Abstract
Background:
Breathing frequency is a sensitive indicator of a neonate patient’s condition and a crucial parameter in the neonatal ICU (NICU). However, conventional methods require sensors to be adhered to the newborn’s sensitive skin. This study assesses a noncontact respiratory monitor, using linear frequency modulation continuous wave radar, to determine the monitor’s accuracy when used in the NICU setting.
Methods:
An observational study was conducted on the noncontact respiration module produced by Mindray, connected to a BeneVisionN12 patient monitor. This unit collected data on breathing frequency; at the same time, the patient breathing frequency was also being measured using a CO2 monitor (accepted standard). Statistical analyses were performed to assess the agreement between the 2 measurements and to assess whether this noncontact monitor is sufficiently accurate for clinical use.
Results:
20 neonatal patients at 2 NICUs were enrolled in the study. The mean bias between the radar-derived and CO2-derived breathing frequency was −0.29 breaths/min (95% CI: −0.53 to −0.04); root-mean-square error was 2.60 breaths/min; concordance correlation coefficient was 0.98; and Pearson correlation coefficient was 0.98. Subgroup analyses grouped by body weight show that the technology has particularly high accuracy and clinical utility for neonates with low body weight (<1.5 kg).
Conclusions:
The noncontact respiration monitoring technology and module developed by Mindray showed a high degree of agreement with the CO2 reference standard and met the standard of accuracy required for clinical use.
Keywords
Introduction
Throughout the process of care, assessment, and treatment for neonates and preterm neonates, breathing frequency is one of the most important indicators for abnormalities in cardiopulmonary function, metabolic status, and the state of the respiratory system.1–3 This is particularly critical for preterm neonates, whose underdeveloped respiratory centers and immature lungs place them at high risk for respiratory disease. Therefore, continuous, real-time monitoring of breathing frequency is a cornerstone of care in the neonatal ICU (NICU), enabling the early detection of tachypnea, apnea, or other respiratory events.4,5 Such vigilance is crucial for supporting timely interventions, including those within the “golden hour” initiatives aimed at improving long-term outcomes. 6 Although automated monitoring is more objective and accurate than manual observation, current techniques in the NICU, including impedance pneumography (via electrocardiogram [ECG] electrodes) and capnography (measuring end-tidal CO2), require direct contact with the patient. These methods, particularly the use of electrodes, can cause medical adhesive-related skin injuries,7,8 skin lesions, or even obstruct circulation, 9 especially during extended monitoring of preterm neonates. Furthermore, patient movement often leads to poor connectivity and additional work load for the treatment team. 10 Although capnography is considered the accepted standard for accuracy, 11 it is used in patients who require continuous monitoring, and intubated patients receiving mechanical ventilation.
In recent years, developmentally supportive care has been recognized as an important model for neonate care. Supporting the healthy development of the body and nervous system in preterm neonates requires minimizing irritation from procedures and equipment while maintaining effective care. 12 Among these, radar has emerged as a particularly promising solution. Radar-based systems offer significant advantages: they are a zero-contact, noninvasive method, can penetrate materials such as acrylic incubator walls and blankets, and are unaffected by ambient lighting or the patient’s skin color.13,14 There have been some initial studies on the use of radar as a noncontact technology for monitoring neonates. Keisuke Edanami et al 15 used a noncontact medical-grade radar sensor in a feasibility study measuring heart rate and breathing frequency in healthy adults, and attempted to use the sensor to measure heart rate in 3 NICU infants. However, this trial did not include the measurement of breathing frequency in neonates, and included a very small patient group. Gabriel Beltrão et al 16 study 12 preterm neonates, with impedance-based measurement of breathing frequency as control. They also showed the feasibility of noncontact radar monitoring for the respiration of neonates in the NICU. However, their radar module requires its own stand and connection to a separate laptop computer. This significantly limits its utility in clinical settings. Won Hyuk Lee 17 included more neonates in the study group, but also compared the performance of the system against impedance monitoring of breathing frequency. The system tested also requires a separate stand and a connection to a laptop, which is a disadvantage for clinical use. As a whole, past studies have generally been lab simulations or clinical feasibility studies. These studies involve only a small number of subjects, and validation has been limited to benchmarking against the impedance-based method. Moreover, the equipment tested was not highly integrated, and its compatibility with clinical environments was low. To address this gap, the present study evaluates a new, highly integrated, noncontact respiration module developed by Mindray. This device is designed for seamless clinical workflow, as it can be placed directly on an incubator and connected to a standard bedside patient monitor. Therefore, the purpose of this trial was to rigorously validate the accuracy of this practical, noncontact system against the accepted-standard measurements from a CO2-based respirometer in the NICU setting.
QUICK LOOK
Current knowledge
Continuous breathing frequency monitoring is a cornerstone of care in the neonatal ICU (NICU). Standard methods require contact sensors, which can cause skin injury and interfere with developmentally supportive care. Noncontact radar technology exists as a promising alternative designed to mitigate these contact-related complications.
What this paper contributes to our knowledge
This study validated a highly integrated, noncontact respiratory monitoring module against the accepted-standard CO2-based measurement in a clinical NICU population. The noncontact technology demonstrated a high degree of agreement. The module also showed high accuracy in the subgroup of neonates with low body weight.
Methods
This study evaluated the performance of a noncontact respiratory monitoring module designed for use in the NICU. The module is placed on top of the incubator and operates without any physical contact with the neonate. Its core technology is based on linear frequency modulated continuous wave (LFMCW) radar. The fundamental principle of radar respiratory monitoring involves emitting an electromagnetic wave and analyzing the reflected signal. The periodic rise and fall of the neonate’s thoracic cage during breathing creates a small displacement (Δx), which in turn induces a proportional phase shift (Δφ) in the reflected wave, governed by the equation Δφ = 4πΔx/λ, where λ is the radar’s wavelength. The LFMCW radar leverages this principle to achieve a high signal-to-noise ratio and accurate motion detection. 18 In this system, the transmitted and received signals are mixed and filtered to produce a difference frequency signal, known as the intermediate frequency (IF) signal, which is then sampled by the hardware to generate the raw data. The subsequent process of extracting the respiratory waveform involves several key steps. First, a Fast Fourier Transform is applied to the IF signal to generate a range-time map, which identifies the specific “range bin” corresponding to the neonate’s chest. Next, the phase sequence from this target bin is extracted. Finally, this sequence undergoes arctangent demodulation and phase unwrapping to correct for phase ambiguities and reconstruct the continuous displacement curve, which represents the neonate’s respiratory waveform. This entire process allows for the precise, continuous, and discomfort-free measurement of breathing.
To assess the efficacy of this noncontact respiration module in a clinical setting, prospective studies were carried out between November 2023 and February 2024 in the neonatal departments at 2 hospitals. After obtaining informed consent, 20 neonates were enrolled in the trial to assess the efficacy of the noncontact respiration module in a clinical setting. The study was self-controlled, with the respiration rate measured by the noncontact monitor (radar-derived) compared against the respiration rate output of a CO2 respirometer (CO2-derived). The statistical analysis included mean bias, root-mean-square error (RMSE), and consistency measures to determine the accuracy of the noncontact monitor. This trial complies with the requirements of China’s Good Clinical Practice for Clinical Trials of Medical Devices (2022), was approved by the ethics committees of Guangdong Women and Children’s Hospital (2023-017) and Shenzhen Maternity & Child Healthcare Hospital (SZFYGCPLS2023014), and was registered at clinicaltrials.gov (ChiCTR2300077892).
The subjects for this trial were pediatric neonates. Informed consent was obtained from both legal guardians of all candidates and they were screened for eligibility. Any candidate not meeting an eligibility criterion was excluded. A total of 20 infant subjects were ultimately enrolled. Demographic information was collected for every enrolled subject (D.O.B., sex, body weight, length, age, twin/single birth, type of delivery, stay in hospital).
Patients were excluded if their clinical condition made the collection of continuous data difficult (patient not in a stable condition, potentially requiring resuscitation, or requiring frequent medical intervention). Enrolled subjects were monitored for as long as they underwent CO2 monitoring. The module was placed or attached by suction cups to the top of the neonate incubator, directly above the upper body of the subject. Diapers, clothing, blankets, ECG electrodes, connecting wires, and other objects on the chest of the subject did not affect the measurement. The module was connected to the patient monitor (Mindray BeneVision N12); when the capnography waveform stabilized and a breathing frequency was displayed, data collection began (Fig. 1).
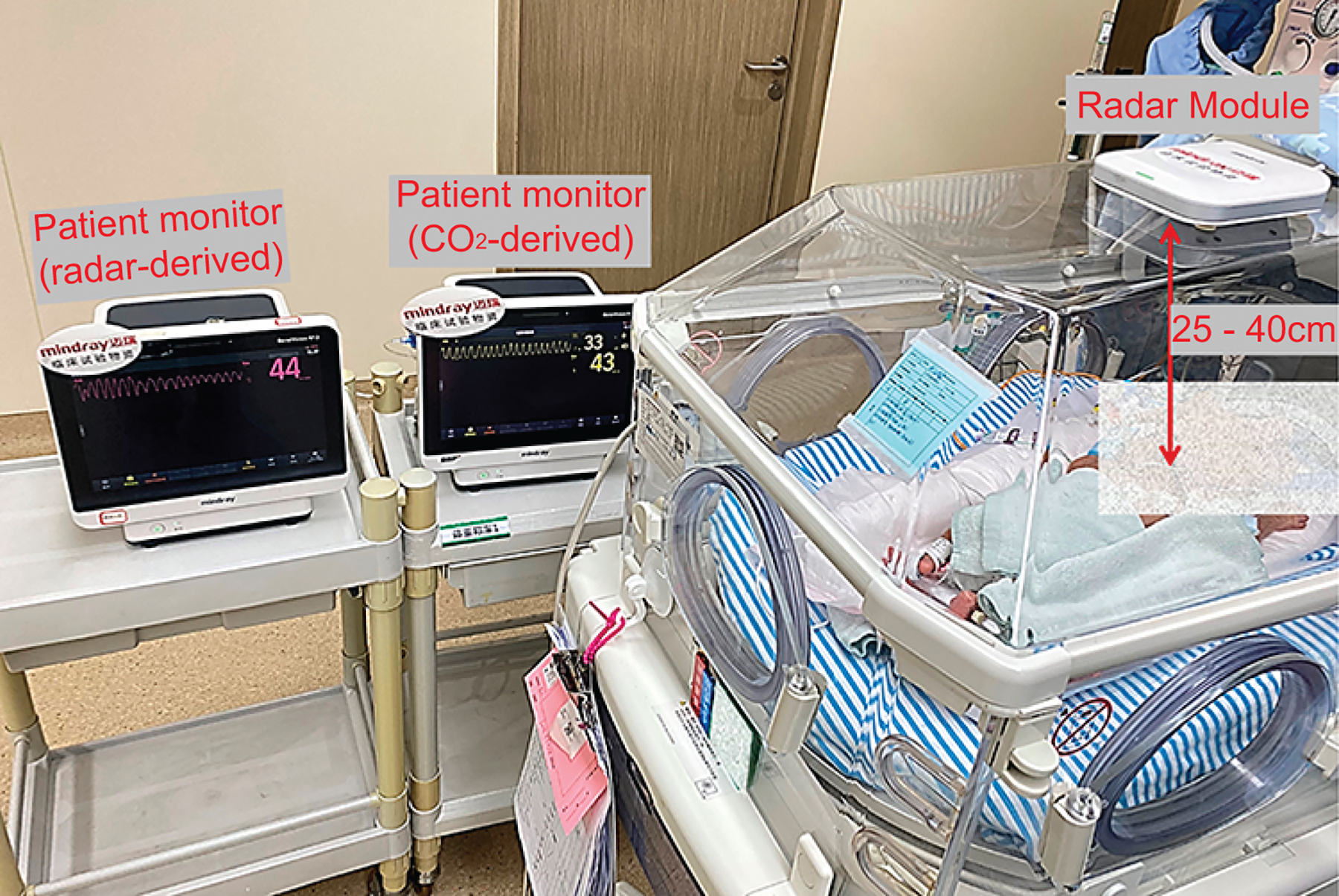
Disposition of equipment during trial. The noncontact monitor is placed on top of the neonate incubator, positioned above the thorax and abdomen of the infant, at a distance of 25–40 cm. Radar-derived breathing frequency and waveform can be observed on a BeneVision N12 patient monitor (left). End-tidal CO2 concentration, CO2-derived breathing frequency, and capnography waveform can be observed on a separate BeneVision N12 patient monitor (right).
After the start of data collection, radar-derived and CO2-derived respiratory frequencies were recorded every minute, along with a description of the subject’s breathing during the preceding minute. Descriptions were: peaceful, some movement, significant movement (repetitive myoclonus, hiccups, or crying), nursing care, and other (specify). Also noted: whether the patient monitor set off the alarm for the infant being out of the incubator (used to assess the accuracy of the monitor’s measurement of patient position). Data collection ended after 30 continuous minutes of data collection. The module was removed from the top of the incubator, the subject’s condition was observed, and if no abnormalities were observed, the test was completed and the patient exited the trial group.
Statistical analysis
Breathing frequency data from the radar module and from the CO2 respirometer were first aligned. The clinical accuracy of the noncontact monitor was assessed by calculating the RMSE between the 2 measurements. Other indicators calculated were the mean bias (μ0), RMSE, upper and lower 95% confidence intervals (U95%, L95%), regression of residuals, intraclass correlation coefficient (ICC), Pearson correlation coefficient (PCC), and concordance correlation coefficient (CCC). CCC combines both precision and accuracy, and is used as the overall measure of consistency between the 2 modes of measurement. PCC is based on linear correlation and assesses the strength of the linear correlation between the 2 modes. ICC assesses intragroup consistency, testing for replicability and consistency across different groups and measurement conditions. These statistics provide the basis for an assessment of the differences between the CO2 measurements made by the test equipment and the reference measurements. In the course of the analysis, we also plotted a Bland–Altman plot to examine the distribution of deviations in breathing frequency data between test equipment and reference equipment. Statistical analysis was carried out using the software packages PyCharm (#PY-212.5457.59) and RStudio (1.2.5001).
Results
Twenty subjects were enrolled in the trial: 8 males and 12 females. All enrolled subjects completed the trial according to protocol. Baseline characteristics of the subjects are shown in Table 1. A total of 600 data points were collected. Data suffering from interference were excluded (128 min with significant movement, 32 with treatment procedures, 18 with invalid CO2-derived data), leaving 422 valid data points.
Baseline characteristics of subjects
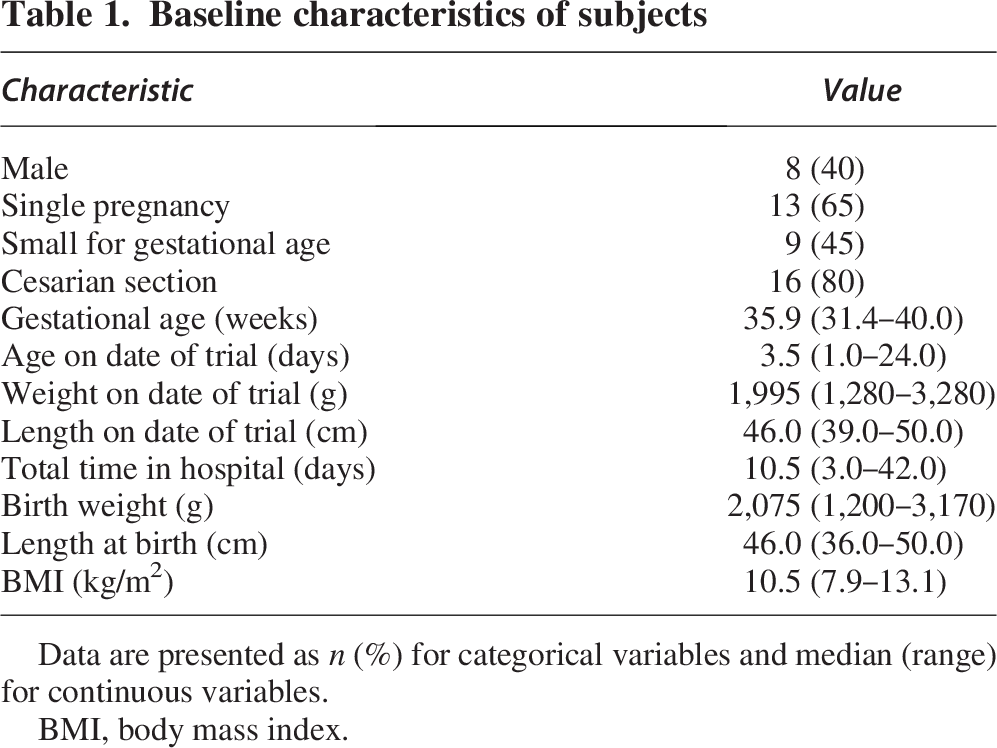
Data are presented as n (%) for categorical variables and median (range) for continuous variables.
BMI, body mass index.
Figure 2 shows a typical example of breathing frequency as measured by the radar-derived and conventional CO2. Data are from a subject age 1 day, gestational age 37 + 6 weeks, weight 2,610 g. The trial found that when the neonate experienced relatively low levels of movement, radar-derived and CO2-derived breathing frequency were in close agreement. The rhythms of the capnography waveform were basically identical. When subjects experienced apnea, the radar recorded the movement of the thoracic cage as close to zero, and the waveform remained close to the central axis. During this episode, CO2 partial pressure was close to zero, and the capnography waveform is at the bottom of the plot. The 2 lines are giving consistent readings. However, during periods of significant movement, both the radar-derived and CO2-derived measurements were affected, and the 2 show quite large disparities. During large movements by the subject, the radar may have been subject to more interference, whereas the CO2-derived recorded a low CO2 pressure and irregular changes, perhaps caused by a poor connection to the sensor or irregular breathing by the neonate.
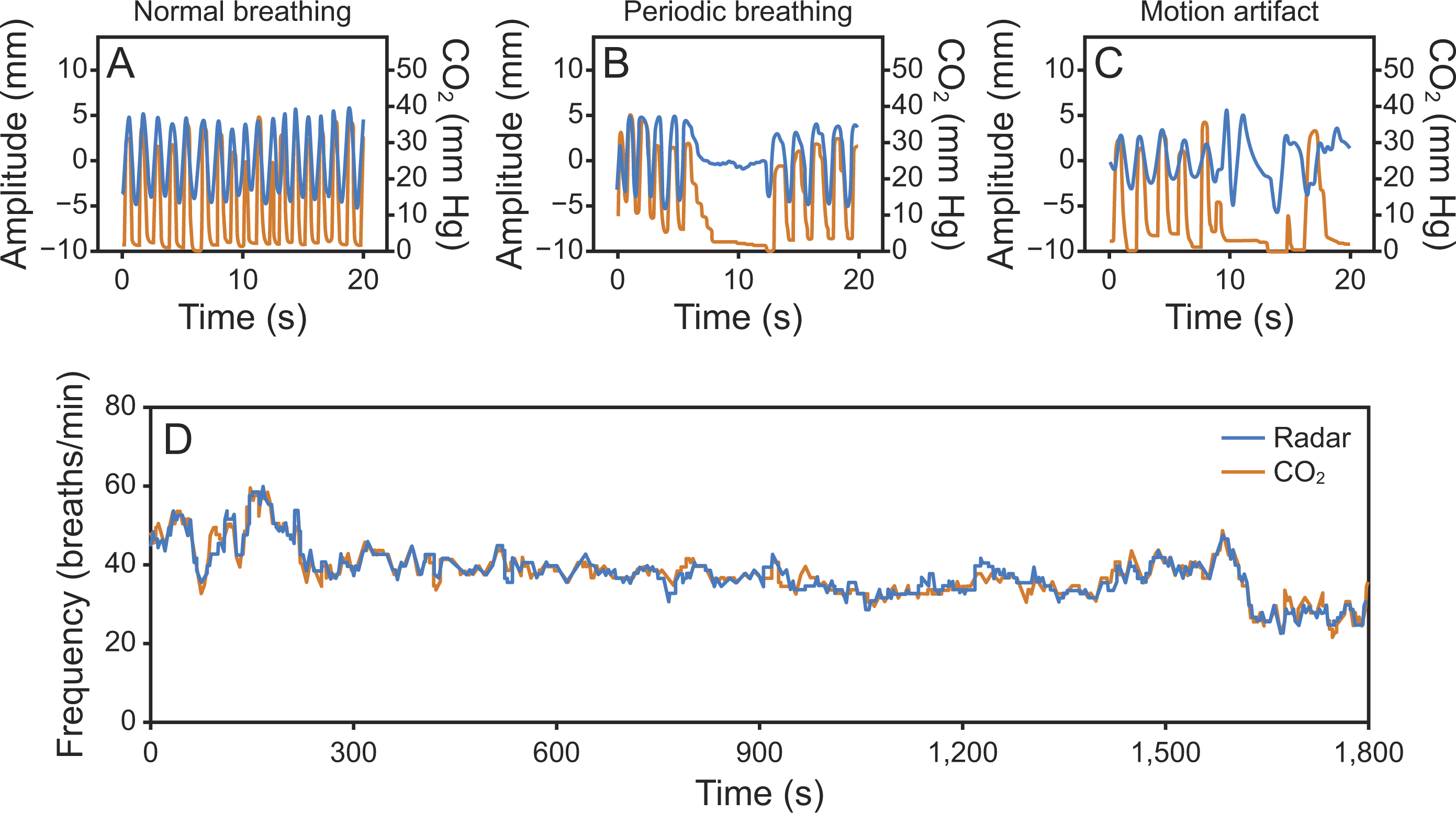
Comparison between radar-derived and CO2-derived breathing frequency and waveform in a sample subject
The statistical measures of consistency between radar-derived and CO2-derived are shown in Table 2. For the purposes of analysis, the subjects were also grouped by body weight and breathing frequency to further explore the performance of the 2 measurement techniques. Grouping by body weight, based on the 2.5 kg criterion used to define low body weight births, the subjects were divided into 2 subgroups: body weight ≥2.5 kg and body weight <2.5 kg. 19 It offers a finer perspective on the accuracy of the measurements under different physiological conditions, and helps us to assess the fitness of noncontact radar measurement of breathing frequency in a variety of clinical scenarios.
Statistical analysis of subgrouped data
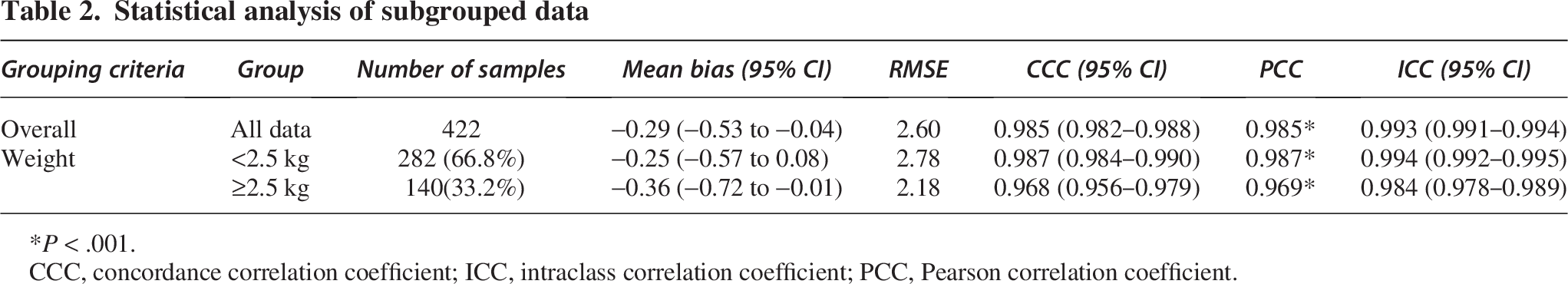
*P < .001.
CCC, concordance correlation coefficient; ICC, intraclass correlation coefficient; PCC, Pearson correlation coefficient.
In the subgroup analysis by body weight, the body weight <2.5 kg subgroup showed excellent agreement between radar-derived and CO2-derived breathing frequency (CCC = 0.987); the body weight ≥2.5 kg subgroup also showed strong agreement (CCC = 0.968) and a smaller RMSE of 2.18 breaths/min. These results suggest that using radar to measure breathing frequency gives precise and accurate results at a range of different body weights, especially with low-body-weight neonates. Overall, noncontact radar measurement delivered breathing frequency results that were highly consistent with CO2 measurements. The agreement was particularly good for the low-body-weight subgroups, and the overall performance was very satisfactory. These results support the effectiveness of a noncontact radar module as an instrument for measuring breathing frequency.
The agreement between the 2 methods for the full trial group is visualized in the Bland–Altman plot in Figure 3. The analysis revealed a mean bias of −0.29 breaths/min. The 95% limits of agreement (LoA) ranged from −5.36 to 4.78 breaths/min, indicating a high level of agreement between radar-derived and CO2-derived breathing frequency. Although most data points fell within these limits, a few outliers suggest that larger deviations can occur under certain conditions. Overall, the distribution is well-balanced and free from systematic error, further supporting the consistency between the 2 measurement techniques.
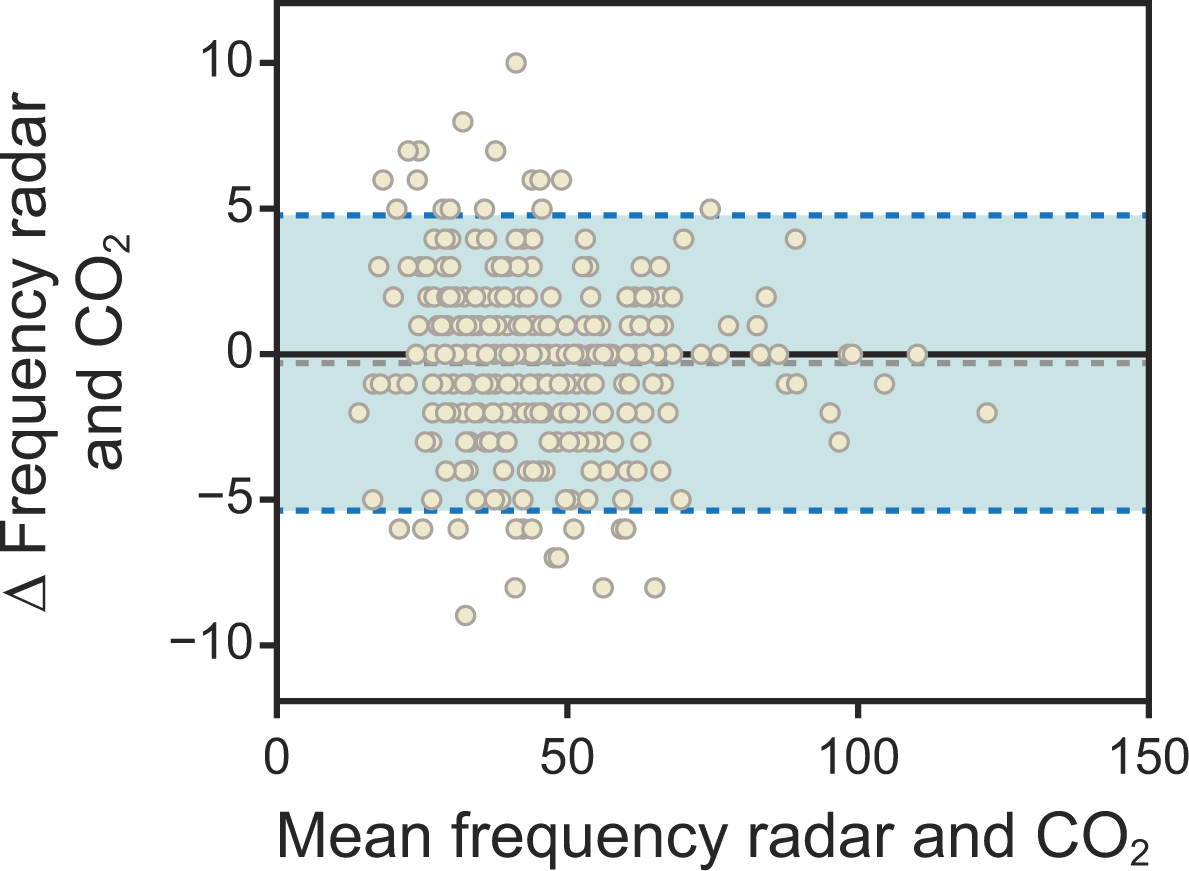
Bland–Altman plot comparing radar-derived and CO2-derived breathing frequency for the full trial group. The plot assesses the agreement between the 2 methods. The dashed gray line indicates the mean bias (−0.29 breaths/min), and the solid black line represents zero difference. The dashed blue lines are the 95% limits of agreement (LoA), which define the range within which 95% of the differences between the 2 methods are expected to lie. The upper limit (ULA) is 4.78 breaths/min, and the lower limit (LLA) is −5.36 breaths/min.
The Bland–Altman plots in Figure 4 detail the agreement within the body weight subgroups. For the low-body-weight (<2.5 kg) subgroup, the data points show a broader spread, particularly at higher mean breathing frequency values. This group had a mean bias of −0.25 breaths/min, with 95% LoA ranging from −5.19 to 5.69 breaths/min. In contrast, the normal-body-weight (≥2.5 kg) subgroup showed a tighter agreement, with a mean bias of −0.36 breaths/min and a narrower 95% LoA from −3.87 breaths/min to 4.60 breaths/min.
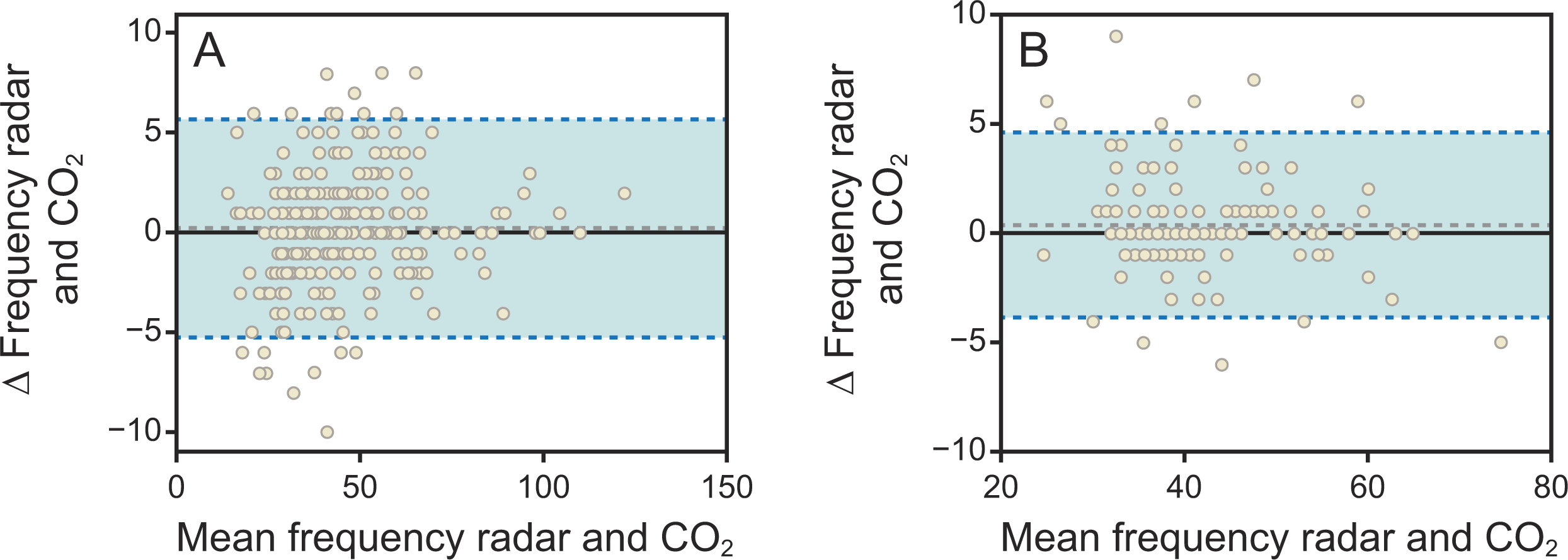
Bland–Altman plots comparing radar-derived and CO2-derived breathing frequency for body weight subgroups. The plots assess the agreement between the 2 methods.
Discussion
The purpose of this trial was to determine the accuracy of a noncontact respiration module in a clinical setting. The trial results show a high degree of agreement between breathing frequency data from the noncontact monitor and data from end-tidal CO2 monitoring. The test and control data had a low mean bias (−0.29 breaths/min) and RMSE (2.60 breaths/min), indicating closer agreement than has previously been shown in similar studies. Despite the promising results, this study has several limitations that must be acknowledged. First, the primary limitation is the relatively modest sample size, which restricts the statistical power for extensive subgrouping; however, we retained the analysis based on body weight. This decision was driven by a key clinical question: the technology’s reliability in low-birth-weight and preterm neonates. These infants, characterized by weaker respiratory muscles and subtle chest wall movements, pose a significant monitoring challenge. Our analysis provides valuable, albeit preliminary, evidence that the noncontact module performs robustly in this vulnerable population. Second, the study’s scope was confined to a specific clinical scenario. The noncontact respiration module was exclusively tested on neonates within incubators, where the device can be conveniently positioned on the top surface. Its applicability for infants cared for in open radiant warmers was not assessed. The use of the device in such an environment would necessitate an additional support structure, such as a dedicated stand, to ensure correct sensor placement. This poses a practical challenge and limits the technology’s universal application across all neonatal ICU (NICU) settings.
Nevertheless, the key finding of this trial is that the noncontact respiration module can accurately monitor neonate breathing without any contact with the patient. The main benefit of this technology is the elimination of physical contact, which in turn reduces the risk of skin injuries, such as pressure sores, and lowers the potential for infection by minimizing the need for frequent sterilization. 20 This directly supports the principle of minimal intervention, a cornerstone of current treatment strategies. 21 By reducing procedural disturbances, the technology helps stabilize the condition of preterm and other high-risk neonates, which is critical for their holistic health and development and ultimately enhances the overall quality of care in the NICU. 22
In addition to detecting breathing frequency in real time, the module used in this trial also outputs a continuous breathing wave. The results section details how the breathing wave can be used to identify apnea. In clinical applications, the monitor can be connected to a patient monitor, and when necessary, an audiovisual alarm can be set in case of an apnea. This will quickly alert the care team and reduce the incidence of irreversible oxygen deprivation injuries. One of the most important issues in clinical care is apnea, bradycardia, and oxygen desaturation (ABD) events, 23 when a neonate simultaneously experiences slowed heart rhythm, lack of oxygen, and a cessation of breathing.24,25 ABD events are typically monitored using an ECG. This device is available in the ICU, but the electrodes can easily irritate the skin of a newborn. Using noncontact monitoring of vital signs, it is possible that noncontact monitoring for ABD events can be achieved. This will make this kind of monitoring easier to deliver, and it can be extended beyond the NICU to other settings, including other hospital wards or home care scenarios. 26
In addition to the primary radar sensor, the module used in this trial also includes a video monitor. The main function of this monitor is to check for the presence of the neonate in the monitor unit’s field of detection (ie, the incubator). When the patient has been removed for standard NICU processes such as washing, examination, or kangaroo care, the unit will automatically register the absence of the patient and stop monitoring. This prevents unnecessary alarms and bad data, which contribute to alarm fatigue. It also reduces unnecessary actions that would have to be taken by the treatment team to silence or turn off alarm functions. In the future, the video components could also be used to judge whether the monitor is correctly positioned relative to the patient. The radar signal needs a line of sight to the patient’s thoracic cage, and in neonates, the breathing signal is weaker than for adults. In preterm neonates, chest movement may be as little as 1 mm. 27 It is therefore important that the monitor is positioned directly above the patient. If either the patient or the monitor is moved too much, the radar signal will weaken and possibly fail to detect the patient’s breathing. In this situation, when the dual sensors detect that the monitor is out of position, it could send an alert to the treatment team, or a mechanical apparatus could be used to reposition the angle of the radar beam and reacquire the target to increase signal quality.
As the technology develops, the range of potential applications for noncontact monitoring technology is expanding significantly. 28 Currently, this technology is primarily used for monitoring breathing frequency, but its potential goes far beyond this single usage. Improved algorithms and greater sensor sensitivity will make this equipment capable of measuring other vital signs, including heart rate and body temperature, and even microscopic movements. Development and trials that are currently underway may expand the use of this equipment to adults and geriatric patients, particularly those with chronic diseases of the respiratory system or sleep disorders. These broader applications could lead to a comprehensive shift in the monitoring of patients of all ages and medical conditions, leading to a major improvement in healthcare. Noncontact technologies are minimally disruptive to patients and offer high value for money. They may be used to monitor multiple patients at once, and so constitute an effective solution for large-scale patient monitoring. 29
Noncontact forms of measurement have great potential for clinical applications. However, there remain a number of challenges that will need to be solved. 30 First, the accuracy of monitoring using noncontact techniques may be affected by many different factors, including the placement of the equipment and environmental interference. In the NICU, patient crying and movement are inevitable, as are frequent procedures. Radar technology may also be affected by echoes from multiple possible radar signal reflections, or by changes in the surroundings and other moving targets. To resolve these issues, future developments should focus on making smarter and better-adapted algorithms for the equipment, to improve performance and mitigate the impact of external factors, or to issue alerts for the treatment team when clear environmental interference is detected. Second, although no adverse incidents were observed during the use of the module, as use of this equipment becomes more common, monitoring its long-term safety is of critical importance. Future research should consider giving priority to assessing the long-term safety of this technology, to ensure its effectiveness and safety over the long term. Data on the ongoing safety and effectiveness will help to build trust in this technology and to support its widespread adoption in clinical practice.
The outcomes of this trial show that the module has good prospects for use in clinical settings, but going forward, there is need for more research into clinical applications. For example, comparative studies on clinical outcomes such as treatment injury or infection risk, or feasibility studies into the identification of different breathing modes from the waveform on a noncontact monitor during diagnosis and treatment of a neonate’s respiratory system. The modules offer an effective and safe option for monitoring neonate breathing. This technology can reduce interference with infant patients and contribute to supportive care for neonates. It has the potential to be widely adopted in the treatment of infants. The goal for future research will be to further improve this technology and to explore broader clinical applications.
Conclusions
This trial demonstrated the accuracy and effectiveness of a noncontact respiration module in NICU settings through a comparison against the accepted-standard CO2-based measurement of breathing frequency. This study demonstrated stronger agreement and smaller errors than have been shown in previous trials. In addition, this study showed a new form of radar and video noncontact monitoring that is more applicable in clinical settings. For the care of neonates, noncontact monitoring of vital signs is a technology with excellent prospects. However, more work remains to be done to increase accuracy, stability, and robustness against interference in complex clinical settings.
Footnotes
Author Disclosure Statement
The authors have no relationships to disclose.
Funding Information
This work was supported by the Guangdong Basic and Applied Basic Research Foundation (Nos. 2023A1515010674 and 2025A1515012722), the Guangdong Province Science and Technology Innovation Popularization Special Project (No. 2023A1414020033), the Guangzhou Science and Technology Key Research and development Program Project (Nos. 2024B03J1297, 2025A03J4281, and 2025A03J4139), the Guangzhou Science and Technology Program Project (No. 202201011272), the Medical Scientific Research Foundation of Guangdong Province of China (No. C2022037), and the Guangdong Medical Research Fund Project (No. A2024312).
