Abstract
Background
Dupilumab and mepolizumab are effective add-on therapies for severe chronic rhinosinusitis with nasal polyps (CRSwNP). However, predictors of response and guidance on biologic selection remain limited. The aims were to identify baseline parameters associated with favorable response to dupilumab or mepolizumab at 6 and 12 months in severe CRSwNP and compare outcomes among treatments.
Methods
We conducted a multicenter, prospective, non-randomized, real-world cohort study across three tertiary hospitals in Spain. Patients with severe CRSwNP initiating dupilumab or mepolizumab were enrolled and followed at 6 and 12 months. Response was defined using EPOS/EUFOREA 2023 criteria, incorporating nasal polyp score (NPS), sinonasal outcome test (SNOT-22), total symptom score (TSS), olfaction (Sniffin’ Sticks Smell Test), systemic corticosteroid need, and asthma control test. Patients were classified as excellent, good, poor, or non-responders based on criteria fulfilled.
Results
Sixty-nine patients were included: 35 received dupilumab and 34 mepolizumab. Asthma was present in 86.8%, and 42.6% had aspirin-exacerbated respiratory disease. Both biologics improved SNOT-22, TSS and NPS at 6 and 12 months (P < .05), with comparable efficacy for most outcomes. Dupilumab showed superior improvement in olfaction (coefficient B [95% CI] mepolizumab vs dupilumab: −6.30 [−9.42; −3.19]). Overall, 84.2% of patients achieved a good/excellent response at 6 months and 89.6% at 12 months. Factors associated with better response included comorbid asthma and shorter time since last surgery.
Conclusions
Dupilumab and mepolizumab improved clinical outcomes in severe CRSwNP, with dupilumab offering greater benefit in olfaction. Asthma and surgical history may help predict response.
Keywords
Introduction
Monoclonal antibodies targeting type 2 (T2) cytokines have emerged as add-on therapy for patients with chronic rhinosinusitis with nasal polyps (CRSwNP) who fail to respond to standard of care (SoC), relapse after endoscopic sinus surgery (ESS) or require more than two cycles of systemic corticosteroids per year. 1 Current biologics approved for T2-CRSwNP include omalizumab (anti-IgE 2 ), mepolizumab (anti-IL-5 3 ), dupilumab (anti-IL-4Rα, inhibiting IL-4 and IL-13 signaling 4 ), and Tezepelumab (anti-thymic stromal lymphopoietin 5 ).
However, evidence on predictors of response to biologics prior to treatment initiation remains limited, and current international guidelines 6 do not provide specific recommendations on whether dupilumab or mepolizumab should be preferred. Elevated blood eosinophil count (BEC) has been proposed as a potential predictor of excellent response at 6 months, 7 but data remain inconsistent, and reliable tools for identifying the most suitable biologic for individual patients are lacking.
We conducted a multicenter, prospective cohort study to evaluate baseline demographic, clinical and laboratory parameters that could assist clinicians in predicting favorable responses to dupilumab and mepolizumab.
The primary aim was to assess associations between baseline factors and the likelihood of good or excellent response to biological therapy at 6 and 12 months. Secondary objectives were to evaluate treatment response at 6 and 12 months according to the EPOS/EUFOREA 2023 criteria, 6 and to compare treatment responses between dupilumab and mepolizumab subgroups at each time point.
Methods
Study Design and Population
We conducted a phase IV, multicenter, prospective, non-randomized, real-world cohort study in three tertiary university hospitals within the public healthcare system in Spain. Patients were evaluated every 6 months, following a standardized schedule. Between February 2022 and February 2025, patients with severe CRSwNP, with or without asthma, treated with mepolizumab or dupilumab were enrolled. Biologic selection was at the discretion of the treating clinician and subject to reimbursement policies at the time.
Inclusion and Exclusion Criteria
Eligible patients initiated with mepolizumab or dupilumab for primary CRSwNP according to EPOS/EUFOREA 2023 criteria, 6 either as primary indication or in the context of asthma with concomitant severe uncontrolled CRSwNP. In Spain, biologic prescription for CRSwNP in public hospitals requires a history of ≥2 prior ESS. 8 All patients, regardless of the primary indication for biologic therapy, underwent a pretreatment evaluation by a rhinologist, which included nasal endoscopy, patient-reported outcome measures and laboratory testing.
Exclusion criteria included secondary CRSwNP, biologic treatment prescribed for asthma in patients with non-severe CRSwNP, and ongoing biologic therapy for T2 inflammation disease at baseline. Patients with less than 6 months of follow-up were excluded.
Flowchart of patient selection is presented in Supplementary Figure S1.
Data Collection and Measurements
Data on demographics, prior surgeries, comorbidities, laboratory tests, and clinical scores were collected at baseline, 6 and 12 months using REDCap.
Clinical parameters assessed to evaluate response to treatment included: total symptom score (TSS, range 0-10); 22-item sinonasal outcome test (SNOT-22, range 0-110); endoscopic bilateral nasal polyp score according to Meltzer scale (NPS, range 0-8); peak nasal inspiratory flow; visual analog scale (VAS) of loss of smell (LoS) (range 0-10); 48-items Sniffin’ Sticks Smell Test (SSST) (Burghardt®, Wedel, Germany). The combined TDI (Threshold, Discrimination, Identification) score classifies function as anosmia (TDI≤16.25), hyposmia (TDI 16.25-30.75), normosomia (TDI 30.75-41.25), and supersmell (TDI≥41.25). 9
T2-inflammatory markers included BEC and total immunoglobulin E (IgE) at each timepoint. BEC was analyzed dichotomously (>300 vs ≤300 cells/µL), 10 across predefined categories (≤150, 150-300, >300 cells/µL), and per 150-cell/µL increment. Systemic corticosteroids intake was also collected (number of courses and cumulative grams of prednisolone).
In patients with comorbid asthma, the asthma control test (ACT) was also evaluated at each visit. 11
Definitions
Indication of Biologic According to EPOS/EUFOREA 2023
According to the EPOS/EUFOREA 2023 guidelines,
6
patients are eligible for biological therapy if they have undergone at least one prior ESS and present with recurrent bilateral nasal polyps, in addition to meeting at least three of the following criteria:
Evidence of T2 inflammation (tissular eosinophilia ≥ 10 eosinophils per high-power-field and/or total IgE ≥ 100 UL/mL and/or blood eosinophilia ≥ 150 cells/µL). Significant loss of smell (anosmia on smell test). Comorbid asthma requiring regular inhaled corticosteroids. Significantly impaired quality of life (QoL), defined as SNOT-22 ≥ 40. Requirement for at least 2 courses of systemic corticosteroids per year or long-term (>3 months) low-dose steroids.
In patients who have not undergone prior surgery due to medical comorbidities, fulfillment of at least 4 of the above criteria is required.
Response to Biologics
Treatment response was evaluated at 6 and 12 months, according to EPOS/EUFOREA 2023 guidelines, 6 using established thresholds for each assessment tool: NPS reduction ≥1 point from baseline 12 ; QoL improvement defined by ≥8.9-point SNOT-22 reduction 13 and/or a total SNOT-22 score <35; olfactory improvement defined by ≥5.5-point increase in the SSST 14 ; absence of systemic corticosteroids use or surgery, and, in patients with asthma, ACT improvement ≥3 points 15 or sustained control with a score ≥20.
According to EPOS/EUFOREA 2023 guidelines, 6 patients were classified based on the number of criteria fulfilled: excellent responders met all criteria; good responders met 3-4/5 (with asthma) or 3/4 (without asthma); poor-responders met 1-2 items, and non-responders met none.
Statistical Analyses
Statistical analyses were performed by an independent analyst blinded to study hypotheses. Continuous variables were summarized as median (Q1-Q3) and categorical variables as absolute and relative frequencies. Baseline characteristics were compared using the Kruskal–Wallis test for continuous variables and the Chi-squared test for categorical variables.
Independent quantile mixed-effects models were fitted to evaluate effectiveness of biologic therapies at 6 and 12 months. In these models, patient was specified as the random factor and time as the fixed factor. To compare outcomes between the dupilumab and mepolizumab groups, independent mixed-effects models were also estimated for each outcome, with baseline outcome value, time, treatment group and the group-by-time interaction included as fixed factors. Logistic regression models for each baseline factor and outcome were applied to examine factors associated with treatment response. Statistical significance was set at P < .05. All analyses were performed with R version 4.3.
Ethical Aspects
The study was conducted in compliance with Declarations of Helsinki and Istanbul and the EU Good Clinical Practice. All participants provided written informed consent. Ethical approval was granted from the local Ethics Committees (protocol code EOM024/23).
Results
Patient Characteristics
Sixty-nine patients were included (median age 52 years); 63.8% (n = 44) were male. Thirty-five received dupilumab and 34 mepolizumab.
Asthma was present in 86.8% (n = 59), and 42.6% (n = 29) had aspirin-exacerbated respiratory disease (AERD). Among patients with asthma, 52.5% (n = 31) had severe asthma, 32% (n = 19) had moderate asthma, and 15.2% (n = 9) had mild-asthma.
All patients received long-term intranasal corticosteroids and high-volume saline irrigations. Thirteen patients (19.4%) were corticodependent. The median of oral corticosteroid courses in the preceding year was 1; up to 25% of patients required 2 (in the mepolizumab group) or 3 (in the dupilumab group) courses. Patients had a median of 2 prior sinus surgeries and a median BEC of 420 cells/μL. Median SNOT-22 and total SSST scores were 60 and 9; mean NPS was 4.93 ± 2.05 (Table 1). Nearly 95% of patients underwent, at minimum, bilateral maxillectomy with complete anterior and posterior ethmoidectomy. Detailed surgical characteristics are reported in Supplementary Table S2.
Baseline Patients’ Characteristics.
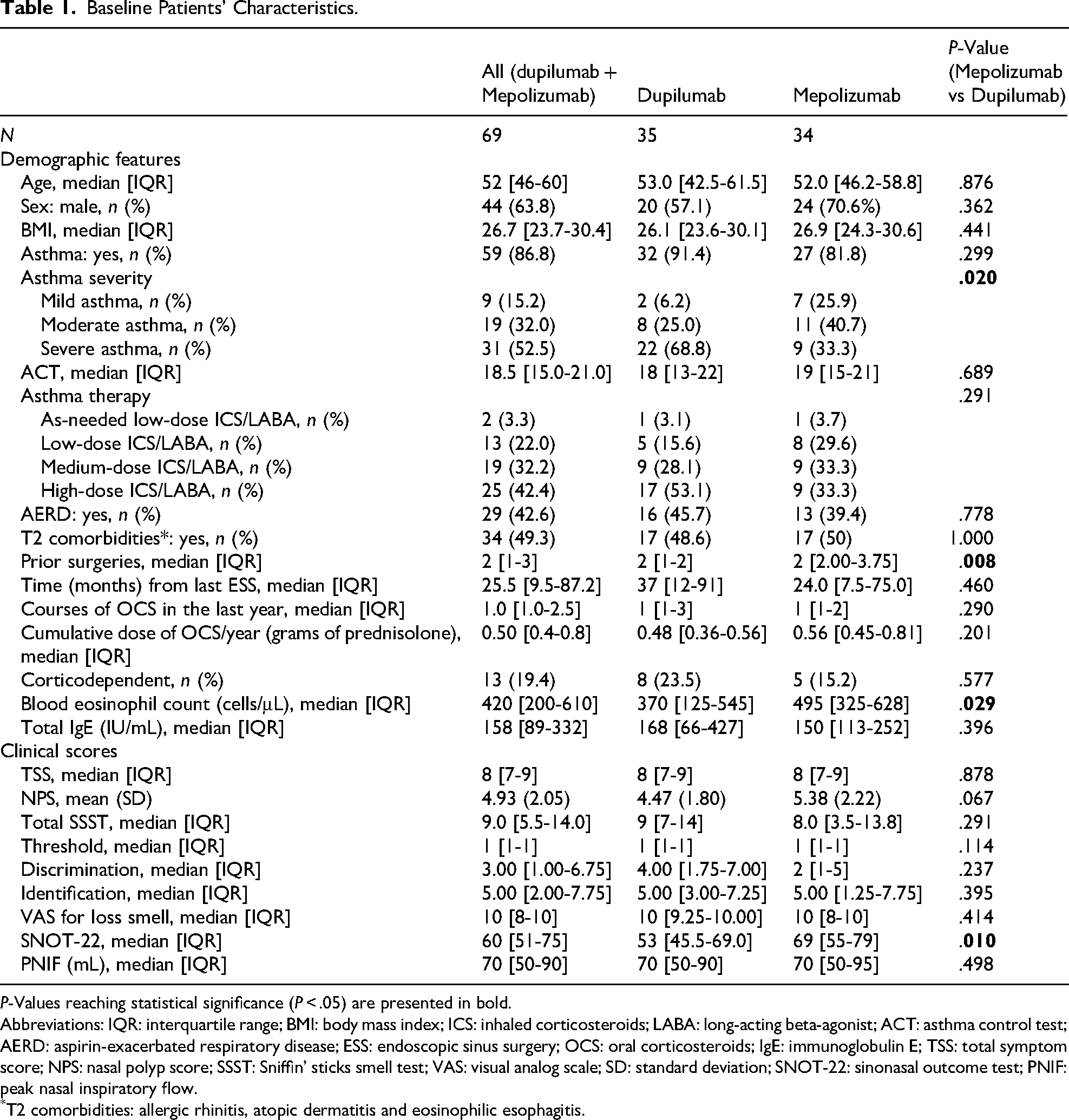
P-Values reaching statistical significance (P < .05) are presented in bold.
Abbreviations: IQR: interquartile range; BMI: body mass index; ICS: inhaled corticosteroids; LABA: long-acting beta-agonist; ACT: asthma control test; AERD: aspirin-exacerbated respiratory disease; ESS: endoscopic sinus surgery; OCS: oral corticosteroids; IgE: immunoglobulin E; TSS: total symptom score; NPS: nasal polyp score; SSST: Sniffin’ sticks smell test; VAS: visual analog scale; SD: standard deviation; SNOT-22: sinonasal outcome test; PNIF: peak nasal inspiratory flow.
T2 comorbidities: allergic rhinitis, atopic dermatitis and eosinophilic esophagitis.
Baseline characteristics were comparable between subgroups, except for prior surgeries (P = .008), BEC (P = .029), and SNOT-22 scores (P = .01), all higher in the mepolizumab group, and asthma severity, which was greater in the dupilumab group (P = .020) (Table 1).
Changes in Clinical Scores
Results of effectiveness to biologics are represented in Figure 1 and summarized in Supplementary Table S1. Notably, no patients were lost to follow-up.
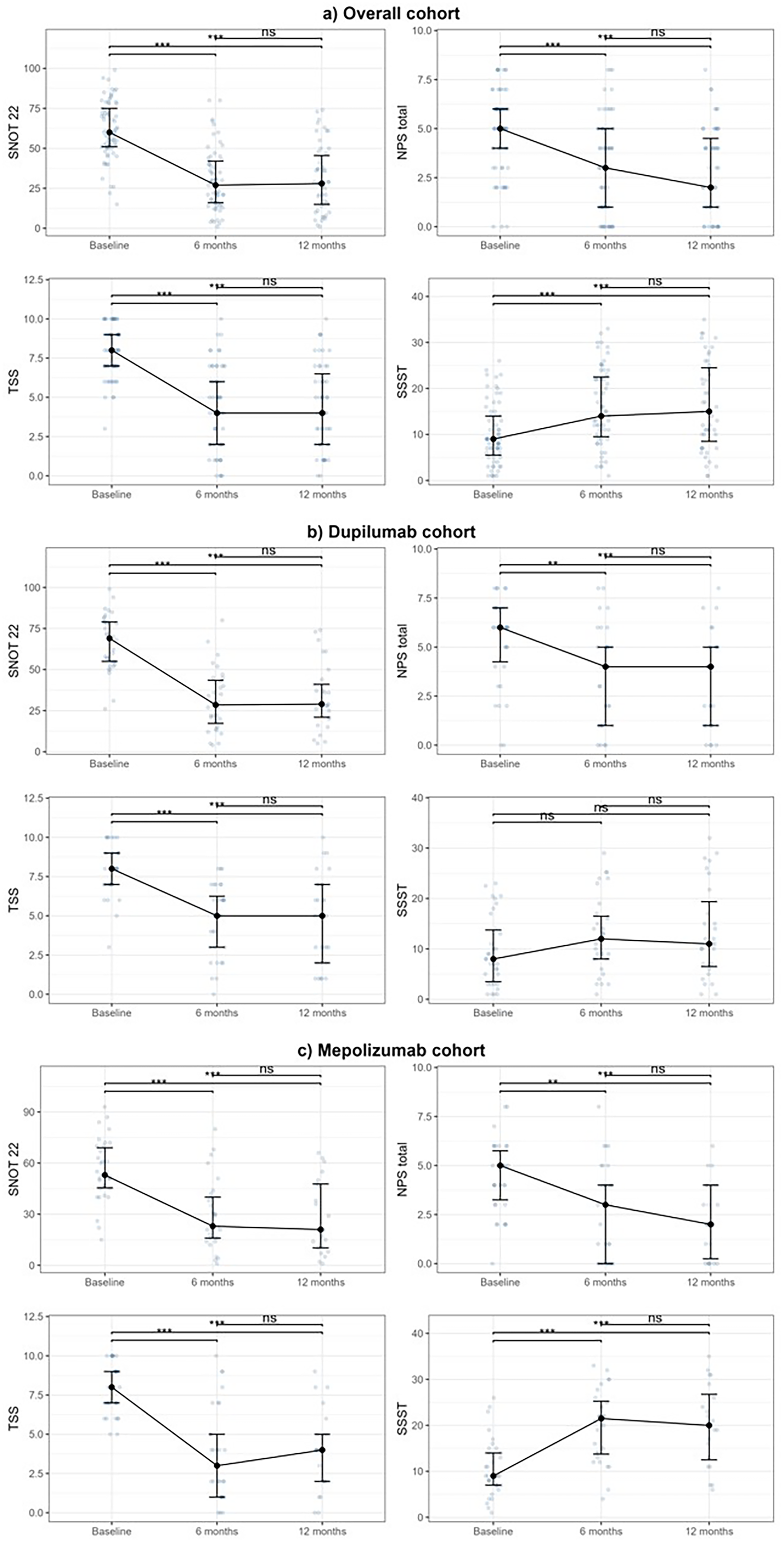
Effectiveness of biologics. Results are presented as median with interquartile range [Q1-Q3] at each timepoint.
Quality-of-Life Outcomes
Biologics significantly improved QoL over the 12-month period. Across all treatments, median SNOT-22 decreased from 60 at baseline to 27 at 6 months and 28 at 12 months (P < .001 vs baseline). TSS decreased from 8 to 4 at both time points (P < .001).
Dupilumab reduced median SNOT-22 from 53 to 23 at 6 months and 21 at 12 months; TSS decreased from 8 to 3 at 6 months and 4 at 12 months (P < .001 for all). Mepolizumab produced similar improvements: SNOT-22 improved from 69 to 30 at 6 months and 29 at 12 months; TSS from 8 to 5 both time points (P < .001 for all).
Mixed quantile regression showed no difference between biologics and confirmed a plateau in SNOT-22 and TSS between 6 and 12 months (Table 2).
Comparative Outcomes of Dupilumab and Mepolizumab Subgroups Assessed by Mixed-Effects Quantile Regression.
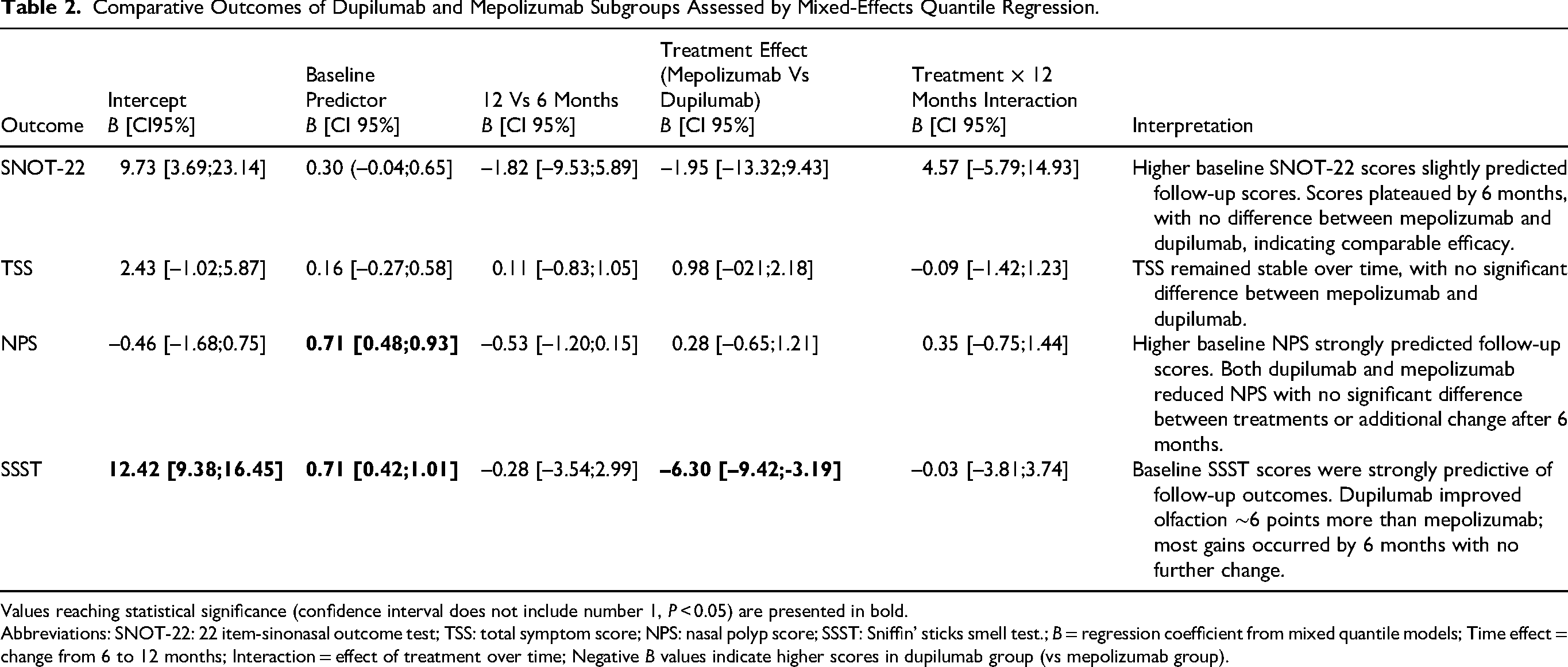
Values reaching statistical significance (confidence interval does not include number 1, P < 0.05) are presented in bold.
Abbreviations: SNOT-22: 22 item-sinonasal outcome test; TSS: total symptom score; NPS: nasal polyp score; SSST: Sniffin’ sticks smell test.; B = regression coefficient from mixed quantile models; Time effect = change from 6 to 12 months; Interaction = effect of treatment over time; Negative B values indicate higher scores in dupilumab group (vs mepolizumab group).
Effectiveness on NPS
NPS improved significantly across 12 months. In the overall cohort, median NPS decreased from 5 at baseline to 3 at 6 months and 2 at 12 months (both P < .001). Dupilumab reduced NPS from 5 to 3 at 6 months (P = .005) and 2 at 12 months (P = .001). Mepolizumab decreased NPS from 6 to 4 at both 6 and 12 months (P = .005 and P = .001).
NPS values plateaued after 6 months, with no difference between biologics, confirming comparable efficacy (Table 2).
Effectiveness on Smell
Olfactory function improved with biologics. In the overall cohort, median SSST scores increased from 9 at baseline to 14 at 6 months and 15 at 12 months (P < .001 and P = .001, respectively). VAS scores for LoS, decreased from 10 at baseline to 7 at 6 months and 8 at 12 months (both P < .001).
Dupilumab showed a pronounced improvement, with SSST scores rising from 9 to 21.5 at 6 months and 20 at 12 months (P < .001 and P = .001), alongside significant gains across all TDI subdomains. VAS scores dropped from 10 at baseline to 5 at 6 months and 6 at 12 months (both P < .001). Mepolizumab showed modest, non-significant changes. No TDI subdomains improvements were observed, and VAS scores remained stable.
Mixed quantile regression showed that most improvement in SSST scores occurred within 6 months and plateaued thereafter. Treatment effect favored dupilumab, with mepolizumab consistently approximately 6 points lower (B [95% CI]: −6.30 [−9.42; −3.19]). No interaction between treatment group and 12-month follow-up was observed, indicating a stable treatment difference (Table 2).
Baseline Factors Associated With Good/Excellent Response
Most significant results of the univariate analyses are summarized in Table 3.
Baseline Factors Associated With Good or Excellent Response to Biologic Therapy in CRSwNP.

Values reaching statistical significance (confidence interval does not include number 1, P < .05) are presented in bold.
Abbreviations: BMI: body mass index, BEC: blood eosinophil count; AERD: aspirin-exacerbated respiratory disease.
At 6 Months
In the overall cohort, asthma (OR 9.64, P = .036) and shorter time since last surgery (OR 0.98, P = .022) were associated with better outcomes. In the dupilumab group, a longer surgery-to-biologic interval showed a borderline association (OR 0.97, P = .050), suggesting that a greater time gap between the last ESS and biologic initiation may negatively impact outcomes. In the mepolizumab group, asthma was associated with good/excellent response (OR 34.5, P = .011). In adjusted analyses, asthma severity and asthma control were not significantly associated with increased odds of favorable outcomes (Supplementary Table S3).
At 12 Months
Asthma remained a robust associated factor in the overall (OR 20, P = .011) and mepolizumab (OR 20, P = .026) cohorts. No baseline factors were significant in the dupilumab group (Supplementary Table S3).
Evaluation of Treatment Response Rates
Treatment response was evaluated according with EPOS/EUFOREA 2023. Only 1 patient (3.7%) was stratified as a non-responder at 12 months in the mepolizumab subgroup. Results are shown in Table 4 and represented in Supplementary Figure S2.
Treatment Response Rates to Biologics at 6 and 12 Months.
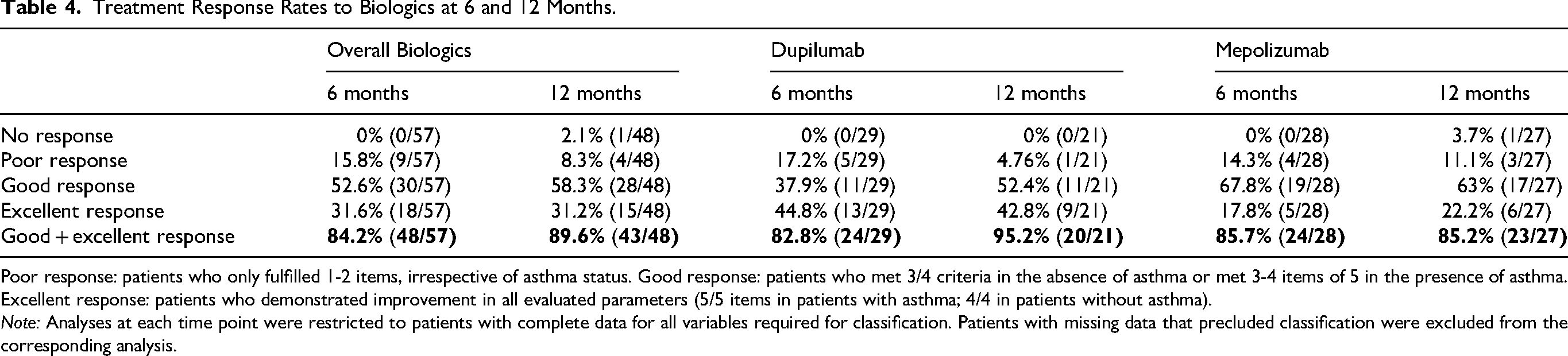
Poor response: patients who only fulfilled 1-2 items, irrespective of asthma status. Good response: patients who met 3/4 criteria in the absence of asthma or met 3-4 items of 5 in the presence of asthma. Excellent response: patients who demonstrated improvement in all evaluated parameters (5/5 items in patients with asthma; 4/4 in patients without asthma).
Note: Analyses at each time point were restricted to patients with complete data for all variables required for classification. Patients with missing data that precluded classification were excluded from the corresponding analysis.
Overall, poor response declined from 15.8% at 6 months to 8.3% at 12 months. Good or excellent responses increased over time, with combined responder rates of 84.2% at 6 months and 89.6% at 12 months. At 12 months, dupilumab response reached 95.2%, while mepolizumab remained 85.2%.
Subgroup analysis showed higher excellent response with dupilumab, whereas mepolizumab patients more often achieved good rather than excellent responses (Table 4). Longitudinal analysis indicated that most patients maintained or improved their response over time, with nearly all showing some clinical improvement by 12 months. Decrease from good/excellent response to poor/no response was anecdotical (n = 1) (Figure 2).
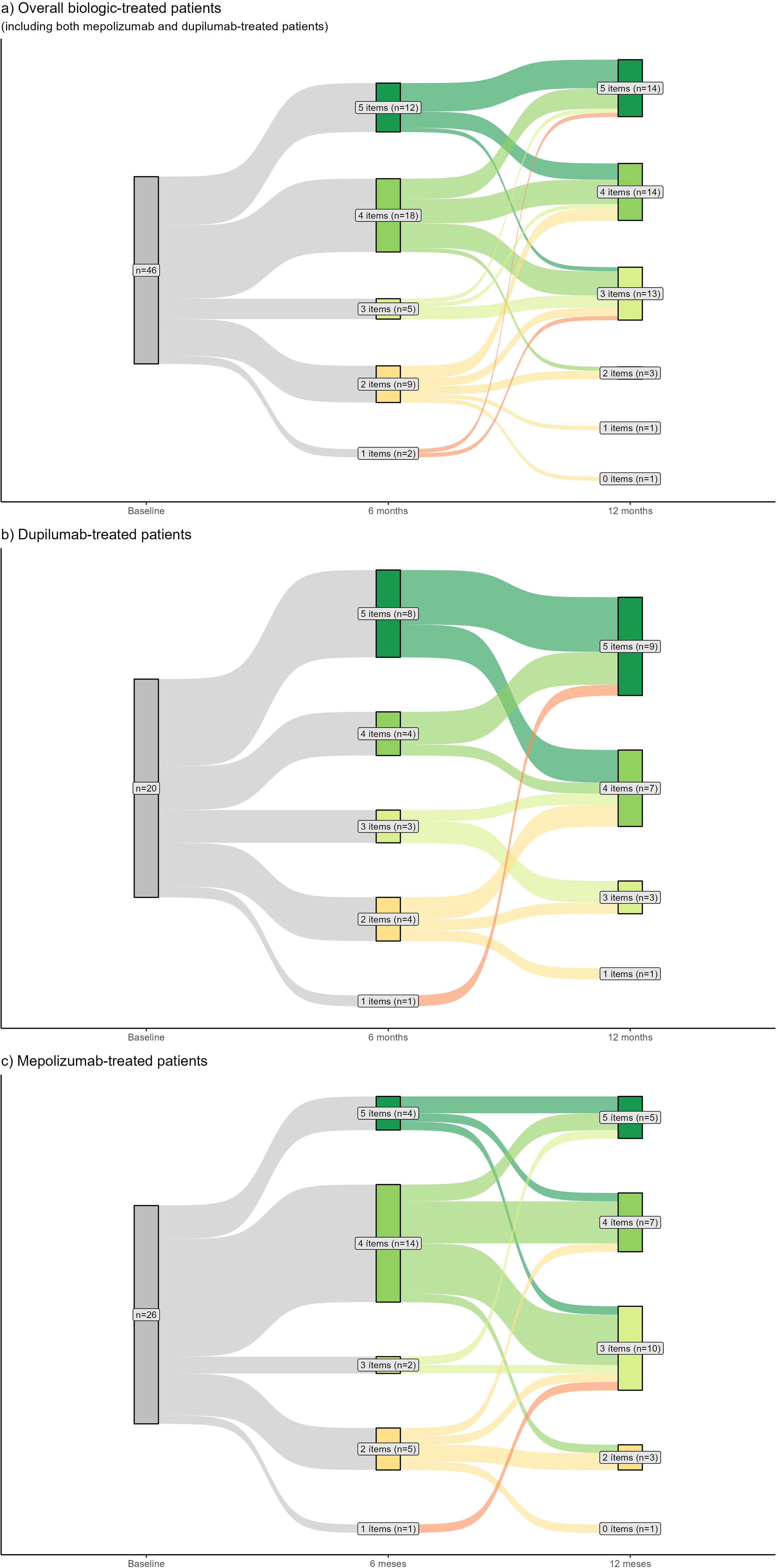
Sankey plot of patients’ response to biologics evolution, at 6 and 12 months. Patients were included only if all data necessary to classify their treatment response was available at both 6 and 12 months; those with missing information, at 6 or 12 months, that precluded categorization were excluded.
Discussion
The advent of biological therapies has expanded therapeutic options for patients with refractory CRSwNP. Following European Medicines Agency approval of dupilumab 4 and mepolizumab, 3 national regulatory agencies progressively authorized their use under country-specific reimbursement policies. In Spain, access and reimbursement is limited to patients with ≥2 prior ESS, 8 a restriction that limits eligibility and complicates real-world recruitment. 16 Within this context, this study represents one of the first multicenter, real-world evaluations of dupilumab and mepolizumab in CRSwNP Spanish cohort.
In our study, consistently with pivotal trials17,18 and real-life evidence on biologics in CRSwNP,7,19–21 both dupilumab and mepolizumab significantly improved QoL and endoscopic NPS, reinforcing their efficacy in heterogeneous real-world populations. Patient-reported outcomes—SNOT-22, TSS—showed marked improvements at 6 months, with no further gains, thereafter, suggesting a therapeutic plateau after 6 months. Recent real-world evidence22–24 suggests that biologic tapering (spacing dose administration), particularly with dupilumab, may be feasible in patients who achieve a sustained good or excellent response after 6 months of treatment. In these studies, clinical benefit achieved during the initial period was generally maintained following dose spacing in selected patients. Nevertheless, current guidelines continue to recommend long-term maintenance therapy, reflecting the limited prospective evidence on de-escalation strategies. Careful patient selection is therefore essential to minimize the risk of relapse after tapering.
One of the most noteworthy findings of our study is that olfactory outcomes differed between treatments. Dupilumab was associated with significant and sustained improvements in olfactory function, whereas mepolizumab produced modest, non-significant changes. Mixed-effects modeling confirmed greater olfactory gains with dupilumab (6.3 points more) sustained through 12 months. Most improvement occurred by 6 months, with no additional change thereafter.
Subdomain-specific SSST analysis showed significant improvement in threshold, discrimination, and identification at both time points for the overall cohort. Dupilumab produced early, sustained gains across all subdomains, whereas mepolizumab showed no significant changes. These findings suggest greater olfactory recovery with dupilumab, warranting longer-term follow-up. Notably, threshold improvement was minimal and did not reach clinical relevance. As threshold reflects peripheral olfactory function, this suggests that biologics primarily enhance odor discrimination and identification, while peripheral recovery is slower—due to the time required for inflammation resolution and neuroepithelium regeneration.25,26 Importantly, VAS scores for LoS paralleled SSST gains, confirming that semi-objective improvements translated into patient-perceived benefit, reinforcing the clinical relevance of these changes.
Baseline predictor analyses yielded interesting results. While a prior French real-life cohort study suggested a week association between elevated BEC and 6-month excellent response, 7 our study identified comorbid asthma as a significant predictor at 6 and 12 months, particularly in mepolizumab group. However, neither asthma severity nor suboptimal asthma control (ACT <20) was associated with better outcomes. In the global cohort, a shorter interval between last ESS and biologic initiation was statistically associated with good/excellent outcomes, suggesting more recent surgery may confer modest benefit, especially in the dupilumab-cohort. At 12 months, no additional predictors emerged, likely due to the low number of poor responders (n = 1).
Response categorization according to EPOS/EUFOREA 2023 showed high overall effectiveness. Excellent disease control was achieved less frequently in our cohort than in previous reports,7,19 likely reflecting differences in treatment distribution, as nearly all patients in those studies received dupilumab. In our dupilumab-cohort, response rates were comparable to those reported in the literature, whereas mepolizumab yielded substantially lower rates, suggesting that dupilumab enables earlier and more frequent achievement of excellent control. These differences could reflect distinct mechanisms of action but may also relate to the role of olfactory improvement—a requisite for excellent response—which appears more consistently achieved with dupilumab. Interpretation of mepolizumab results warrants caution, as these patients had higher baseline BEC, SNOT-22 scores and more prior surgeries, likely reflecting its earlier approval in Spain and use in patients unable to defer treatment initiation while awaiting access to other biologics.
Longitudinal analysis revealed that most patients maintained or improved their response categories between 6 and 12 months. These dynamics underscore the sustained clinical benefits of biologic therapy and support its long-term value.
Given the high costs of biologics, we evaluated the proportion of patients with meaningful benefit (≥3 response criteria), thus combining good and excellent responders. At 6 months, 84.2% met this threshold, rising to 89.6% at 12 months. Rates were similar at 6 months for dupilumab and mepolizumab (82.8% and 85.7%) but diverged at 12 months (95.2% vs 85.2%).
EPOS/EUFOREA guidelines advise continuing biologics if any improvement is observed at 6 months and reassessing at 12 months. Limited response may still justify continuation through shared clinician-patient decision, when alternatives are lacking, 6 reflecting current therapeutic constraints in uncontrolled CRSwNP and aligning with real-world practice. We strongly support the recommendation to reassess at 12 months, as some biologics require extended time to achieve full effect, and premature withdrawal may preclude assessment of their full potential.6,27
Strengths of our study include the prospective design, application of EPOS/EUFOREA 2023 criteria, comprehensive assessment of subjective and objective outcomes, 12-month follow-up and absence of loss to follow-up. A key methodological advantage is the use of the full 48-item SSST, providing a comprehensive and semi-objective evaluation of smell, aligned with COMET initiative recommendations, 28 unlike other studies that relied on abbreviated versions.19,22,29 In contrast with prior studies, 7 that combined dupilumab and mepolizumab into a single cohort for analysis, we evaluated each agent separately, providing greater granularity in treatment-specific effects.
Limitations include those inherent to an observational design, most notably the lack of randomization, which may have caused baseline differences that restrict direct comparisons. The low number of non-responders, especially among the dupilumab-cohort, prevented robust analysis of associated factors. The lack of standardization of the extent of prior ESS before the use of biologics may have introduced heterogeneity in surgical burden assessment. Due to regulatory requirements for biologics in Spain (≥2 prior ESS), patients with less advanced disease and fewer or no prior surgeries, may be underrepresented, which could limit the generalizability of the findings to this population. Larger prospective head-to-head studies are needed to identify reliable predictors of response and optimize treatment selection.
Conclusions
Both biologics demonstrated comparable improvements in overall symptom control and NPS reduction, but dupilumab yielded superior olfactory recovery. Comorbid asthma, particularly with mepolizumab, and shorter interval since last surgery predicted favorable outcomes.
Supplemental Material
sj-docx-1-ajr-10.1177_19458924261431418 - Supplemental material for High Response Rates and Predictors in Patients with CRSwNP Treated With Mepolizumab and Dupilumab: Results From a Prospective, Multicenter Cohort Study
Supplemental material, sj-docx-1-ajr-10.1177_19458924261431418 for High Response Rates and Predictors in Patients with CRSwNP Treated With Mepolizumab and Dupilumab: Results From a Prospective, Multicenter Cohort Study by Mireia Golet, Paula Cruz Toro, Albert Llansana Ríos, Carlota González Lluch, Laura Pardo-Muñoz, Ignacio Clemente, Aina Brunet and Xavier Gonzalez-Compta in American Journal of Rhinology & Allergy
Footnotes
Acknowledgments
The authors would like to thank the Biostatistics Unit from IDIBELL, especially Judith Peñafiel, for the contributions to the statistical analyses that supported this study. They also thank the CERCA Programme/Generalitat de Catalunya for institutional support.
ORCID iDs
Ethics Considerations
The study was conducted in compliance with Declarations of Helsinki and Istanbul and the European Union Good Clinical Practice. All participants provided written informed consent for data collection and analysis, in accordance with EU Regulation 2016/679. Ethical approval was granted from the Ethics Committee of Bellvitge University Hospital (protocol code EOM024/23), and subsequently from the ethics committees of the two other participating centers (Hospital Universitari Germans Trias i Pujol and Hospital Universitari Mútua de Terrassa).
Authors’ contributions
MG performed conceptualization, data curation, investigation, methodology, study design, methodology, writing the original draft preparation, revision and editing.
PCT did conceptualization, supervision, methodology, writing the original draft, revision and editing.
ALR performed conceptualization, data curation, and writing the original draft.
CGL, LPM, and IC did conceptualization, data curation and investigation, writing, revision and editing of the manuscript.
ABG performed conceptualization, investigation, and writing the original draft.
XGC did conceptualization, supervision, methodology, investigation, writing the original draft, revision and editing.
All authors have read and approved the final version of the article before submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG, PCT, LMP, and IC have received honoraria for consultancy and conferences from Sanofi and GlaxoSmithKline (GSK). ALR has received honoraria for conferences from Sanofi and GlaxoSmithKline (GSK). CGL and AGB have no conflict of interest. XGC has received honoraria for consultancy, conferences or clinical trials from AstraZeneca, Aldo, Sanofi, GlaxoSmithKline (GSK), Merck and Novartis.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
