Abstract
Background
Studies evaluating health-related quality of life (HRQOL) in patients with hereditary hemorrhagic telangiectasia (HHT) have expanded rapidly in the past decade. These studies have evaluated QOL aspects ranging from the general QOL for patients living with HHT to intervention-specific outcomes. However, few tools have been fully validated across the spectrum of disease manifestations and interventions in HHT.
Objective
In this scoping review, we aim to map the literature on HHT-QOL metrics, identify gaps, inform future QOL research, and facilitate future metric development.
Methods
We analyzed articles in English that assessed at least 1 measure of general HRQOL, including physical health, mental health, social health, or intervention-specific QOL in patients with HHT. Searches across 2 bibliographic databases (PubMed and Scopus) yielded 186 articles after duplicates were removed. Sixty-three studies met eligibility criteria: 22 prospective studies (34.9%), 20 retrospective studies (31.7%),
Results
The
Conclusions
Comparison of results across studies remains challenging given the heterogeneity in outcomes measures. Further development of HHT-specific patient-reported outcomes instruments that capture the global illness experience of HHT is needed.
Keywords
Introduction
Hereditary hemorrhagic telangiectasia (HHT) is a genetic disorder with an autosomal dominant inheritance pattern that has a prevalence of 1 in 5000 to 8000 individuals. 1 It is characterized by arteriovenous malformations (AVMs) and telangiectasias with associated bleeding-related complications throughout the body, including but not limited to the nose, skin, lungs, liver, gastrointestinal tract, and central nervous system.2–4 HHT affects persons of all races, ethnicities, genders, and ages indiscriminately. 5 Individuals with severe forms of HHT have a limited selection of therapeutic choices, and even with treatment, on average their life-expectancy is 3 years shorter than that of the general population. 6 The manifestations of HHT lead to significant morbidities, such as chronic blood loss (from chronic epistaxis or gastrointestinal bleeding), intracranial abscesses, and acute hemorrhage from the nose or gastrointestinal tract. 7 Patients with HHT also frequently experience heightened psychological, social, and occupational stress as a consequence of their disease. 8 The complex and heterogenous presentation of HHT makes the evaluation of the disease's impact on patients’ quality of life (QOL) challenging.
Patient-reported outcome measures (PROMs) and QOL instruments are needed to evaluate the illness experience of HHT and the effectiveness of HHT treatments. These tools enable clinicians to comprehend HHT's physical, psychological, and social impacts on their patient's QOL from the patient perspective. Healthcare professionals can increase treatment efficacy, identify areas for improvement, and adapt interventions to individual patients by integrating PROMs and QOL instruments into clinical trials and everyday care. The tools enable patients to actively participate in their care by expressing their concerns and priorities to healthcare personnel. PROMs and QOL tools are thus essential for HHT research and patient care.
In recent years, as medical research has increasingly emphasized health-related QOL (HRQOL), researchers have worked to better understand the experience of those living with HHT.8–12 However, the literature describing the effect of HHT on individuals’ physical health, mental health, and social functioning, the effect of specific symptoms on QOL, and intervention-specific QOL is still incomplete. Furthermore, the inconsistent use of different outcome measures makes comparisons between studies difficult. Comprehensive and accurate QOL tools for evaluating current treatment methods for patients with HHT are needed, and these tools may motivate improvements in care pathways for individuals with HHT. Without assimilated evidence, it remains unclear where further research is needed. Therefore, the purposes of this scoping review were to (1) summarize the HRQOL literature related to HHT, (2) identify gaps in the literature, and (3) recommend steps for future research.
Methods
Literature Review Process
We conducted our scoping review using the Arksey and O’Malley methodological framework. 13 PubMed and Scopus, 2 bibliographic databases, were queried for studies using keywords relevant to QOL, patient-reported outcomes, and HHT (see Supplemental Material). The timeframe of the final list of publications was 2004 to 2022. Two independent reviewers screened abstracts and full texts. A total of 186 unique articles were identified in the initial search, and 69 studies were selected for full-text review. After full-text screening, we selected 63 articles that described QOL metrics in individuals with HHT. At the 14th HHT International Scientific Conference in October 2022, we identified 2 additional articles, which made a total of 65 articles (Figure 1).
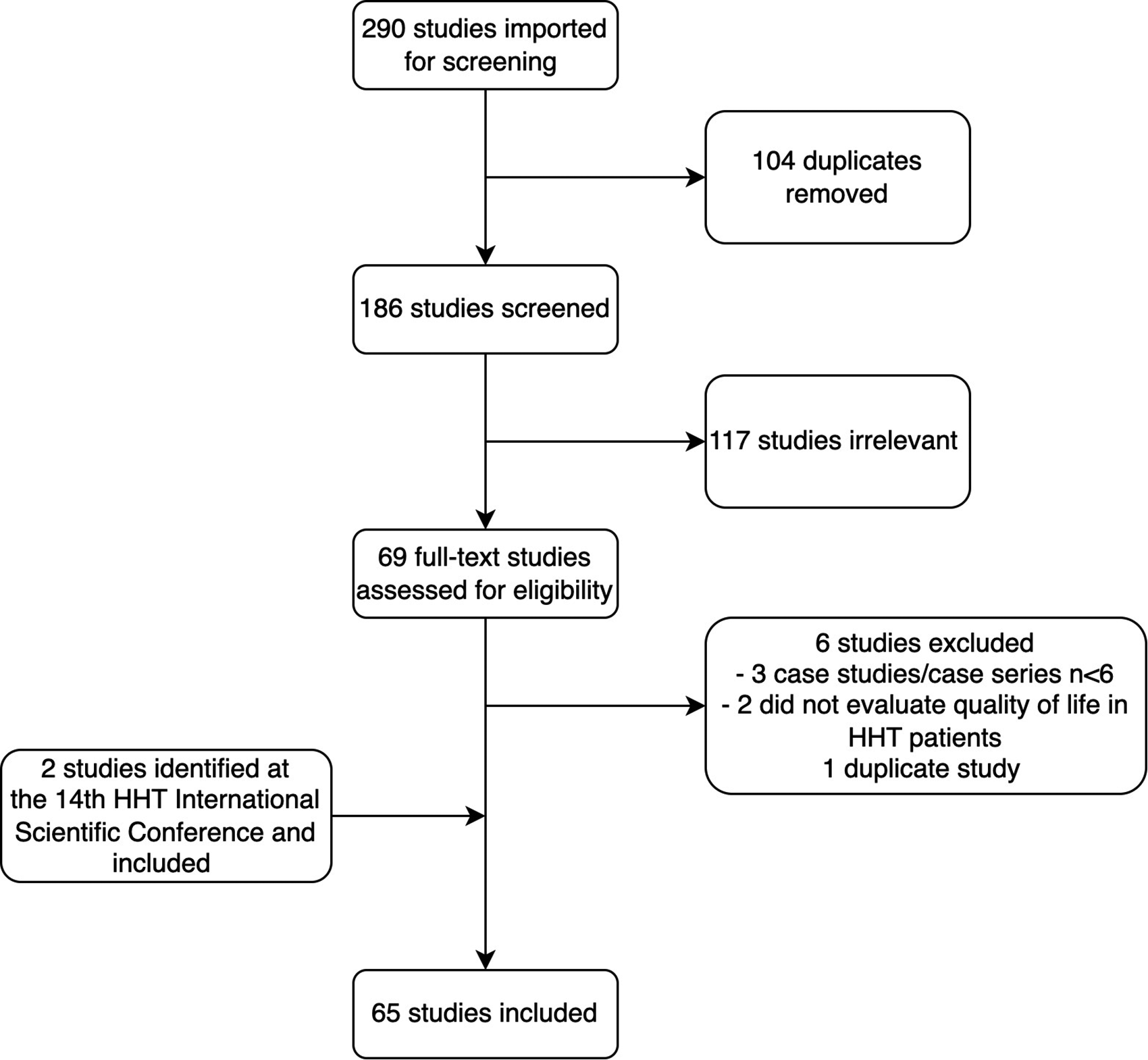
Flowchart demonstrating literature search and study selection.
Data Abstraction
Data abstraction was performed between August 2021 and October 2022. Data collected from each of the eligible articles included first author, year, study country, sample size, study design, aims of study, study type, outcomes measured, and instruments used to assess each outcome (Tables 1 and 2). Instruments included any validated tools, custom-made instruments/questionnaires, and numerical scales that were reported in the 65 articles.
65 Studies Describing Use of Quality of Life Metrics in Patients with HHT, 2004-2022.
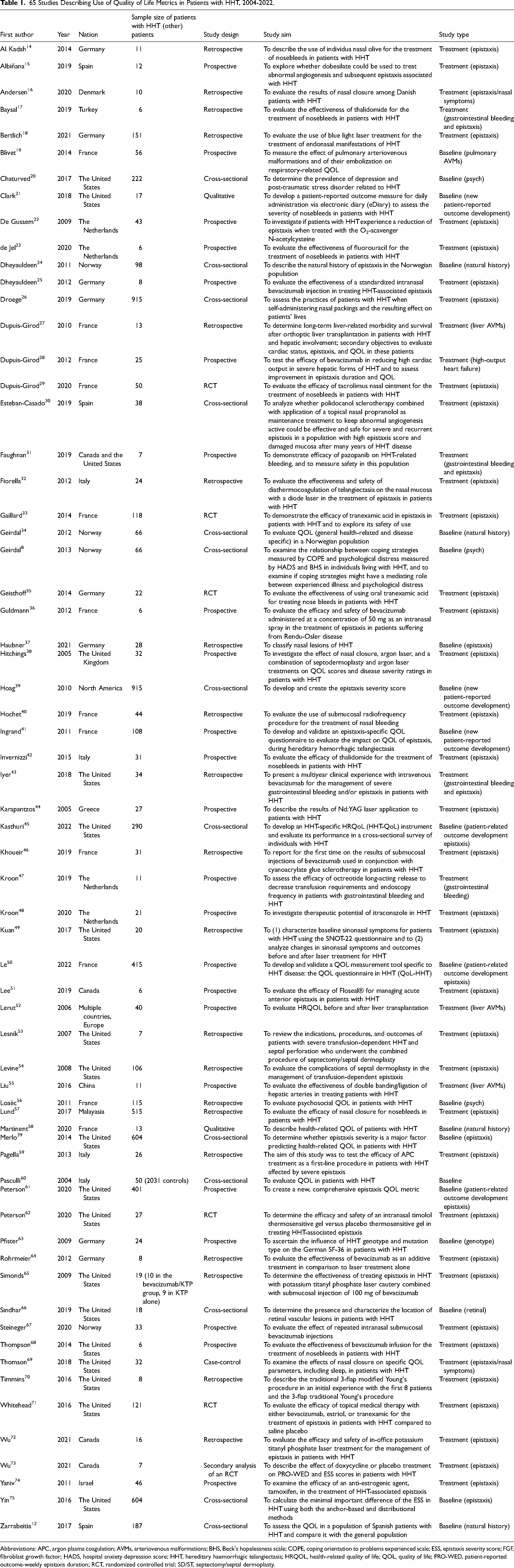
Abbreviations: APC, argon plasma coagulation; AVMs, arteriovenous malformations; BHS, Beck's hopelessness scale; COPE, coping orientation to problems experienced scale; ESS, epistaxis severity score; FGF, fibroblast growth factor; HADS, hospital anxiety depression score; HHT, hereditary haemorrhagic telangiectasia; HRQOL, health-related quality of life; QOL, quality of life; PRO-WED, patient-reported outcome-weekly epistaxis duration; RCT, randomized controlled trial; SD/ST, septectomy/septal dermoplasty.
Health-Related Quality of Life Patient-Reported Outcome Measures in the Hereditary Hemorrhagic Telangiectasia Literature.
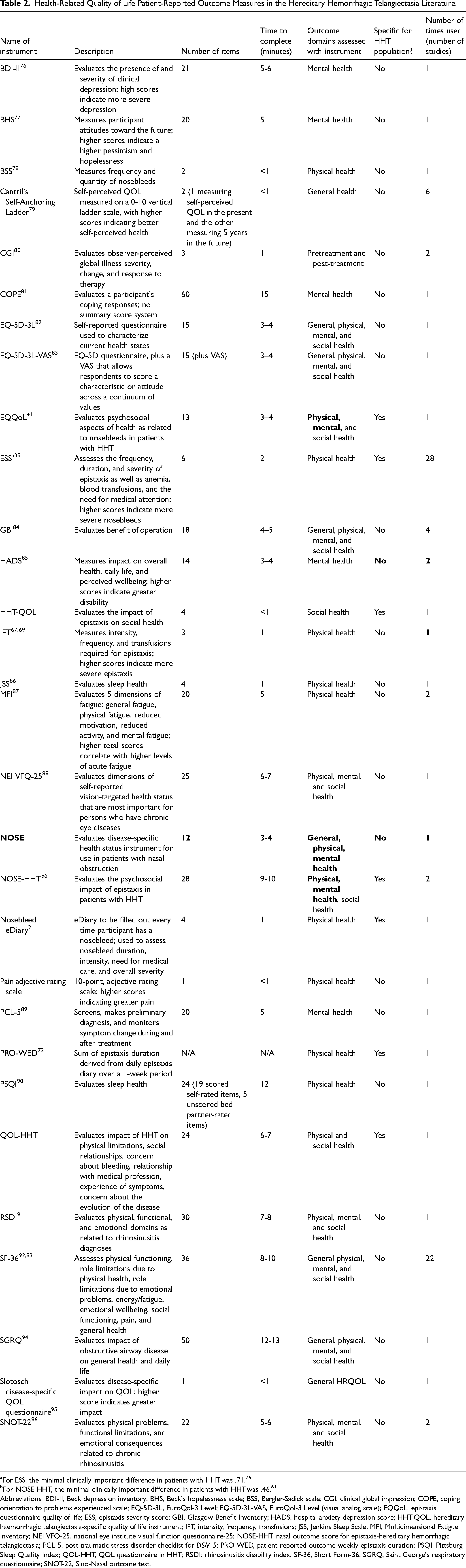
For ESS, the minimal clinically important difference in patients with HHT was .71. 75
For NOSE-HHT, the minimal clinically important difference in patients with HHT was .46. 61
Abbreviations: BDI-II, Beck depression inventory; BHS, Beck's hopelessness scale; BSS, Bergler-Sadick scale; CGI, clinical global impression; COPE, coping orientation to problems experienced scale; EQ-5D-3L, EuroQol-3 Level; EQ-5D-3L-VAS, EuroQol-3 Level (visual analog scale); EQQoL, epistaxis questionnaire quality of life; ESS, epistaxis severity score; GBI, Glasgow Benefit Inventory; HADS, hospital anxiety depression score; HHT-QOL, hereditary haemorrhagic telangiectasia-specific quality of life instrument; IFT, intensity, frequency, transfusions; JSS, Jenkins Sleep Scale; MFI, Multidimensional Fatigue Inventory; NEI VFQ-25, national eye institute visual function questionnaire-25; NOSE-HHT, nasal outcome score for epistaxis-hereditary hemorrhagic telangiectasia; PCL-5, post-traumatic stress disorder checklist for DSM-5; PRO-WED, patient-reported outcome-weekly epistaxis duration; PSQI, Pittsburg Sleep Quality Index; QOL-HHT, QOL questionnaire in HHT; RSDI: rhinosinusitis disability index; SF-36, Short Form-36; SGRQ, Saint George's respiratory questionnaire; SNOT-22, Sino-Nasal outcome test.
We defined 5 outcome domains that were assessed in QOL studies. Three of them—physical health, mental health, and social health—were in line with the PROMIS adult self-reported health domains. 97 Social health was defined as social functioning (i.e., the ability to participate and satisfaction with social roles), including but not limited to work and social relationships. 98 Two additional domains, general HRQOL and intervention-specific QOL, were also added. We defined intervention-specific QOL as QOL directly related to an intervention experience and/or its outcomes. Each instrument reported in the collected studies was assigned to 1 or more of the 5 outcome domains (Table 3). Some instruments satisfied criteria for multiple domains.
QOL and Patient-Reported Outcome Measures by Domain.
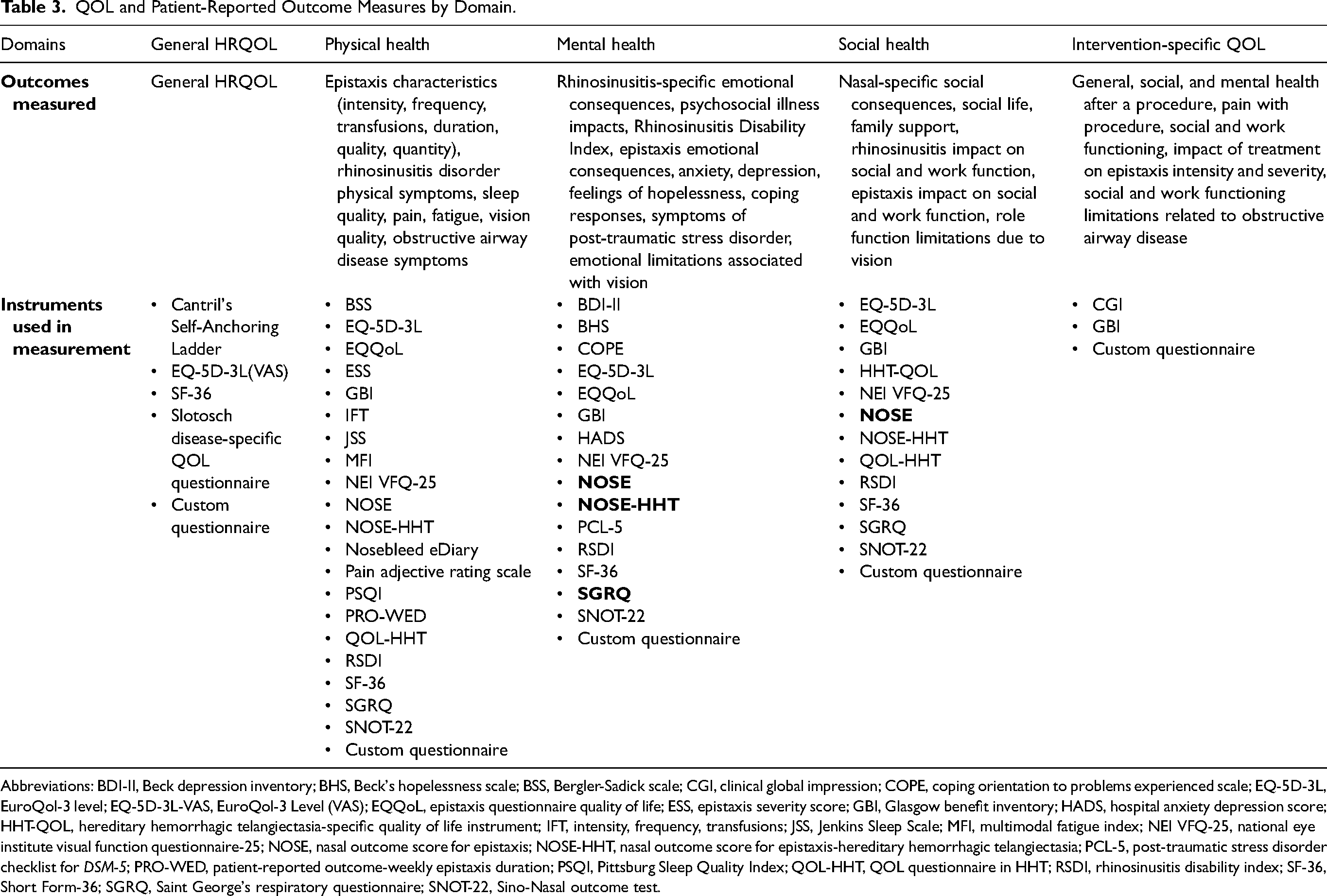
Abbreviations: BDI-II, Beck depression inventory; BHS, Beck's hopelessness scale; BSS, Bergler-Sadick scale; CGI, clinical global impression; COPE, coping orientation to problems experienced scale; EQ-5D-3L, EuroQol-3 level; EQ-5D-3L-VAS, EuroQol-3 Level (VAS); EQQoL, epistaxis questionnaire quality of life; ESS, epistaxis severity score; GBI, Glasgow benefit inventory; HADS, hospital anxiety depression score; HHT-QOL, hereditary hemorrhagic telangiectasia-specific quality of life instrument; IFT, intensity, frequency, transfusions; JSS, Jenkins Sleep Scale; MFI, multimodal fatigue index; NEI VFQ-25, national eye institute visual function questionnaire-25; NOSE, nasal outcome score for epistaxis; NOSE-HHT, nasal outcome score for epistaxis-hereditary hemorrhagic telangiectasia; PCL-5, post-traumatic stress disorder checklist for DSM-5; PRO-WED, patient-reported outcome-weekly epistaxis duration; PSQI, Pittsburg Sleep Quality Index; QOL-HHT, QOL questionnaire in HHT; RSDI, rhinosinusitis disability index; SF-36, Short Form-36; SGRQ, Saint George's respiratory questionnaire; SNOT-22, Sino-Nasal outcome test.
Results
Study Designs of Eligible Studies
Among the 65 eligible articles, there were
Outcomes Assessed by Study Objective
Twenty-nine pre-existing QOL instruments were used in the studies (Table 2). In addition, custom instruments were developed and used in 18 studies. Thirty-four studies used more than 1 instrument.
Outcomes Assessed by Domain
Among the 63 quantitative studies, 56 (88.9%) assessed physical health,
Measurement Properties of Instruments Validated for HHT.

Abbreviations: BHS, Beck's hopelessness scale; COPE, coping orientation to problems experienced scale; EQQoL, epistaxis questionnaire quality of life; ESS, epistaxis severity score; HADS, hospital anxiety depression score; HHT-QOL, hereditary hemorrhagic telangiectasia-specific quality of life instrument; NOSE-HHT, nasal outcome score for epistaxis-hereditary hemorrhagic telangiectasia; QOL-HHT, QOL questionnaire in HHT.
General Health-Related Quality of Life
General HRQOL was assessed in 31 studies (50.8%).17–19,23,25,27–32,34,37,38,41,43,44,46–48,55,60–64,67,68,74,75,100 Among them, 4 pre-existing instruments were used (Table 3). None of the 4 were fully validated in an HHT population. The Short Form-36 (SF-36) (
Physical Health
Physical health was assessed in 56 studies (88.9%).12,14–17,19,22–32,34–44,46–51,53–55,57,59–75,100 Twenty pre-existing instruments were used, and 15 studies used custom instruments.22,24,32,34,35,38,39,42,50,53,54,57,59,65,71 The Epistaxis Severity Score (ESS) was the most used instrument (28; 50.9%), followed by SF-36 (
Mental Health
Mental health was assessed in
Social Health
Social health was assessed in 39 studies (61.9%).12,16,17,19,22,23,25,27–29,31,34,38,41,44,45,47–50,52,53,55–57,60–63,66–68,70,74,75,100 Among them, 12 pre-existing instruments were used, and 7 studies used custom instruments. The SF-36 (22; 57.9%) was the most used tool, followed by the GBI (4; 6.4%), the NOSE-HHT (2; 5.1%), and the SNOT-22 (2;
Intervention-Specific QOL
Intervention-specific QOL was assessed in 13 studies (21.3%).14,16,26,33,46,51,53,54,57,61,62,64,70 The GBI (4; 30.8%) was the most used, followed by the Clinical Global Impression (CGI) scale (2; 15.4%). Seven studies used custom instruments. None of the instruments used in 13 studies were validated in individuals with HHT.
Discussion
This scoping review summarizes the multidimensional outcome measures currently used for evaluating HRQOL in individuals with HHT. Patient-centered care relies on patients’ perspective on their disease as a whole and the changes in manifestations and experience of the disease following treatment and interventions.101–103 Use of pre-existing QOL metrics and patient-reported outcomes is critical to collecting this data in a systematic and reproducible fashion.104–106 Currently, there are many custom outcomes used as measures of QOL in people with HHT. This heterogeneity in QOL outcomes makes selecting appropriate outcome measures and comparing across studies challenging.107,108
In this scoping review, the data were organized into the PROMIS domains of physical health, mental health, and social health.97,109 We found that critical aspects of QOL in individuals with HHT are not captured within these PROMIS domains; therefore, we decided to also include both a general QOL domain and an intervention-specific QOL domain. Physical health was the most frequently assessed domain, with epistaxis being the most often assessed symptom. The evaluation of the physical health domain was likely more common because physical complaints are more readily reported by patients and more easily elicited by physicians in a clinical encounter compared to complaints about mental health and social functioning.110–113 Additionally, patients with HHT may predominantly report physical concerns. 114 Mental health and social health were the next most frequently evaluated QOL domains. HHT is a chronic illness, which can lead to significant stress over time and contribute to mental distress; however, accurate and reproducible assessment of mental health can be difficult.12,20,115,116 In our literature search, we found that anxiety and depression were assessed often. Social health investigations included social support and role limitations. In this review, although most studies evaluated pretreatment and post-treatment QOL, only 5 studies were RCTs. RCTs are widely regarded as the gold standard of study design, and more such studies are needed to improve our understanding of changes in pretreatment and post-treatment QOL. 117
Although studies used a wide variety of outcome measures, most used generic measures or disease-specific questionnaires not specifically designed for or validated in patients with HHT. Several studies developed custom, nonvalidated measures that were then used for analysis. Ideally, validated instruments specific to the domain(s) under investigation, rather than one-off, nonvalidated instruments, should be used. For instruments that assess many domains, a clear categorization of scores by domain would enable the examination of multiple domains independently. Five of the validated outcome measures currently available for assessing patients with HHT focus on assessing the impact of epistaxis on HRQOL (Table 2), which is logical given that epistaxis may be the only presenting symptom of HHT and it has a prevalence of >90% among patients with HHT. However, these epistaxis-specific measures fall short of capturing the full breadth of disease-related symptoms that individuals with HHT frequently experience. More investigation is needed to expand the scope of HRQOL outcome measures for individuals with HHT. To achieve this, there is a need for content elicitation and validation to identify the full range of symptoms affecting patients with HHT. We believe that this scoping review is a crucial step toward expanding validated outcome measures for individuals with HHT.
A 2022 study examined more than 400 patients with HHT at 20 specialist centers in France. The investigation led to the creation of a 24-item questionnaire with high-quality content validity and psychometric analysis, including tests of construct validity and reliability. 50 This is the first holistic HHT-QOL measure that focuses on the illness experience beyond epistaxis and includes the following evidence-based domains: physical limitations, social relationships, concern about bleeding, relationship with the medical professional, experience of symptoms, evolution of the disease. To date, the questionnaire has only been tested in French, and neither a minimal clinically significant difference nor clinical relevance has been identified. Further psychometric validation in a diversity of populations is needed.
Our study has some limitations. By keeping our search terms broad and using 2 large bibliographic databases, we attempted to capture all studies that evaluated HRQOL or patient-reported outcomes in patients with HHT; however, it is possible that studies were omitted inadvertently. Further, only studies published in the English language were included on analysis, thus biasing our results to those studies published outside English-speaking countries.
This review highlights the current heterogeneity in QOL metrics and patient-reported outcomes used to measure HRQOL in patients with HHT. We identified 31 unique instruments and cataloged them under 5 domains. Only 7 of these instruments (ESS, EQQOL, Nosebleed eDiary, PRO-WED, NOSE-HHT, HHT-QOL, and QOL-HHT), 6 of which focus on the impact of epistaxis on QOL, were validated in patients with HHT. Although epistaxis is seen in most individuals with HHT, the disease has many other manifestations because of its impact on multiple organ systems. The clinical features and symptoms include but are not limited to gastrointestinal bleeding, symptoms due to visceral AVMs, and neurological dysfunction secondary to brain AVMs. This review can serve as a reference for clinicians and researchers seeking guidance in choosing outcome HRQOL instruments for patients with HHT. Importantly, we want to emphasize the need for validation of pre-existing instruments in the HHT population around the world and for development of tailored, disease-specific instruments in multiple languages that target symptoms beyond epistaxis. Overall, the heterogeneity of outcome measures used in the HRQOL literature makes it challenging to compare results across studies and select appropriate metrics for individuals with HHT. Development and validation of a core set of outcome measures tailored to the complex clinical reality of HHT is urgently needed to advance research aimed at improving patient-centered outcomes and QOL for patients living with HHT.
Supplemental Material
sj-docx-1-ajr-10.1177_19458924231207123 - Supplemental material for Health-Related Quality of Life Outcome Measures in Individuals With Hereditary Hemorrhagic Telangiectasia: A Scoping Review
Supplemental material, sj-docx-1-ajr-10.1177_19458924231207123 for Health-Related Quality of Life Outcome Measures in Individuals With Hereditary Hemorrhagic Telangiectasia: A Scoping Review by Anna J. Gong, Tushar Garg, Adham Khalil, Prateek C. Gowda, Stephen C. Mathai, Nicholas R. Rowan, Christian A. Merlo and Clifford R. Weiss in American Journal of Rhinology & Allergy
Footnotes
Acknowledgements
For their editorial assistance, including submission on the authors’ behalf, we thank Sandy Crump, MPH, Denise Di Salvo, MS, and Rachel Box, MS, in the Editorial Services group of The Johns Hopkins Department of Orthopaedic Surgery.
Declaration of Conflicting Interests
The authors would like to declare the following conflict of interest with respect to the research, authorship, and/or publication of this article: Unrelated funding from Cure HHT.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
