monoclonal antibodies applied were CD34, CD45, CD90, CD117, CD73, CD166, CD105, and CD47 (BD Biosciences, San Diego, CA). The samples and/or unlabeled controls were included for each antibody and used to set the electronic gates on the flow cytometer. All data were obtained in a BD FACSCalibur flow cytometer and analyzed with CellQuest PRO software (BD Biosciences) with a mean of 20,000 events ( Fig. 1F ).
Culturing of CD90+ Cells
An aliquot of 2.5 × 104 CD90+ cells were isolated from the mobilized peripheral blood cultured in Dulbecco’s modified Eagle’s medium (DMEM; Gibco Life Technologies, Carlsbad, CA), 20% autologous serum, and 1% antibiotic-antimycotic (Gibco). Cells were cultured in an incubator at 37°C with 5% CO2 in 75 cm2 culture flasks for 15 days.
Cell Seeding in Polyurethane Scaffold
After 15 days of culture, cells were seeded in the polyurethane meniscal scaffold by adding approximately 20 × 106 cells ( Fig. 1G ). The surface of the scaffold was sealed in order to avoid cell loss with the use of fibrin glue covering all the implant surfaces ( Fig. 1H ). Then was left in culture media in the incubator for 2 days to promote cell adhesion. The scaffold was kept in sterile conditions and sent to the operating room for arthroscopic implantation ( Fig. 2 ).
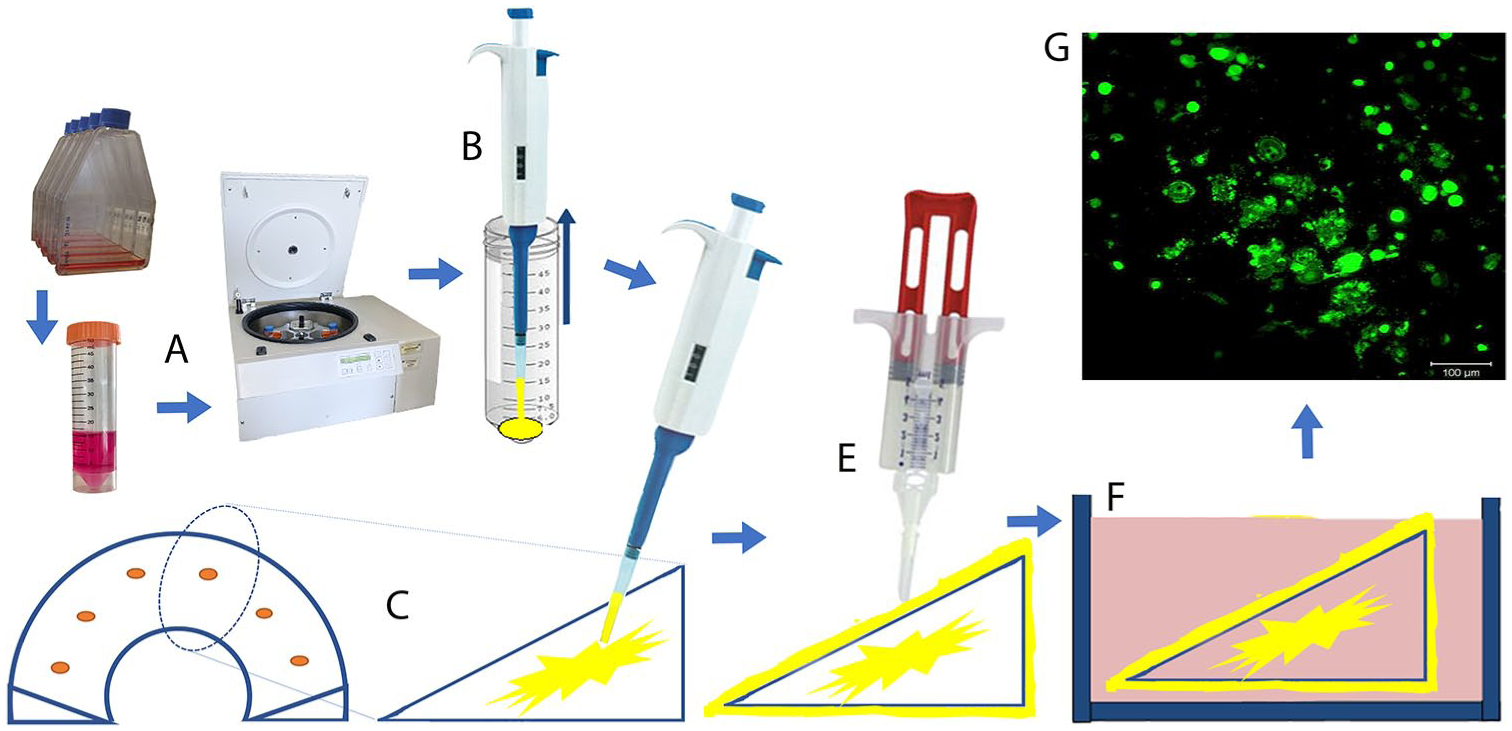
Cell seeding in the polyurethane meniscal scaffold. (
Surgical Technique: Arthroscopic Implantation
All cases were performed arthroscopically under regional anesthesia by the same surgeon (AOM) via standard anterolateral and anteromedial portals. Routine knee arthroscopy was performed documenting chondral surfaces, ligament stability, and state of the meniscus to confirm the appropriate indication for meniscal substitution. At the time of the index surgery, remnants or damaged sections of the previous meniscectomy were excised, leaving a residual meniscal rim in the vascular zone and straight walls in the adjacent native meniscal tissue. The size of the defect was measured by arthroscopy, then the implant was trimmed on the back table 10 mm larger than the defect size to prevent over-tensioning of the sutures when fixed to native meniscus ( Fig. 3A ). The scaffold was delivered into the joint through a 10-mm cannula and sutured to the capsule and native meniscus borders. For the posterior horn, all-inside fixation with nonabsorbable suture (Fast-Fix, Smith & Nephew, Andover, MA) was used. For fixation of the scaffold in the meniscus body, an inside-out technique with absorbable PDS suture and a protector meniscus device (Arthrex, Naples, FL) was used. For the anterior horn, an outside-in fixation with PDS suture was used with a micro-suture lasso device (Arthrex, Naples, FL) ( Fig. 3B to F ).
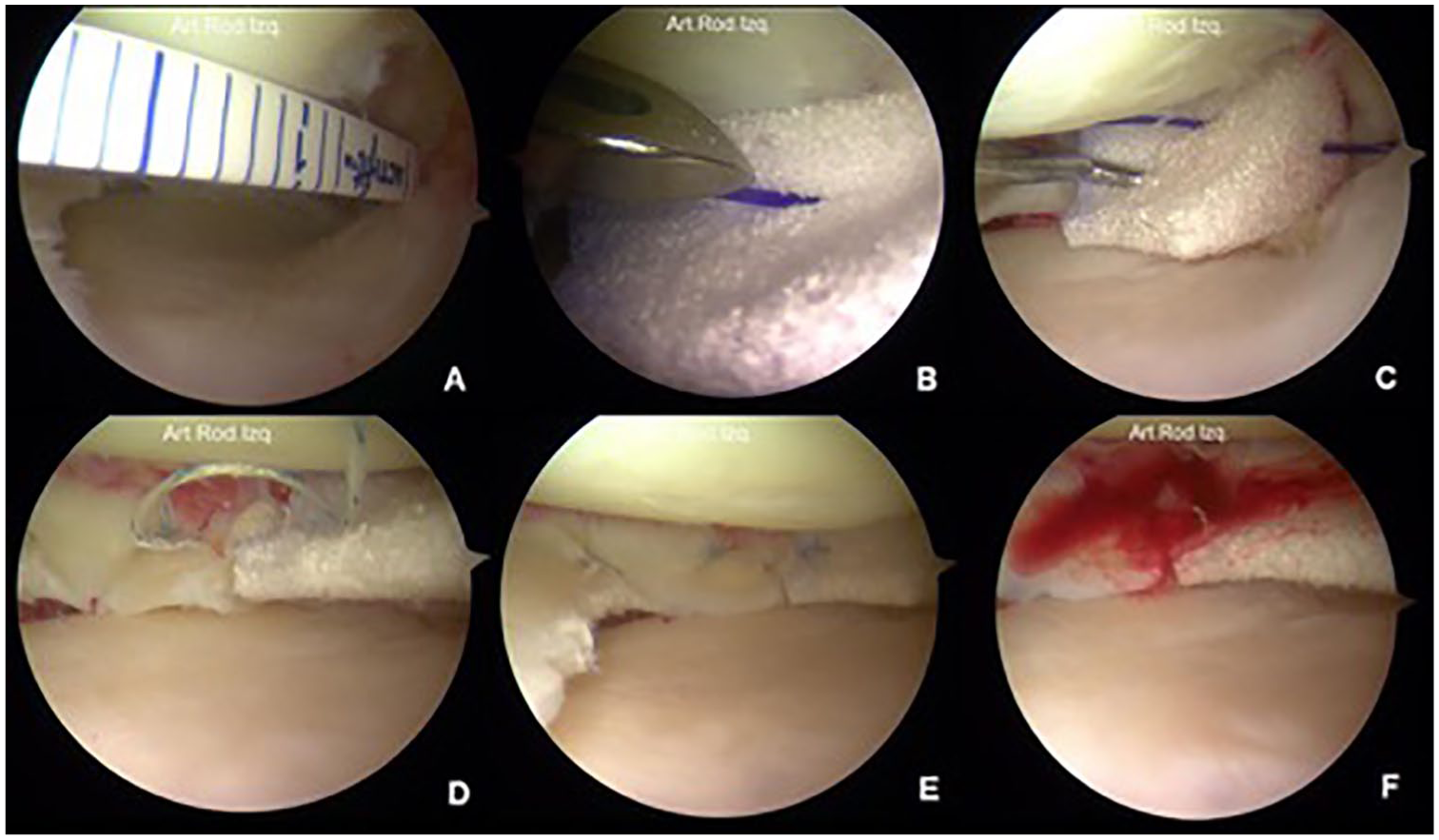
Surgical technique of medial meniscus substitution in the posterior horn with polyurethane implant enriched with MSCs. (
Cartilage status in the tibia plateau and femoral condyle was assessed with the International Cartilage Repair Society (ICRS) scoring system. For patients in the APS group, the scaffold was opened in the operating room without any processing. For the patients in the MPS group, the cultured scaffold was transported from the biotechnology laboratory to the operating room in sterile conditions soaked in DMEM culture media with 10% autologous serum and 1% antibiotic-antimycotic.
Rehabilitation Protocol
All patients received postoperatively a knee brace locked from full extension to 90° of flexion for 6 weeks, which was removed 3 to 4 times per day to perform self-assisted passive range-of-motion exercises. During the first 4 weeks, the range of motion was limited from 0° to 90°, then allowed to increase to 130°. After 6 weeks, the brace was unlocked and worn for comfort only. Walking was allowing but with non–weight-bearing during the first 2 weeks. At 3 to 4 weeks, transitioning from partial to total weight-bearing was allowed. After 12 weeks, muscle strengthening and unrestricted activity was performed.
Imaging Evaluation
Magnetic resonance imaging (MRI) evaluation was performed using T2 mapping at 3, 6, 9, and 12 months postoperatively using a 1.5 Tesla clinical imaging system (GE Healthcare, Milwaukee WI), using an 8-channel HD knee array (GE Healthcare). The cartilage quality in the involved compartment was evaluated by 2 independent radiologists using T2 mapping (FuncTool 4.5.1, GE Healthcare) to identify deterioration signs of the articular cartilage adjacent to the meniscal scaffold. Those values were calculated by taking 2 regions of interest (ROI): weight-bearing area of the femoral condyle (FC) and tibial plateau (TP) in sagittal planes ( Figs. 4 and 5 ).
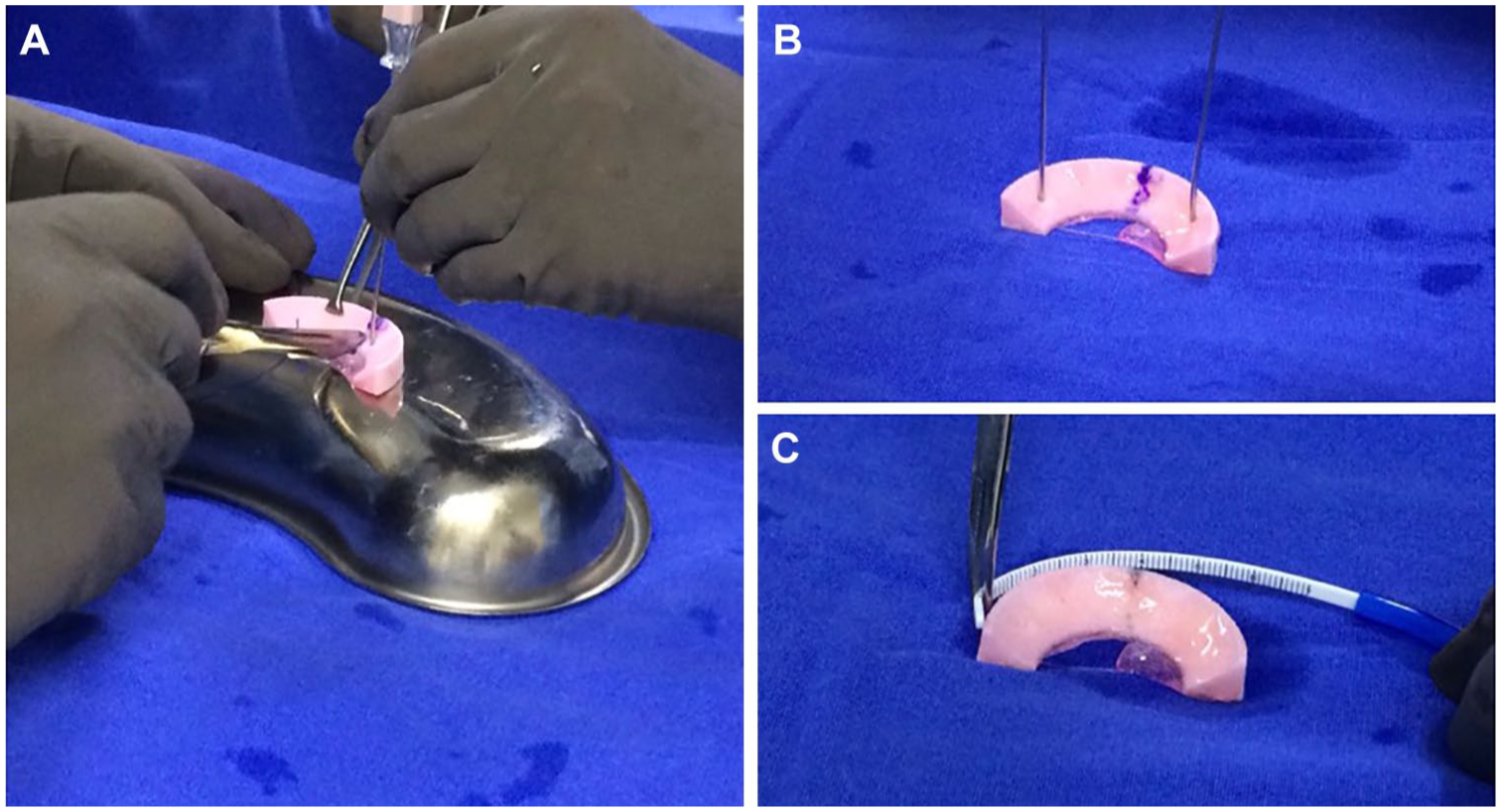
Polyurethane implant enriched with MSCs and sealed with fibrin glue. (
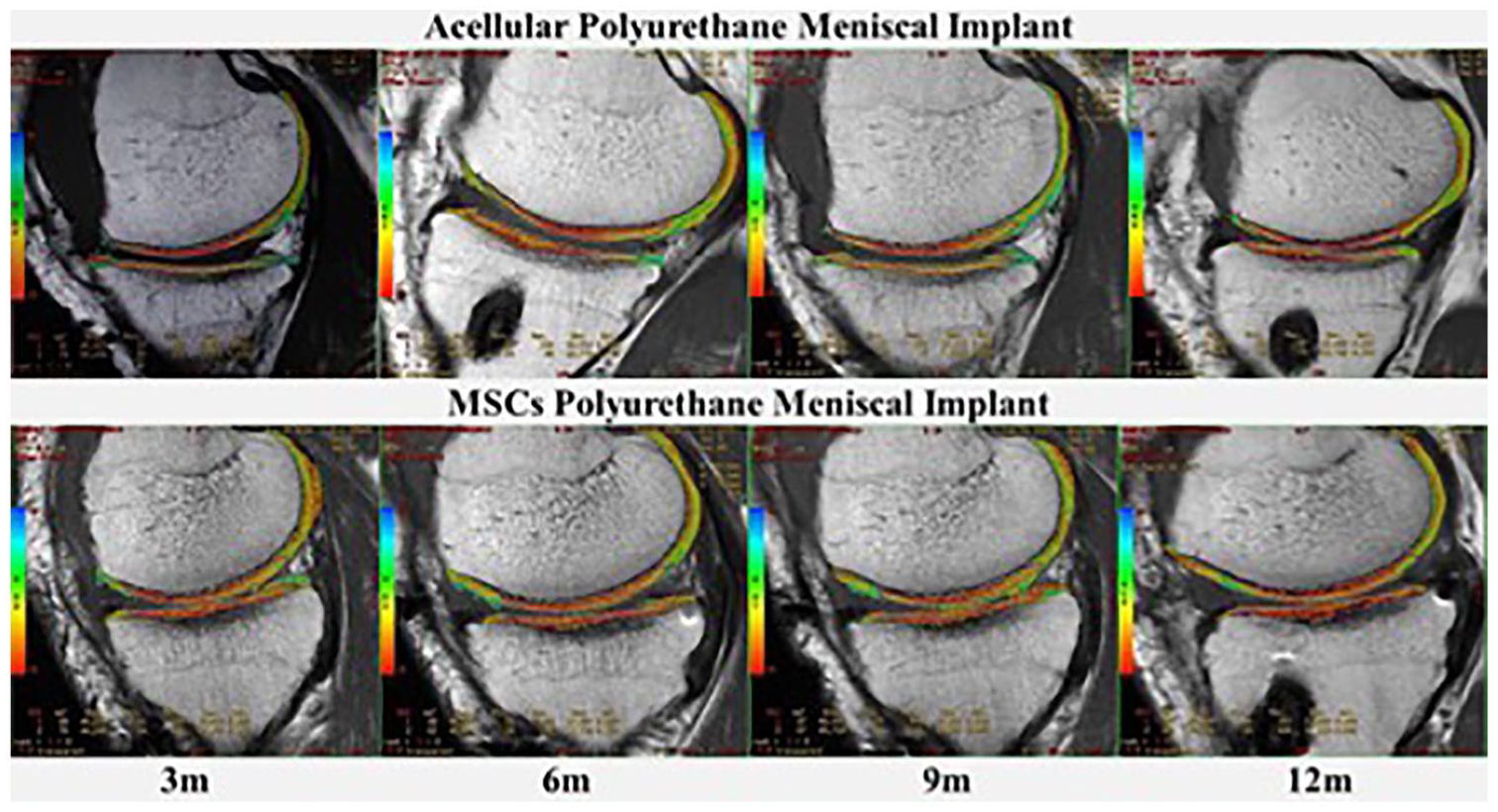
Evaluation of the cartilage status after meniscal substitution by T2 mapping. In the acellular group the tendency in the tibial plateau was toward decrease significantly from 3 to 12 months. Cartigram values in the MSCs group slightly increased at 9 months but at 12 months returns to the initial measurements. This difference tends to be significantly lower in acellular patients than cellular implants at final outcome (P = 0.18).
Clinical Evaluation
Before surgery and postoperatively (3, 6, 9 and 12 months), all patients underwent clinical follow-up examinations and Lysholm functional scores were used to assess the outcome.
Statistical Analysis
Data were analyzed with SPSS v22 (IBM, Armonk, NY). Descriptive statistical analysis was performed for demographic variables in both groups. An intragroup analysis was performed for T2 mapping using a 2-way variance analysis (Friedman test) for related samples. The sphericity of the variance-covariance matrix was evaluated first by the Mauchly sphericity test, taking W < 0.05 to reject the sphericity hypothesis. In the case the sphericity hypothesis was rejected, the univariate statistical analysis of Greenhouse-Geisser was used. In contrast, when W > 0.05 (spherical variance), the multivariate contrast was applied with the statistical analysis of the Hotelling trace. Each follow-up between intervention groups was also compared with the nonparametric Mann-Whitney U test. A value of P < 0.05 was taken as the level of significance for all the tests.
Results
Demographics
A total of 17 patients were included in the study; 13 men and 4 women with a mean age of 36.0 years (±7.6; range 21-47). Eleven patients (64.70%) received the polyurethane scaffold enriched with MSC (MPS), and 6 patients (35.3%) were treated with acellular polyurethane scaffolds (APS). In the APS group, all 6 patients were men (100%), while in the MPS group, 7 cases (63. 6%) were men and 4 cases (36.4%) women. Nine right knees (52.9%) and 8 left knees were treated (47.1%). Preoperatively, demographic characteristics of all patients were analyzed by gender. As noted above, the intervention groups differed significantly in the distribution of patients by gender since in the APS group 100% of patients were male compared with 63.6% in the MPS group (P = 0.04). They also differed in the distribution; in APS group 83.3% knees were right versus 36.4% in the MPS group (P = 0.05) ( Table 1 ).
Preoperative Characteristics of the Patients According to Gender.
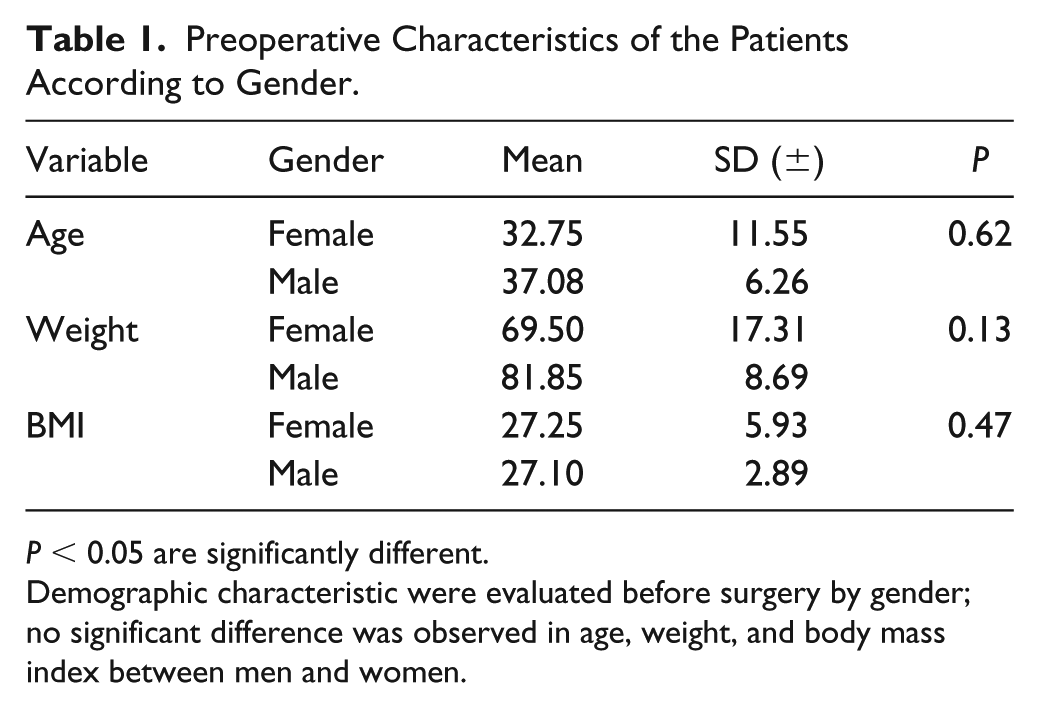
P < 0.05 are significantly different.
Demographic characteristic were evaluated before surgery by gender; no significant difference was observed in age, weight, and body mass index between men and women.
Influence of Gender on Scaffold Size and Basal T2 Mapping
The mean post-meniscectomy time in the APS group was 31.8 months compared with 14.2 months in the MPS group (P = 0.09). Analyzing further by gender, men had longer post-meniscectomy times (22.27 ± 21.7 months) compared with women (14.0 ±6.16 months) but the difference was not statistically significant (P = 0.48). The defect size and resulting scaffold size were significantly larger in men (1.74 ± 4.59, 4.91 ± 1.76) than in women (1.65 ± 4.20, 2.37 ± 0.75) (P = 0.001). The mean scaffold size in the APS group was 55 mm compared with 35 mm in MPS group (P = 0.8). Basal T2 mapping did not show a significant difference when evaluated by gender in either femoral condyle (male 47.92 ± 4.41, female 51.42 ± 8.84, P = 0.78) or tibial plateau (male 44.19 ± 4.75, female 42.09 ± 5.05, P = 0.62). However, the age of the patients correlated with the femoral T2 mapping showing a positive coefficient (0.471), meaning that the older the patient, the higher the initial femoral cartigram (P = 0.05).
Concomitant Lesions
A total of 6 patients received concomitant ACL reconstruction (35.29%)—4 patients from the APS group (66.7%) and 2 patients from the MPS group (18.2%) (P = 0.04). Three of the 6 patients with ACL reconstruction had additional cartilage lesions in the medial femoral condyle that required concomitant treatment. Two patients were treated with MSC seeded in a polyglycolic acid (PGA) mesh (Neoveil, Gunze Ltd., Tokyo, Japan) and one patient was treated with microfracture technique (MFx). The meniscal lesions in these patients were also in the medial compartment. The 2 patients with cartilage lesions treated with MSCs + PGA mesh were in the APS group, while the patient treated with MFx was in the MPS group.
The total number of patients with cartilage lesions in both groups was 10 (58.82%)—2 in the APS group (11.76%) corresponding to 33.3% of the patients in the APS group, and 8 patients (47.05%) in the MPS group corresponding to 72.72% of this group. Chondral lesions were classified according to the ICRS score system, observing that 5 lesions (29.4%) were grade 4 (concomitant repair technique was performed), 3 lesions (33.3%) were grade 3, and 1 lesion (11.1%) was grade 2. The mean size of cartilage lesions was 9.5 mm in the APS group and 10.72 mm in the MPS group.
Quality of Adjacent Cartilage Evaluated with T2 Mapping after Meniscal Substitution
The evolution of tibial cartilage means-adjusted for age, height, months of meniscectomy, and scaffold size showed no significant differences between treatment groups. It was noted that tibial T2 mapping in the MPS group increased slightly at 9 months (42.6 ± 3.1) but decreased at 12 months (43.3 ± 4.2) to the same level as at 3 months (43.3 ± 3.3). In the APS group, T2 mapping in the tibial plateau showed a significant decrease from 3 to 12 months (45.2 ± 6.1 vs. 41.4 ± 4.1). This difference tends to be significantly lower in acellular patients (41.4 ± 4.1) compared with MSCs implants (43.3 ± 3.3) at the final time point (P = 0.18) ( Fig. 4 ).
Analyzing only the male patients, in the APS group at 12 months, the mean of the T2 mapping in tibial plateau was significantly lower compared with the male MPS patients (41.4 ± 4.1 vs. 43.3 ± 4.2, P = 0.01). In femoral T2 mapping, the differences were few between treatments and evolved in a similar way over time. Nonetheless, at 12 months it increased in the MPS group compared with the APS group (47.8 ± 3.4 vs. 45.3 ± 4.9, respectively). In both groups, persistent higher values of T2 mapping in the femoral condyle compared with the tibial plateau for men and women were observed ( Table 2 ).
Quality of Adjacent Cartilage Evaluated with T2 Mapping after Meniscal Substitution.
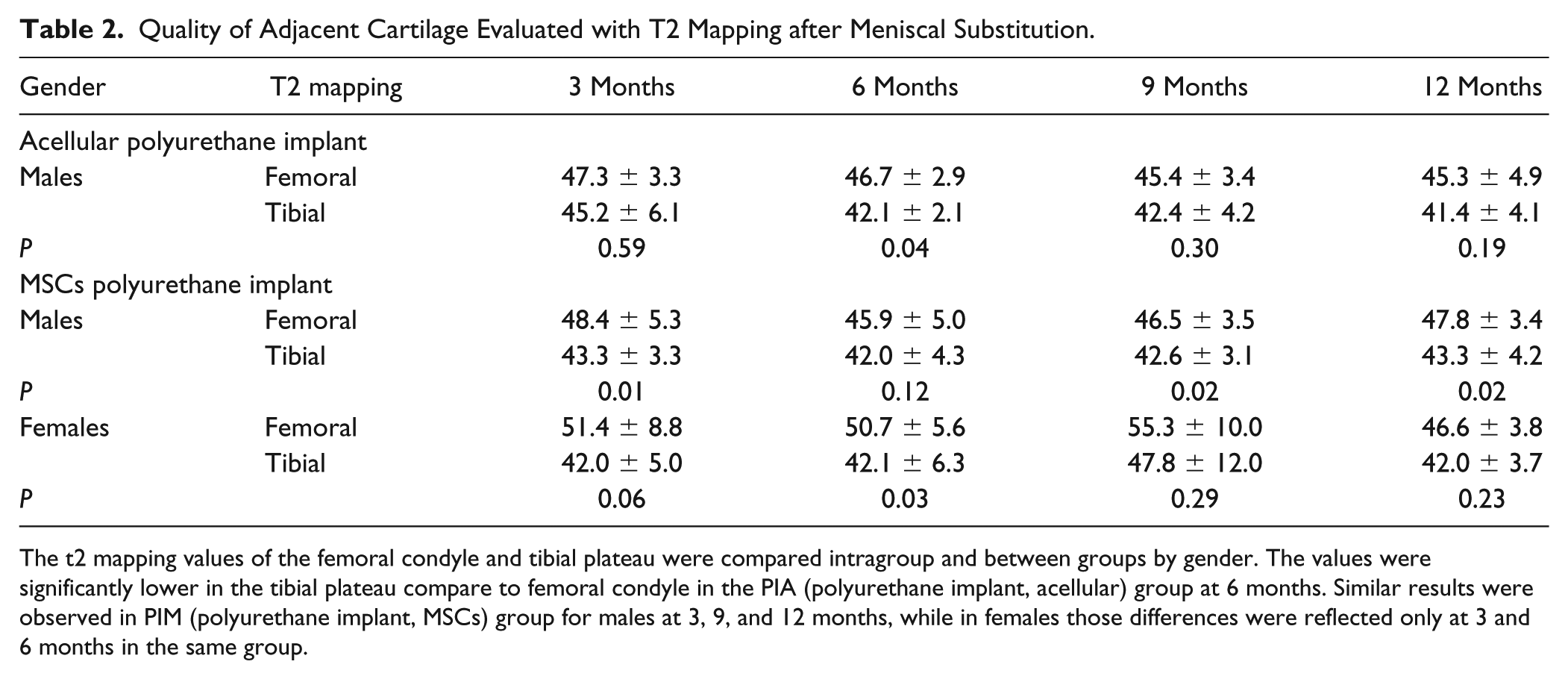
The t2 mapping values of the femoral condyle and tibial plateau were compared intragroup and between groups by gender. The values were significantly lower in the tibial plateau compare to femoral condyle in the PIA (polyurethane implant, acellular) group at 6 months. Similar results were observed in PIM (polyurethane implant, MSCs) group for males at 3, 9, and 12 months, while in females those differences were reflected only at 3 and 6 months in the same group.
Comparison of Intergroup Variables
Due to the small sample size, it was not possible to perform a multivariate analysis to control post hoc the influence of age, gender, size, knee side, presence or absence of ACL injury, months of meniscectomy, and scaffold size on the evolution of the T2 mapping at 12 months. Therefore, only a step-by-step analysis was carried out considering some of the variables as a whole ( Table 3 ).
Comparison of Variables between Groups (PIA vs. PIM).
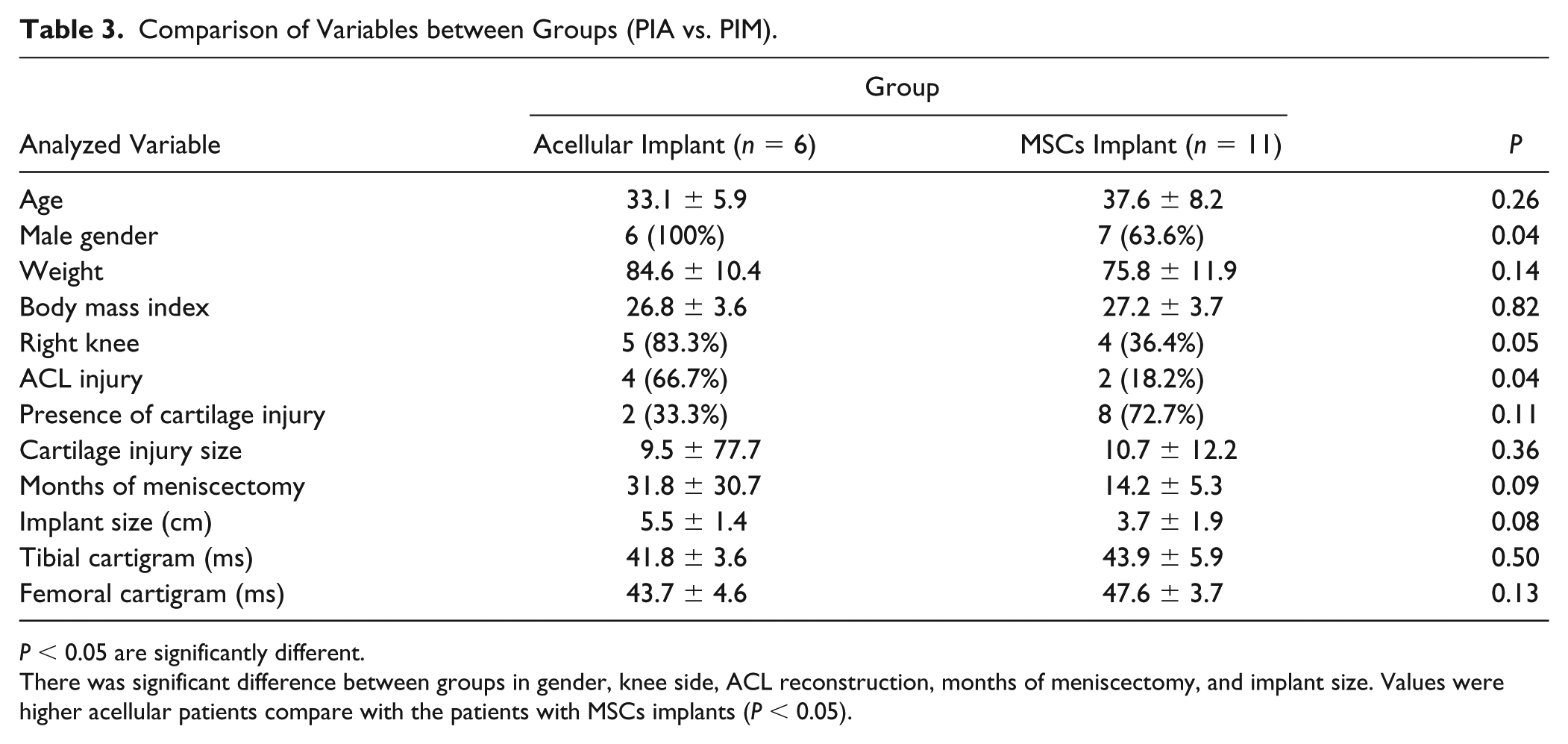
P < 0.05 are significantly different.
There was significant difference between groups in gender, knee side, ACL reconstruction, months of meniscectomy, and implant size. Values were higher acellular patients compare with the patients with MSCs implants (P < 0.05).
Patients in the MPS group were older (37.6 ± 8.2 years) than those in acellular group (33.1 ± 5.9 years), but this difference was not statistically significant (P = 0.26). There was a statistically significant gender difference between groups (P = 0.04). All patients in the APS group were male (100%), and in the MPS group, the males accounted for 63.6% of the cases and females 36.4%.
Weight and BMI were not significantly different between groups (P > 0.05). The number of operated right knees in the APS group (83.3%) was significantly higher than in the MPS group (36.4%) (P = 0.05). Meniscal substitution without any other concomitant treatment was performed only in one patient in every group. The presence of ACL reconstruction in the APS group was significantly higher (66.7%) compared with the MPS group (18.2%) (P = 0.04).
Cartilage lesions in the APS group were found in 2 patients (33.3%) compared with 8 patients (72.7%) in MPS group (P = 0.11). However, the 2 patients with cartilage lesions in APS group were scored as ICRS-4 and were repaired by MFx in one patient and MSC+PGA scaffold in the second patient. Three patients (30%) in the MPS group had also cartilage injuries ICRS-4 that were treated with MFx. Patients in the APS group had significantly more months post-meniscectomy (31.8 ± 30.7 months) than those with MSCs (14.2 ± 5.3 months) (P = 0.09). There was a correlation with the time of meniscectomy, and the required size of the implant was significantly larger in more chronic patients than in those with less months of meniscectomy (PIA 5.5 ± 1.4 months, PIM 3.7 ± 1.9 months, P = 0.08).
Lysholm Scale Analysis
In both groups, Lysholm scores increased significantly from preoperative values to 12 months. In the APS group, the increase was 26.5% (66.6 vs. 84.3, P = 0.008) while in the MPS group it was 11.2% (75.0 vs. 83.4, P = 0.01). However, there was no statistically significant difference between groups all time points ( Table 4 ). However, the initial Lysholm scores correlated very strongly with scaffold size (correlation coefficient = −0.494, P = 0.04) and with chondral lesion size (correlation coefficient = −0.595, P = 0.11). Since larger scaffold sizes correspond to males, gender may be a possible confounding variable. It should be noted that the initial Lysholm scores were significantly lower in men (66.8 ± 22.1) compared with women (89.2 ± 14. 2) (P = 0.04). Due to an analysis of variance and covariance adjusting the change in Lysholm scores from preoperative values to 12 months, comparison between groups by gender could only be carried out for males and showed that, at 12 months, Lysholm scores were lower in the PIA group (86. 9 ± 5.4) compared with the PIM group (88.5 ± 5.0), but this difference was not statistically significant (P = 0.82). No significant difference was observed between men and women in the MPS group at 12 months (P = 0.07).
Clinical Evolution Evaluated with Lysholm Comparing Acellular versus MSCs Implant.
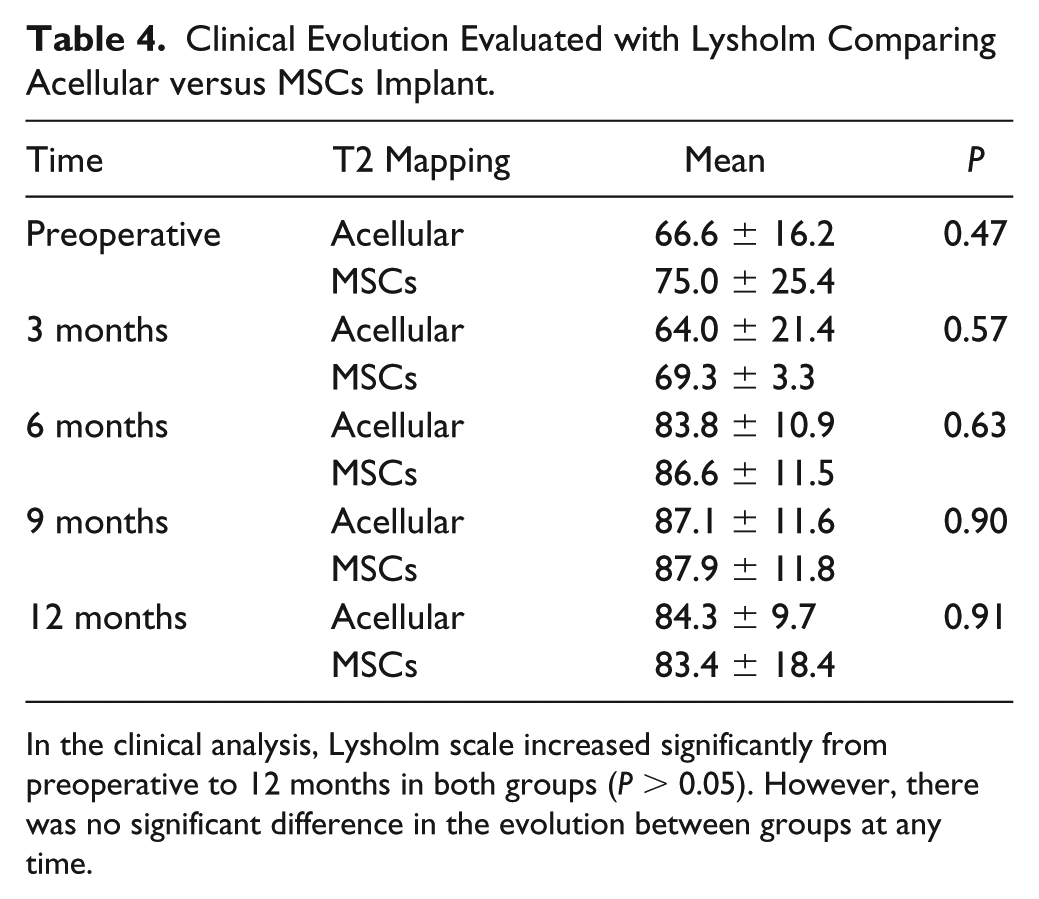
In the clinical analysis, Lysholm scale increased significantly from preoperative to 12 months in both groups (P > 0.05). However, there was no significant difference in the evolution between groups at any time.
Implant Failures
Implant failure needing reoperation and postoperative complications were evaluated in the clinical assessment. Any required reoperation or persistence of preoperative symptoms (pain and functional impairment) was likely caused by a mechanical failure of the device or nonintegration of the scaffold with the meniscal remnant. No implant failures were observed at 12 months.
Discussion
Untreated meniscal injuries progressively destabilize the knee and ultimately lead to degenerative osteoarthritic changes. Therefore, meniscus preservation and regeneration remains essential to maintain the functional integrity of the knee joint.19,20 Partial meniscal replacement appears to have a significant beneficial effect in relieving pain, enhancing knee function, and delaying articular cartilage deterioration. 21 Interest has been growing in the use of stem cells to replace meniscal tissue.22,23 These typically involve a stem cell–seeded scaffold to regenerate meniscus-like tissue. Studies have demonstrated meniscal regeneration using seeded scaffolds in rats, rabbits, and pigs.24-26 Lu et al. demonstrated a substantial improvement in meniscal healing compared with controls, which essentially exhibited no healing. 26
In this study, we investigated whether a polyurethane meniscal scaffold alone or in combination with autologous MSCs obtained from peripheral blood would enhance healing, promote meniscus-like tissue ingrowth, and preserve the articular adjacent cartilage as evaluated by T2 mapping on both articular surfaces. In the tibial plateau of the MPS group, values increased slightly at 9 months but returned to the initial values at 12 months (P > 0.05), while in the APS group, a clear decrease from 3 months to 12 months was observed (P > 0.05). This difference tended to be significantly lower in the APS group compared with the MPS group loaded at 12 months (P = 0.18). In the femur, few differences were observed between treatments. A slight increase at 12 months in the MPS group compared with the APS group was noted (47.8 ± 3.4 vs. 45.3 ± 4.9, P > 0.05). However, is important to note that initial T2 mapping in the tibial plateau was always lower than the femur in both groups. In the APS group, the tibial-femoral difference was only significant at 6 months (P = 0.04). It is important to consider the evaluation by gender, as only men were operated in the APS group, whereas in the MPS group significant tibial-femoral differences in T2 mapping were maintained from 3 to 12 months in both genders. In the case of femoral T2 mapping, the comparisons were biased because females had higher values than males.
We found that the meniscal replacement either alone or with addition of MSCs achieve significant and encouraging improvement in knee scores when compared with baseline values (P < 0.05). However, in the evaluation of clinical results at the endpoint, there was no significant difference between groups (P > 0.05). Our prospective study has an obvious source of bias in that 10 of the 17 patients underwent another procedure in addition to the scaffold implantation. Thus, although clinical benefits were recorded, we cannot determine with certainty that they were directly related to the scaffold, ligament repair, cartilage repair, or the addition of MSC to the meniscal scaffold.
The Actifit Study Group reported a multicenter case-series study of 52 patients treated with the Actifit scaffold alone. Significant improvements were recorded for pain and function (IKDC [International Knee Documentation Committee], KOOS [Knee Injury and Osteoarthritis Outcome Score], and Lysholm scores). In addition, the condition of the cartilage (ICRS grade) remained unchanged or improved in 92.5% of patients. We are currently working to record the cartilage condition by second-look arthroscopy and compare it to the condition at the time of scaffold implantation as part of a further analysis.
Our study results are expected as the polyurethane scaffold has already been shown to be beneficial in the short and medium term. 27 However, Leroy et al. 15 and Bulgheroni et al. 28 reported a decrease in scaffold dimensions over time which leads to concern about the scaffold’s capacity to cover the femoral and tibial articular surfaces. It is hypothesized that this could progressively lead to osteoarthrosis. It is important to note that the results of these studies have been inconclusive, and further long-term studies are required to assess the full clinical potential of these scaffolds.
Our results show that the addition of MSCs to the polyurethane meniscal scaffold shows no additional clinical benefit at 12 months. Nonetheless, we remain optimistic that for a specific patient population, the Actifit scaffold offers a clinical benefit. This conjecture is supported by earlier studies and our preliminary data. In addition, longer term studies are required since no complete clinical, MRI, and T2 mapping assessment were achieved within the current study period of 12 months.
Limitations
The small number of patients, lack of randomization, length of follow-up, and subjectivity of the function of MSC compared with acellular scaffolds because of bleeding during the surgery are the main limitations in this study. The lack of biopsy samples for histologic examination as an outcome measure may be considered as an additional limitation.
Conclusions
Implantation of a polyurethane meniscal scaffold maintains normal T2 mapping values in adjacent cartilage at 12 months. The addition of MSCs did not show any advantage in articular cartilage protection over acellular scaffolds. Clinical improvements are significant from preoperative values to 12 months in both groups, but there is no significant benefit in MSC-loaded scaffolds. Longer term studies are needed to assess the full benefits of MSC and meniscal scaffolds.
Footnotes
Acknowledgments and Funding
This work was supported by the Mexican National Council of Science and Technology (CONACyT, Mexico City, Mexico) Grants SALUD-2011-1-162387, PDCPN-2013-01-215138, and PDCPN-2013-01-216779. We would like to acknowledge and thank Orteq® Sports Medicine (OSM) who donated the Actifit® meniscal scaffolds used in this study. The authors express their gratitude to Ivonne Trigueros-Anaya, Gloria González-Vellano, Leticia Balderas-López, Ana María Godinez-Monroy, Catalina Cano-Alvarado, Carmen González Vellano (Arthroscopy Surgical Nursing team); Mónica Saldaña García, Cecilia Nieto-Gómez, Miguel Antonio Cervera-Bustamante (Blood Bank); and Karina Martinez Rodriguez (Biomedical Engineering) for their assistance in conducting this study.
Declaration of Conflicting Interests
The author(s) declare no conflicts of interest with respect to research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from the National Institute of Rehabilitation “Luis Guillermo Ibarra Ibarra,” Mexico City, Mexico approval number 1/11 bis.
Informed Consent
Written informed consent was obtained from all patients for this prospective, longitudinal, comparative, and analytic study.
