Abstract
Long-term oxygen therapy (LTOT), that is, home oxygen prescribed for at least 15–18 hours per day, improves survival in patients with COPD and severe hypoxemia. LTOT does not benefit all severely hypoxemic patients, however, a large proportion of patients who receive home oxygen do not benefit from therapy, whereas a number of patients who would benefit from home oxygen are denied therapy because they do not meet the current prescription criteria that are accepted worldwide. To overcome this problem, we suggest that the practice of LTOT prescription be individualized. Biomarkers of cellular or vascular dysfunction should complement the mere measurement of PaO2 or SpO2 to decide who should receive LTOT. In this article, we present clinical vignettes to illustrate the application of this proposal and describe a protocol for a clinical trial designed to test its validity.
Introduction
It is a startling fact that between 2011 and 2013, one-fourth of the 5.75 million Medicare beneficiaries with COPD received supplemental oxygen for at least 6 months. 1 Although criteria for long-term oxygen therapy (LTOT) exist and are accepted worldwide, a report has indicated that as many as 50% of those who receive home oxygen do not meet them. 2 Although many factors (including oxygen availability, imprecise reimbursement policies, and patient preferences) may explain poor guideline adherence, this situation suggests that the indications for LTOT that clinicians perceive may not correspond to the established prescription criteria. In a recent publication, we supported the notion that prescribing LTOT solely on a PaO2 threshold may be inappropriate. 3 We asserted, based on published LTOT trials in COPD, that current oxygen prescription criteria are poorly validated and, as indicated in a National Heart, Lung, and Blood-sponsored workshop report, “overstate their scientific basis.” 4 Furthermore, although the Nocturnal Oxygen Therapy (NOTT) and the British Medical Research Council (BMRC) oxygen trials showed a mortality benefit, these studies are 45 years old and were conducted when the COPD disease and management landscape were dramatically different.5,6 There is reason to suspect that if the trials were repeated now, the mortality benefit may not be as impressive.
Prescribing LTOT only to those who need it is important because oxygen is cumbersome and expensive. On the one hand, starting LTOT while hypoxemia is only moderate is usually pointless since it confers no clear survival benefit. 7 On the other hand, initiating LTOT late in the course of the disease, when end-organ dysfunction is established, may be in vain since subgroup analyses of the NOTT indicated that the benefits of LTOT are hard to demonstrate in those with advanced disease. 5 There seems to be a window of opportunity where the benefits of LTOT may be optimized.
David Flenley, who led the BMRC trial, asserted that the rationale of oxygen therapy is “not to make the patient pink, nor even to make his arterial blood have a normal content of oxygen, but to provide his mitochondria with enough oxygen to allow them to operate an efficient aerobic metabolism.” 8 Hypoxemia (ie, low blood PaO2) must therefore be distinguished from hypoxia (ie, insufficient tissue oxygen supply). Accordingly, we proposed to work toward abandoning PaO2 as the main criterion for oxygen prescription and to adopt a more physiological approach in determining who would really benefit from LTOT, in accordance with the principles of personalized medicine. We hypothesized that products released during reactions to chronic cellular hypoxia may serve as biomarkers to complement the usual measurement of PaO2 or SpO2 as measured by pulse oximetry or the emerging technology of near-infrared spectroscopy (a direct real-time assessment of tissue hypoxia)9,10 in personalizing oxygen prescription in COPD, at a time before end-organ dysfunction becomes clinically apparent. 3 In this communication, we provide more explanation on how this proposal might work.
Biomarkers of Chronic Hypoxia
A biomarker is “a characteristic objectively measured as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to therapeutic intervention.” 11 Biomarkers may be found in any body fluid or tissue. We conducted a scoping review of the literature that identified several potentially useful biomarkers of chronic hypoxia in patients with stable chronic lung diseases. 3 The most studied included circulating levels of hemoglobin, erythropoietin, brain natriuretic protein, and endothelin-1, as well as macroalbuminuria. Other candidate biomarkers less well studied include hypoxia-inducible factors, ischemia-modified albumin, heat shock proteins, fibroblast growth factor, high-sensitivity cardiac troponin, lactate-to-pyruvate ratio, and vascular endothelial growth factor.
Current knowledge only indicates that the most studied biomarkers have a relation to the state of chronic hypoxia. Their ability to determine the need for LTOT in COPD has not been specifically investigated, and insufficient information is currently available to support a definitive proposal. Nevertheless, the following clinical vignettes illustrate, from fictitious cases, our futuristic view of the potential utilization of biomarkers in deciding who should or should not be prescribed LTOT. We wish to emphasize that the management suggestions presented below are forward-looking and should not be used to replace current strategies until these new paradigms are validated.
Clinical Vignettes
Clinical vignette #1
Mr Smith is a 72-year-old who carries a diagnosis of severe COPD. He has noted over the last 2–3 months progressive painless swelling of both ankles and legs, without any symptoms of exacerbation of COPD. Acral cyanosis is noted upon physical examination, which additionally confirms pitting edema on both lower limbs. Lower venous extremity Doppler ultrasound quickly rules out deep venous thrombosis. Arterial blood gas measurement at room air demonstrates severe hypoxemia (PaO2 is 51 mm Hg), PaCO2 is 46 mm Hg, and pH is normal. Chest radiograph shows no evidence of left heart failure.
This patient presents severe hypoxemia with end-organ dysfunction (right heart failure). Oxygen therapy is clearly indicated. In this case, the measurement of biomarkers of chronic hypoxia (eg, hemoglobin, brain natriuretic peptide, endothelin-1) is only optional. The disease is at its end stage. According to the NOTT, 5 survival benefit of LTOT is uncertain, and the prognosis is poor.
Clinical vignette #2
Mrs Johnson is a 75-year-old who has suffered from severe COPD for at least 15 years. During her annual visit at the respiratory clinic, she reports no recent symptoms of COPD exacerbation. She is active. Physical examination is within normal except for reduced breath sounds on auscultation. SpO2 at rest is 90%. No biomarker of chronic hypoxia is found in her plasma or urine: hemoglobin, erythropoietin, brain natriuretic protein, and endothelin-1 are within the normal range. Her urinalysis is normal; no microalbuminuria is detected.
This patient is moderately hypoxemic at rest. She is representative of those who participated in the Long-term Oxygen Treatment Trial, in which long-term supplemental oxygen did not result in a longer time to death or first hospitalization. 12 There is no indication that any reaction to chronic hypoxia has been activated as her biomarker profile is normal. We conclude that she does not need oxygen at home. Follow-up is scheduled for 1 year.
Clinical vignette #3
Mr Williams received a diagnosis of severe emphysema 10 years ago at the age of 61. He reports that a COPD exacerbation was successfully treated with oral corticosteroids and antibiotics 4 months ago. He is sedentary and severely dyspneic (modified Medical Research Council dyspnea scale: 3 on a 0–4 scale). He has never noted ankle or leg swelling. Physical examination shows a typical pink-puffer phenotype. SpO2 at rest is 88%. Arterial blood gases are measured: PaO2 and PaCO2 are 53 and 42 mm Hg, respectively; pH is normal. Hemoglobin, erythropoietin, brain natriuretic protein, and endothelin-1 are all within normal values; no microalbuminuria is detected.
This vignette illustrates the case of a patient with uncomplicated severe hypoxemia. Biomarkers of chronic cellular hypoxia are negative. Although he would qualify for LTOT based on his PaO2, our hypothesis would indicate that he is unlikely to benefit from oxygen if the objective is to improve survival. Close follow-up with repeated measurement of hypoxia biomarkers would be in order.
Clinical vignette #4
Mrs Brown, aged 68, has been brought to the sleep laboratory because she complains of chronic fatigue. Her husband reports that she snores while sleeping. She suffers from severe COPD and has not noted any change in her symptoms for at least 6 months. Physical examination is unremarkable. Her sleep study rules out obstructive sleep apnea. However, oximetry demonstrates significant nocturnal oxygen desaturation (75% of the recording time with a saturation <90%; mean saturation: 83%) with nonperiodic variation in saturation throughout sleep. Her hemoglobin is slightly elevated (16 g/dL; hematocrit: 49%). Circulating erythropoietin levels measured at 10:00 AM and while she was in the sleep laboratory (2:00 AM) are about the same and are both elevated. Brain natriuretic peptide is also elevated.
Isolated nocturnal oxygen desaturation may be equated to moderate hypoxemia. 7 The International Nocturnal Oxygen (INOX) trial provided no indication that nocturnal oxygen has a positive or negative effect on survival or progression to LTOT in patients with COPD. 13 However, this patient has more severe nocturnal hypoxemia than the average patient who participated in the INOX trial. Her biomarkers (elevated hemoglobin and brain natriuretic peptide, loss of erythropoietin circadian cycle) suggest that she suffers from chronic hypoxia. A trial of nocturnal oxygen is suggested, with repeated measures of biomarkers after a 3-month trial of nocturnal oxygen.
Clinical vignette #5
Mr Jones is a 68-year-old man with a diagnosis of severe obstructive chronic bronchitis. He has been clinically stable over the last 3 months. Arterial blood pressure is normal. Physical examination does not indicate any signs of right heart failure. SpO2 is 90%, which prompts the measurement of arterial blood gases. Results are as follows: PaO2: 61 mm Hg; PaCO2: 49 mm Hg; pH: 7.39. Several biomarkers of chronic hypoxia are found in his plasma and urine: hemoglobin is rising (from 13 to 15.5 g/dL over a 3-month period); erythropoietin, brain natriuretic peptide, and endothelin-1 are elevated, and microalbuminuria is detected. Glycated hemoglobin and serum creatinine are normal.
Because of chronic respiratory failure, hypercapnia and hypoxemia often coexist. Hypercapnia may also determine the response to oxygen. Most patients enrolled in the BMRC trial were severely hypercapnic. 6 Not often appreciated is that, in the NOTT, subgroup analysis showed no significant LTOT mortality effect in normocapnic patients, whereas the largest mortality difference was seen in hypercapnic patients. 5 This patient does not meet the usual criteria for LTOT prescription. However, his biomarker profile clearly indicates that hypoxemia, although moderate, is detrimental and leads to tissue hypoxia. A trial of oxygen may be offered. Consideration could also be given to noninvasive ventilation if hypercapnia was more severe. 14
Genetics and Response to Oxygen
Significant variations in the cellular response to hypoxemia can be detected between normal humans, and this may be partly related to differences in genetic background. 15 In a subanalysis of the Long-term Oxygen Treatment Trial, single-nucleotide polymorphisms (representing DNA sequence variations) showed significant interaction between oxygen treatment and the trial primary outcome. 16 Many of these variations are near arylsulfatase B (ARSB), an oxygen-responsive gene that regulates mitochondrial functions. This study suggests that not all patients respond the same way to oxygen therapy. If confirmed, this information may become extremely useful in selecting candidates for oxygen therapy.
Putting It All Together
Figure 1A illustrates the traditional method of LTOT prescription based on PaO2 measurement. In several jurisdictions, SpO2 is also used to diagnose severe hypoxemia and to prescribe LTOT in COPD, although this practice is not based on evidence from randomized trials.
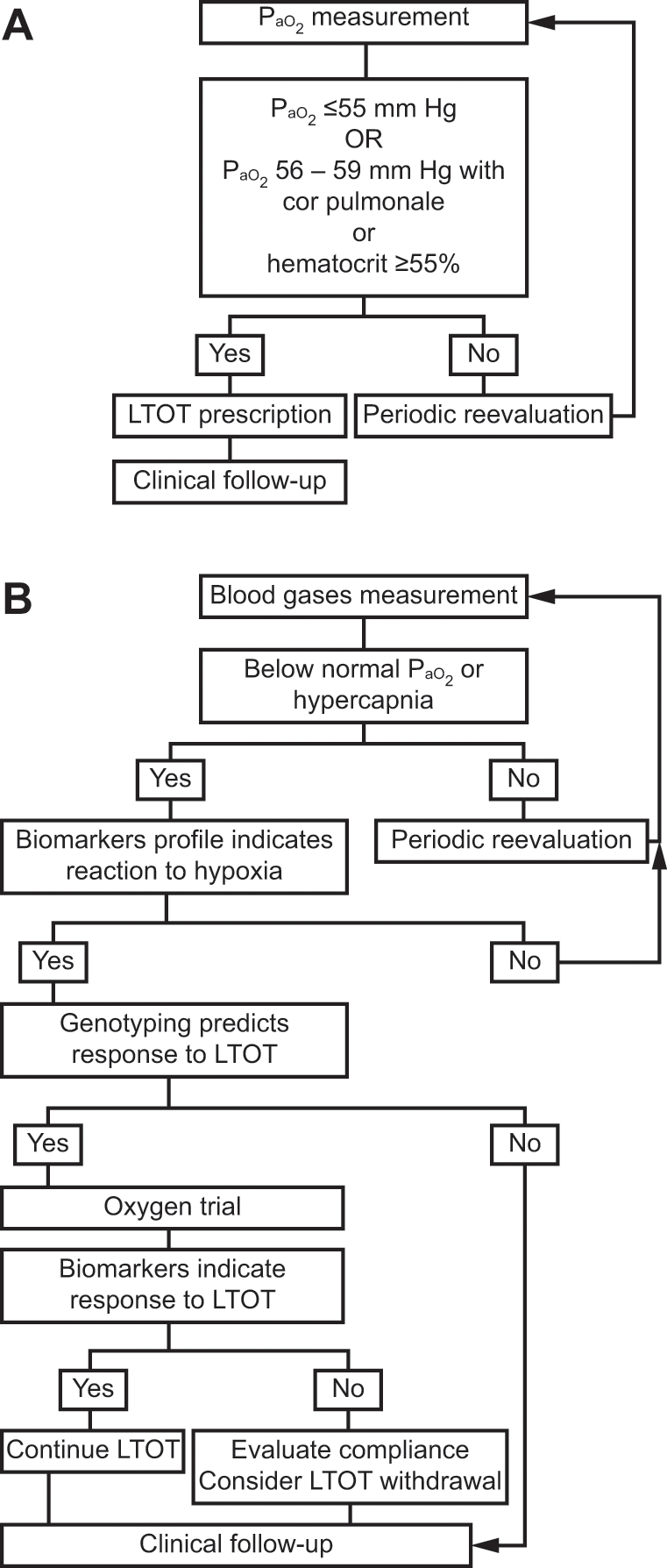
The practice of LTOT prescription in stable COPD.
Figure 1B illustrates our forward-thinking perspective of the potential of biomarkers and genetics to guide LTOT prescription in COPD. Patients at risk for clinically important hypoxemia are screened with arterial blood gas measurement. Clinical judgment is used to determine whether further investigation is needed when PaO2 is below normal and/or hypercapnia is noted, without consideration of threshold values. Biomarkers may or may not indicate a reaction to chronic hypoxia. Since none of the biomarkers we identified from our review of the literature is acceptably accurate on its own to detect the state of cellular hypoxia, they most likely will be measured in bundles and interpreted in a stepwise manner, guided by their respective sensitivity and specificity, after careful consideration of individual comorbidities. Changes in biomarkers after the introduction of LTOT may demonstrate adherence and favorable response to therapy, whereas lack of change may indicate poor compliance or resistance to oxygen therapy. The measurement of these biomarkers would occur in the same conditions as those that currently apply for the determination of the need for LTOT in nonsmoking patients, away from an episode of exacerbation. Genotyping may eventually add another layer to this decision process by prospectively identifying responders and nonresponders to oxygen therapy.
What Might a Clinical Trial to Test This Hypothesis Look Like?
Of course, we acknowledge that our proposal needs validation. Once biomarkers of chronic hypoxia are well validated and their measurement methods are standardized, our proposal will need to be submitted to the test of randomized trials. 3 The objective of these confirmatory trials will not be to test the effectiveness of LTOT to improve survival and quality of life or to reduce hospitalizations. Rather, the aim will be to compare two strategies to select patients most likely to benefit from LTOT (ie, Fig. 1A vs Fig. 1B). A research question formulated according to the Population/Intervention/Comparator/Outcome framework could read as this: “In patients with moderate-to-severe COPD at risk of clinically important hypoxemia, is selecting patients for LTOT based on biomarkers of chronic hypoxia more efficient than the current selection strategy based on PaO2 thresholds to improve survival?”
Because clinical research on oxygen in COPD is difficult and takes time, pragmatic trials (ie, trials designed to evaluate the effectiveness of interventions in routine practice conditions 17 ) would be best suited to answer this question. Pragmatic trials usually compare a novel intervention to usual care and are conducted in a way that is as close as possible to usual practice. The intensity of measurement and the follow-up of participants in the trial are also similar to the typical follow-up in usual care. The trial’s primary outcome is directly relevant to participants. Pragmatic trials must therefore be distinguished from explanatory trials. The former are intended to inform a clinical or policy decision in real-world clinical practice, whereas the latter aim at confirming a physiological or clinical hypothesis. 18
In the proposed trial, defining the study population is methodologically challenging, since the indications for intervention (LTOT) in the two strategies are not necessarily intended for the same disease states. The indication for LTOT may become apparent at different points in time in the course of the disease, depending on the patient selection strategy. To overcome this problem, broader inclusion criteria should allow patients to be enrolled before biomarkers are detected, before PaO2 crosses the limit of 55 mm Hg, and before end-organ dysfunction is noted. These patients would be considered “at risk” of developing significant hypoxemia, which is the case for most of those who are followed in respiratory or COPD clinics.
Once patients are randomized, follow-up at predefined intervals will allow the detection of biomarkers of chronic hypoxia in those allocated to the novel strategy, and of severe hypoxemia in those allocated to the traditional method of LTOT prescription. LTOT will be started when convincing evidence of reaction to chronic hypoxia is noted in the intervention group or when PaO2 reaches LTOT prescription threshold in the control groups. Randomization may occur at the individual level or at the level of clusters if multiple sites are involved.
Primary outcome should be survival from randomization and from LTOT inception in the prespecified subgroup of patients ending up on LTOT. Secondary outcomes may consist of the proportion of patients in each group ending up on LTOT, cost-effectiveness, health-related quality of life, and measures of health care utilization (including hospitalization). Cost may indeed be an issue. However, since LTOT is a major cost driver in the management of COPD, 19 we would predict that the cost of inappropriate prescriptions of LTOT most likely outweighs that of the use of biomarkers to select LTOT candidates. Outcome data may be collected from databases often maintained in respiratory clinics and/or national registries such as those that exist in Sweden or Denmark.20,21
Although we can only speculate on the results of such a trial, a better selection of patients based on the physiology of chronic hypoxia is likely to reduce the proportion of patients who will receive LTOT. Those patients should be more likely to respond to oxygen. In the main survival analysis involving all randomized patients, equivalent survival in both groups is expected. In the subgroup analysis of those who end up on LTOT, survival is likely to be better in the intervention group, the treatment effect being diluted in those allocated to usual care, many of them improperly receiving LTOT. Such results would reflect the success of precision medicine.
It must be acknowledged that such a study may raise ethical concerns in the minds of clinicians, given the results of the NOTT and the BMRC trials.5,6 Unavoidably, some participants in the intervention group who present at some point with a PaO2 less than 55 mm Hg will not be assigned supplemental oxygen until their biomarker profile shows evidence of reaction to chronic hypoxia. We would provide several arguments favoring the conduct of the trial, even if this situation is very likely to occur. First, the PaO2 ≤55 mm Hg threshold was chosen rather arbitrarily by the NOTT investigators as the main inclusion criterion following the observation that tissue hypoxia is noted when PaO2 approximates 50 mm Hg. 22 Of note, the mean PaO2 at study entry was 51 mm Hg in both the NOTT and the BMRC trials. Second, long-term survival has been observed in patients with very severe hypoxemia (mean PaO2: 45 mm Hg) who declined oxygen therapy. 23 Third, if our hypothesis is true, biomarkers of chronic hypoxia should be detected when hypoxemia becomes significant, which will prompt LTOT prescription. Close follow-up of these patients may be desirable.
Summary
Our preliminary proposal has the merit of encouraging clinicians and researchers to “think outside the box” and to consider the value of establishing a new clinical pathway. More than twenty randomized trials of oxygen in COPD have been conducted since publication of the NOTT and the BMRC trial. Most used PaO2 and/or SpO2 thresholds as inclusion criteria. All subsequent trials have failed to demonstrate long-term clinically important benefits (eg, mortality, hospitalization, or exacerbation reduction) of LTOT in a variety of indications (including oxygen in moderate hypoxemia for isolated nocturnal or exercise-induced desaturation). It is time for oxygen therapy to progress from the status quo that has persisted for more than 40 years. We strongly believe that a change in paradigm should be sought. We have also proposed steps to the scientific validation of a novel strategy to select candidates for oxygen therapy. 3 The measurement of biomarkers of chronic cellular hypoxia in COPD to determine who should or should not receive LTOT may offer an opportunity to practice precision (or personalized) medicine to better serve our patients.
Footnotes
Author Disclosure Statement
Y.L. and F.M. report participation in Innovair, a company that holds shares in OxyNov, the owner of FreeO2, an automated oxygen delivery system; R.C. has served on an Advisory Board for Inogen.
Funding Information
No funding was received for this article.
