Abstract
Objective
Glypican-3 possesses a possible action in regulation of bone growth and development implicated in osteoarthritis (OA) pathology. Therefore, this study aimed to investigate glypican-3 in plasma and synovial fluid of knee OA patients and to determine the possible association between glypican-3 levels and radiographic severity.
Design
A total of 80 knee OA patients and 80 healthy controls were recruited. Glypican-3 levels in plasma and synovial fluid were measured using enzyme-linked immunosorbent assay. The severity of knee OA was assessed by radiographic grading according to the Kellgren-Lawrence classification. Relative mRNA expression of glypican-3 in 10 inflamed synovial tissues from OA patients and 10 noninflamed synovial controls was quantified using real-time polymerase chain reaction.
Results
Plasma glypican-3 levels were significantly lower in OA patients than in healthy controls (P = 0.03), whereas synovial fluid glypican-3 levels were remarkably greater than in paired plasma samples of OA patients (P < 0.001). Subsequent analysis demonstrated that plasma and synovial fluid glypican-3 levels were inversely associated with the radiographic severity of knee OA (r = −0.691, P < 0.001; r = −0.646, P < 0.001, respectively). Furthermore, there was a positive relationship between plasma and synovial fluid glypican-3 levels in knee OA patients (r = 0.515, P < 0.001). Additionally, overexpression of glypican-3 mRNA was observed in inflamed synovium of OA patients (P = 0.021).
Conclusions
The present study revealed that plasma and synovial glypican-3 levels were negatively associated with radiographic severity of knee OA. Glypican-3 could emerge as a potential biomarker for reflecting the severity of knee OA and might play a plausible role in the pathophysiology of degenerative joint disease.
Introduction
Osteoarthritis (OA) is the most common form of degenerative joint disease that affects elder people worldwide. The disease is characterized by destruction of articular cartilage with joint space narrowing, osteophyte formation, subchondral bone sclerosis, and synovial inflammation, leading to severe pain, joint swelling, increased physical disability, and eventually to joint replacement surgery. 1 Currently, the diagnosis of the affected joint is based on clinical and radiographic criteria; however, radiographic assessment does not always relate to patients’ perception of symptoms.2,3 Moreover, the current limited options available for treatment are pharmacological methods, in which they are mainly used for pain relief and cannot help reverse any of the damage within a patient’s joints. 4 Indeed, it has been suggested that the pharmacological treatments are not very effective for long-term management of knee OA. 5 These facts highlight the importance of not only developing more effective disease-modifying treatments but also identifying potential biomarkers to aid with earlier diagnosis to prolong the period before patients experience significant disability. 6 In this context, a better understanding of what causes OA development could lead to identifying potential biomarkers and new therapeutic targets. Although the precise etiology and pathogenesis of OA are still not completely understood, it is generally accepted that bone remodeling is one of the most pathogenic events driving the progression and development of knee OA. 7 Consequently, the possible role of molecule-mediated bone remodeling is of great interest to many researchers for development of biochemical markers indicating the onset of this biological process.
Glypican-3, one of 6 different proteoglycans within the glypican-related integral membrane proteoglycan family, functions as a co-receptor to bind the external surface of the plasma membrane by a glycosyl-phosphatidylinositol bond (GPI). 8 Glypican-3 is known to play important roles in regulating a range of biological processes, including cell growth, differentiation, and survival during embryonic development via its interaction with various growth factors such as Hedgehogs, fibroblast growth factors, Wnts, and bone morphogenetic proteins (BMPs).9,10 With such its primary role in mammalian development, glypican-3 was found to be expressed in embryonic tissues—predominantly fetal articular cartilage, 11 but its expression is limited in adult tissues. 12 In addition to its effect on embryonic morphogenesis, glypican-3 has been previously implicated in the process of bone remodeling via the activity of BMP-2. 13 These previous findings led us to speculate that glypican-3 may have an immense potential to be a molecular predictor for dysregulated development of bone, which may be involved in the onset of OA. Although the pathogenic role of glypican-3 in knee OA is yet to be elucidated, growing evidence indicates that glypican-3 could be used as a sensitive and specific marker for early diagnosis of various multifactorial diseases—especially hepatocellular carcinoma.14,15 It therefore seems plausible that glypican-3 may serve as a potential biomarker for knee OA being a chronic multifactorial disease, and it could be detected in the circulation, which can be collected relatively easily and noninvasively from patients.
As far as we know, no previous study has investigated glypican-3 with regard to its relationship with radiographic severity in primary knee OA. Hence, the current study aimed to investigate plasma and joint fluid glypican-3 in knee OA subjects compared with plasma glypican-3 in healthy controls and envisaged the association of plasma and synovial fluid glypican-3 and the severity of knee OA.
Materials and Methods
Study Population
The experimental protocols were approved by the Ethical Committee on Human Research (IRB No. 533/54) of our institution. Written inform consent was provided by all participants in the present study. Participants with overarching disorders, including diabetes mellitus, reports of long-term steroid treatment, any conditions of arthritis, previous knee injury and/or infection, malignancy, or other chronic inflammatory disorders were eliminated from this investigation.
Standard weightbearing anteroposterior radiography of the affected knees was performed when each subject maintained full knee extension. Evaluation of radiologic severity was carried out by 2 examiners who were blinded to the findings with regard to the Kellgren and Lawrence (KL) classification. 16 The radiographic change of knee OA was categorized into 5 grades (0 to IV): grade 0 (normal findings with no X-ray changes), grade I (possibility of osteophytic lipping), grade II (clear osteophytes and plausible narrowing of joint space), grade III (medium multiple osteophytes, clear narrowing of joints space and some sclerosis), grade IV (large osteophytes, severe narrowing of joint space, bone sclerosis and clear deformity of bone contour). The presence of radiographic knee OA was delineated as KL grade ≥II. Control group was delineated as acquiring none of radiographic hip and knee OA.
Laboratory Methods
Following a 12-hour overnight fast, peripheral venous blood specimens were obtained from every subject 1 day before surgery, centrifuged to remove cells and debris, and kept instantly at −20°C for subsequent measurement. Synovial fluid samples were collected from the most severe knee during diagnostic or therapeutic arthroscopy or total knee replacement. The specimen was then centrifuged to exclude cells and joint debris and then kept at −20°C for further analysis. Double-blinded quantitative measurement of circulating and synovial fluid glypican-3 levels were assessed using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, USA). According to the manufacturer’s instruction, 100 µL of recombinant human glypican-3 standards, plasma and synovial fluid samples were pipetted into every well of a microplate, which was precoated with specific antibody to glypican-3. After incubating for 2 hours at room temperature, all wells were washed completely 3 times with washing reagent. Subsequently, glypican-3 conjugate was pipetted into every well and incubated for 2 hours at room temperature. After 3 washes, substrate solution was added into each well and then the microplate was incubated for 20 minutes at room temperature without light. Ultimately, all reactions were terminated by the stop reagent and the optical density was evaluated with automated microplate reader at 450 nm. The intensity of color derived is clearly proportionate to the quantity of glypican-3 in the samples. A standard optical density–concentration curve was depicted for the determination of glypican-3 value (range 0.313-10 ng/mL).
Quantitative Real-Time Polymerase Chain Reaction for mRNA Expression of Glypican-3
To determine mRNA expression of glypican-3 in inflamed and noninflamed synovial tissues obtained from knee OA patients using real-time polymerase chain reaction (PCR), total RNA was isolated from synovium biopsies using RNeasy Mini Kit (Qiagen, Hilden, Germany), with cDNA that was reverse transcribed by using TaqMan Reverse Transcription Reagents (Applied Biosystems, Inc., Foster City, CA, USA). Real-time PCR was performed using QPCR Green Master Mix HRox (biotechrabbit GmbH, Hennigsdorf, Germany) on StepOnePlus Real-Time PCR System (Applied Biosystems, Inc., Foster City, CA, USA). Primers used for glypican-3 and glyceraldehyde 3-phosphate dehydrogenase (GADPH) amplification were as follows: glypican-3 forward primer 5′-GATACAGCCAAAAGGCAG-3′, glypican-3 reverse primer 5′-ATCATTCCATCACCAGAG-3′; GADPH forward primer 5′-GTGAAGGTCGGAGTCAACGG-3′, and GADPH reverse primer 5′-TCAATGAAGGGGTCATTGATGG-3′. Real-time PCR conditions were performed as follows: (initial step) 95°C for 10 minutes, followed by 40 cycles of 95°C for 15 seconds, and then 56°C for 1 minute. Relative mRNA expression of glypican-3 was normalized to GADPH as an internal control and was determined using 2−∆∆Ct method.
Statistical Analysis
Statistical analyses were accomplished with the statistical package for social sciences (v. 22.0, IBM Corp., Armonk, NY). Student unpaired t test was used to compare the means of 2 independent groups, and 1-way analysis of variance (ANOVA) was employed to compare the means of more than two independent groups. Pearson’s correlation coefficient was used to examine the relationship between circulating and synovial fluid glypican-3 levels and the radiographic severity. All data were shown as a mean ± standard error of the mean (SEM). Statistical significance for differences and correlations was set at P < 0.05.
Results
Baseline Clinical Characteristics
A total of 160 participants were enrolled in this study. Eighty knee OA subjects were recruited in accordance with the criteria of the American College of Rheumatology (ACR). Eighty gender- and age-matched control volunteers showed no symptoms or signs of OA in all hip and knee joints. This case-control study consists of 80 knee OA subjects (66 women and 14 men) and 80 gender- and age-matched healthy controls (58 women and 22 men). The OA group had an average age 71.1 ± 0.8 years (range 51-84 years). The control group had an average age of 69.8 ± 1.2 years (range 50-80 years). The detailed demographic features of the participants are shown in Table 1 . There was no statistically significant difference in age and gender between knee OA subjects and control participants. Mean plasma glypican-3 value was significantly lower in OA subjects than in plasma controls (65.5 ± 7.3 vs. 97.9 ± 10.5 pg/mL, P = 0.03). As illustrated in Figure 1 , mean glypican-3 value in synovial fluid of knee OA patients was significantly 2-fold higher than that in corresponding blood samples (136.8 ± 10.5 vs. 65.5 ± 7.3 pg/mL, P < 0.001).
Baseline Clinical Characteristics of Patients with Knee Osteoarthritis (OA) Patients and Controls.
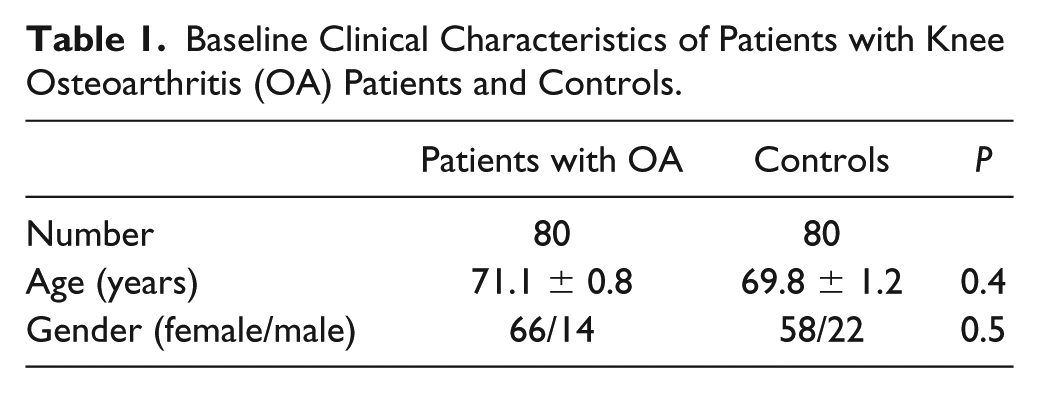
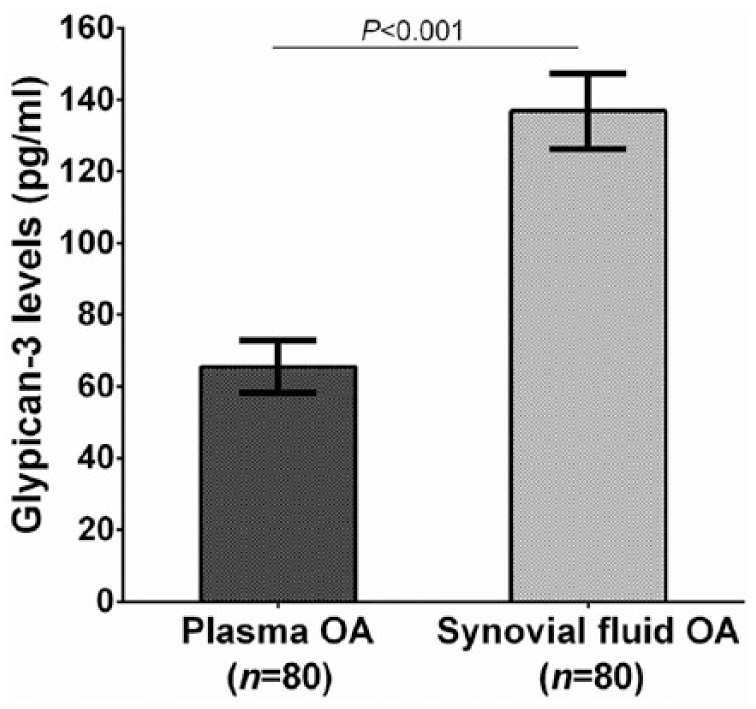
Plasma and synovial fluid glypican-3 levels of patients with knee osteoarthritis (OA).
Correlation Between Circulating and Synovial Fluid Glypican-3 and OA Severity
To explore the association between circulating and synovial fluid glypican-3 concentrations and OA severity, the cohort was categorized into 3 groups corresponding to KL grading ( Table 2 ). Of the 80 knee OA subjects recruited in the study, 13 exhibited a KL score of 2, 32 had a KL score of 3 and 35 had a KL score of 4. The groups were analyzed with respect to both the plasma OA and joint fluid OA glypican-3 levels ( Fig. 2 ). Both the circulating and synovial fluid glypican-3 concentrations presented significant decreases with the KL grading of the OA patient groups (r = −0.691, P < 0.001 and r = −0.646, P < 0.001, respectively; Figs. 2 and 3 ). Further analysis revealed a positive relationship between plasma glypican-3 levels and synovial fluid glypican-3 levels in patients with knee OA (r = 0.515, P < 0.001) ( Fig. 4 ).
Plasma and Synovial Fluid Glypican-3 in Patients with OA and P Values for Differences Among Kellgren and Lawrence Subgroups.
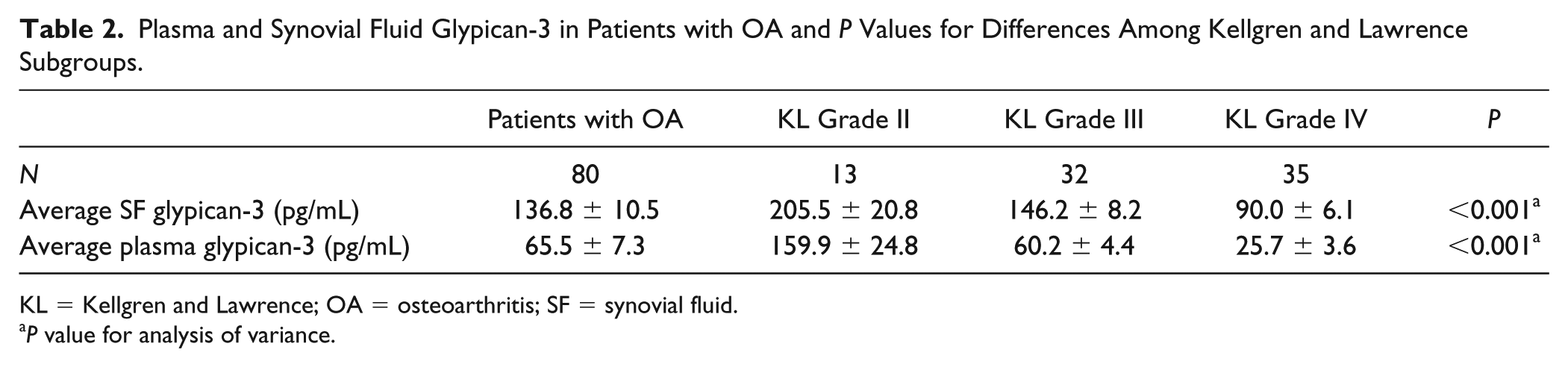
KL = Kellgren and Lawrence; OA = osteoarthritis; SF = synovial fluid.
P value for analysis of variance.
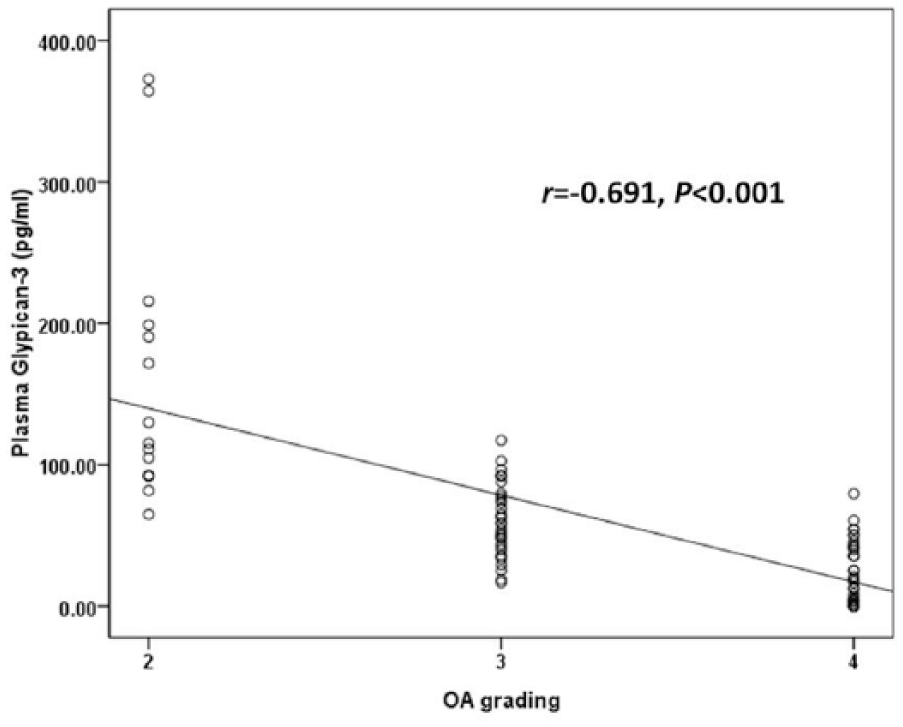
Negative correlation between plasma glypican-3 levels in patients with osteoarthritis (OA) and disease severity classified according to Kellgren and Lawrence grading scale (r = −0.691, P < 0.001).
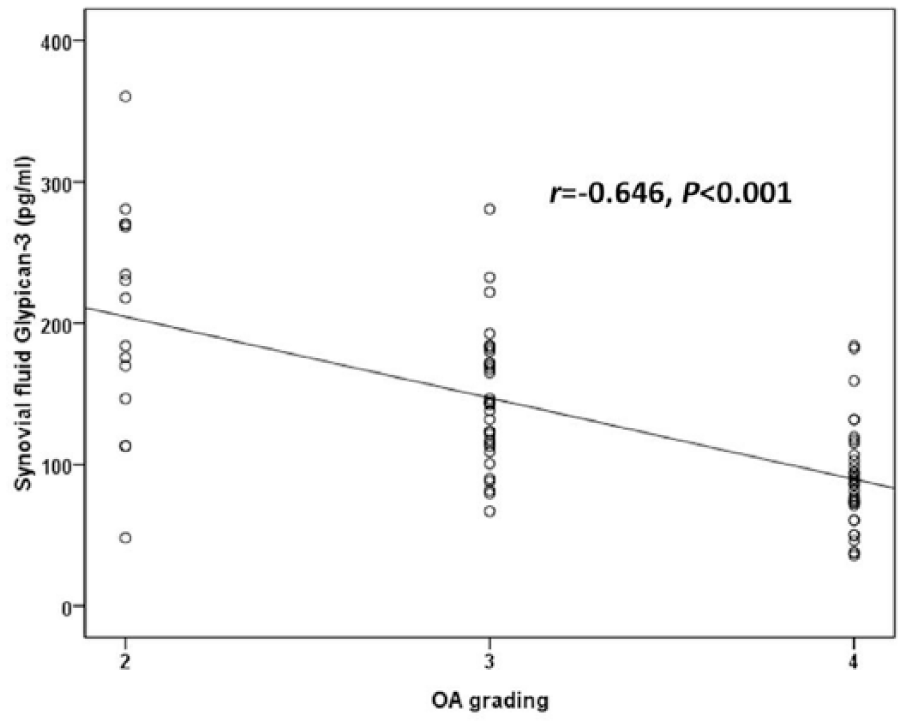
Negative correlation between synovial fluid glypican-3 levels in patients with osteoarthritis (OA) and disease severity classified according to Kellgren and Lawrence grading scale (r = −0.646, P < 0.001).
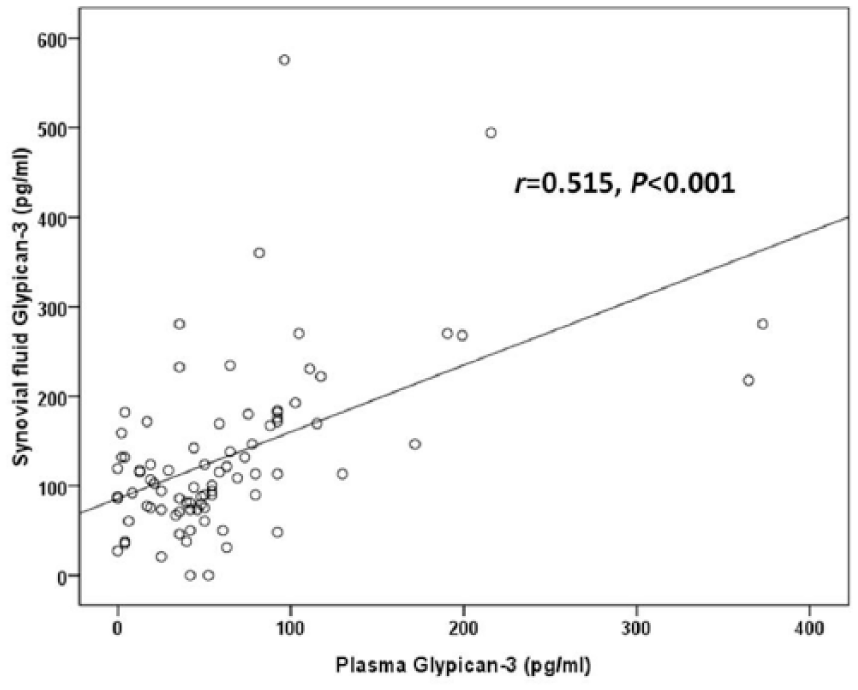
Positive correlation between plasma and synovial fluid glypican-3 levels in patients with osteoarthritis (OA) (r = 0.515, P < 0.001).
Relative mRNA Expression of Glypican-3
To examine transcriptional expression of glypican-3 in synovial tissues of patients with knee OA, relative glypican-3 mRNA expression was quantified by real-time PCR in inflamed synovial tissues from patients with knee OA (n = 10) and noninflamed synovial controls (n = 10). Quantitative real-time PCR showed that relative glypican-3 mRNA expression was significantly higher in inflamed synovium from patients with OA than in noninflamed synovial controls (P = 0.021) ( Fig. 5 ).
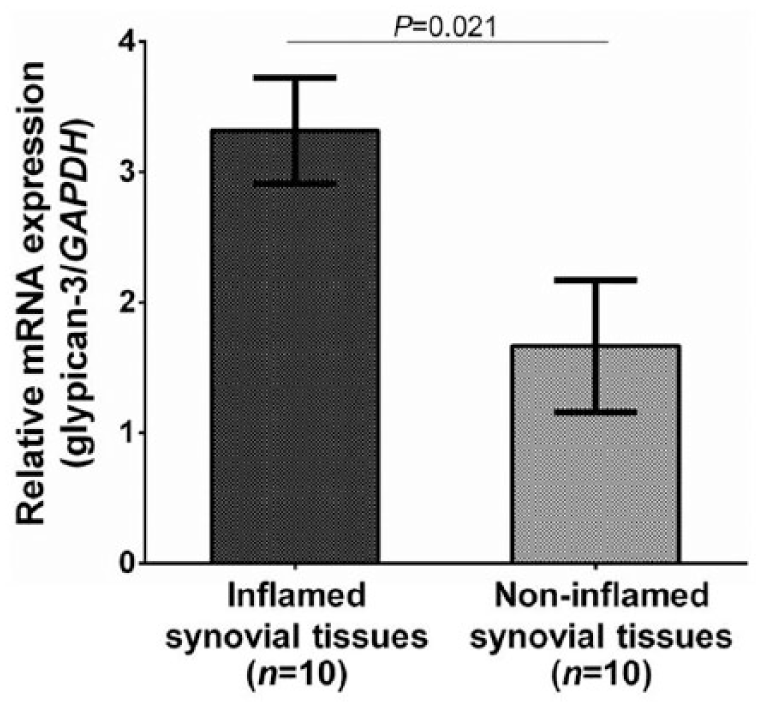
Upregulated mRNA expression of glypican-3 normalized by GAPDH in inflamed synovial tissues from patients with knee OA compared with that in noninflamed synovial tissues (P = 0.021).
Discussion
Given a leading cause of chronic pain toward disability, knee OA, a debilitating degenerative joint disease, is becoming a major public health leading to economy loss. 17 Since currently reliable treatments could not stop the onset of OA and are mainly focused on pain relief and slowing down the disease progression, patients with advanced OA often undergo total joint arthroplasty. In this regard, early diagnosis is actually of paramount important to improving the clinical outcome of patients and reducing the need for joint replacement surgery. Nevertheless, until now, there is no any effective and useful biomarker for detecting OA at the early stage. It is therefore imperative to identify and develop biochemical markers for early diagnosis of OA. In order to achieve early diagnosis and prognosis in knee OA, the present study investigated glypican-3 levels in both plasma and synovial fluid obtained from patients with knee OA, in whom glypican-3 has been reported to regulate bone formation that may involve the development of knee OA.18,19 Herein, we found a reduction of plasma glypican-3 levels in patients with OA. Furthermore, plasma glypican-3 levels were positively associated with synovial fluid glypican-3 levels; however, synovial fluid glypican-3 levels were remarkably increased in patients with OA. Notably, plasma and synovial fluid glypican-3 levels were inversely correlated with the radiographic severity of knee OA. We subsequently performed real-time PCR to determine mRNA expression of glypican-3 in synovium and observed that upregulation of glypican-3 mRNA in inflamed synovium of knee OA. All aforementioned findings support the notion that glypican-3 might be used as a biochemical marker for predicting the progression of knee OA.
It has been recognized that glypican-3 exerts its potential roles in regulation of a number of physiological processes, including cellular growth, differentiation, and apoptosis through a variety of signaling pathways, including fibroblast growth factors, Hedgehogs, Wnts, and BMPs. 8 Given that these 4 signaling cascades have been discovered to be the major mechanisms controlling bone growth and development,19-22 it is not surprising that glypican-3 may take a fundamental part in the pathogenic mechanism of knee OA. With regard to the effect of glypican-3 on bone biology, it has been shown that glypican-3 inhibits the activity of BMP-2-induced osteogenesis. 13 Furthermore, an experimental study by Viviano et al. 24 highlighted the role of glypican-3 in regulation of osteoclast differentiation, through which glypican-3 knockout mice can cause a reduction in osteoclast differentiation and an abnormal persistence of hypertrophic chondrocytes at embryonic stage. Based on this evidence, it is apparent that glypican-3 may have a possible effect on bone remodeling. Although the association of glypican-3 in both the circulation and synovial fluid with the severity of knee OA has as yet not been investigated, there is considerable published data measuring glypican-3 levels in the circulation of chronic multifactorial diseases—particularly cancerous patients. For example, Wang et al. 25 measured circulating glypican-3 levels in patients with primary hepatocellular carcinoma (HCC). They found that circulating glypican-3 levels were remarkably higher in patients with HCC than in patients with other liver diseases and in control subjects. Apart from its utility as a disease-specific biomarker for HCC, circulating glypican-3 levels have been reported to be increased in acute respiratory distress syndrome patients with severe pneumonia when compared with healthy controls. 9 The previous findings contrast our main results observed in this study, in which the conflicting results may be ascribed to differences in disease pathogenesis. In the present study, it appeared that plasma glypican-3 levels in patients with knee OA were significantly decreased when compared with healthy controls and were positively correlated with its levels in synovial fluid. Instead, synovial fluid glypican-3 levels were considerably greater than in paired plasma samples. Our aforementioned findings indicate that there is a significant reduction in systemic production of glypican-3, while its local production was increased in patients with knee OA. Even though the precise mechanisms regulating the systemic and local production of glypican-3 remain poorly understood, the possible reason for reduced glypican-3 levels in plasma may be attributed to the limited release of glypican-3 from the local tissues such as inflamed synovium, cartilage, and subchondral bone into the circulation. This assumption has been supported by our results revealing overexpression of glypican-3 mRNA in inflamed synovium from OA patients as compared with noninflamed synovial controls. In addition, as to its primary role in mammalian development, 26 age appears to be one of the most important factors for regulating glypican-3 production. It has been documented that glypican-3 mRNA expression was reduced in the adult. 12 Another possible explanation for an increase in synovial fluid glypican-3 levels may be presumably attributable to either enhanced secretion of glypican-3 residing in extracellular matrix responsible for injured arthritic tissues, increased glypican-3 clearance, which in turn exceeded its synthesis, or both. To support these hypotheses, previous study demonstrated the presence of glypican-3 overexpression in fetal cartilage, 27 thereby establishing a possible action of glypican-3 in cartilage development. It should be noted, however, that the precise mechanisms regulating glypican-3 production not only glypican-3 reduction in the circulation but also its enhancement in synovial fluid of patients with knee OA remain to be elucidated. Interestingly, we demonstrated that glypican-3 is expressed at higher level in inflamed synovium from patients with knee OA than noninflamed synovial controls. This finding suggests that glypican-3 may be a crucial mediator of the processes implicated in OA pathophysiology.
In addition to altered systemic and local production of glypican-3 in patients with knee OA, the possible association of plasma and synovial fluid glypican-3 levels with the severity of knee OA has been explored in this research. To our knowledge, this is the first report to describe that both plasma and synovial fluid glypican-3 levels were negatively correlated with the radiographic severity of patients with knee OA. This finding was in accord with our previous investigation, which illustrated the inverse relationships between both plasma and synovial fluid sclerostin levels and the radiographic severity of knee OA. 28 Likewise, Orita et al. 29 highlighted a negative correlation between interleukin-6 levels in synovial fluid of patients with knee OA and their radiographic grades. Despite lack of previous data supporting our finding regarding the association of glypican-3 with severity of knee OA, through its binding with a wide spectrum of morphogens and growth factors, especially BMP, glypican-3 has been reportedly implicated in regulation of bone remodeling process,13,17,18,23 which is an important mechanism involved in knee OA pathology. Considering an inverse link between the systemic and local levels of glypican-3 and knee OA severity, it is tempting to speculate that reduced production of glypican-3 might reflect the susceptibility of an imbalance between bone resorption and bone formation contributing to the pathogenesis of OA. The real-time PCR study showed overexpression in inflamed synovium from patients with OA. Synovitis is likely to be facilitating factor in the release of glypican-3 into the joint fluid.
This study inevitably had some inherent caveats, which should be considered when evaluating the findings presented herein. First, the study is a cross-sectional in design with a relatively small number of participants; such a study cannot establish definite cause-and-effect relationships. It is recommended that multicenter prospective longitudinal investigations with larger sample sizes will help to elucidate any relationships. Second, due to ethical reasons, synovial fluid samples were not collected from healthy controls, leading to no direct comparison between synovial fluid glypican-3 of controls and that of patients with OA. Last, we did not examine functional impairment, pain, and clinical symptoms in these patients. Additional studies are warranted to determine whether glypican-3 associates with functional impairment (Western Ontario and McMaster Universities Osteoarthritis Index [WOMAC] score) and pain (visual analogue scale). Besides, insufficient assessment of potential confounders, for example, age, gender, and medical comorbidities need to be taken into account.
In summary, this study is the first to provide evidence showing that synovial fluid glypican-3 was remarkably higher with respect to paired plasma in patients with knee OA. Furthermore, plasma and synovial fluid glypican-3 levels were inversely associated with the radiographic severity in knee OA. These findings supported the hypothesis that glypican-3 could be used as a possible biochemical marker reflecting the degenerative process of primary knee OA. However, further investigations that elucidate the molecular mechanism regulating the role of glypican-3 in the development and severity of knee OA are necessary to gain insight into the discovery of novel therapeutic agents against OA.
Footnotes
Acknowledgments and Funding
The authors thank the Ratchadapisek Somphot Endowment Fund (Chulalongkorn University) and the Research Chair from National Science and Technology Development Agency (NSTDA). The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Ratchadapiseksompotch Fund, Faculty of Medicine, Chulalongkorn University, grant number RA61/104
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocols were approved by the Ethical Committee on Human Research (IRB No. 533/54) of our institution.
Informed Consent
Written inform consent was provided by all participants in the present study.
Trial Registration
Not applicable.
