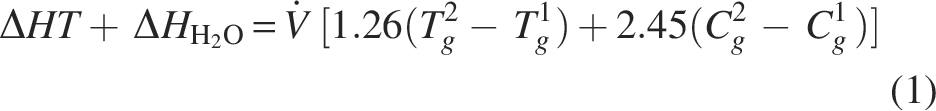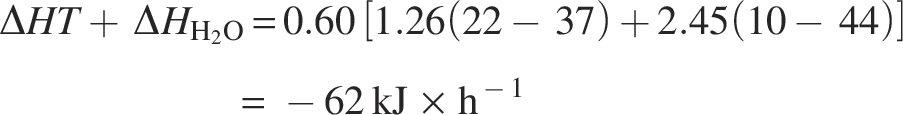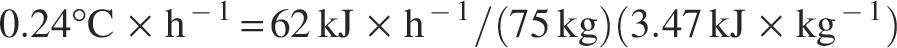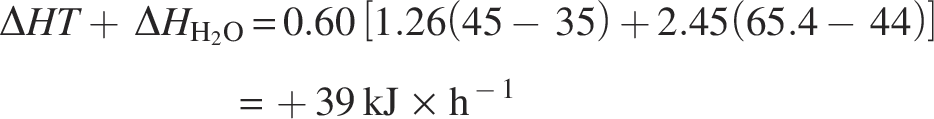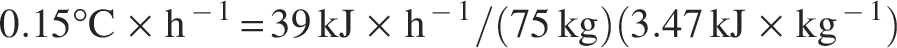Abstract
Hypothermia, defined as a core body temperature ≤35°C, significantly increases morbidity and mortality in mechanically ventilated patients across numerous care settings. Physiologically, the upper airway conditions inspired gases to body temperature and humidity, minimizing heat energy loss and preventing mucosal damage. Instrumentation, such as endotracheal intubation, bypasses this natural mechanism, leading to considerable heat and moisture loss, potentially exacerbating hypothermia risks in critically ill patients. Active humidifiers and heat and moisture exchangers represent common strategies to mitigate airway heat loss, yet their effectiveness as a method to assist in whole-body rewarming is controversial. Emerging technologies indicate renewed interest in airway-based warming devices, especially for prehospital and military trauma scenarios, but robust clinical validation remains necessary. This narrative review evaluates the feasibility and effectiveness of airway-based thermoregulation through inhalation of heated, humidified gases.
This is a visual representation of the abstract.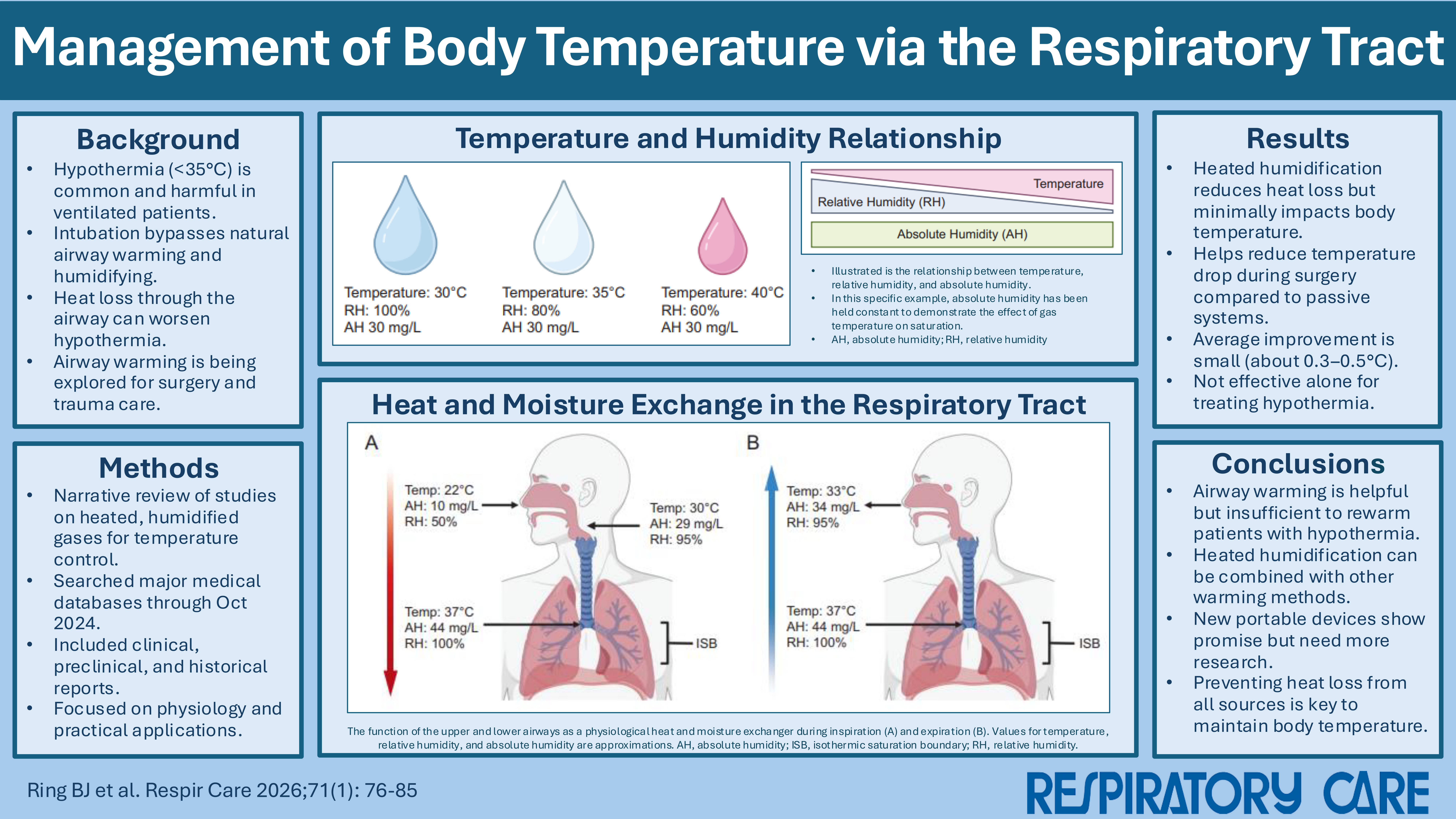
Introduction
Hypothermia is defined as a body temperature ≤35°C and is a risk factor for poor outcomes in patients suffering from its downstream physiologic effects.1,2 Various conditions disrupt thermoregulation and contribute to a positive feedback loop of adverse physiological changes (ie, coagulopathy, metabolic derangements, and poor tissue oxygenation). 3
Hypothermia can be categorized as intentional or unintentional and caused by iatrogenic, therapeutic, infectious, endocrine, trauma, environmental exposure, or surgical interventions, all of which may not be mutually exclusive in the critically ill patient. Whether intentional or unintentional, hypothermia in mechanically ventilated patients is a relatively common occurrence in intensive care settings and is a risk factor for increased morbidity and mortality, extended hospital stays, infections, and cardiovascular issues.4–6 Data suggest that the incidence of hypothermia in mechanically ventilated patients ranges from 5% to 13%, with variations influenced by factors such as the patient’s underlying condition, the duration of mechanical ventilation, and the environment within the intensive care unit. 4 In the perioperative space, the incidence of unintentional hypothermia is reported to occur in nearly 54% of patients upon admission to an ICU. 7
In the prehospital environment, unintentional hypothermia is a risk factor for numerous poor outcomes in patients suffering from a major trauma-related event.1,2 As a result, preventive measures, including body warming strategies and temperature monitoring, are crucial to mitigate the incidence and impact of hypothermia in patients receiving mechanical ventilation and improve overall clinical outcomes. 8 The importance of maintaining body temperature to improve patient outcomes has been recognized since the initial exploration of resuscitation efforts.
When drowning was a prominent cause of death in the early 18th century, rewarming the victim after exposure was considered compulsory during resuscitation. 9 In conjunction with what would be considered questionable practices today (eg, hanging drowned victims by their legs to expel water from the lungs), resuscitation involved simultaneous, nontargeted interventions, contrary to today’s precision-focused care paradigm. Among these interventions, both invasive and noninvasive rewarming of hypothermic drowning victims included the use of heated insufflated gas. This dated practice raises a contemporary question: Is the airway a reasonable target for thermoregulation during mechanical ventilation?
This review explores thermoregulation via inhalation of heated, humidified gas at or above physiological levels. This review also covers the basic physiology of airway heat and moisture exchange, reviews equipment for conditioning inspired gas during mechanical ventilation, and examines relevant literature on airway-based thermoregulation in perioperative and trauma care.
Methods
A narrative review was performed to examine the role of inhaled heated and humidified gases in pulmonary heat transfer and core temperature thermoregulation. Electronic databases, including PubMed, EMBASE, and CINAHL, were searched from inception through October 2024. Search terms included combinations of “hypothermia,” “rewarming,” “heated humidified gas,” “pulmonary heat exchange,” “thermoregulation,” and “mechanical ventilation.” Both preclinical and clinical studies were included, along with historical reports and review articles deemed relevant to the physiologic principles and clinical application of respiratory heat exchange. Reference lists of retrieved articles were manually reviewed to identify additional relevant studies.
Because this is a narrative review, no formal grading of evidence or assessment of study quality was conducted. Articles were selected based on their relevance to the topic and contribution to understanding the mechanisms and potential utility of rewarming strategies via the airway.
Physiology of heat and humidification within the airway
Absolute humidity (AH) refers to the mass of water vapor per unit air volume, whereas relative humidity (RH) is the ratio of actual water vapor to the maximum possible at a given temperature. Because warmer air holds more moisture, RH changes with temperature even if AH remains constant (Fig. 1).
Illustrated is the relationship between temperature, relative humidity, and absolute humidity. In this specific example, absolute humidity has been held constant to demonstrate the effect of gas temperature on saturation. AH, absolute humidity; RH, relative humidity.
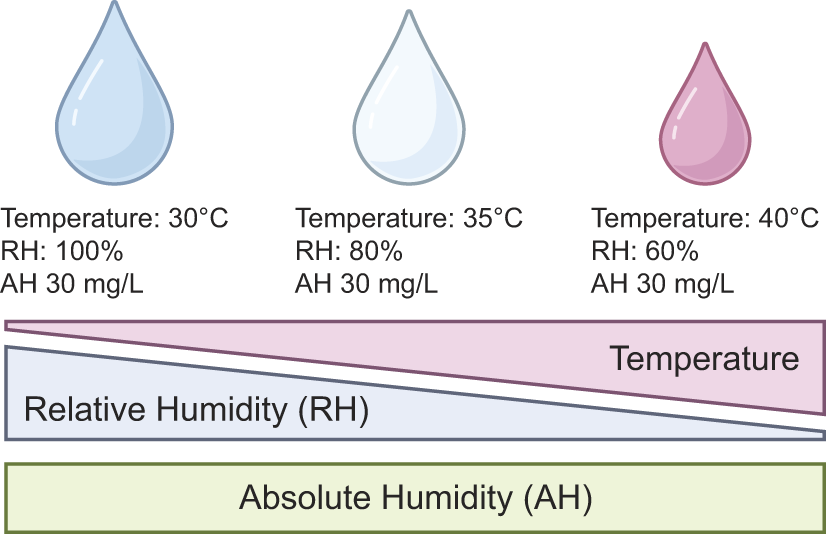
The upper airway, particularly the nasopharynx, is responsible for filtering, warming, and humidifying inspired gas before it reaches the lower respiratory tract. Vascularized nasal turbinates and moist mucous membranes facilitate this conditioning process, ensuring that gases entering the lower airways are near body temperature and fully saturated with water vapor.10–12 By the fourth or fifth generation of bronchi, known as the isothermic saturation boundary, inspired gases typically reach alveolar conditions: 37°C, 100% RH, and an AH of approximately 44 mg H2O/L (Fig. 2A). 11 During exhalation, the fully conditioned exhaled gas cools and condenses on the airway mucosa, scavenging some water and heat for the next breath cycle (Fig. 2B). As such, the respiratory tract is a countercurrent heat and moisture exchanger. 11
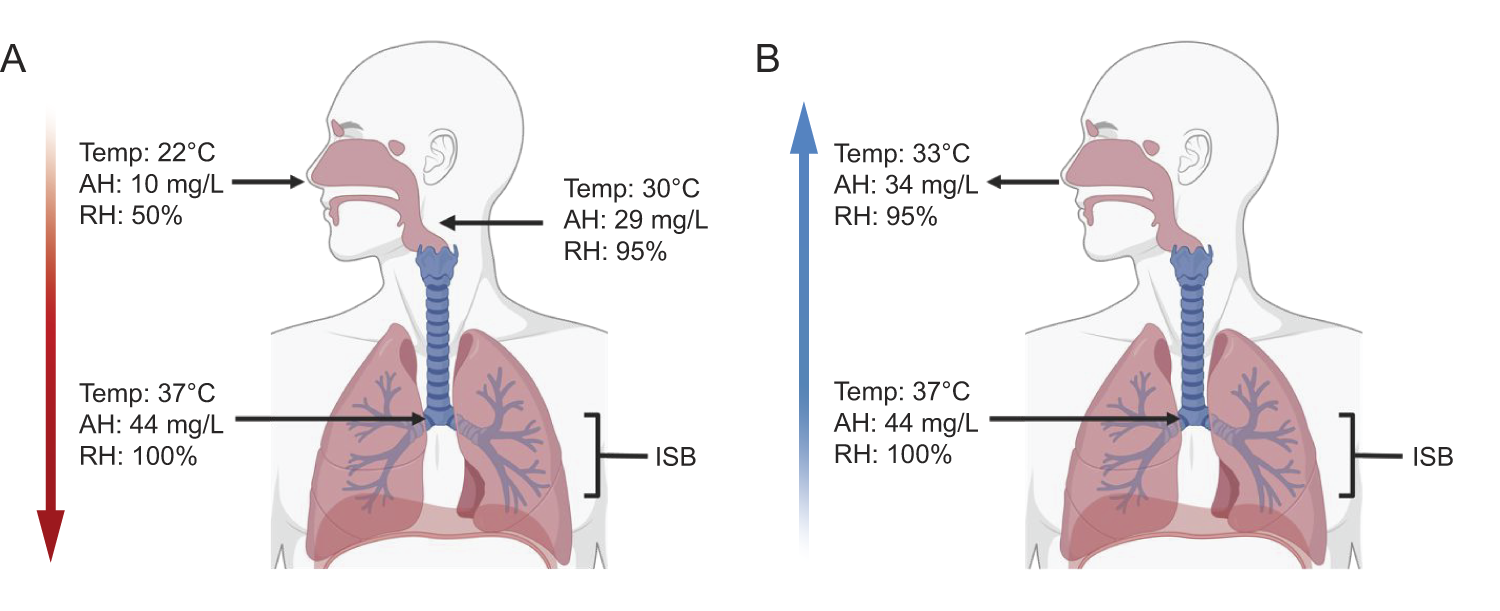
The function of the upper and lower airways as a physiological heat and moisture exchanger during inspiration
Artificial airways, mechanical ventilation, and inspired gas conditioning
Instrumentation of the airway (eg, endotracheal intubation) bypasses the proximal mechanisms that support the conditioning of inspired gases, altering the function of the airway (Fig. 3). Additionally, inhalation of cold and dry gases disrupts the normal physiological function of the airway and contributes to iatrogenic heat, or energy, loss in critically ill patients.13–15 Current guidelines from the American Association for Respiratory Care suggest using passive or active devices that meet the basal physiological requirements for proper airway function during invasive ventilation. 16
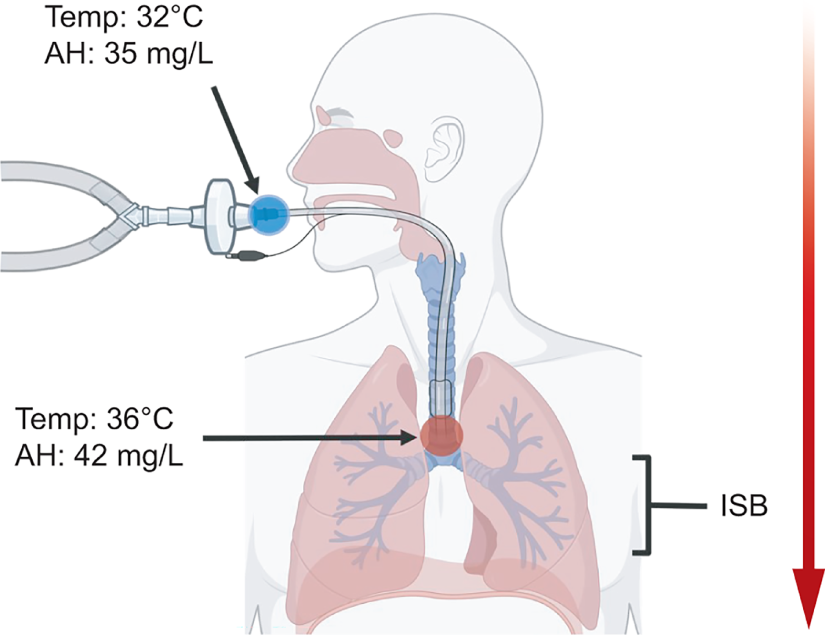
The alterations of the airway’s function related to inspired gas conditioning during endotracheal intubation in a normothermic patient. Moisture and heat are scavenged from the expired gas on the patient’s side of the heat and moisture exchanger (blue dot). This gas is available during inspiration and at physiologically appropriate temperature and humidity at the isothermic saturation boundary (red dot). Values for temperature and absolute humidity are approximations from Lellouche et al. 17 AH, absolute humidity; ISB, isothermic saturation boundary.
Appropriate airway physiological requirements can be met by using devices such as heat and moisture exchangers (HME) placed proximal to the endotracheal tube (ETT) or heated humidifiers (HH) in the inspiratory limb of the mechanical ventilation circuit. Regardless of the device used to condition inspired gases during mechanical ventilation, the minimum standards for gas humidification devices vary. According to the American Association for Respiratory Care, the chosen modality must be able to supply, at minimum, an AH of 30 mg H2O/L, a gas temperature of 34°C, at 100% RH. 16 Of note, these thresholds are rarely achieved during independent HME testing. 17
Passive and active devices
Passive devices
Passive humidification uses HMEs—hydrophobic or hygroscopic devices—that capture exhaled heat and moisture for delivery with the next inhaled breath. Because HMEs do not actively add heat, their performance depends on the patient’s exhaled temperature, which dictates the maximum water content of the gas. Noteworthy, HME performances are heterogeneous and often insufficient for normothermic and hypothermic patients.17,18 That said, hypothermia is commonly a contraindication for HME use.16,18
Active devices
Active HH are effective for providing optimal humidity levels in ventilator circuits regardless of patient characteristics. However, HH may provide excess free water to the lower airway in a hypothermic subject, although there is limited evidence identifying adverse outcomes related to this phenomenon.18,19 Despite perceived consistency, ambient and ventilator outlet temperatures can alter the performance of active humidification devices (ie, higher ambient and ventilator outlet temperatures reduce the AH provided to the patient) and provide inadequate conditioning of inspired gases.20,21 Regardless, active humidifiers are often preferred over passive humidifiers in situations where consistent levels of humidity are required, such as low-tidal volume ventilation, excessive secretions, or when the patient’s condition increases the risk of airway desiccation. 16
Total Body Heat Loss and Gain via the Airway
To conceptualize the role of AH, inspired gas temperature, and the thermodynamic properties of the airway concerning heat transfer during water evaporation or condensation, the following equation (Equation 1) illustrates the transfer of heat within the airway while breathing ambient air and air that is conditioned to physiologic values:
The equation above, derived by Ralley et al
22
represents the additive effect of the heat transfer by gas and because of water in the airway. The heat transfer of gas is represented as
From a clinical perspective, assuming a 75 kg patient in respiratory distress receiving mechanical ventilation with a tidal volume of 450 mL and breathing frequency of 22 breaths/min, receiving room temperature dry gas (22°C with 50% RH and 10 mg H2O/L of AH) without conditioning, the humidity deficit can be calculated from Equation 1. The body’s metabolic cost to condition the gas to 37°C with 100% RH and 44 mg H2O/L of AH is illustrated as follows: Parameters:
Based on this calculation, the patient utilizes approximately 62 kJ/h to appropriately condition the dry room temperature gas supplied via mechanical ventilation through the endotracheal tube. Assuming a basal metabolic rate of approximately 300 kJ/h, 21% of heat is lost via the airway in this scenario.
To contextualize the rate of temperature change (ΔΤ), the amount of heat transfer (Q), mass of the patient (m), and specific heat of tissue (c) must be known to utilize the following equation (Equation 2):
Utilizing Equation 2, if the specific heat of tissue is approximately 3.47 kJ/kg,
23
the respiratory heat loss in this 75 kg patient will decrease total body temperature by 0.24°C/h; however, the average specific body heat of tissues is debated.
24
Relative to normal breath parameters (ie, V˙E of 7.0 L/min of conditioned-inspired gas), total respiratory energy loss is approximately 10–12% of the total heat produced by the body at rest. Breathing dry air increases these energy losses as there are greater evaporation rates and heat exchange in the respiratory tract.
If the same patient presented in this theoretical clinical scenario developed mild hypothermia (ie, ≤ 35°C), inspired gas heated to 45°C would provide the following change in body temperature (from Eq. 1):
Considering the same metabolic conditions as previously described, it would take 7 hours to increase their body temperature approximately 1°C (rate of 0.15°C/h) if receiving inspired gas conditioned to 45°C at 100% RH (from Equation 2).
Thresholds for inspired gas temperature are not clear in human subjects concerning airway injury, 25 but there are currently no available devices in the United States that will deliver inspired gas >45°C, regardless.
Despite their relevance, the presented theoretical scenarios are extreme and do not account for inspiratory and expiratory flows, air flow dynamics (ie, laminar or turbulent gas flow), water evaporation rates across the lung, or other sources of body heat loss (eg, radiation, convection, conduction, and evaporation), or the logarithmic relationship between heat transfer and the rate of temperature change. For comparison, Table 1 lists other thermoregulation techniques and associated rewarming rates that are often used in acute and critical care settings. Given the previous theoretical scenarios, the thermoregulation techniques listed in Table 1 appear more effective when compared with utilizing the airway as a target for rewarming or maintaining normothermia.
Common rewarming techniques for hypothermic patients in acute and critical care settings
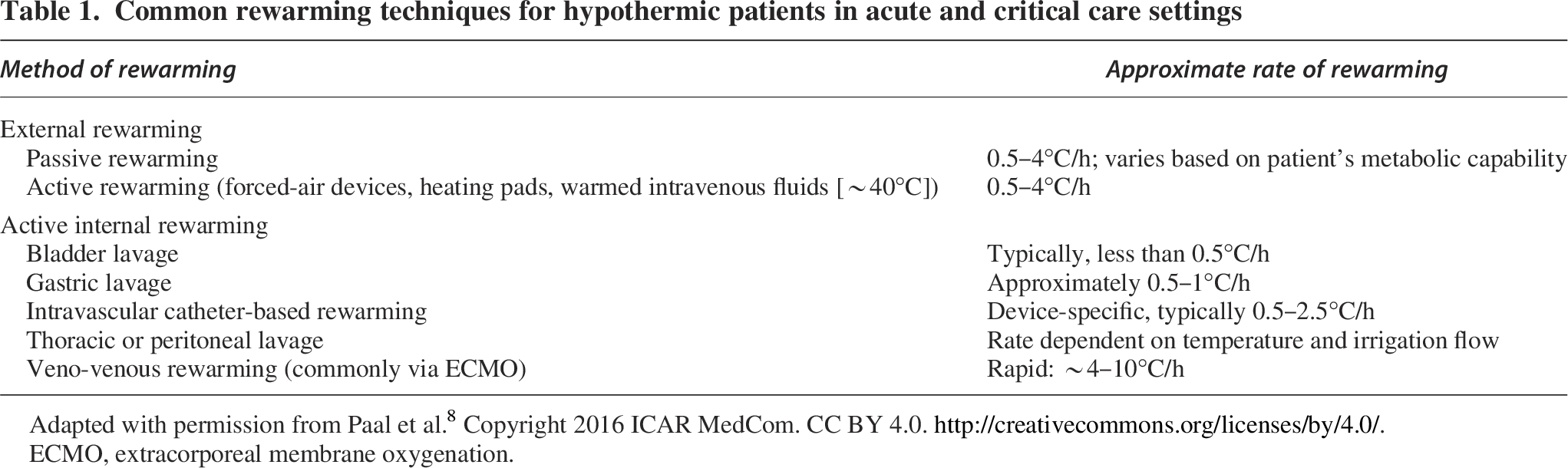
Adapted with permission from Paal et al. 8 Copyright 2016 ICAR MedCom. CC BY 4.0. http://creativecommons.org/licenses/by/4.0/.
ECMO, extracorporeal membrane oxygenation.
Rewarming Through the Airway: Methods and Outcomes
Theoretically, the amount of heat energy that conditioned-inspired gases can supply through an artificial airway is insufficient to increase total body temperature from a hypothermic state, as previously discussed. However, in practice, thermoregulation via the airway is of interest in perioperative and trauma-related patient populations.
Perioperative hypothermia
Hypothermia is a concern in the perioperative setting, where patients are vulnerable to reduced body temperature because of exposure and anesthetic effects. 26 There are 2 phases of heat loss in the perioperative period. The first phase consists of the redistribution of heat from the body’s central compartment to the periphery because of the peripheral vasodilation caused by the administration of anesthetic drugs. The second phase contributes to the most heat lost in the perioperative period because of body heat loss to the environment through convection, evaporation, conduction, and radiation. Anesthetics independently disrupt the physiological thermoregulatory response (ie, vasoconstriction and maintenance of core body temperature). Perioperative hypothermia is further exacerbated by low ambient temperature, administration of room temperature fluid/blood products, use of room temperature irrigation fluids, open abdominal and thoracic cavities, and intra-/postoperative blood loss.27–29
The consequences of perioperative hypothermia are grave and contribute to poor outcomes in surgical patients. Poor outcomes associated with perioperative hypothermia consist of excessive intra/post-operation blood loss, surgical wound infection, delayed emergence from anesthesia, increased incidence of mortality, postoperative shivering, and other events that reduce the likelihood of successful recovery from surgical procedures.30–34
Measures to counteract these adverse effects are taken to maintain normothermia, such as using warming blankets, warmed intravenous and irrigation fluids, and forced-air devices to regulate the patient’s temperature. Although the use of temperature monitoring and warming are integral components of perioperative care thermoregulation, the incidence of hypothermia in the perioperative period remains high.15,35
Perioperative temperature management via the airway
Providing heated and humidified gas to the airway to maintain normothermia in the perioperative space has been explored. Still, there is a lack of consensus on providing physiologically conditioned gases after airway instrumentation as a reasonable adjunct intervention for thermoregulation.36,37 The inherent heterogeneity of patients, equipment, temperature measurement sites, procedural conditions, and relevant outcome measures that fall under the perioperative umbrella is represented in the focus of this review (Table 2).
Clinical studies evaluating active airway humidification and core temperature outcomes in perioperative and critical care settings
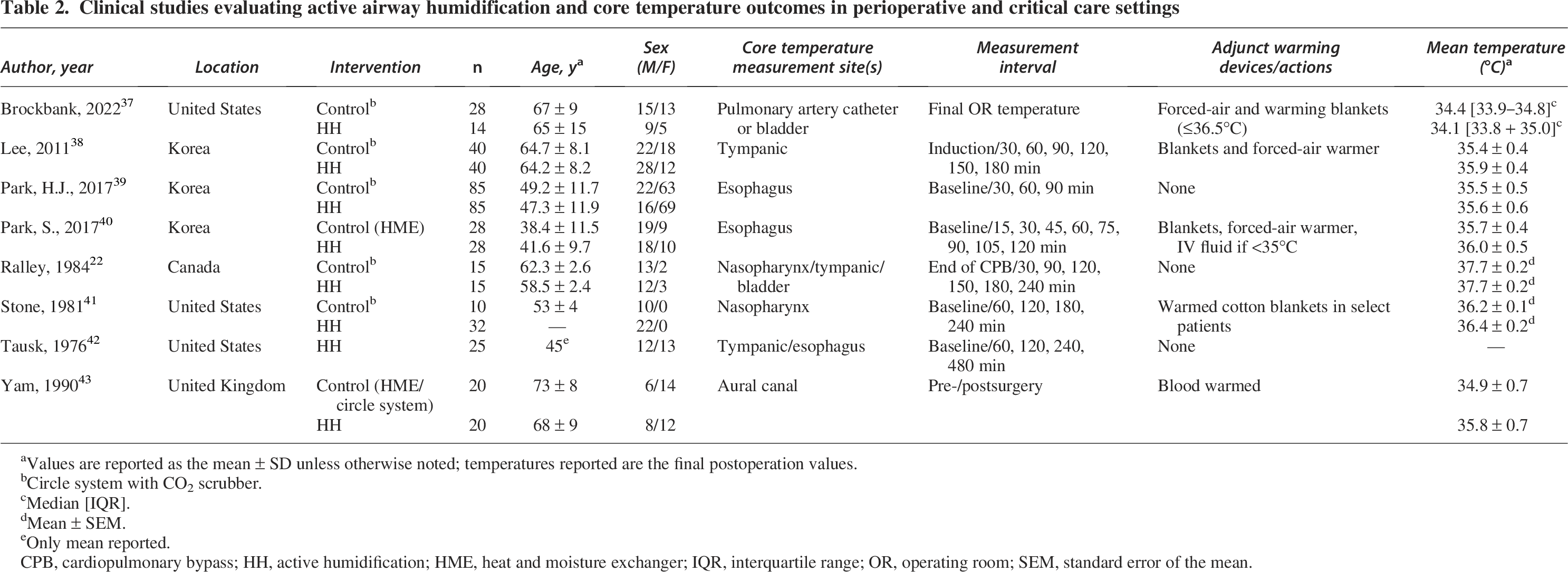
Values are reported as the mean ± SD unless otherwise noted; temperatures reported are the final postoperation values.
Circle system with CO2 scrubber.
Median [IQR].
Mean ± SEM.
Only mean reported.
CPB, cardiopulmonary bypass; HH, active humidification; HME, heat and moisture exchanger; IQR, interquartile range; OR, operating room; SEM, standard error of the mean.
Active humidification versus circle systems
Early investigations in the perioperative space commonly utilized anesthesia circle systems, which are closed circuits that recycle gases exhaled by the patient after CO2 has been removed, as the control when comparing the efficacy of thermoregulation with active humidification systems. The requisite CO2 absorber provides a degree of inspired gas conditioning similar to that of a poor-to-moderate-performing HME and theoretically plays a role in body heat loss because of airway desiccation.17,44
Studies dating back to the mid-20th century highlighted the functionality of delivering physiological levels of heat and humidity to anesthetized patients undergoing surgical procedures and thermoregulation. In 1976, Tausk et al reported the effectiveness of a prototype active humidifier in maintaining normothermia in 25 subjects receiving lengthy surgical procedures (mean = 9.0 h). 42 When supplying inspired gas to the airway at 37°C and 100% RH, 56% of the subjects had a measurable increase in body temperature, 20% maintained normothermia, and 24% had a decline in body temperature when measured from rectal, esophageal, or tympanic sites. 42 All subjects in the work by Tausk et al received some type of external thermoregulatory support in the form of draping or covering of exposed nonsurgical sites.
Within the decade of the work by Tausk et al, Stone et al published similar results when utilizing an active humidification system delivering inspired gas heated to physiologic levels in 45 surgical subjects with a procedure duration ≥3 h. 41 Subjects receiving heated/humidified gas throughout the surgical procedure did not have a clinically important drop in body temperature or an incidence of hypothermia when compared with the use of a circle system anesthesia ventilator. 41 The findings by Stone et al were consistent regardless of the presence or absence of other warming adjuncts (ie, warming blankets and body surface coverings); however, a nasopharyngeal temperature probe was utilized to trend body temperature changes, and more importantly, clinical advantages other than temperature stability were not evaluated. Nasopharyngeal and esophageal temperature probes may reflect changes secondary to the proximity to the inspired gas.
Findings concerning the reduction or maintenance of core body temperature were corroborated in contemporary work by Park et al, 39 which demonstrated the maintenance of normothermia in subjects undergoing an elective thyroidectomy receiving HH-inspired gas. 39 Compared with a group receiving conventional treatment, the HH group had a statistically significantly higher core temperature at 30 and 60 min of procedure time via tympanic membrane measurements. 39 Despite this core temperature difference, both groups were moderately hypothermic throughout the procedure, and it is not clear if there was a rewarming effect beyond 90 min. 39 Furthermore, a thyroidectomy does not require exposure of large areas of the body, so the reduction of convective, radiation, evaporative, and conductive heat loss is limited in the work by Park et al. The lack of contribution of these common sources of heat loss during surgical procedures may confound the impact of HH on body temperature regulation.
Active heat and humidification versus heat and moisture exchanger
Heat and moisture exchangers are often considered an energy-saving device and presented as an inexpensive tool to condition inspired gases in mechanically ventilated patients. An HME does not create heat but instead retains a portion of the exhaled heat from the patient to condition the inspired gas. Under this principle, the use of an HME during surgical procedures as a device to prevent unintended hypothermia has been featured in multiple investigations.
In a cohort of 60 elderly subjects receiving total hip arthroplasty, Yam et al demonstrated that an HME did not provide any clinical or statistically significant retention of total body heat compared with subjects without artificial humidification (ie, circle system alone). 43 Comparatively, net tympanic temperature change with HH averaged −0.3°C at the end of the procedure. Groups with only the CO2 absorber in the circle system or HME had net end-procedure tympanic temperature of −1.3°C and −1.2°C, respectively, thus demonstrating the HH attenuating heat loss. 43 The duration of the surgical procedures for all subjects was approximately 2 hours, so there was an appropriate duration to incorporate the effect of anesthesia induction, patient preparation, and body surface area exposed to the environment.
A similar investigation was performed by Park and colleagues and found no statistically significant differences in core body temperature 2 hours postanesthesia induction between HME and HH groups. 40 Despite this, the HME group had a greater drop in core body temperature versus the HH group, albeit not clinically important, and the study was underpowered. 40 From esophageal temperature measurements, approximately 64% of subjects in the HME group were hypothermic during procedures compared with the HH group (54%). 40
Regardless of the prevalence or incidence of hypothermia in the perioperative milieu, there is evidence of attenuation of the magnitude of the core temperature drop when HH is utilized versus HME or a circle system during surgical procedures ≥2 h.38–41,43
Maintenance of body temperature and perioperative outcomes
The use of HH to reduce the general incidence of perioperative adverse events is not well investigated. Still, there is a modicum of evidence that HH may reduce the incidence of perioperative blood loss and transfusion requirements in addition to other strategies of thermoregulation. 38 Lee et al demonstrated 38 the reduction of transfusion requirements in a group of 80 subjects without evidence of pre-enrollment coagulopathy receiving posterior lumbar spine fusion. Forty subjects were allocated to an HH group, with the remaining subjects managed on a conventional anesthesia circuit (ie, circle system).
In the group receiving HH, the mean intra-operative blood loss was significantly lower than in the control group; 25% of subjects in the HH group required transfusion of blood products in the perioperative period compared with 57% of those in the control group. 38 There was a nadir tympanic temperature of 35.9 ± 0.4°C in the HH group at 120 min, which was constant until 180 min of measurement. A core body temperature trend was present in the control group with a temperature of 35.5 ± 0.5°C at 120 min and a nadir of 35.4 ± 0.5°C at 180 min. 38 Despite the findings by Lee et al, there was no difference in postoperative blood loss or hospital stay between the control and HH groups.
Trauma, Hypothermia, and Thermoregulation via the Airway
Thermoregulation of trauma patients may serve to improve outcomes within the context of reversing the trauma triad of death. 45 The lethal triad of metabolic acidosis, hypothermia, and coagulopathy conspires to exacerbate one another and is prevalent in severe trauma cases. 45 Of the pillars of the triad, hypothermia is a strong predictor of mortality in trauma, with a 2.7-fold increase in the odds of death at 24 hours. 46 Furthermore, life-saving efforts for trauma cases exist in the causal pathway of hypothermia (ie, intubation, massive blood loss, exposure, patient transport anesthesia, surgical procedures).2,47
Body temperature loss begins at the point of the trauma-related event and may remain tenuous until postsurgical intervention. 48 In the prehospital environment, polytrauma, Glasgow coma scale ≤8, motorcycle/bicycle accidents, volume of fluid administration during transport, time of day, prolonged preclinical transport, and environmental conditions are prominent risk factors for the development of hypothermia for trauma cases presenting to the emergency department. 47 Despite hypothermia presenting a predictor of death in trauma, there has been limited development of active rewarming equipment used to support thermoregulation in the prehospital setting. As such, investigations concerning temperature regulation of trauma cases in prehospital, austere, and combat environments via the airway are also limited.
Rewarming techniques supported by the Wilderness Medical Society (WMS) for the treatment of out-of-hospital hypothermia include the use of HH-inspired gas, but it is unclear if this suggestion considers a bypassed upper airway. 49 Regardless, this intervention is not suggested as a singular approach to rewarming the hypothermic patient by the WMS, as there is limited evidence available in human subjects that supports rewarming via the airway as an effective intervention for correcting hypothermia in the prehospital environment.50–52 In fact, when compared with shivering, inspiration of HH gas was not as effective a rewarming as shivering in exposure-related hypothermia. 51
Technological Developments of Thermogenic Airway Adjuncts
Although there is a paucity of evidence to support the use of HH as a sole means of thermoregulation in the hypothermic trauma patient, there has been a recent introduction of devices targeting the development of airway rewarming devices that can be deployed during prolonged casualty care in military applications. These advancements suggest a renewed interest in the airway’s role as an alternative pathway for thermoregulation in critically ill patients. In 2022, Stevens et al presented a prototype device that is a battery-powered thermogenic airway adjunct to minimize heat loss via the airway postinstrumentation in a trauma, prehospital, or battlefield application. 53 Although there are no data confirming the efficacy of this device in human subjects, the development of such technology circumvents the cumbersome and unrealistic application of heated passover humidification devices in the context of a prehospital hypothermic trauma patient. 54
Summary
The excessive incidence of hypothermia in the perioperative and prehospital trauma milieu drives interest in utilizing the airway as a potential conduit for thermoregulation. Reduced equipment burden and unencumbered accessibility provided by utilizing the airway as a target for thermoregulation may be appealing in the prehospital and perioperative environments, respectively. However, the heterogeneous body of literature concerning patient populations, procedures, and applications using passive or active humidification devices for rewarming does not appear to be efficacious as an independent intervention.
The gestalt of thermoregulation should encompass interventions to reduce total body heat loss from multiple sources. Preventing total body heat loss because of conduction, convection, radiation, and evaporation from the mechanically ventilated patient, in addition to providing heated and humidified inspired gases at physiologic conditions (37° and 100% RH) to support thermoregulation, appears to be the most reasonable approach. The use of heated humidification to actively rewarm a hypothermic subject is not supported by the evidence.
Footnotes
Author Disclosure Statement
Dr. Ring serves on the editorial board forRespiratory CareDr. Lellouche received research funding from different companies working in the field of humidification to evaluate HH/HME (Fisher & Paykel, Vincent Medical, Dräger, Sedana). Development of the VentilO Application (implementation and optimization of protective ventilation—dead space), partly financed by Fisher & Paykel. Consultancy Medtronic: Series of presentations on humidification during respiratory support (Medtronic medical education program). Mr. Chatburn serves on the editorial board for Respiratory Care is a consultant for IngMar, Ventis, Promedic, and University of Cincinnati. Dr. Goodman has nothing to report. Mr. Branson discloses a consulting relationship with Lung Pacer LLC and is the Editor-in-Chief of R
Funding Information
No funding was received for this article.