Abstract
Caspase-2 can induce apoptosis in response to extrinsic and intrinsic signals. Unlike other caspases, this protein is not expressed solely in nonnuclear compartments; a subpopulation is constitutively localized in the nucleus. As one of the most evolutionarily conserved caspases, caspase-2 may have roles in multiple cellular processes. However, its contribution to nonapoptotic processes remains a mystery. In this study, we show that caspase-2 activity is important for proliferation by cells of the androgen-dependent prostate cancer cell line LNCaP. LNCaP cells expressing either a dominant-negative (dn) form of caspase or an siRNA against caspase-2 had lower androgen receptor (AR)–dependent proliferative responses than control cells, and application of the siRNA resulted in downregulation of the expression of both AR-dependent prostate-specific antigen (PSA) and AR-dependent reporter luciferase. Also, caspase-2 formed complexes with the cell cycle regulatory proteins cyclin D3, CDK4, and p21/Cip1, and caspase-2 regulated AR transactivation by inhibiting the repressive function of cyclin D3. Taken together, these results reveal, for the first time, that caspase-2 is involved in cell cycle promotion and AR activation. Given that prostate cancer cells depend on AR activity in order to survive, the fact that our data indicate that caspase-2 positively regulates AR activity suggests that caspase-2 has potential as a target in the treatment of prostate cancer.
Introduction
Caspase-2 is one of the most conserved caspases, 1 and like the precursor forms of other initiator caspases, procaspase-2 contains a long prodomain. This prodomain of caspase-2 is most closely related in structure to that of caspase-9; both contain a caspase recruitment domain (CARD). 2 Like procaspase-9, which is activated within the apoptosome, caspase-2 can be activated within the PIDDosome complex. 3 However, it has been suggested that procaspase-2 may not require an adaptor protein to oligomerize. Studies using a noncleavable mutant form of caspase-2 revealed that dimerization is the key event that drives the initial procaspase-2 activation and that processing is not required. 4 Following dimerization, procaspase-2 undergoes autocatalytic cleavage that promotes its further stabilization and enhances its catalytic activity. Although caspase-2 zymogen does not require this cleavage for the initial acquisition of activity, inter-subunit cleavage is essential to generate the level of activity required for the induction of cell death. 4 Subcellular fractionation studies have revealed that procaspase-2 resides in the Golgi complex, mitochondria, nucleus, and cytoplasm. Notably, procaspase-2 is the only procaspase with a constitutive presence in the nucleus.5,6 Its nuclear import is regulated by two nuclear localization signals (NLSs) in the prodomain. Additionally, when overexpressed as a GFP fusion protein, the catalytically inactive caspase-2 mutant or the prodomain can form higher order structures, mostly in the nucleus.7,8 Overexpression of caspase-2 with mutated NLS leads to apoptosis; however, the significance of the nuclear pool of caspase-2 remains enigmatic. 7
Several cyclin-dependent kinases play a role in the G1-to-S transition. Each holoenzyme complex contains minimally a regulatory subunit (a cyclin) and a catalytic subunit (a cyclin-dependent kinase [CDK]).
9
During progression through G1, the levels of D-type cyclins increase, and these proteins associate with and activate CDK4 or CDK6 in a mitogen-regulated manner.
10
This first wave of cyclin D–dependent kinase activity is followed in late G1 by an increase in cyclin E–CDK2 activity.11,12 Current evidence suggests that the S phase–promoting function of cyclin D3– and cyclin E–associated kinases relates to their ability to phosphorylate pRb and release E2F transcription factors from an inactive or repressive Rb-E2F complex.
13
Outside of this role in the cell cycle, cyclins have CDK-independent functions. Cyclin D1 is known to bind estrogen receptor (ER)–α and to activate its transactivation function in the absence of ligand.14,15 In contrast, cyclins D1 and D3 interact with the androgen receptor (AR)
The known CKIs are grouped into two gene families, Ink4 and Cip/Kip, according to structural similarities. Currently, the Cip/Kip family is known to include the following three members: p21/Cip1, p27/Kip1, and p57/Kip2. These proteins act as stoichiometric inhibitors of the cell cycle, and although they inhibit all G1 complexes
A limited number of publications indicate that caspase-2 may contribute to regulation of the cell cycle and transcription machineries. Recently, it has been shown that the cell cycle regulator cyclin D3 interacts with caspase-2. 23 It has also been established that caspase-2 can repress the myocyte enhancer factor-2C transcription factor (MEF2C) by regulating the cleavage of histone deacetylase 4 (HDAC4). 24
To date, the nonapoptotic cellular functions of caspase-2 are not fully understood. We have previously shown that the level of caspase-2 expression is androgen dependent. 25 The goal of this work was to identify the nonapoptotic role caspase-2 plays in androgen-dependent LNCaP cells. We found that caspase-2 activity is important for the proliferation of LNCaP cells in response to AR ligand dihydrotestosterone DHT. Additionally, caspase-2 regulates the activity of AR and the cell cycle by forming a complex with cyclin D3, CDK4, and p21/Cip1. Taken together, these findings highlight some earlier unknown functions of caspase-2.
Results
Proliferative response to DHT in LNCaP is caspase-2 dependent
Previous studies have shown that culturing LNCaP cells under SFC results in early G1 arrest. 26 The typical growth curve of LNCaP cells in response to androgens is biphasic: low doses (in the pM range) stimulate proliferation, whereas higher concentrations (in nM to μM ranges) cause a progressive decline in cell growth.27,28 Recently, we showed that caspase-2 is androgen regulated: high concentrations of DHT led to decreased expression of caspase-2 (but not other caspases) and inhibited apoptosis induced by TNF family ligands. 25 Furthermore, chromatin immunoprecipitation revealed that AR interacts with the androgen-responsive elements (AREs) in intron 8 of the caspase-2 gene.
In order to determine the role of caspase-2 in the response to DHT treatment, we assessed the proliferation of LNCaP-Puro (control), LNCaP Caspase-2dn, and LNCaP siCaspase-2 cells by measuring [3H] thymidine uptake (Fig. 1A). We found that the proliferative response to DHT in LNCaP Caspase-2dn and LNCaP siCaspase-2 cells was approximately 2-fold lower than that in control cells. These results were confirmed by ViaCount analysis 72 hours after treatment with 10 pM DHT (Fig. 1B). The results obtained from direct counting of cells also showed that cells expressing Caspase-2dn proliferated less rapidly than their control counterparts in response to DHT treatment and that LNCaP siCaspase-2 cells failed to respond to DHT treatment altogether. Thus, our results show that reducing the levels of active caspase-2 downregulates the proliferative response to DHT. This suggests that the proliferation of LNCaP cells in response to DHT is dependent on caspase-2.
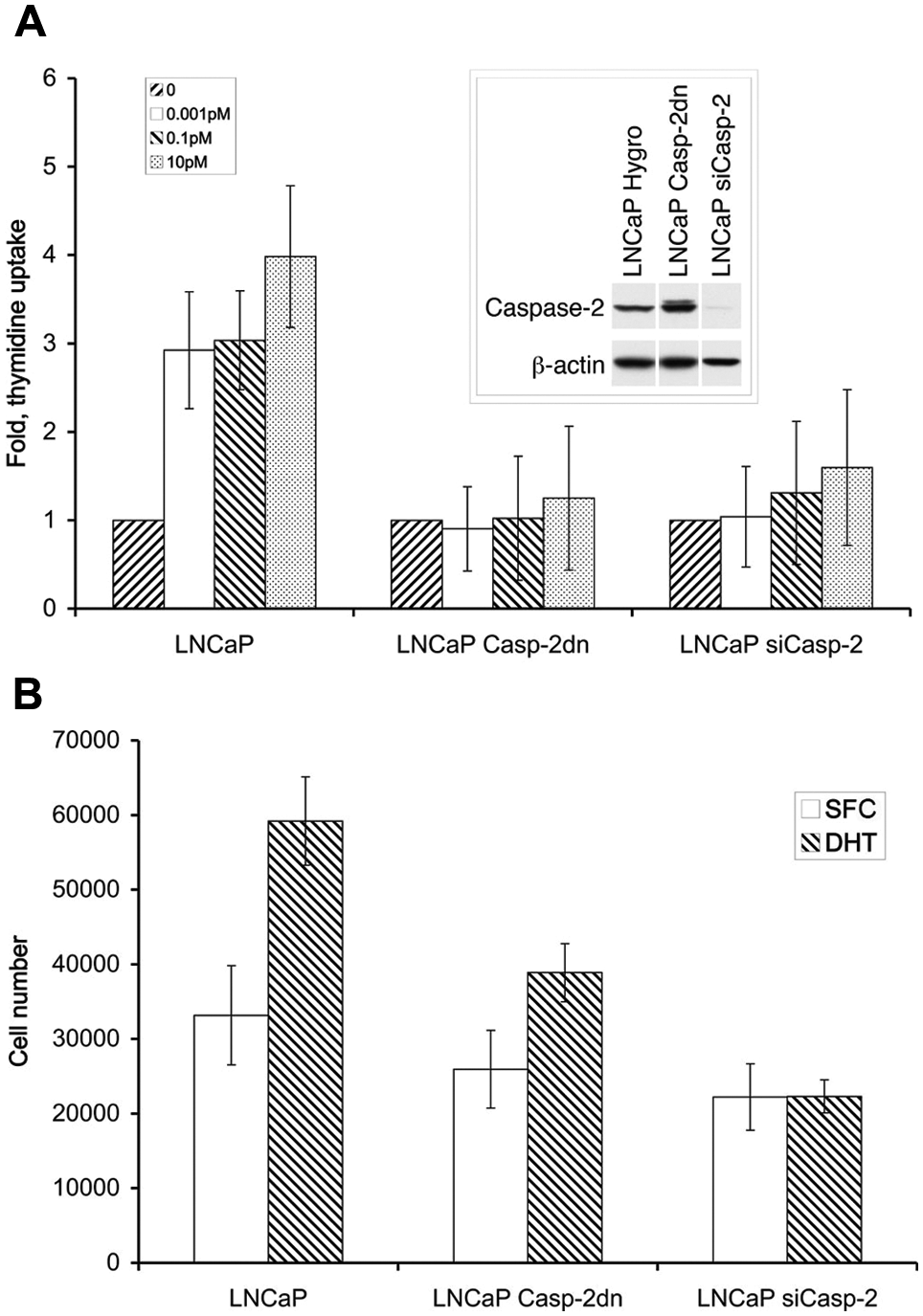
The proliferative response to DHT in LNCaP is caspase-2 dependent. (
AR activity depends on caspase-2 levels
Based on the results described above, we hypothesized that caspase-2 regulates AR activity. We tested this hypothesis by measuring the levels of one of the well-known AR-dependent proteins, prostate-specific antigen (PSA), in culture medium conditioned by LNCaP and LNCaP siCaspase-2 cells (Fig. 2A). The cells were incubated under SFC for 48 hours and treated with 10 pM or 1 nM DHT for 24 hours. We found that cells with reduced caspase-2 expression also had reduced levels of PSA. Thus, AR activity depends on caspase-2 expression. In the next set of experiments, we measured AR-dependent transactivation following DHT treatment in LNCaP and LNCaP siCaspase-2 cells transfected with a luciferase reporter gene whose expression is regulated by probasin androgen-responsive elements in the promoter. 29 Here also, we found lower AR activity in cells expressing reduced levels of caspase-2 (Fig. 2B). This association between caspase-2 inhibition and low AR activity strongly suggests that caspase-2 is a positive regulator of AR activity.
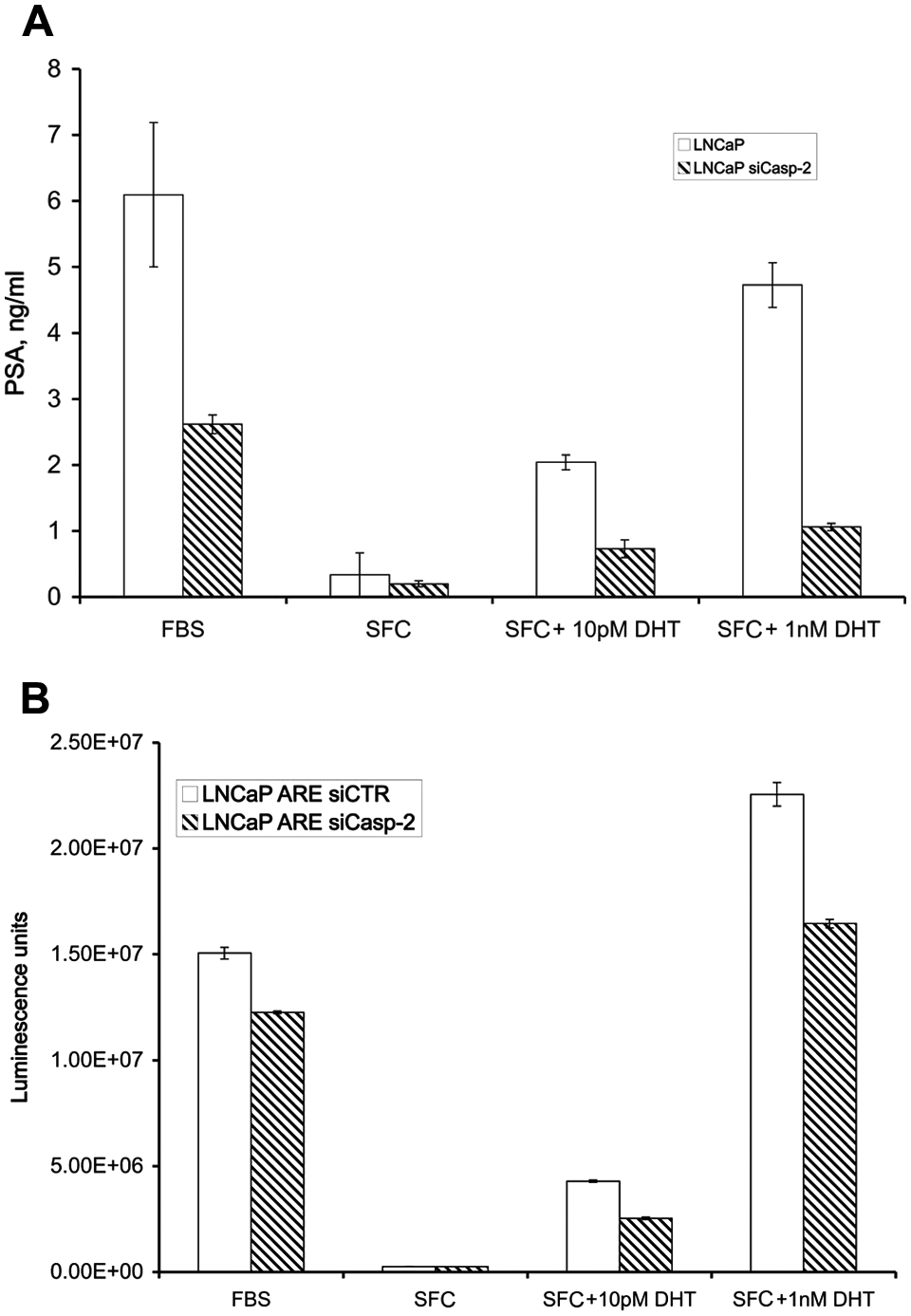
Caspase-2 regulates AR transactivation. (
AR–cyclin D3 interaction depends on caspase-2
Recently, Olshavsky
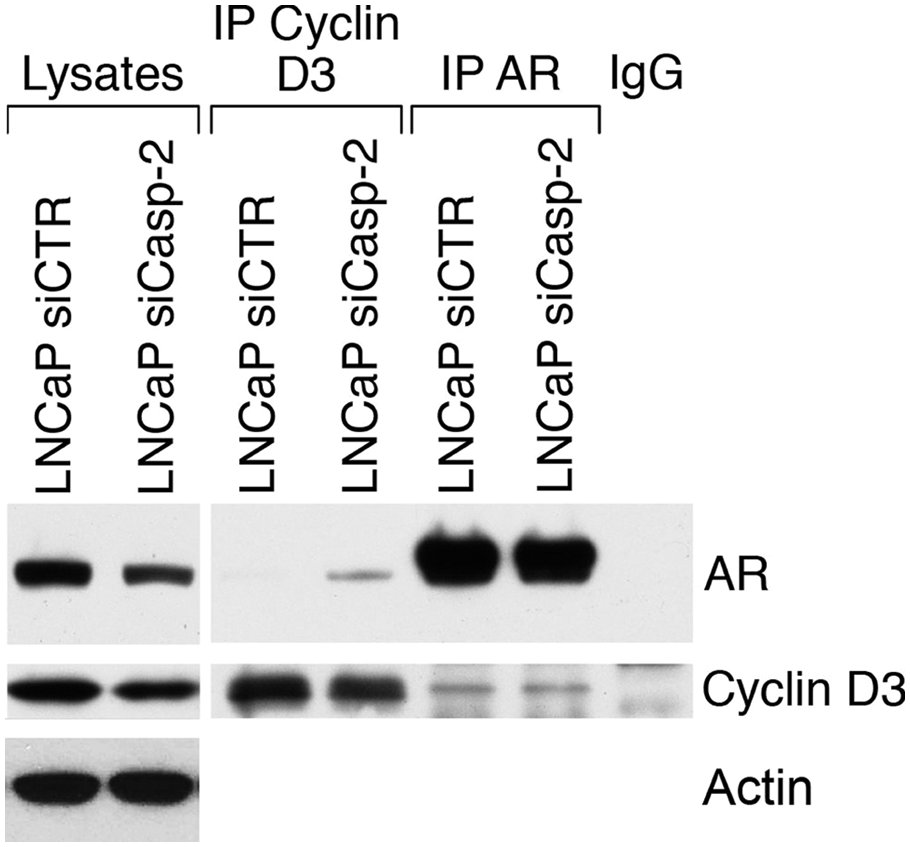
Caspase-2 regulates the AR–cyclin D3 interaction. Equal amounts of cell lysates from untreated LNCaP-siCTR and LNCaP siCaspase-2 cells were subjected to immunoprecipitation analysis using mouse anti-AR and mouse anti–cyclin D3 antibodies. The same antibodies were used to visualize proteins by Western blot analysis. The figure is representative of three experiments.
Caspase-2 forms a complex with cyclin D3, CDK4, and p21/Cip1
We next sought to determine how caspase-2 regulates binding between cyclin D3 and AR. Immunoprecipitation analysis using anticaspase-2 antibodies and subsequent Western blot analysis revealed that this protein can associate physically with cyclin D3, CDK4, and p21/Cip1 in LNCaP cells. We also found that DHT treatment leads to an increase in the association between caspase-2 and both cyclin D3 and CDK4 and to dissociation of caspase-2 and p21/Cip1 (Fig. 4A). Taken together, these results indicate that caspase-2 is involved in a complex (es) with three well-known regulators of the cell cycle. These data also indicate that AR activation increases the association of caspase-2 with proteins that promote the cell cycle while decreasing its association with an antagonist of the cell cycle (Fig. 4A). p21/Cip1 is a universal inhibitor of cyclin/CDK family members, and its overexpression inhibits the proliferation of mammalian cells. 30 AR activation initiates dissociation of p21/Cip1 from the caspase-2/cyclin D3/CDK4 complex and promotes cell proliferation (Fig. 4A). In order to check the involvement of caspase-2 in p21/Cip1 downregulation, we cultured LNCaP-Hygro and LNCaP siCaspase-2 cells under SFC and exposed them to 10 pM DHT for 72 and 120 hours. The results of Western blot analysis at these time points indicate that caspase-2 inhibition led to restoration of p21/Cip1 in response to DHT treatment (Fig. 4B). Thus, caspase-2 may increase activation of the cyclin D3/CDK4 complex by downregulating p21/Cip1.

Caspase-2 forms a complex with cyclin D3, CDK4, and p21/Cip1. (
Cyclin D3 can associate with AR and repress AR activity through CDK4-independent mechanisms. Cyclin D3–mediated AR repression requires an association with HDAC3; inhibition of HDAC3 activity using the histone deacetylase inhibitor (HDI) TSA abrogates cyclin D3 repression. 18 Immunoprecipitation analysis of caspase-2 and anti-p21/Cip1 indicates that TSA treatment promotes the formation of complexes that incorporate caspase-2, cyclin D3, and p21/Cip1 (Fig. 5A). Based on the amounts of cyclin D3 and p21/Cip present in caspase-2 containing complexes before and after TSA treatment, we can conclude that inhibiting HDAC activity increases the amount of cyclin D3 and p21/Cip1 bound to caspase-2. Based on the results described above, we suggest that caspase-2 acts as an anchoring protein for cyclin D3, a function that also can promote cyclin D3 ability to interact with CDK4 by decreasing the p21/Cip1 inhibitory effect. At the same time, caspase-2 may promote AR activity by preventing cyclin D3 from exerting its repressive function when present in a complex with p21/Cip1. We also found that caspase-2 can form a complex with this set of proteins in the androgen-independent cell line DU145, following treatment with TSA (Fig. 5B). This fact indicates that the involvement of caspase-2 in cell cycle and/or cyclin D3 regulation is a universal mechanism rather than LNCaP specific.

TSA-induced formation of caspase-2/cyclin D3/p21 complexes inhibits HDAC activity. Immunoprecipitation analysis. (
Discussion
In higher organisms, caspase biology is very complex, with more than 14 caspases collectively able to cleave a large variety of substrates. It is therefore assumed that caspases are intimately involved in many aspects of cellular function rather than playing roles only in apoptosis. 31 Indeed, caspases in higher organisms have acquired functions in regulating cellular processes such as cell proliferation and cell cycling.31-33 Nevertheless, how an individual caspase maintains its tissue and stimulus specificity during apoptosis in addition to participating in other cellular processes remains unknown.
Here, we describe nonapoptotic functions of caspase-2 in the androgen-dependent prostatic carcinoma cell line LNCaP. Our results indicate that proliferation of these cells in response to DHT treatment is caspase-2 dependent (Fig. 1). These data are in agreement with results obtained by Alam
Among the caspases, caspase-2 is the second of the mammalian caspases to have been identified35,36 and the best conserved from an evolutionary standpoint.
37
Caspase-2 has two nuclear localization signals and is capable of translocating to the nucleus.
7
Moreover, activation of the nuclear pool of caspase-2 results in apoptosis-related mitochondrial changes.8,38 The recent discovery that cyclin D3 interacts biochemically with caspase-2 led to the suggestion that this interaction might stabilize caspase-2.
23
Cyclin D3 is also linked to attenuation of the ligand-dependent proliferation-promoting function of AR in AR-dependent prostate cancer cells.
18
Zong
Cells from exponentially growing cultures that were subsequently cultured under SFC were characterized by a significant increase in the G1 population, reflecting G1 arrest, and the addition of DHT reversed this effect. 26 To elucidate the mechanism by which caspase-2 regulates cyclin D3 with respect to the cyclin D3–caspase-2 link in the regulation of AR activity, we decided to check for the possible involvement of caspase-2 in cell cycle regulation. We found that caspase-2 was able to form a complex with cyclin D3, CDK4, and p21/Cip1 in cells cultured under SFC, suggesting that caspase-2 is involved in the regulation of the cell cycle. Moreover, DHT treatment, which promotes the G1-S transition, 26 led to increases in the levels of cyclin D3 and CDK4 bound to caspase-2 and to a decrease in the amount of p21/Cip1 bound to caspase-2 (Fig. 4A). Furthermore, Western blot analysis of proteins involved in cell cycle regulation revealed that the most profound effect of caspase-2 inhibition was on p21/Cip1 (Fig. 4B); DHT treatment led to the disappearance of p21/Cip1 in control cells but to an accumulation of p21/Cip1 in cells expressing an siRNA targeting caspase-2. Thus, in LNCaP, p21/Cip1 levels depend on caspase-2, and caspase-2 may also cleave p21/Cip1. Taken together, our results indicate that caspase-2 is a previously unidentified member of a network that governs the cell cycle.
Based on our findings, we conclude that caspase-2 has two cyclin D3–related functions. First, caspase-2 negatively regulates binding between cyclin D3 and AR, and second, when bound to cyclin D3, caspase-2 promotes the cell cycle. Thus, caspase-2 regulates the switching of cyclin D3 between AR-containing and CDK4-containing complexes. Other studies have shown that cyclin D3–HDAC3 complex represses AR activity and that inhibiting HDAC3 activity abrogates cyclin D3 inhibitory function.17,18 Our finding that TSA, an HDAC inhibitor, induces formation of the caspase-2/cyclin D3 complex (in both LNCaP and DU145 cells) (Fig. 5) indicates that cyclin D3 moves from an AR-containing complex to a caspase-2–containing complex when HDAC3 activity is inhibited.
The dimerization of procaspase-2 is the key event that drives caspase-2 activation. Following this dimerization, procaspase-2 undergoes an autocatalytic cleavage that stabilizes this dimerization and further enhances its own catalytic activity. 4 How cells regulate caspase-2 activity, keep it at a certain level, and avoid initiation of apoptosis is one of the main questions. Notably, cyclin D3 has been implicated in the activation of caspase-2. 23 Coexpression of cyclin D3 with caspase-2 in human cells leads to enhanced apoptosis compared to that observed when only caspase-2 is overexpressed. Additionally, the expression of cyclin D3 increases the amount of active caspase-2. Based on these findings, it was suggested that an interaction between cyclin D3 and caspase-2 may stabilize the latter. 23 In another study, p21/Cip1 was shown to inhibit caspase-2 activity in a p21/Rb/E2F pathway–dependent manner. 40 Collectively, results obtained by us and others indicate that the activities of proteins that depend on caspase-2 may regulate the activity of caspase-2.
Based on our own results and data reported in the literature, we propose the following model to explain caspase-2–based regulation of AR activity (Fig. 6). According to this model, caspase-2 binds to cyclin D3, thereby neutralizing the inhibitory effect of cyclin D3 on AR. CDK4 promotes the cell cycle by interacting with the caspase-2/cyclin D3 complex. Binding of p21/Cip1 to the caspase- 2/cyclin D3/CDK4 complex, in turn, inhibits the cell cycle. Binding of p21/Cip1 to the caspase-2/cyclin D3 complex, however, has the opposite effect, inhibiting caspase-2 activity as it relates to apoptosis (Fig. 6). This model would account for how caspase-2 might link cell signaling pathways central to two vital cellular processes: apoptosis and the cell cycle.
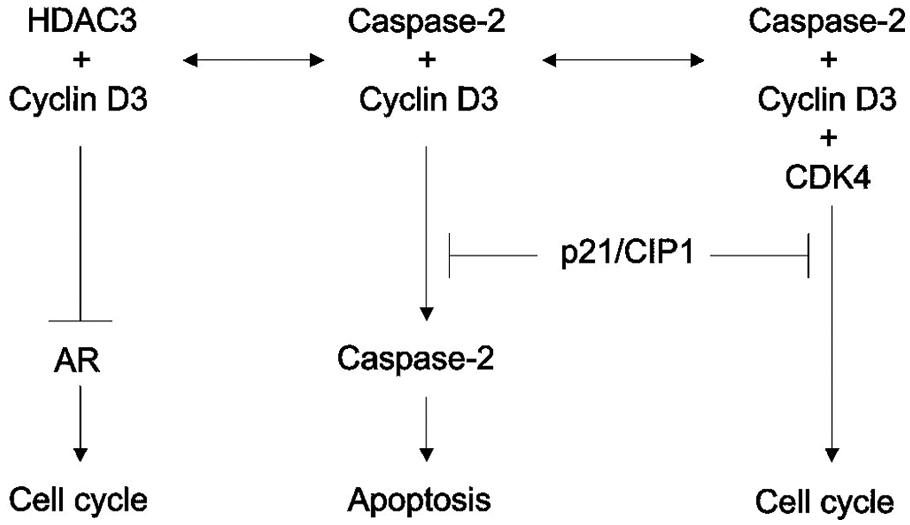
Links between caspase-2 and cell signaling pathways that regulate apoptosis and the cell cycle. Critical elements of the suggested model are schematically represented (see details in the Discussion).
Materials and Methods
Cell culture and transfection
The human prostate cancer cell lines LNCaP and DU145 were maintained in RPMI 1640 (HyClone Laboratories Inc., Logan, UT) supplemented with 10% fetal bovine serum. A steroid-free condition (SFC) was achieved by using phenol red–free RPMI (Gibco, Invitrogen, Carlsbad, CA) supplemented with 10% charcoal-stripped fetal bovine serum. The Effectene transfection reagent kit (Qiagen GmbH, Hilden, Germany) was used for transfection. The vector used for silencing caspase-2 and the hairpin forms of caspase-2 siRNAs were previously described.25,34 Plasmid DNA expressing Caspase-2dn (pMaRXmycflag/caspase-2 cis/Puro), a catalytically inactive form of caspase-2 (Cys303Ser), was a kind gift from Dr. Y. Lazebnik (Cold Spring Harbor Laboratory, Cold Spring Harbor, NY) and was described earlier. 41 The control cell lines LNCaP-RNA H1/Hygro and LNCaP-Puro were as sensitive to treatment as the LNCaP cell line.
DHT treatment
For proliferation assays, cells were plated at a density of 7,000 cells/well in a 96-well flat- bottomed plate for 72 hours under SFC alone and after with DHT present at the indicated concentrations. Twenty-four hours later, 1 μCi/well [ 3 H] thymidine was added, after which the cells were incubated a further 24 hours and then harvested. Cells were harvested onto glass fiber filter paper, and the incorporated radioactivity was measured by liquid scintillation counting. All samples were measured in triplicate in each of four independent experiments. In experiments where the ViaCount reagent was used (Guava Technologies/Millipore, Billerica, MA), cells were plated at a density of 30,000 cells/well in a 12-well flat-bottomed plate and incubated 72 hours under SFC and in the presence of 10 pM DHT. Following this incubation, cells were harvested from each well and counted in triplicate (1,000 cells per count). All samples were measured in duplicate in two independent experiments. For immunoprecipitation experiments, cells at approximately 70% confluence were incubated under SFC for 72 hours, after which DHT was added to a concentration of 10 pM and the cells were incubated for a further 48 hours.
Preparation of total cell lysate, immunoprecipitation, and Western blot analysis
Cells were detached from culture flasks by trypsinization, washed twice with PBS, and incubated in lysis buffer (20 mM Tris-HCl, pH 7.2, 1% Triton X-100, 100 nM phenylmethylsulfonyl fluoride [PMSF], 0.2 g/L EDTA tetrasodium salt, and protease inhibitors) on ice for 20 minutes, followed by centrifugation at 16,000
For immunoprecipitation, cells were lysed in IP buffer (30 mM Tris-HCl, pH 7.5, 150 mM NaCl, 0.1% (v/v) Triton X-100, 10% (v/v) glycerol, 100 nM PMSF, and protease inhibitors) on ice for 20 minutes, after being subjected to centrifugation at 16,000
The following primary antibodies were used: rabbit and mouse anticaspase-2 (generous gift from Dr. Y. Lazebnik), anti-actin (Sigma), anti-CDK4 (Cell Signaling Technologies Inc., Beverly, MA), anti-p21 (Calbiochem/Millipore, Billerica, MA), anti–cyclin D3 (BD Transduction Laboratories, San Jose, CA), and anti-AR (BD Pharmingen, Franklin, NJ). The blots were counterstained with goat anti-mouse or goat anti-rabbit IgG conjugated with HRP (Pierce, Rockford, IL). The immunoreactive bands were visualized by incubating the membrane in enhanced chemiluminescence reagent (Pierce).
Luciferase reporter assay
AR transactivation was measured using an LNCaP-expressing luciferase reporter cassette (LNCaP-Luc) described earlier. 29 The LNCaP-ARE-siCTR and LNCaP-ARE-siCaspase-2 cell lines were generated by lentiviral infection of the LNCaP-Luc cell line with pLSL-Puro constructs expressing the siRNA caspase-2 hairpins described earlier25,41 or siCTR hairpins bearing a scrambled sequence GGCATTATCGAATTGGGTTA. Luciferase activity was estimated by harvesting cells (by trypsinization), washing them with PBS, and lysing them in reporter lysis buffer (200 μL). Luciferase bioluminescence activity was measured using the luciferase assay system (Promega, Madison, WI). Sample aliquots (2 μg/20 μL) were assayed for light emission with a 20/20n luminometer (MLX Dynex Technology Inc., Franklin, MA).
Measurement of PSA expression
LNCaP-hygro and LNCaP siCaspase-2 cells were cultured in 24-well plates for 48 hours under SFC and then treated for 24 hours with DHT at the indicated concentrations. PSA levels in the culture medium were estimated by using the Free PSA ELISA kit according to the manufacturer’s instructions (MP Biomedicals, Orangeburg, NY). The amount of PSA was calculated using a calibration curve based on varying PSA concentration (provided by the manufacturer) and was normalized to the number of cells per well. Cells were counted using the ViaCount reagent (catalog number 4000-0040), obtained from Guava Technologies.
Footnotes
Acknowledgements
The authors thank Dr. Michael B. Cohen and Dr. C. Michael Knutson for continuous support during data collection and preparation of the article. They also thank Dr. Y. Lazebnik (Cold Spring Harbor Laboratory) for providing antibodies to caspase-2 and the expression vector with mutated caspase-2, and Natalya Guseva, Sean Martin, and Oksana Zagorodna for helpful discussions and critical review of the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported by funds from the Department of Pathology, The University of Iowa, Iowa City.
