Abstract
Background
Frontal sinus surgery remained a challenge of restenosis or obliteration of the drainage pathway caused by the scarring and neo-osteogenesis after mucosal stripping and bone drill-out. The pedicled or free nasal mucosal flap is typically used to repair the exposed bone surface to avoid or reduce recurrence.
Objective
This study aimed to explore the histopathological mechanism of mucosal flaps repairing bare bone after mucosal resection and bone drill-out in the rabbit model.
Methods
Thirty New Zealand white rabbits were used. Sixteen rabbits were selected as the experimental group, and Staphylococcus aureus was used to establish the CRS model (CRS group). Fourteen healthy rabbits were allocated to the control group (NCRS group). Each group was divided into two subgroups with or without mucosal flap repair (CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP, respectively). The bony anterior and lateral walls of the maxillary sinus of each rabbit were abraded by the drill. The bare bone was then covered with a flap in FLAP subgroups. Bone remodeling and mucosal morphological changes were observed and compared by histopathological hematoxylin and eosin and Masson staining.
Results
In the CRS-NFLAP subgroup, the regenerated epithelium lacked typical structure, accompanied by numerous inflammatory cell infiltration and collagen deposition. Conversely, the inflammatory reaction was mild in the CRS-FLAP subgroup, and there was less collagen deposition. The restored mucosal structure was like the normal mucosa. The epithelium in the NCRS-NFLAP subgroup was partially exfoliated, with few cilia, goblet cells, and glandular structures. Compared with the NCRS-NFLAP subgroup, the CRS-NFLAP subgroup showed significant bone remodeling with enhanced activity of osteoblast and osteoclast cells.
Conclusions
Pedicled mucosal flap repair could significantly reduce local mucosal and bone remodeling in a rabbit model of CRS.
Keywords
Introduction
Draf IIb and Draf III frontal sinus surgeries, also known as frontal sinus drill-out procedures, have been instrumental in resolving complex and refractory frontal sinus diseases.1-3 The surgical approach is predominantly used in recurrent and refractory sinusitis cases, particularly chronic sinusitis with nasal polyps and asthma. 4 Furthermore, it is also applied to treat inverted papilloma, osteoma of the frontal sinus, and cerebrospinal fluid leakage. Using the drill-out procedure to an open frontal sinus aims to obtain a wide field of operation of the large sinus and establish an available postoperative drainage channel. 5
But in fact, there were some dilemmas in the frontal drill-out procedures, which included postoperative restenosis and neo-osteogenesis caused by mucosal striping and bone exposure. 6 Moreover, the existing neo-osteogenesis in sinusitis has been shown to be a risk factor for postoperative sinus restenosis. 6 In 2007, Tran et al. 7 reported that the restenosis rate of the frontal sinus was 33%, and the degree of stenosis exceeded 60% in 28.6% of patients. Therefore, the exposed bone surface was recommended to be covered with a free mucosal flap to reduce sinus restenosis or obliteration. In 2012, Hildenbrand et al. 8 found that the frontal sinus ostium could operate at 94% of the usual maximum efficiency with a free nasal septal mucosal flap after 25.6 months of follow-up. In contrast, the frontal sinus ostium is reportedly markedly compromised when a mucosal flap is not used. 9 In addition, pedicled mucosal flaps of the middle turbinate and the nasal septum have also been reported to repair the exposed bone surface of the frontal recess, which can reduce the formation of new bone and mucosal scarring, thus effectively reducing the restenosis rate of the frontal sinus ostium.10,11 Although the clinical outcomes were satisfactory, the specific mechanisms behind the inhibition of tissue remodeling after applying mucosal flaps need further study. From this point of view, we performed an animal experiment using a rabbit model of sinusitis to observe the tissue remodeling of sinus bone after grinding injury repaired with or without mucosal flap histopathologically.
Materials and Methods
Experimental Animals and Groups
Appropriate IRB approval from the Experimental Animal Ethics Association of Capital Medical University was obtained for this study (AEEI-2018-227). Thirty New Zealand white rabbits were purchased from Beijing Long'an Experimental Animal Breeding Center [Certificate No: SCXK2019-0006]. There were no specific requirements regarding the sex of these rabbits, and their weight varied from 2.5 to 2.7 kg. Sixteen rabbits were established the CRS model (CRS group). Fourteen healthy rabbits were allocated to the control group (NCRS group). Each group was divided into two subgroups with or without mucosal flap repair (CRS-FLAP, n = 8, CRS-NFLAP, n = 8), NCRS-FLAP, n = 7 and NCRS-NFLAP, n = 7, respectively).
Experimental Modeling
Sinus inflammation and congenital maxillofacial bone malformation were excluded by 64-slice spiral sinus CT (PHILIPS Brilliance, matrix 512 × 512) on rabbits before modeling (Figure 1A). The suspension was derived from the international standard Staphylococcus aureus strains (No. ATCC 25923) and was made with normal saline at a 108 CFU/mL concentration. General anesthesia was induced using 2.5% pentobarbital sodium administered through an auricular vein. A 1 cm incision was made on the anterior lateral wall of the maxillary sinus, and then the subcutaneous tissue was separated into the sinus lumen. A 0.3 mL suspension of Staphylococcus aureus was injected into the maxillary sinus under a nasal endoscope. Finally, the sinus ostium was closed using a cotton ball, and the incision was sutured (Figure 1C). CT showed a soft tissue density shadow in the maxillary sinus four weeks later, indicating successful modeling after surgical treatment (Figure 1B and D).
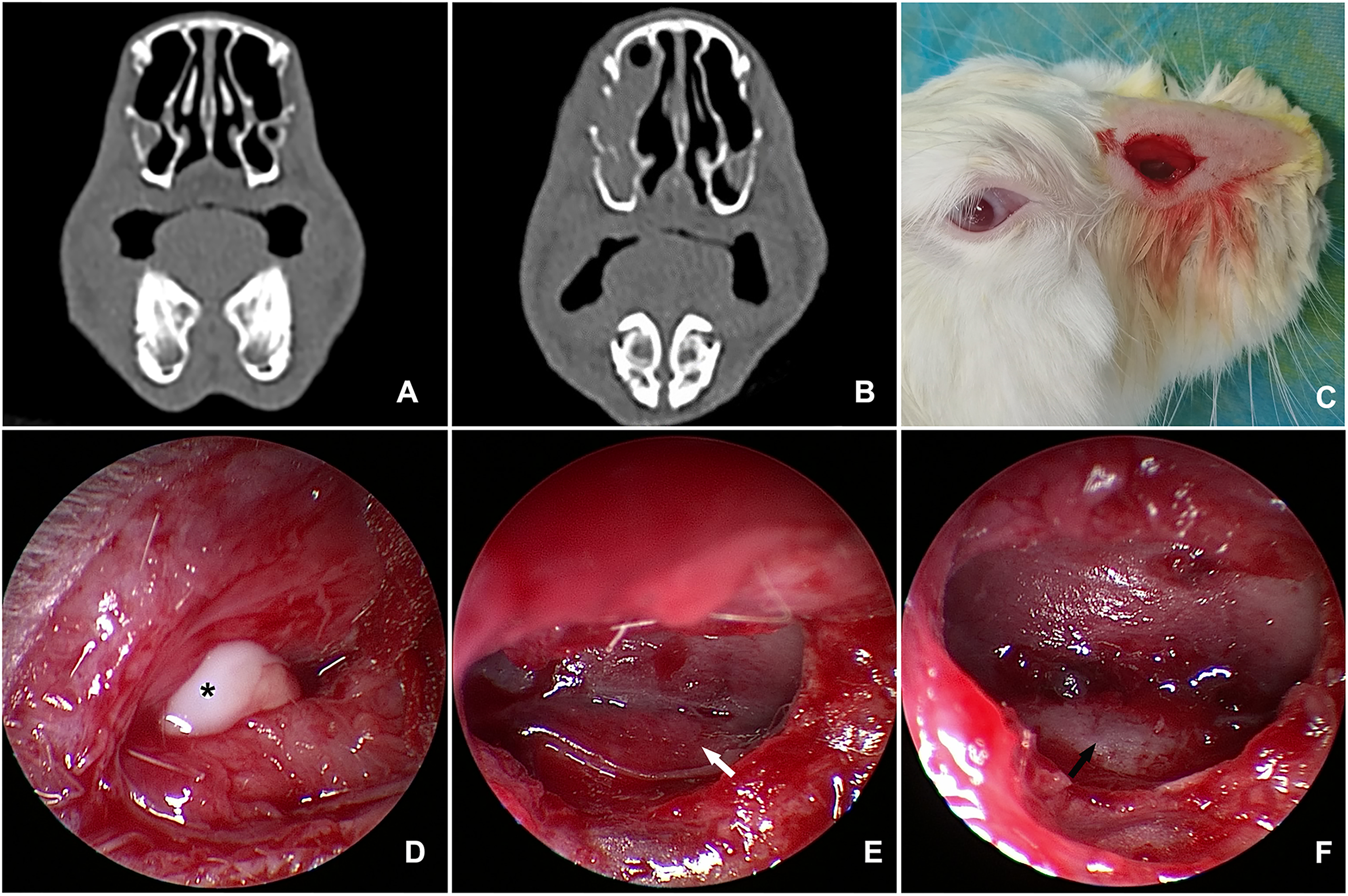
(a) Head CT (horizontal position) of experimental rabbits before modeling; (b) Head CT (horizontal position) of experimental rabbit after modeling; (c) Diagram of external incision and surgical approach in experimental rabbits; (d) The maxillary sinus was entered through the approach shown in Figure 1C. *: White purulent discharge in the sinus; (e) Pedicled mucosal flap was retained after bone drill-out (white arrow); (f) Mucosal flap was not retained after bone drill-out, and the exposed bone surface was visible (black arrow).
A 10 mm skin incision was made on the anterolateral side of the head of the experimental rabbits, and the subcutaneous tissue was separated. The 2.7 mm endoscope was introduced into the sinus from the boneless area of the anterolateral side of the maxillary sinus. The mucosa of the anterolateral wall was elevated and turned forward with an elevator, and about 10 mm long pedicled mucosal flap in front was made. After we ground the local denuded bone (Strong 90®, Nanchang, China, cutting bur, diameter = 2.35 mm, 18,000 RPM), the mucosal flap was repositioned to cover the bare bone or resected in the FLAP (Figure 1E) and NFLAP (Figure 1F) subgroups, respectively. The experimental rabbits were euthanized 8 weeks after the operation, and their heads were cut. The site of bone grinding was identified, and a midline incision was made. After the tissue samples from the maxillary sinus were collected, they were fixed using 10% neutral formaldehyde, decalcified, embedded in paraffin, and sequentially sliced using a microtome (4 μm). Six samples were randomly selected from each group; three were subjected to hematoxylin and eosin (HE) and Masson staining, respectively.
Dyeing Index Measurement
Mucosal thickness was defined as the distance between the mucociliary epithelium and the periosteum, including the epithelium, basement membrane, and lamina propria. Mucosal thickness was measured using Image J software (Public Domain, 1.53J). Semi-quantitative measurement of the collagen fiber area in the blue-stained area was performed using Image J software, and the average value of the three fields was taken. Professional pathologists determined the grading of all sections.
Bone Grade Scoring Criteria
The bone grade was determined as follows: Grade 0: normal bone, no apparent bone hyperplasia or destruction; Grade 1: mild periosteal thickening without obvious bone hyperplasia or destruction; Grade 2: moderate periosteal thickening and the presence of osteoblasts, osteoclasts, and bone absorption or remodeling or both; Grade 3: moderate to severe periosteal thickening, increased activity of osteogenic and osteoclast cells, bone absorption or remodeling or both, and formation of osteoid and adhesive lines; and Grade 4: white blood cells appear, causing osteomyelitis and bone destruction. 12
Statistical Analysis
We used SPSS 25.0 software for statistical analyses. The bone remodeling grade, mucosal thickness, and subepithelial collagen fiber area percentage were compared in pairs among the four subgroups using the Mann–Whitney U rank-sum test. A P-value of <.05 was considered statistically significant.
Results
Comparison of Mucosal Morphology among the Four Subgroups
HE stains found that the epithelium of the regenerated mucosa was absent in the CRS-NFLAP subgroup. In contrast, the epithelium in the NCRS-NFLAP subgroup was partially exfoliated, with few cilia, goblet cells, and glandular structures. However, in CRS-FLAP and NCRS-FLAP subgroups, the restored mucosal structure was similar to that of the normal mucosa. Furthermore, unlike in CRS-FLAP and NCRS-NFLAP subgroups, in the CRS-NFLAP subgroup, the glandular structures disappeared. Many lymphocytes and plasma cells infiltrated the lamina propria, and tissue hyperplasia of collagen fibers increased. However, the inflammatory response in FLAP groups was milder. The pathological characteristics of mucosal morphology are shown in Table 1 and Figure 2.
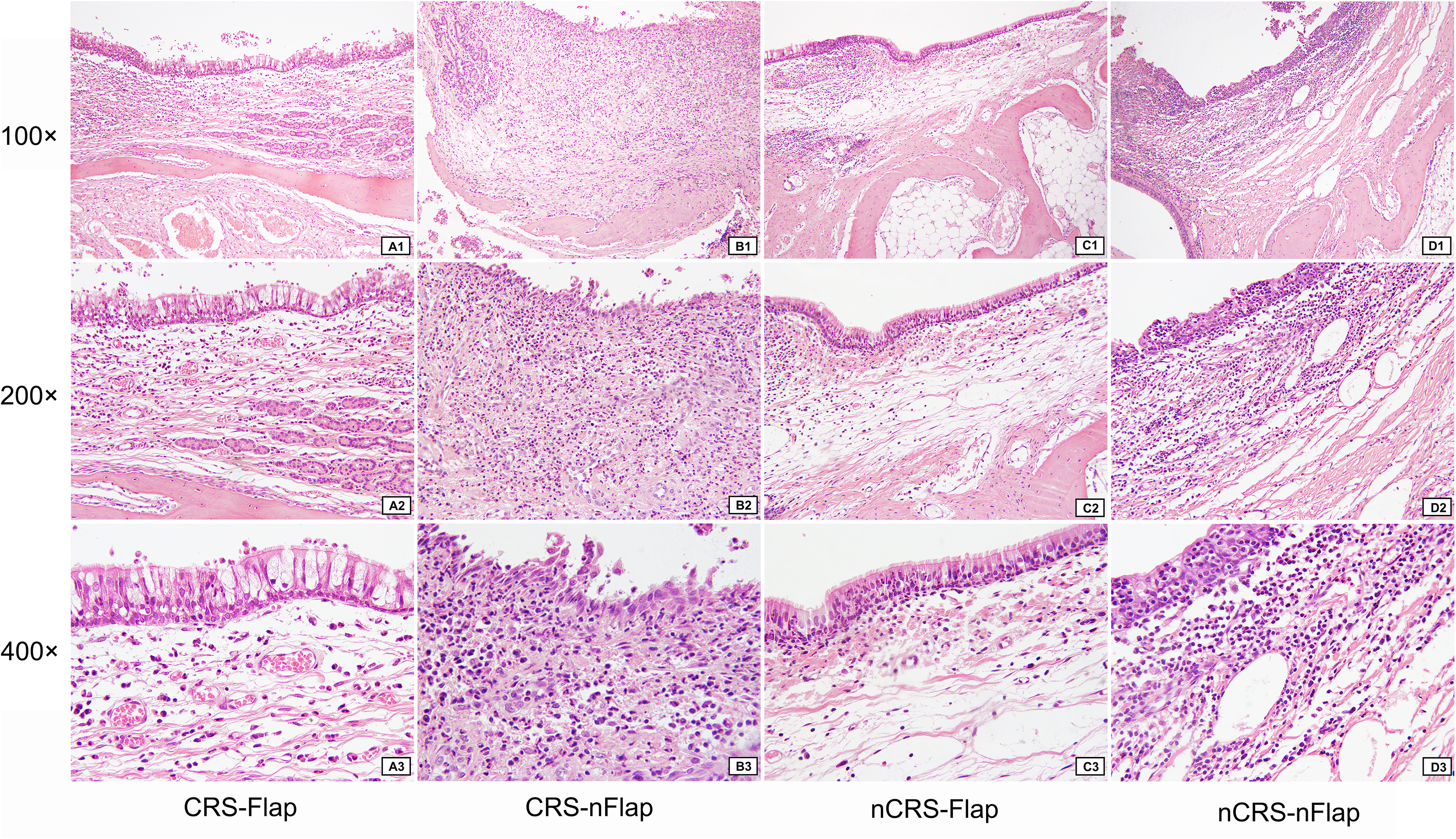
The morphology of repaired mucosal epithelium in the four subgroups at the maxillary sinus drill-out site (HE stains). Panels (a–d) represent CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP, respectively. 1–3 illustrates 100 × , 200 × , and 400 × magnification, respectively.
Histopathological Characteristics in the Four Subgroups.

The mucosal thickness of epithelialization at the drill-out site of the maxillary sinus was 256.55 ± 116.66 μm, 380.76 ± 95.01 μm, 182.72 ± 17.54 μm, and 241.78 ± 51.13 μm, in the CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP subgroups, respectively. The comparison analysis demonstrated that there was a statistical difference between CRS-FLAP and CRS-NFLP subgroups (P = .04) and a significant statistical difference between the CRS-NFLAP and the NCRS-NFLAP subgroup (P = .004) (Figure 3).
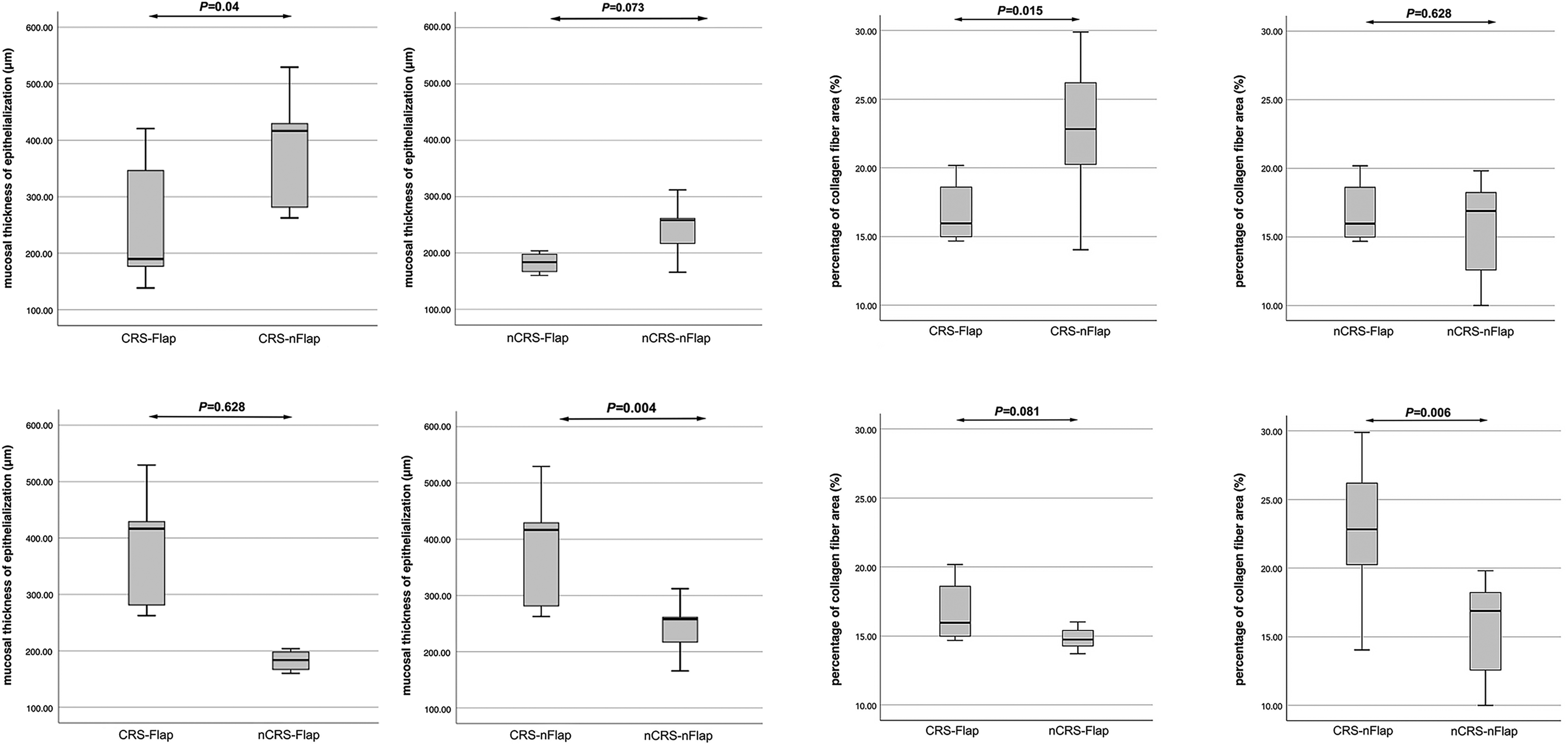
Comparison of the mucosal thickness of epithelialization and percentage of collagen fiber area.
Comparison of Collagen Distribution among Four Subgroups
Masson staining found more fibers in CRS groups than in normal groups (Figure 4). Semi-quantitative measured results showed that the percentage of collagen fiber area in the four subgroups was as follows: 16.74 ± 2.09, 22.80 ± 4.83, 14.81 ± 0.83, and15.47 ± 3.88, in the CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP subgroups, respectively. There was a statistical difference in proportion between CRS-FLAP and CRS-NFLAP subgroups (P = .015). And the proportion in the CRS-NFLAP was also higher than that in the NCRS-NFLAP subgroup (P = .006) (Figure 3).
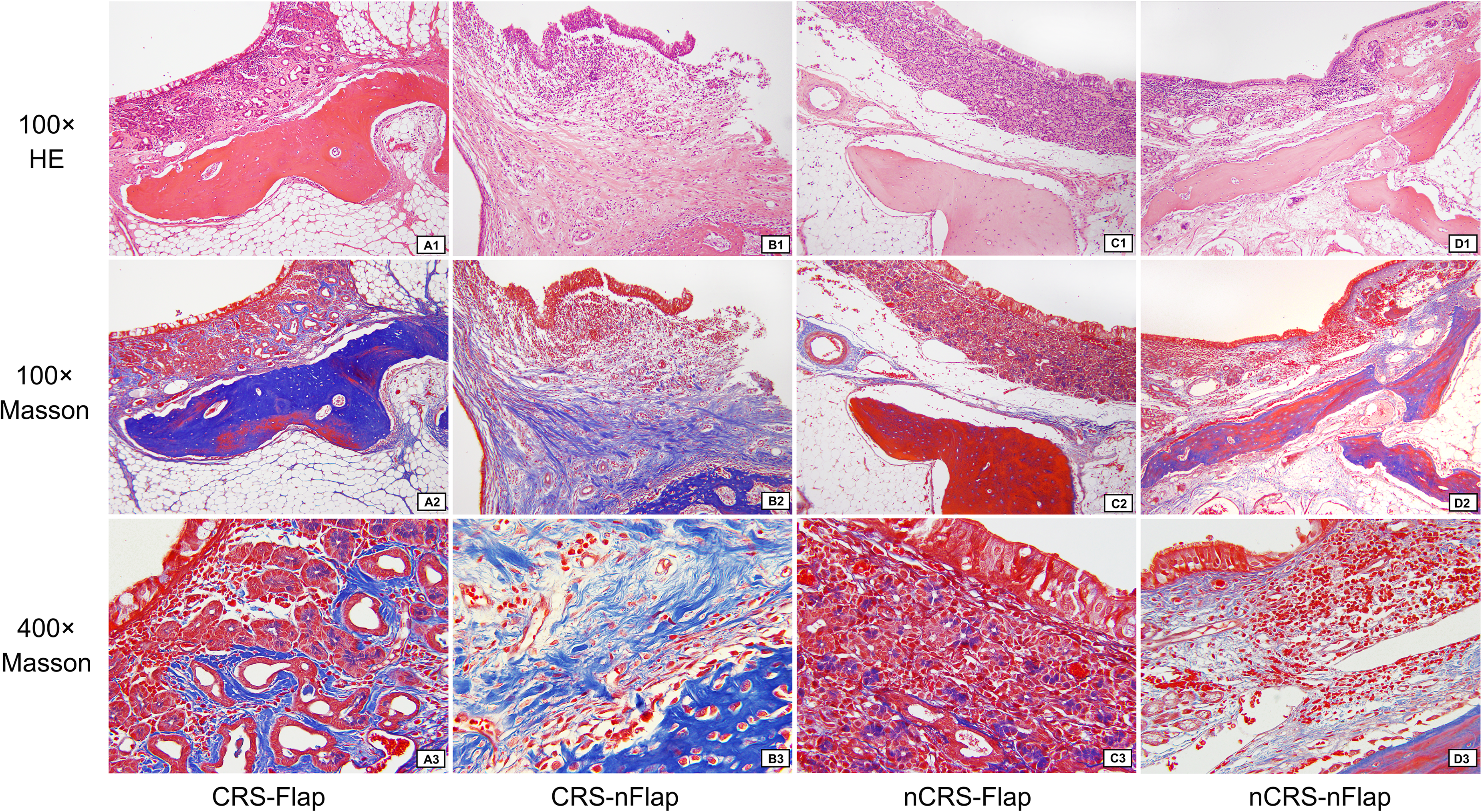
The distribution of submucosal collagen fibers in the four subgroups at the maxillary sinus drill-out site (Masson staining). Panels (a–d) represent CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP, respectively. 1–3 represents 100 × magnification with HE staining, 100 × magnification with Masson staining, and 400 × magnification with Masson staining, respectively.
Comparison of Bone Morphology among the Four Subgroups
HE stains revealed evident bone remodeling in the CRS-NFLAP subgroup compared to other subgroups, with enhanced activity of osteogenic and osteoclast cells, increased number of osteoclasts, higher osteoid deposition, and adhesion line and woven bone formation (Figure 5). Furthermore, the bone grade statistically significantly differed between the NCRS-NFLAP subgroup and the CRS-NFLAP subgroup (P = .014) and between the CRS-FLAP subgroup and the CRS-NFLAP subgroup (P = .038); however, it did not significantly differ between the CRS-FLAP subgroup and the NCRS-FLAP subgroup (P = .281) (Table 2).
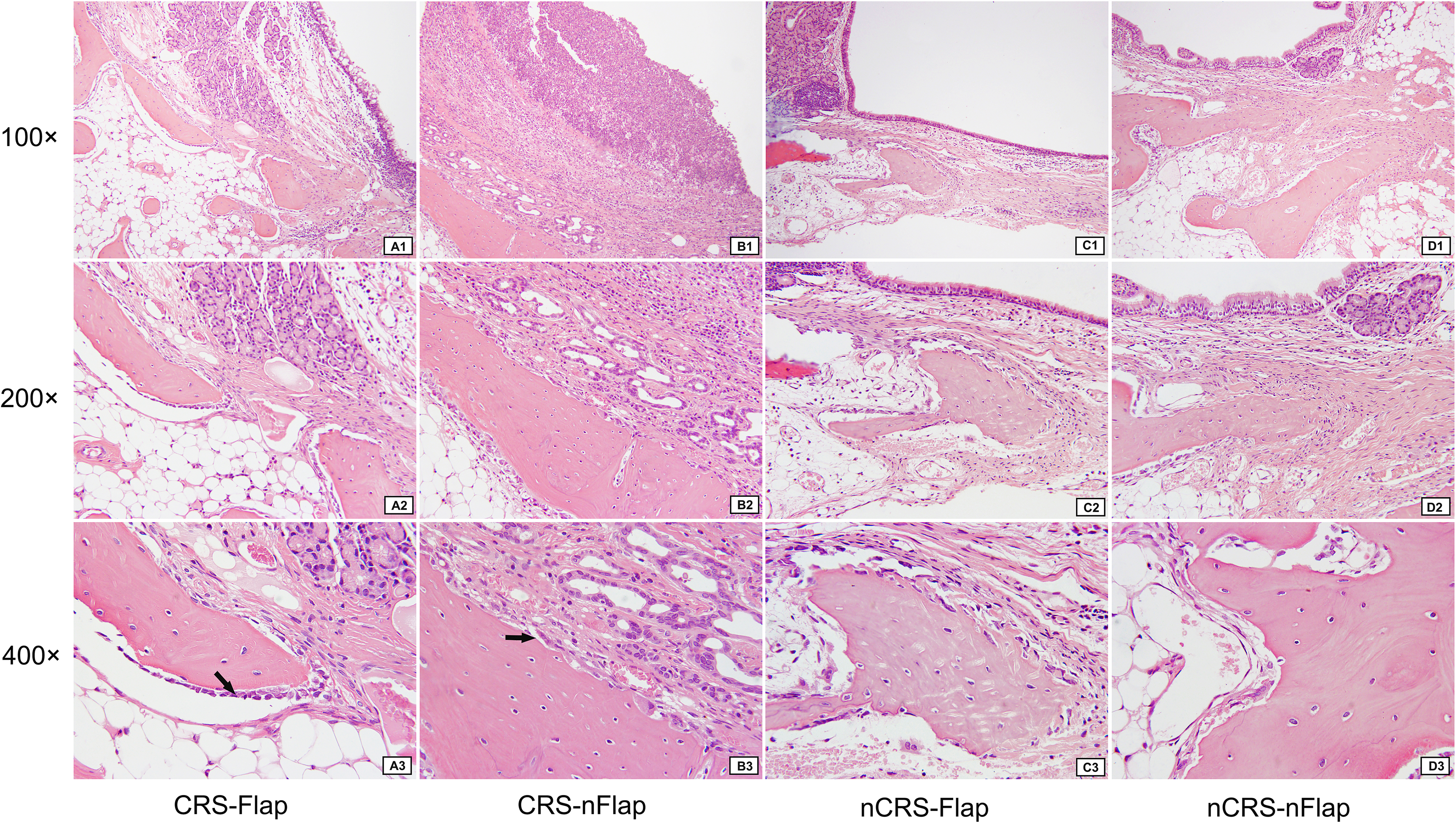
Histopathological morphology of the bone in the four subgroups at the maxillary sinus drill-out site (HE stains). Panels (a–d) represent CRS-FLAP, CRS-NFLAP, NCRS-FLAP, and NCRS-NFLAP, respectively. 1–3 illustrates 100 × , 200 × , and 400 × magnification, respectively. A3: Black arrow: osteoblasts in rows; B3: Black arrow: Osteoblasts and osteoclasts with markedly enhanced activity. The number of osteoblasts increased significantly.
Comparison of Bone Histopathological Grading After Maxillary Sinus Drill-out in the Four Subgroups of Experimental Animals.

Mann–Whitney U-test analysis for bone grading in each group: A versus B: P = .038*; C versus D: P = .71; A versus C: P = .281; B versus D: P = .014*.
*P < .05, the difference was statistically significant.
Discussion
Since Draf 13 proposed frontal sinus surgery classification in 1991, the surgical technique has continued to develop and has gained widespread acceptance. Draf IIb and Draf III procedures reportedly offered a high success rate, low recurrence and reoperation rates, and better long-term disease control. 14 Our study evaluated the potential benefits of mucosal flaps on maintaining mucosal integrity and reducing neo-osteogenesis in a rabbit model. Therefore, we conducted a preliminary investigation into the pathological mechanism in which the mucosal flap may have an effect of inhabitation on bone remodeling.
Theoretically, following mucosal tissue injury caused by various factors such as inflammation or trauma, a tissue remodeling reaction occurs, including an increase in goblet cells, thickening of the basement membrane, an increase in inflammatory cells, and proliferation of mucosal collagen fibers. The structure of the mucosal epithelium formed during tissue repair after loss of the bone surface's mucosa is disordered. 15 At 8-week post-injury in the CRS-NFLAP subgroup, the bone surface covered by the remodeling mucosal epithelium lacked mucosa and exhibited minimal cilia, goblet cells, and glands (Figure 2). Simultaneously, a more pronounced inflammatory response was observed with increased infiltration of lymphocytes and plasma cells into the lamina propria of the remodeling mucosa. However, in the NFLAP subgroup of the NCRS subgroup, although the new mucosal epithelium was also observed, a smaller number of cilia, goblet cells, and gland structures were present (Figure 2), and the inflammatory reaction was mild. The inflammatory response in the NCRS-FLAP subgroup was even lighter, indicating that inflammation is an influencing factor for epithelial remodeling and repair.
In addition to the above observation of basic mucosal structure, this study assessed histomorphological changes by measuring mucosal thickness after mucosal repair. The results indicated that in the NFLAP group, the remodeling mucosa's thickness was significantly greater in the CRS subgroup than in the NCRS subgroup (P = .004), suggesting that sinusitis may exacerbate the proliferative reaction of the mucosal remodeling process.
When comparing the subgroups of Flap and NFLAP in the CRS group, a statistically significant difference was found with a P-value of .04. This confirms the role of inflammation, as stated above. However, in the Flap group, whether in the CRS subgroup or the NCRS subgroup, the complete mucoperiosteum-containing mucosal flap was used to repair the exposed bone surface. This resulted in an autologous mucosal epithelium transplantation effect rather than remodeling the mucosal epithelium of the wound. The restored mucosal epithelium structure was similar to normal mucosa, as Figures 2, 4, 5 show. Comparison between the groups resulted in a P-value of .628. The difference was not statistically significant, suggesting that applying a mucosal flap can play a role in wound repair by inhibiting tissue remodeling and having a structural basis for mucociliary clearance due to its intact mucosal epithelial structure. These results demonstrate the important role of mucosal flap in wound repair.
The Masson staining of collagen fibers revealed that the CRS group had a higher proportion of collagen fibers in the lamina propria than the healthy control group (NCRS group), along with more inflammatory cell infiltration, and the CRS-NFLAP subgroup had a higher proportion of collagen fibers (P = .015) (Figures 3 and 4).
The CRS-NFLAP subgroup was found to have more collagen fibers in the lamina propria compared to the healthy subgroup (P = .006). However, there was no significant difference in the histopathological observation of the collagen fiber area in the lamina propria between the CRS-Flap and the NCRS-FLAP (P = .081) (as shown in Figure 3). This suggests that, on the one hand, the mucosal flap may have a potential inhibitory effect on mucosal tissue remodeling; on the other hand, it also implies that existing inflammation is an essential factor exacerbating tissue remodeling.
The histopathological bone score also demonstrated that the CRS subgroup had a significantly higher bone score than the NCRS subgroup in the NFLAP subgroup, with a statistically significant difference (P = .014). This finding is consistent with our previous study on rabbit sinusitis animal models, which suggested that sinus inflammation is directly proportional to the extent of bone remodeling.16,17 Notably, mucosal loss can stimulate an inflammatory response, which may exacerbate the bone remodeling reaction. Nevertheless, there was no significant difference in histopathological bone score between CRS and NCRS subgroups in the Flap group (P = .281), indicating that the mucosal flap exerted a substantial inhibitory effect on bone remodeling.
Conclusions
This animal study suggests that bone remodeling following bone drilling is characterized by mucosal scarring and new bone formation, with mucosal loss being the primary cause. Mucosal loss can elicit a more pronounced inflammatory response, while inflammatory factors may exacerbate the remodeling reaction. Repairing exposed bone surfaces with a mucosal flap can help prevent tissue remodeling after the procedure and prevent sinus ostium stenosis or atresia. This mechanism may be related to the contact inhibition phenomenon observed during tissue repair. 18 Furthermore, preserving the complete mucosal flap provides a basis for restoring mucociliary clearance function. It is important to note that this is an animal experimental observation study. Further, clinically controlled studies are required to validate the efficacy of the mucosal flap in preventing or reducing sinus stenosis or atresia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Special Fund of the Pediatric Medical Coordinated Development Center of Beijing Hospitals Authority, The Capital Health Research and Development of Special, Talent Training Fund of Beijing Tongren Hospital, National Natural Science Foundation of China, (grant number XTYB201825, 2020-1-2051, 2019-YJJ-ZZL-032, 81770977 and 82171107)
Zoology Ethics
This study was approved by the Experimental Animal Ethics Association of Capital Medical University (ethics approval number: AEEI-2018-227). New Zealand white rabbits were purchased from Beijing Long'an Experimental Animal Breeding Center [Certificate No: SCXK2019-0006].
