Abstract
Objective
The aim of this study was to evaluate the effects of a lyophilized inactivated culture (LIC) from Bifidobacterium longum CBi0703 in a spontaneous model of osteoarthritis (OA) in Dunkin Hartley guinea pigs. Histology of cartilage and synovial membrane was the primary outcome. Biomarkers were also considered to evaluate the treatment efficacy.
Design
LIC (1 µg/kg) with or without vitamin C (1 mg/kg) were tested in Dunkin Hartley guinea pigs spontaneously developing OA and compared with control (sterile water; CTL). Treatment was initiated orally in 16-week-old animals over a period of 12 weeks. Histological lesions of articular cartilage and synovial membrane were scored according to the OARSI (Osteoarthritis Research Society International) recommendations. Four biomarkers (Coll2-1, PIINAP, Fib3-2, and osteocalcin) were measured in animal sera.
Results
The global OARSI score increased with time in all group but no significant difference between groups was observed. When score items were analyzed individually, a significant lower score of cartilage structure was observed in the LIC + vitamin C group compared with CTL (P < 0.0001). Synovial membrane showed a mild inflammatory reaction that was not affected by the treatment. LIC significantly decreased serum levels of Coll2-1 (P = 0.0004 vs. CTL), a marker of type II collagen degradation and LIC + vitamin C significantly increased PIINAP (P = 0.0003), a marker of type II collagen synthesis. The ratio Coll2-1/PIINAP was significantly decreased in both LIC groups (P < 0.001).
Conclusion
Lyophilized inactivated culture of B. longum CBi0703 administrated orally over a period of 12 weeks decreased cartilage structure lesions and decreased type II collagen degradation suggesting a potential prophylactic effect on OA development.
Introduction
Osteoarthritis (OA) is definitely a major issue in public health due to the wide proportion of the affected population, the duration of evolution, and the lack of treatment. OA is a disease of the entire joint affecting both articular and peri-articular tissues, 1 but OA may be of different clinical phenotypes. The main phenotypes described in the literature are traumatic inflammatory, aging, metabolic syndrome, and biomechanical malalignment. Each of these could require a specific intervention and/or treatment. 2 The currently available interventions aim at reducing the symptoms (i.e., pain and function) but none of them are able to maintain or restore the articular structures. This far, all the recommendations refer to the control of symptoms.3-6 There is a need for new treatment targeting structural changes associated with OA.
Recently, a close connection between intestinal inflammation and chronic inflammation of the joints was demonstrated giving new therapeutic perspectives. 7 This relationship is now documented for spondyloarthritis,8,9 rheumatoid arthritis (RA),10,11 and OA. 12 Two major hypotheses have been advanced to explain the relationship between inflammation and mucosal immune system. The first refers to the bacterial translocation, 13 which corresponds to an increased permeability of the gut wall lumen and to the exposure of immune system to commensal microbiota that are not physiologically linked to a disease. The second suggests that chronic inflammatory arthritis could be associated with microbiome dysbiosis, which corresponds to an imbalance between pathogen and commensal bacteria resulting from stress, dietary components, drugs like antibiotics or nonsteroidal anti-inflammatory drugs or inflammatory bowel diseases.14-16 This second mechanism involves the recruitment of lymphocytes from the gut or activated macrophages to the joints 17 and lipopolysaccharides (LPS). LPS content depends on the nature of the microbiome and its biological activity requires the toll-like receptor (TLR) pathway.18,19 Finally, and interestingly, it was suggested that the administration of probiotics could not only act to reduce inflammation and pain 20 but also modulate the efficacy of a treatment against OA. 21
Altogether these findings suggest that measures that could correct intestinal permeability and/or joints microinflammation could be effective modalities to treat or prevent chronic arthritis like OA. One approach is the administration of a cocktail of metabolites derived from probiotics. 22 .
Bifidobacterium longum CBi0703 has recently been characterized 23 and proven to promote bifidobacterial growth. This specific strain has been selected for its action on immune response and possible applications in inflammatory disorders. This has been extensively detailed by Scuotto et al. 23 This strain was shown to colonize the lowest parts of intestine where they reduced the bacterial translocation. 24 In addition, it was shown that the tested product, when administered orally, reduced the translocation as well as the spread of bacteria (WO 2004/093898 and WO 2006/040485). The present study relied on the hypothesis that OA pathogenesis, due to its chronic inflammatory status, 25 could also be associated to dysbiosis and that treatment of OA patients with specific probiotics could lead to patient improvement. 24
The lyophilized inactivated culture (LIC) from B. longum CBi0703 23 was tested in Dunkin Hartley guinea pigs, a strain spontaneously developing OA.26-29
The evaluation of articular structure according to OARSI recommendations in the guinea pig spontaneous model 28 and the measurement of the serum levels of several pertinent biomarkers in guinea pigs were considered to evaluate the efficacy of this new intervention.
The aim of the present study was to evaluate the potential protective effect of LIC administered at low dose during OA development in Dunkin Hartley guinea pigs.
Methods
Animal Models
Forty-one male Dunkin Hartley guinea pigs (16 weeks old at the time of treatment initiation, T0) (Harlan, the Netherlands) were involved in this study (see Fig. 1 for details regarding the study design). Animals were randomly divided into 4 groups (n = 5 to 12 per group; n = 5 for CTL [control] T0 reference group, n = 12 for placebo and test groups).
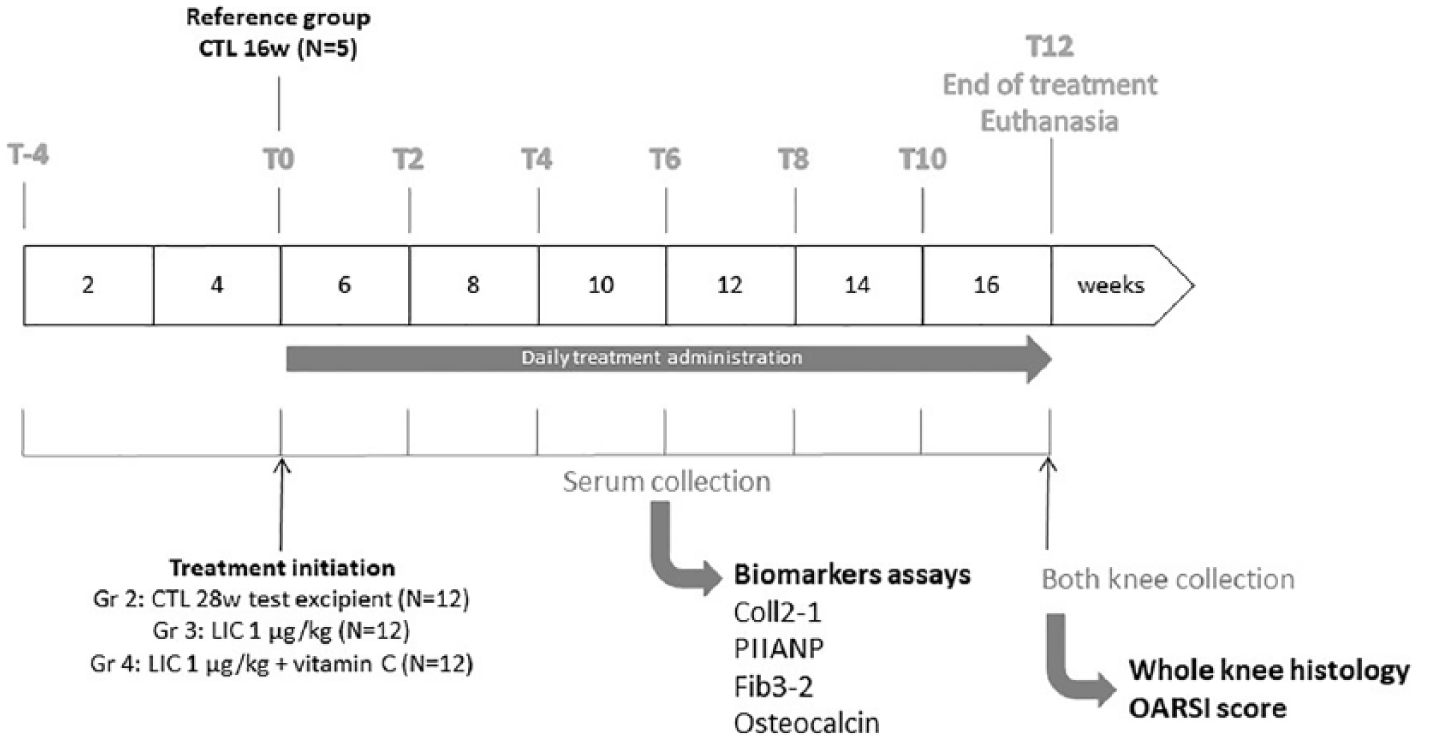
Study design.
The study was submitted to and approved by the Vet-Agro Sup (Ecole Nationale Vétérinaire de Lyon) Ethical Committee. An application for the project authorization was submitted to the MESR (Ministère de l’Enseignement Supérieur et de la Recherche).
Animals were housed at Biovivo Veterinary School (Marcy L’Etoile, France)–accredited animal facilities. Animals were housed collectively in pen (1 pen/group) in a climate- (temperature 21°C±2°C, humidity ≥35%) and light- (12-/12-hour light/dark cycle) controlled room with free access to food and drinking water. Animals were fed a standard diet (Safe 114, #U8540G10R, Safe, France).
All procedures used for these studies conformed to the national ethic chart for animal experimentation.
Treatment Administration
Treatment was initiated in guinea pigs on T0 after an acclimatization period of 4 weeks and for 12 weeks ( Fig. 1 ). Animals were 16 weeks old at the time of treatment initiation. Treatment (1 mL/kg) was administered orally, directly at the back of the mouth, every day for 12 weeks (T12). LIC was administered at the dose of 1 µg/kg/d with or without vitamin C (1 mg/kg/d). Vitamin C has been added by the manufacturer to obtain a joint health claim. The treatment groups were compared with control group (sterile water, 1 mL/kg/d).
Histological Evaluation of Articular Cartilage and Synovial Membrane
Whole knee joints were fixed in 4% paraformaldehyde (PFA) right after dissection and were processed as recommended by Kraus et al. 28 Frontal histological sections (5 µm) were performed with a standard microtome on paraffin-embedded samples after appropriate decalcification (DC2 decalcifier, QPath).
Three sections 200 µm apart were performed in weightbearing area and stained with Safranin-O and Fast green. An additional slide was stained with toluidine blue.
Each slide was scored according to the OARSI (Osteoarthritis Research Society International) score for cartilage and synovial membrane modification. 28
Serum Samples Collection and Biomarkers Assays
Serum was prepared from T-4 (when guinea pigs were 12 weeks old, before treatment initiation) and then every 2 weeks during the treatment administration in Dunkin Hartley guinea pigs from T0 to T12 ( Fig. 1 ). Blood puncture was performed under volatile anesthesia (isoflurane) in the superior vena cava with a 23G hypodermic needle. Blood was let for clotting at least 1 hour at room temperature after puncture. The clot was removed before centrifugation in order to avoid hemolysis. Samples were then centrifugated at 1500 × g for 10 minutes in a refrigerated centrifuge (+4°C) and kept frozen (−20°C) until assay.
Frozen sera were shipped to the centralized biomarker platform (Artialis S.A., Liège, Belgium) for biomarker testing at the end of the study. Samples have been assayed in pooled test series using validated immunoassay assays and according to written procedures provided by the manufacturer.
Coll2-1 (Artialis S.A., Liège, Belgium), Fib3-2 (Artialis S.A., Liège, Belgium), PIIANP (Merck Millipore, Overijse, Belgium), and osteocalcin (Quidel, San Diego, CA, USA) were measured in the sera of all guinea pigs.
Statistical Analysis
All collected samples were analyzed. Except otherwise specified, data from histological scores were analyzed using Kruskal-Wallis test for nonparametric values followed if positive by Dunn’s multiple comparison test.
Histological score for cartilage is presented either for the whole joint (sum of all compartments, mean of the 3 evaluated slides) or for a specific compartment (mean of the 3 evaluated slides). The evaluated criteria are also analyzed separately. Both knees were considered independently (n = 10-24 depending on the study group).
Histological score of synovial membrane is presented for the whole joint (mean of the 3 evaluated slides) as the global score or each evaluated criterion separately. Data given in the text are mean ± standard deviation (mean ± SD).
A mixed model will be applied to the data from biomarkers assays to test for differences in time evolution between the 3 study groups (CTL, LIC, and LIC + vitamin C). The covariates included in this model were the time and the interaction with the treatment indicator. This statistical approach allows the comparison of response curves between treatments while accounting for repeated data within each guinea pig. This model considered the analysis of longitudinal evolution of Coll2-1, PIINAP, Coll2-1/PIIANP, Fib3-2, and osteocalcin. Calculations were carried out on the maximum number of data available. Results were considered to be significant at the 5% critical level (P < 0.05). Data analysis was carried out using SAS (version 9.3 for Windows) statistical package. Data are mean ± SE (mean ± standard error). Results from PIIANP and Fib3-2 assays were log transformed.
A Spearman correlation for nonparametric values was performed between the variation of biomarkers level (T12-T0/T0) and the OARSI global score.
Results
Effects on Cartilage Degradation in Dunkin Hartley Guinea Pigs during the Spontaneous Development of OA
OARSI global score significantly increased between T0 (16-week-old animals; CTL T0) and T12 (28-week-old animals; CTL T12) ( Fig. 2 ). The development of moderate OA lesions was symmetrical in right (36.31 ± 6.04) and left knees (35.93 ± 4.25) with the medial compartment (19.05 ± 3.37) slightly more affected than the lateral compartment (17.07 ± 2.31).
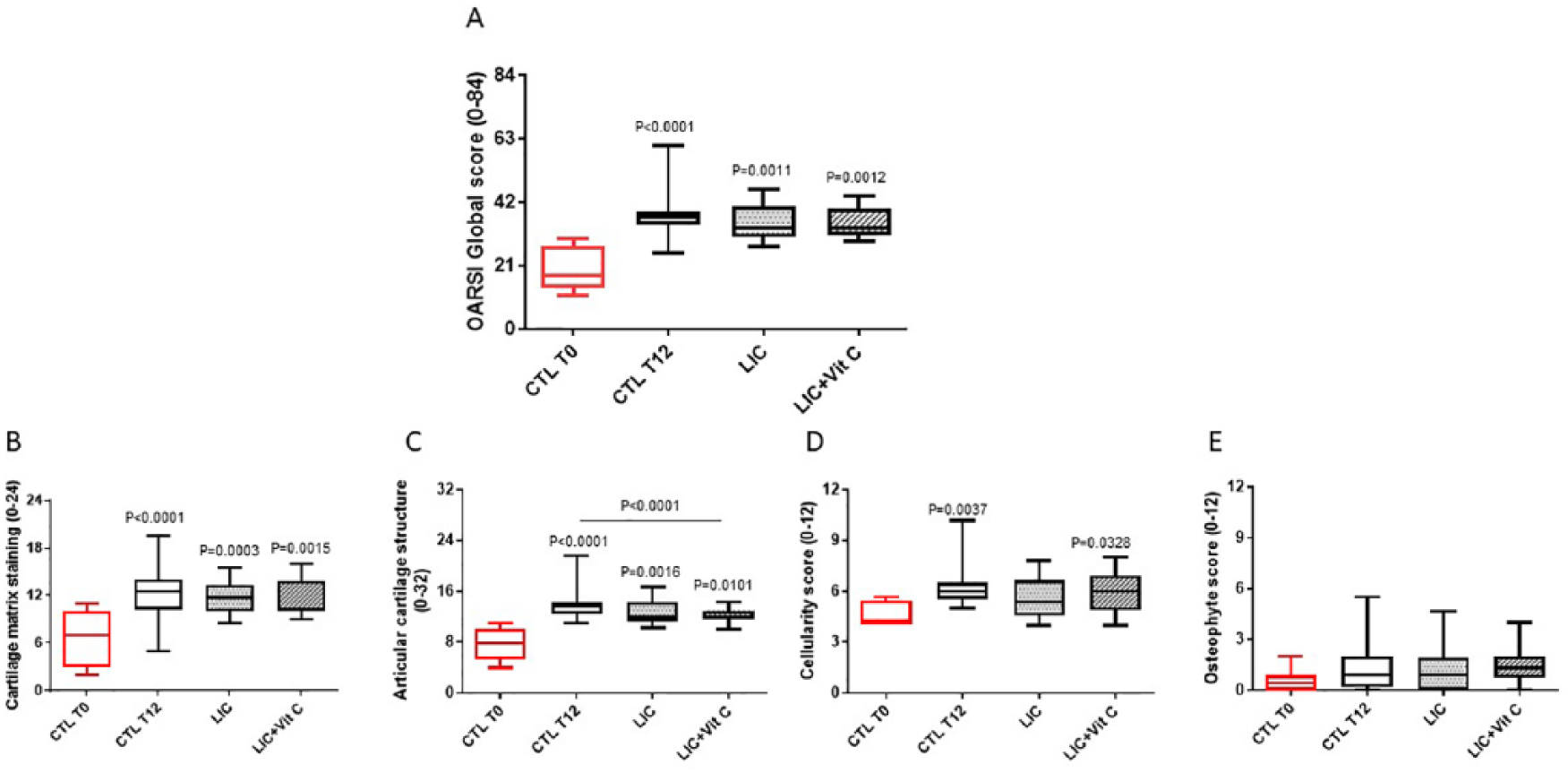
Global histological score of cartilage according to OARSI (Osteoarthritis Research Society International) recommendations
28
in 16-week-old (CTL [control] T0, reference group, untreated, n = 8, right and left knees evaluated) and 28-week-old Dunkin Hartley guinea pigs (CTL T12; placebo or lyophilized inactivated culture [LIC; 1 µg/kg/d] with or without vitamin C [1 mg/kg/d] groups; n = 24, right and left knees evaluated). (
Compared with controls, there were no significant differences in global scores between any of the treatment groups ( Fig. 2A ). When OARSI score items were analyzed individually, a significant lower cartilage structure change was observed in the LIC + vitamin C group compared with CTL T12 ( Fig. 2C ), suggesting a protective effect of the combination. Other criteria of evaluation (i.e., cellularity or osteophyte formation) were not significantly modified by the tested products. Note that the scores for osteophyte score and tidemark (data not shown) revealed no differences between study groups.
Noteworthy when considering the medial tibial plateau alone ( Fig. 3 ), where lesions appeared first and were the most severe, only CTL worsened significantly (P = 0.048). However, no significant difference between groups was observed at T12. The individual analysis of the OARSI score items ( Fig. 3C-F ) showed that matrix staining (P = 0.0275) and structure (P = 0.0084) scores were significantly increased in CTL. In contrast, these histological items were not significantly modified in LIC with or without vitamin C groups. The cellularity and osteophytes items were not significantly modified between T0 and T12 in the CTL groups and no treatments effects were observed on these items.
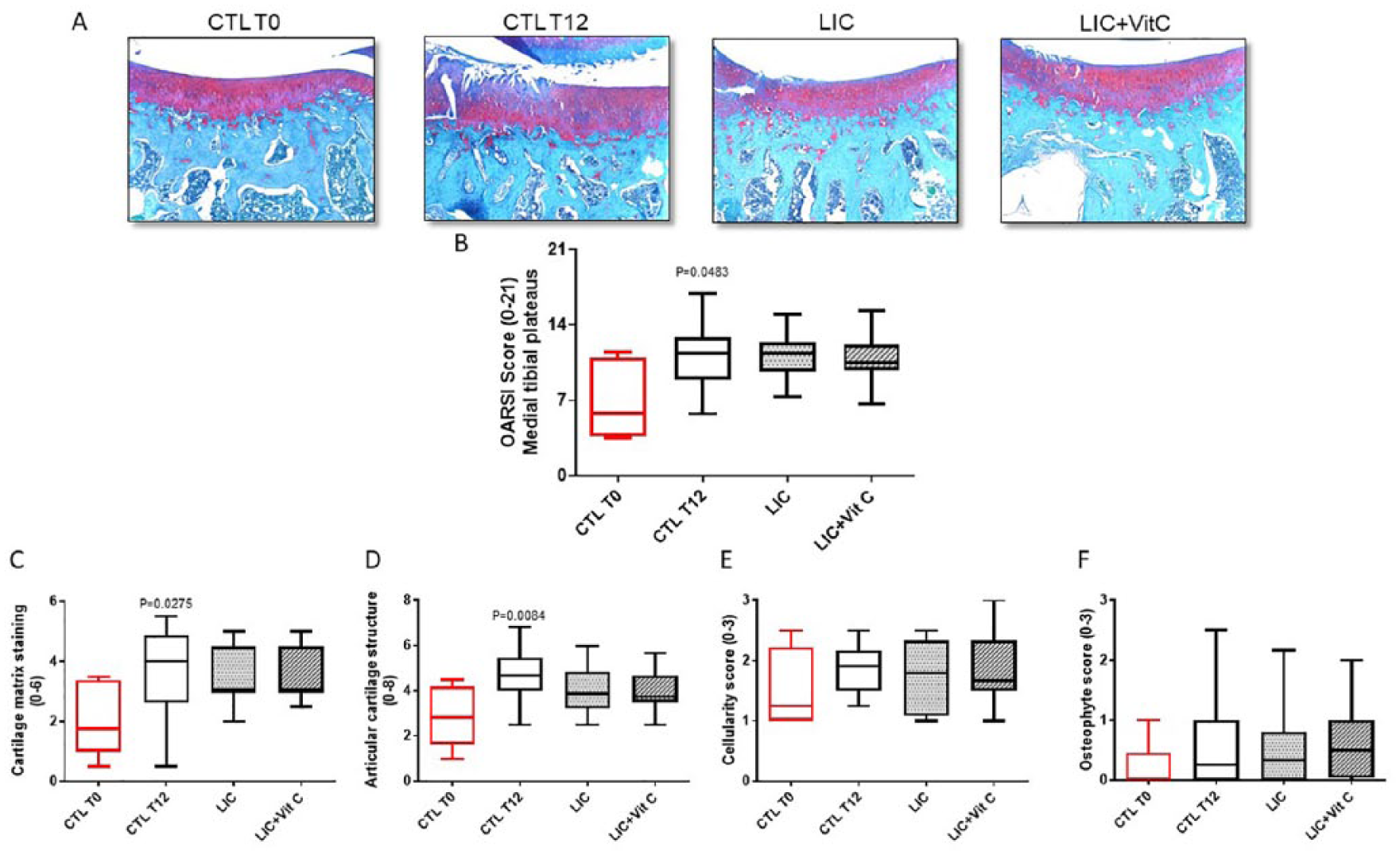
Histological score of cartilage obtained on the medial tibial plateau according to OARSI (Osteoarthritis Research Society International) recommendations
28
in 16-week-old (CTL [control] T0, reference group, untreated; n = 8, right and left knees evaluated) and 28-week-old Dunkin Hartley guinea pigs (CTL T12; placebo, or lyophilized inactivated culture [LIC; 1 µg/kg/d] with or without vitamin C [1 mg/kg/d] groups; n = 24, right and left knees evaluated). (
Inflammatory Status of the Synovial Membrane
The OARSI score for synovial membrane ( Fig. 4 ) revealed the development of a mild inflammatory reaction at T12. This inflammatory reaction was characterized by a significant increase of the villous hyperplasia criteria in all groups ( Fig. 4C ). Synovial lining hyperplasia and cellular infiltration scores tended to increase with time, but changes failed to reach significance ( Fig. 4A-D ). None of the inflammatory criteria were significantly modified by LIC or LIC + vitamin C.
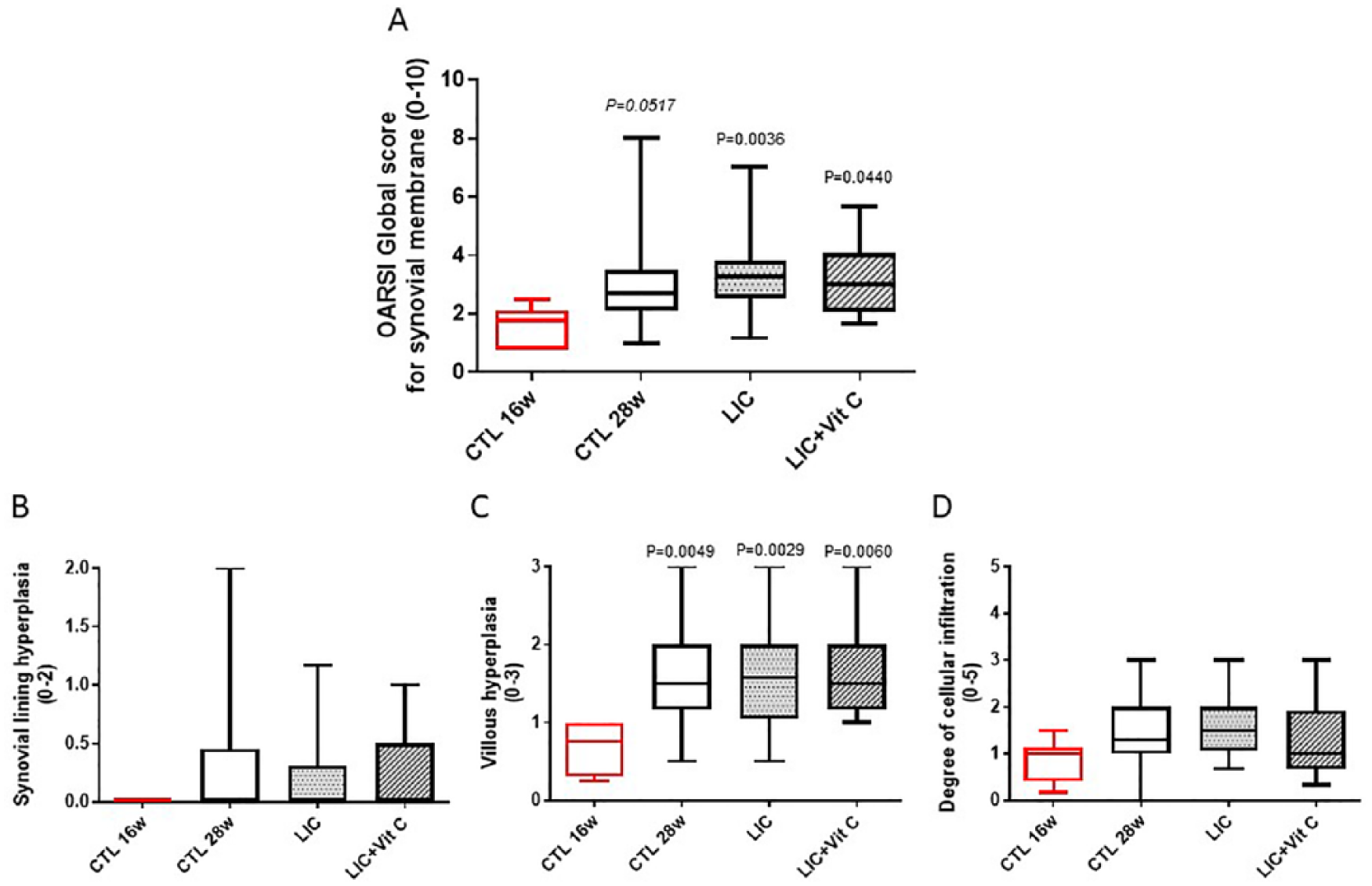
Histological score of the synovial membrane according to OARSI (Osteoarthritis Research Society International) recommendations
28
in 16-week-old (CTL [control] T0, reference group, untreated; n = 8, right and left knees evaluated) and 28-week-old Dunkin Hartley guinea pigs (CTL T12; placebo, lyophilized inactivated culture [LIC; 1 µg/kg/d] with or without vitamin C [1 mg/kg/d] groups; n = 24, right and left knees evaluated). (
Effect on Serum Levels of Specific Biomarkers in Guinea Pigs during the Spontaneous Development of OA
The serum level of Coll2-1 ( Fig. 5A ), a marker of type II collagen degradation, throughout the study was found to be stable over time (P = 0.15) in the LIC group, whereas Coll2-1 level increased over time in the LIC + vitamin C (P = 0.0002) and CTL (P = 0.0023) treatment groups. Coll2-1 time curve was significantly lower in LIC than in CTL group (P = 0.0004) while LIC + vitamin C and CTL time curves were superimposed (P = 0.69).
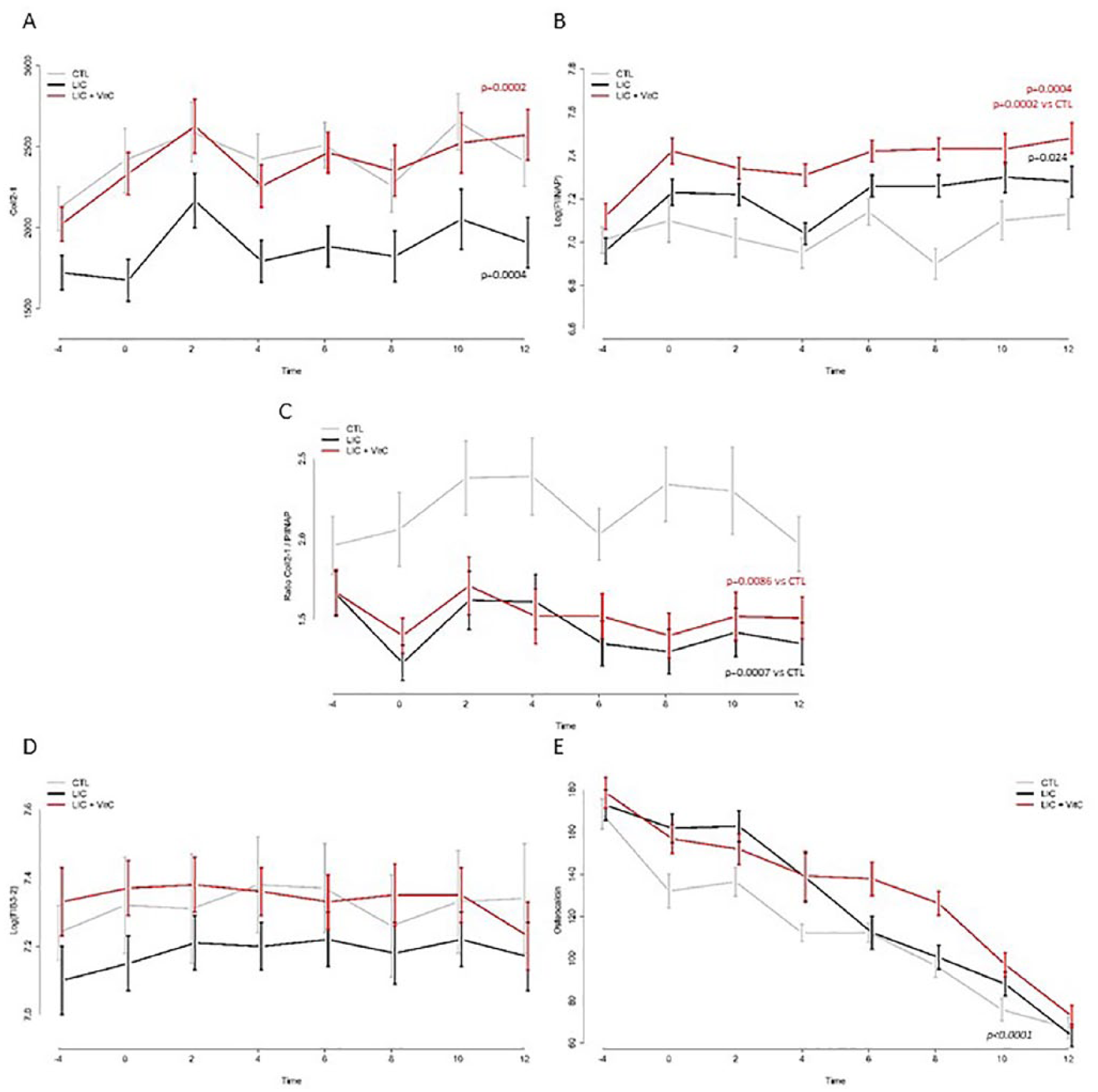
Biomarkers levels in GUINEA pigs sera between T-4 (when guinea pigs were 12 weeks old) and T12 (when guinea pigs were 26 weeks old and after 12 weeks of oral treatment) in CTL (control), lyophilized inactivated culture (LIC), and LIC + vitamin C groups: (
Interestingly, the levels of serum PIIANP ( Fig. 5B ), a marker of type II collagen synthesis was found to be stable over time (P = 0.67) in the CTL group, PIINAP level increased over time in the LIC (P = 0.024) and in the LIC + vitamin C (P = 0.0004) treatment groups. As compared with the CTL group, PIIANP time curve was found to be significantly higher in the LIC + vitamin C (P = 0.0003) group; no difference was observed between LIC and CTL groups (P = 0.14).
When considering the ratio Coll2-1/PIIANP ( Fig. 5C ), no significant time evolution was observed (time effect in CTL group, P = 0.64; LIC, P = 0.066; LIC + vitamin C,P = 0.28). However, when compared with the CTL group, the ratio between Coll2-1/PIIANP was significantly lower with LIC (P = 0.0007) and LIC + vitamin C (P = 0.0086).
The Fib3-2, a fragment of fibulin-3 a glycoprotein present in cartilage extracellular matrix, serum levels ( Fig. 5D ) did not evolved over time (time effect in CTL group, P = 0.41; LIC, P = 0.71; LIC + vitamin C, P = 0.35). The 3 groups were found to be comparable (CTL vs. LIC,P = 0.74; CTL vs. LIC + vitamin C, P = 0.93).
The serum levels of osteocalcin, a marker of bone formation ( Fig. 5E ) were found to decrease significantly over time (P < 0.0001). The decrease was comparable in each group (control vs. LIC, P = 0.84; control vs. LIC + vitamin C, P = 0.12). Serum osteocalcin was not affected by both treatments.
Interestingly, the OARSI global score was significantly correlated with the variation of the Coll2-1/PIIANP ratio (r = 0.2794, P = 0.0494).
Tolerability of the Treatment
No adverse events were reported. The daily clinical observation was normal. To ensure the tolerability of LIC, several organs (i.e., liver, kidney, spleen) were analyzed (data not shown). No observations related to the product were reported.
Discussion
This study was designed to document the potential prophylactic effect of LIC against the spontaneous development of OA in Dunkin Hartley guinea pigs. To our knowledge, this is the first time that B. longum lyophilized inactivated culture was tested in an OA animal model.
This study was developed in the model that fitted the best the preventive action of a dietary supplement and the targeted physiopathology. The Dunkin Hartley guinea pig model is validated, is accepted by the scientific community, and is well described. 28 This model of developing spontaneous knee OA is very attractive because it mimics well the pathophysiological processes observed in aging-related human OA. 30 The appearance of joint pathology in both guinea pigs and humans is associated to a variety of well-recognized OA risk factors, including body weight, mechanical load, ligament laxity, and high bone turnover. In this model, cartilage lesions in the knee are bilateral and symmetric, and appear first, and are more pronounced, in the medial knee compartment in the area not covered by the meniscus. Cartilage destruction is associated with mild inflammation of the synovial membrane.28,29 In this study, the lesions observed in control animals were in accordance with the published data.27,28 They ensured the validity of the developed model and consequently of the obtained results. The parameters of evaluation are considered as gold standard parameters (i.e., histology). Moreover, the results were further documented with the assay of several pertinent biomarkers in the serum of guinea pigs. 31
LIC was administered in a prophylactic manner in guinea pigs from the age of 16 to 28 weeks. The treatment was initiated at the time of the appearance of the first cartilage lesions.27,28,32 LIC was shown to be not only well tolerated but also efficient at preventing the development of OA. Interestingly, treatment efficacy was more pronounced on the medial plateaus where the lesions appeared first and were more severe. This conclusion was based on the observation that in the medial compartment histological lesions significantly increased while treated groups did not reach statistical significance. Of course, this finding needs to be confirmed in longer follow-up and in other OA models in which OA is induced by other mechanisms like mechanical stress. Additionally, biomarkers confirmed that treatments might positively interfere with cartilage metabolism. Indeed, the ratio Coll2-1/PIIANP was in favor of cartilage matrix synthesis and correlated with the global histological score. LIC induced a decrease of Coll2-1, a marker of type II collagen degradation.33,34 Several studies have demonstrated that treatment in both human and animals could decrease its serum levels.29,35,36 On the contrary, LIC increased PIINAP (type IIA procollagen N-propeptide), a marker of cartilage anabolism through the synthesis of type II collagen. 37 The balance between catabolism (reflected by Coll2-1 levels) and anabolism (reflected by PIIANP levels) was in favor of anabolism with the treatments. Of course, we cannot exclude an effect of vitamin C, since only the effect of LIC + vitamin C treatment was significant on PIIANP levels. Furthermore, vitamin C supplementation has been shown to stimulate collagen synthesis in this animal model, which may explain the significant increase in PIIANP with LIC + vitamin C. 38 Unfortunately, this cannot be confirmed due to the absence of a vitamin C control group. We recognized that the absence of vitamin C control group is a major limitation of this study. Interestingly, among the tested biomarkers, cartilage metabolism seemed to be the only one target of the treatments. Indeed osteocalcin, a marker of bone turnover, was not affected by the treatment. Osteocalcin decreased in all groups throughout the study duration. This result was in accordance with the previous published results in the same model 39 showing the involvement and the importance of subchondral bone in this model development. Altogether, these results obtained with biomarkers assays revealed a positive effect of LIC on cartilage metabolism.
The role of microbiome is more and more evoked and investigated in OA pathophysiology.7,12 It was recently shown with Lactobacillus casei in patients suffering from bilateral primary knee OA. 40 Indeed after 6 months of treatment this double blind randomized controlled trial showed a significant improvement of patient symptoms. In the present study, LIC showed a significant trend toward cartilage structure preservation. The potential effect of LIC was proven on efficacy parameters (i.e., structure and biomarkers). Hence, to act on the microbiota could play a role in the prevention of cartilage degradation occurring during spontaneous OA development in a context of mild inflammation.
These results are promising, if confirmed and completed, to consider LIC as a dietary supplement. Dietary supplement or nutraceuticals could indeed provide with an interesting alternative for the prevention in OA.41,42 Of course, the mechanisms of action of LIC remains questionable. Because of the low concentration of the administered LIC (1 µg/mL), this is difficult to conclude to a direct effect on dysbiosis. Nevertheless, one may suggest a potential indirect role of LIC on immunity. Indeed, Collins et al. 43 suggested that microinflammation may be linked to circulating LPS through the activation of the TLR. However, previous works 23 have shown that the B. longum strain did not interact with TLR. By this way, LIC could have induced cartilage protection when other bacteria or debris would induce its degradation through the stimulation of LPS-TLR (-2 and -6). The protection of cartilage by the treatment was indirectly demonstrated by the measurement of the biomarkers of cartilage.
However, we have to interpret these data carefully. Indeed, one major limitation to this study is that the microbiome and the effect on dysbiosis were not studied because of the collective housing system. It would be interesting to test LIC in animals suffering of dysbiosis. This should be further documented to better understand the mechanism of action of probiotics.
In summary, LIC showed a positive effect on type II collagen metabolism evaluated by measuring specific biomarkers in the serum of OA animals, but no significant effect was observed on structural changes in OA joint tissues. Before concluding on the potential therapeutic efficacy of LIC, more evidence of its efficacy should be collected including LIC effects on animals with objectified dysbiosis.
Footnotes
Authors’ Note
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Author Contributions
Conception and design: YH, AN, SP
Analysis and interpretation of the data: all authors
Drafting of the article: YH
Critical revision of the article for important intellectual content: all authors
Final approval of the article: all authors
Provision of study materials or patients: YH
Statistical expertise: YH, SP, AN
Administrative, technical, or logistic support: YH
Collection and assembly of data: YH, AN, SP
Acknowledgments and Funding
The authors thank Dr. Christelle Boileau for her assistance in manuscript preparation and ARTIALIS SA (Liège, Belgium) for the conduct of in vivo studies. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Biose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AN, SP, AP and MR are employees of Biose. YH received speaker fees from Biose.
Ethical Approval
The study was submitted to and approved by the Vet-Agro Sup (Ecole Nationale Vétérinaire de Lyon) Ethical Committee.
Animal Welfare
All procedures used for these studies conformed to the national ethic chart for animal experimentation.
