Abstract

Introduction
The diffusing capacity of the lung for carbon monoxide (DLCO) provides an important measurement of the transfer of gas from inhaled air to the bloodstream. Accurate DLCO measurements are essential for diagnosing and managing lung diseases. 1 Although American Thoracic Society/European Respiratory Society (ATS/ERS) standards 2 define acceptable maneuvers, the impact of common errors beyond slow or submaximal inhalation, variable breath-hold times, changes in intrathoracic pressure (exceeding ± 40 to 50 cm H2O 3 ), and improper alveolar sampling remains under characterized. We prospectively simulated seven errors to quantify their directional effects on DLCO, alveolar volume (VA), and carbon monoxide transfer coefficient (KCO).
Methods
This study employed a within-subject experimental design and enrolled three experienced pulmonary function technologists, each acting as their own control. The UCSF Institutional Review Board approved the study (approval number 23-38743) and the subjects provided informed consent. Each error’s three trials with two associated control efforts were performed in separate sessions for each error type. Single-breath DLCO followed ATS/ERS standards, including the four-minute minimum between efforts. The equipment to measure DLCO was manufactured by nSpire. The test gas composition was 0.3% CO, 0.3% CH4, 21% O2, and balance nitrogen. We simulated seven common errors observed clinically (Fig. 1): (1) inspiratory leak; (2) early inhale; (3) leak during breath-hold to <85% of vital capacity (VC), the threshold specified in the ATS/ERS technical standard as secondary criteria for acceptability and as grade B (VC was from forced spirometry efforts performed in the same session); (4) leak during breath-hold but maintenance of ≥85% of VC; (5) leak around the mouthpiece with a closed valve; (6) leak during exhalation; and (7) nonzeroed sensors. Primary outcomes were DLCO, VA, and KCO relative to the session’s control values. Given the small sample size, heterogeneous baselines between subjects, and expected variability within a subject and error type due to the inherent difficulty in replicating the error exactly, we report within-subject qualitative changes rather than inferential statistics.
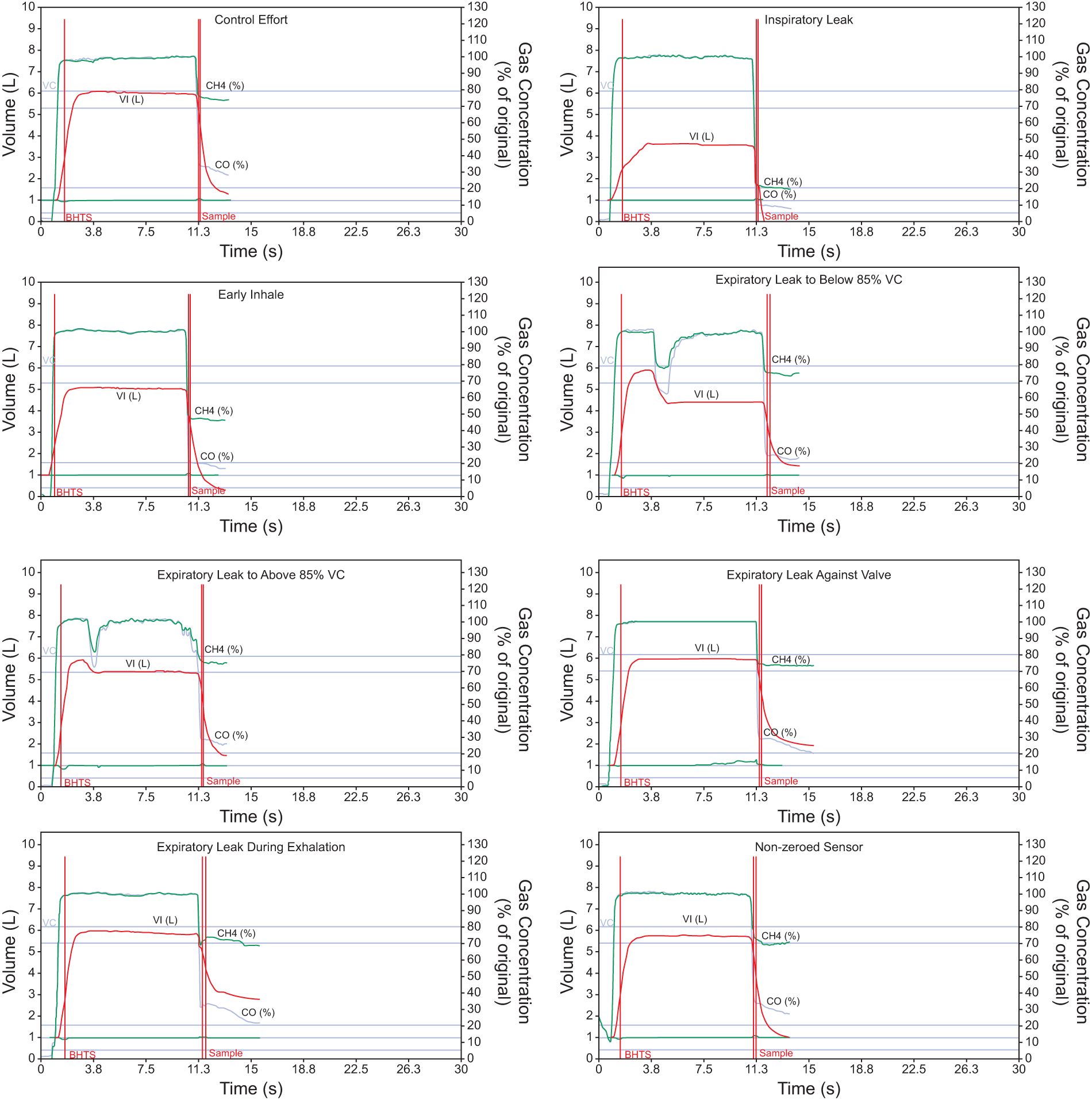
Representative single-breath DLCO tracings for all errors. Sampled concentrations of methane (CH4%, green) and carbon monoxide (CO%, blue) rise during inspiration, plateau over a ∼10-second breath-hold (starting at the vertical red line marking breath-hold start [BHTS]), then wash out on expiration. The red trace shows spirometry throughout the maneuver, with residual volume arbitrarily set at 1 L per manufacturer settings. The upper horizontal blue line labeled VC denotes the predicted threshold for 100% vital capacity obtained from forced spirometry testing, and the lower blue horizontal line represents 85% of VC (ATS/ERS Grade B). The thin green tracing at the 1.0 L Y axis indicates mouth pressure (two horizontal blue lines above and below the 1.0 L line indicate ±60 cm H2O). The left vertical axis is the volume of inspired air in liters; the right vertical axis is the gas concentration based on a percentage of the test gas concentration. The bold red lines on the right indicate the alveolar sample window.
Results
Errors clustered into three patterns (Fig. 2).
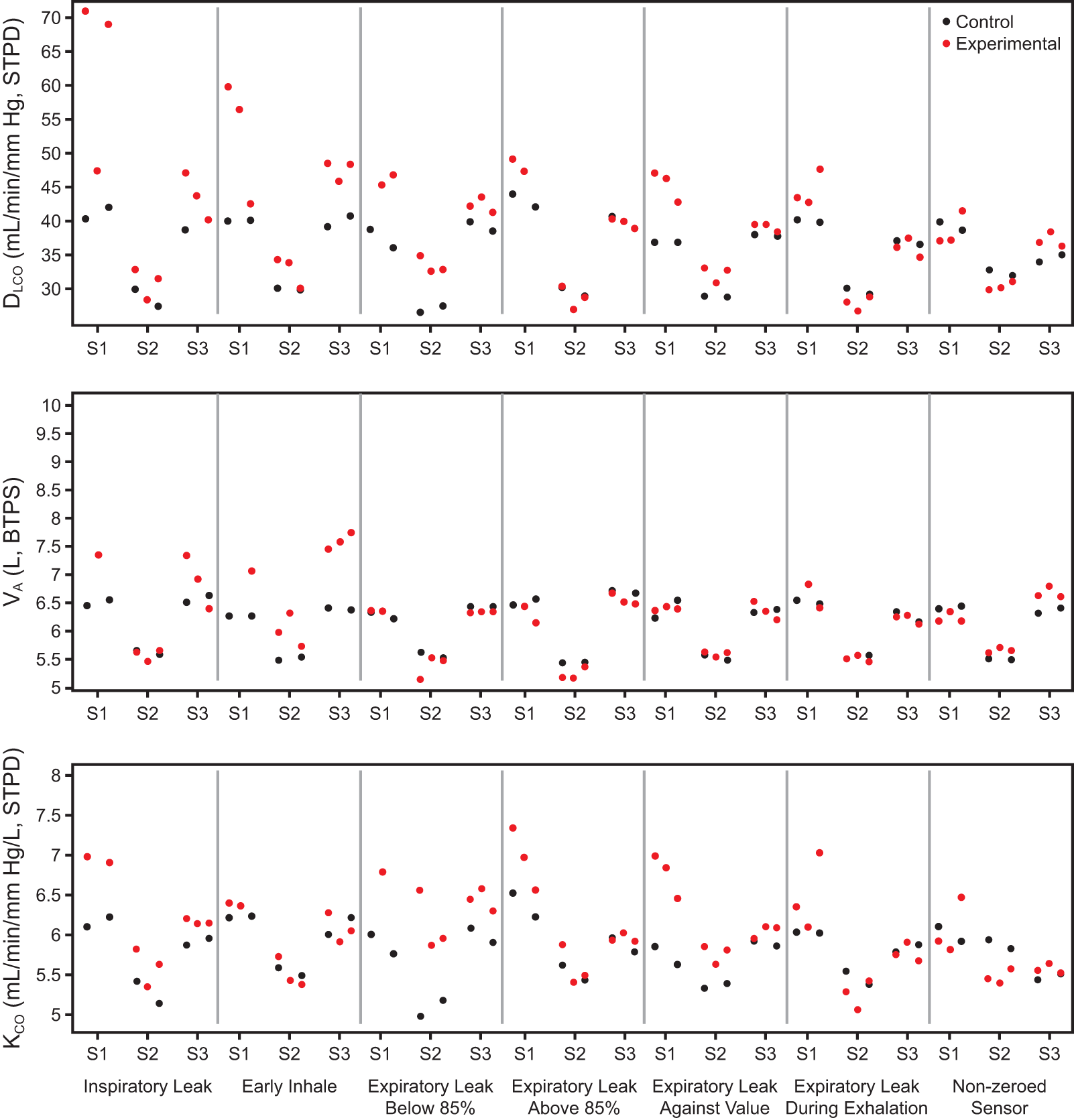
DLCO, VA, and KCO across all errors. Scatter plots display DLCO, VA, and KCO values for three subjects (S1–S3) across all tested error types (black: control efforts; red: experimental efforts). Shifts relative to control represent measurement artifacts introduced by the maneuver rather than changes in underlying lung function.
Increased DLCO due to dilution/volume artifacts: Inspiratory leak and early inhale generally increased DLCO, driven primarily by increases in VA; KCO increased slightly with an inspiratory leak and did not change consistently with an early inhale.
Increased DLCO due to loss of lung volume artifacts: Breath-hold leaks through the circuit to <85% of VC and leaks around the mouthpiece against a closed valve consistently increased DLCO with stable VA and increased KCO, consistent with the known volume dependence of KCO. 4
Breath-hold leaks with lung volume maintained at ≥85% of VC produced smaller, inconsistent DLCO shifts, typically with slight decreases in VA. Leaks during exhalation and nonzeroed sensors were variable across subjects and produced directionally inconsistent changes in all three metrics.
In brief, inspiratory leak and early inhale → ↑DLCO via ↑VA; breath-hold leak <85% VC and closed-valve leak → ↑DLCO via ↑KCO; other errors inconsistent.
Discussion
Our simulations show that commonly observed deviations in pulmonary function testing can systematically inflate DLCO through two mechanistic archetypes. First, dilution/volume artifacts through inspiratory leaks and early inhalation primarily increase VA with little change in KCO, yielding higher DLCO (DLCO = KCO × VA). Second, loss of lung volume artifacts through breath-hold leaks to <85% VC and leaks against a closed valve leave VA similar or slightly lower but raise KCO, again inflating DLCO. The magnitude of DLCO inflation exceeds reported inter-session variability. 5 Errors that preserved lung volume at ≥85% VC, leaks during exhalation, and nonzeroed sensors produced small and directionally inconsistent shifts that have the potential to be device- and subject-dependent.
During an inspiratory leak or early inhale, ambient air mixes with the test gas before the alveolar sample is drawn. CO and CH4 concentrations measured in the alveolar sample decrease as external air dilutes these gases, falsely indicating gas mixing in a larger lung volume. In addition, an inspiratory leak underestimates the inspired volume (VI) as measured by the spirometer. The net impact of a low measured VI with CH4 dilution was an increase in the VA. CO continues to diffuse during the breath-hold while CH4 remains inert, producing a larger fall in alveolar CO than CH4, an increased CH4:CO ratio in the alveolar sample, and consequently a higher KCO. In practice, this yields an increased DLCO with unchanged VA and unchanged or increased KCO.
Exhalation to <85% VC or loss of lung volume via leak with a closed valve (where the amount of volume lost is unknown) reduces alveolar volume. KCO is known to increase at lower lung volumes 3 due to proportionally greater recruitment of capillary blood volume and alveolar-capillary surface area relative to the loss of ventilated volume. The nSpire equipment in this study uses the highest inspiratory volume attained during the maneuver to calculate VA regardless of a subsequent decline in volume. Hence, VA is similar, but KCO increases, so DLCO increases. Exhalation to <85% VC accentuates this effect; when subjects remained ≥85% VC, changes were smaller and less consistent, aligning with the volume-dependence of KCO.
Because this study involved only three healthy technologists on a single manufacturer and model of equipment, with characteristics that may differ from modern systems, our observations should be considered hypothesis-generating. The nSpire equipment has a valve that is closed manually by operator trigger. In contrast, a system that automatically closes the valve will not lead to leaks with a decline in lung volume visible on the spirometry tracing, but there will still be a risk of a leak against a closed valve. These findings serve as useful educational tools for identifying quality-control issues rather than as generalizable conclusions for larger patient groups. Effects in patients with lung disease, different dead-space fractions, or altered hemoglobin may differ. Future work should quantify effect sizes across devices and patient phenotypes, and develop automated QC flags.
In conclusion, common technical errors can inflate DLCO through two predictable mechanisms: VA inflation (dilution) and KCO elevation at low volume. An elevated DLCO may generate concern 6 and a normal DLCO is generally reassuring, yet these data show that it can be artifactually high due to subtle errors. Teaching these subtleties and embedding them into quality control practices can prevent reporting or misinterpreting erroneous data.
Footnotes
Author Disclosure Statement
N.R.B. received a payment from Jaeger Medical for a one-time webinar on implementation of spirometry reference equations.
Funding Information
No funding was received for this article.
