Abstract
Keywords
Case Report
A previously healthy 21-year-old man presented to an outside hospital with sudden lower extremity weakness, nausea, and vomiting after consuming two alcoholic beverages the prior evening. Upon waking, he experienced nausea, vomiting, and painless paresis in his upper and lower extremities, with 2/5 proximal strength and preserved distal reflexes. Sensation was intact. Cervical spine MRI ruled out spinal cord compression. Initial potassium was 2.0 mEq/L (normal: 3.6-5.1), and EKG showed U waves. He received 60 mEq IV and 40 mEq enteral potassium. Agitation, tachycardia (130 beat per minute), and further proximal weakness (1/5) developed, leading to ICU admission. He experienced worsening tachypnea, weak cough, and difficulty managing secretions, requiring emergency intubation.
The patient was airlifted to our neuro-ICU with serum potassium rapidly rising to 6.8 mEq/L (see Figure 1). To lower potassium, 3 rounds of insulin and dextrose were administered within 8 hours. Metabolic panels and EKGs were assessed hourly. Vasopressors were employed to manage hypotension from potassium-wasting diuresis. Strength improved as potassium normalized. Electrolyte and treatment timeline: Potassium levels and therapies administered during the first 40 hours after patient presentation to medical care is shown. The blue shading represents normal laboratory potassium range. Therapies and the time they were administered relative to time from presentation are shown. Abbreviations: D50: 50% IV dextrose; IV: intravenous; PO: oral; PTU: propylthiouracil.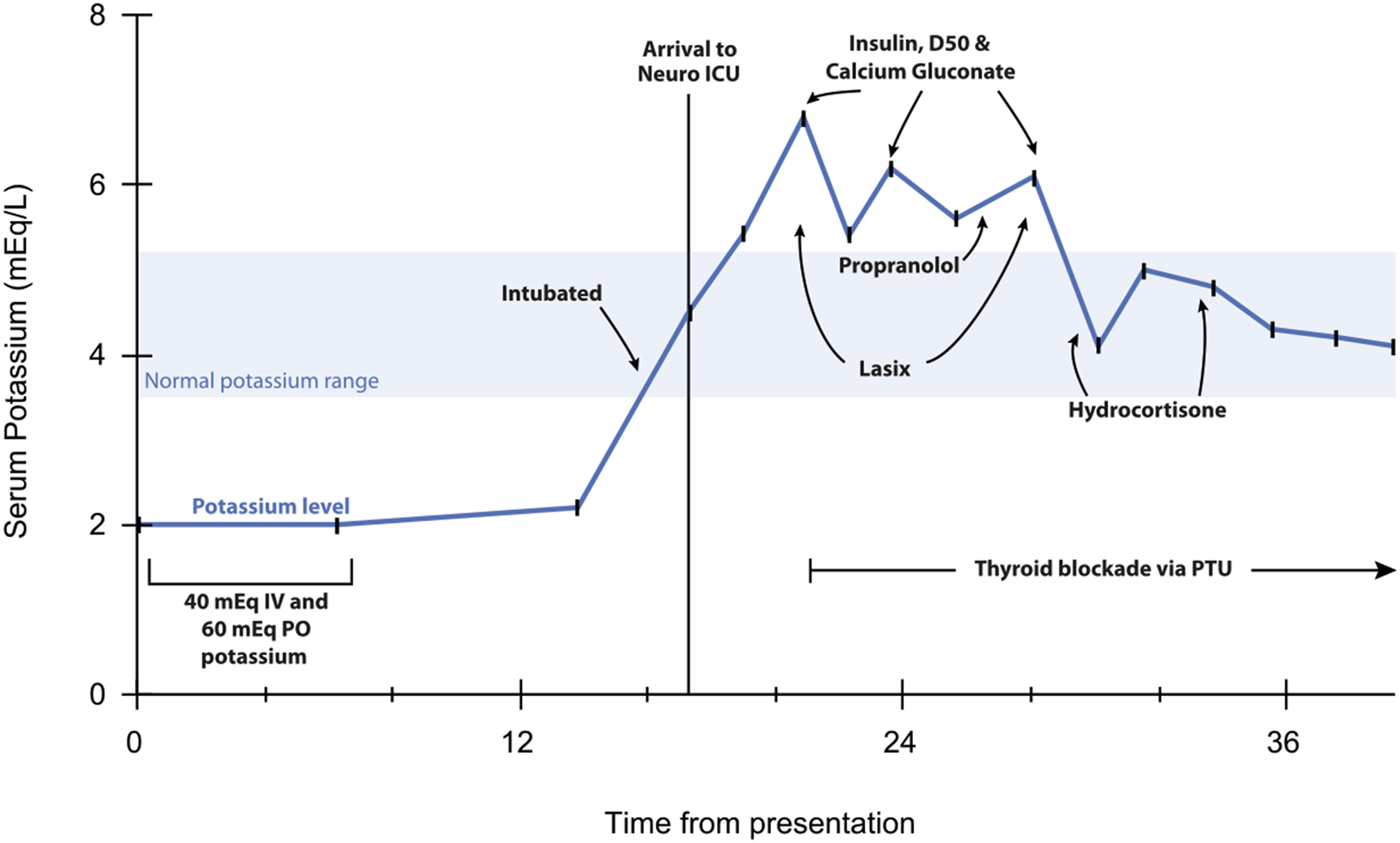
Thyroid function tests confirmed thyrotoxicosis: undetectable TSH, elevated thyroxine (16.84 μg/dL, normal 4.0-11.7), and elevated triiodothyronine (2.04 ng/mL, normal 0.87-1.78). Treatment included propylthiouracil, propranolol, and hydrocortisone. Antibody testing later showed elevated thyroid peroxidase and TSH receptor antibodies. The patient had hypoxemia and multifocal pneumonia from SARS-CoV-2 and possible bacterial superinfection and was treated with antibiotics, remdesivir, and dexamethasone. He recovered and was discharged a few days later.
Post-discharge, he had 2 ER visits for leg soreness and weakness that resolved with potassium repletion. Outpatient genetic testing for hypokalemic periodic paralysis was recommended but declined due to cost. There was no family history of similar symptoms.
Discussion
We report a 21-year-old man with rapid-onset paralysis from thyrotoxic periodic paralysis (TPP), requiring intubation—an exceptionally rare TPP complication,1-3 whose treatment was complicated by severe rebound hyperkalemia. While TPP can resemble familial hypokalemic periodic paralysis (FHPP), distinguishing between the 2 is essential as management strategies differ. We discuss the key similarities and differences, along with evaluation and optimal management strategies for TPP.
Pathophysiology
Unlike TPP, which requires both a genetic predisposition and thyrotoxicosis, FHPP is a genetic channelopathy that does not require hyperthyroidism for symptoms to develop. Both cause episodic, self-limited muscle weakness. FHPP is autosomal dominant, caused by mutations in the CACNA1S and SCN4A channels, leading to altered muscle excitability during hypokalemia, whereas TPP is an acquired condition linked to thyrotoxicosis that typically affects East Asian men. 1 Various mutations may increase susceptibility to TPP including those of the inward rectifier potassium (Kir) channel genes, KCNJ2 and KCNJ18, and the GABA-receptor (GABRA3).1,4,5
Clinical Features and Evaluation
TPP often presents in the second to third decade of life. Attacks tend to diminish in frequency in the fourth decade and often abate by age fifty. 1 Patients may awaken with sudden onset of symptoms. Prodromal symptoms, if present, might include fatigue and muscle heaviness on the previous day.1,2 Proximal muscles and lower limbs are first and most often affected; ocular, bulbar and respiratory muscles are often spared except in severe cases.1,2,6 Hypotonia and areflexia are common findings in all hypokalemic periodic paralysis types.
FHPP and TPP share common triggers, including carbohydrate-rich foods, strenuous physical activity followed by inactivity, high salt intake, infection, stress, trauma, cold, alcohol, glucocorticoids and anesthesia (Figure 2 depicts physiology and triggers of TPP)1,3,7 Exercise and carbohydrate rich meals cause intracellular potassium shifts by triggering rises in epinephrine and insulin, respectively.
8
In TPP, thyroid hormone likely sensitizes the hypokalemic action of epinephrine and insulin.
9
Hyperthyroidism leads to hypokalemia. Thyroid hormone increases beta-adrenergic responsiveness causing increased transcription and activity of sodium-potassium ATPase pumps2,3 resulting in a relative shift of potassium ions intracellularly and decreased serum potassium. Hypokalemia is thought to cause muscle inexcitability through poorly understood mechanisms, including paradoxical depolarization.
1
Thyrotoxic periodic paralysis physiology and acute treatment. (A) Schematic depiction of physiologic processes that result in hypokalemia leading to muscle weakness. Black arrows depict processes that can worsen hypokalemia-related weakness. Red items depict treatments that can lead to improvement in hypokalemia-related weakness. Purple items depict external factors that can worsen hypokalemia-related weakness. Right center depicts an EKG with a red box around a U-wave which can be in hypokalemia. (B) Treatment algorithm for acute thyrotoxic periodic paralysis and possible rebound hyperkalemia. Abbreviation: TPP: thyrotoxic periodic paralysis.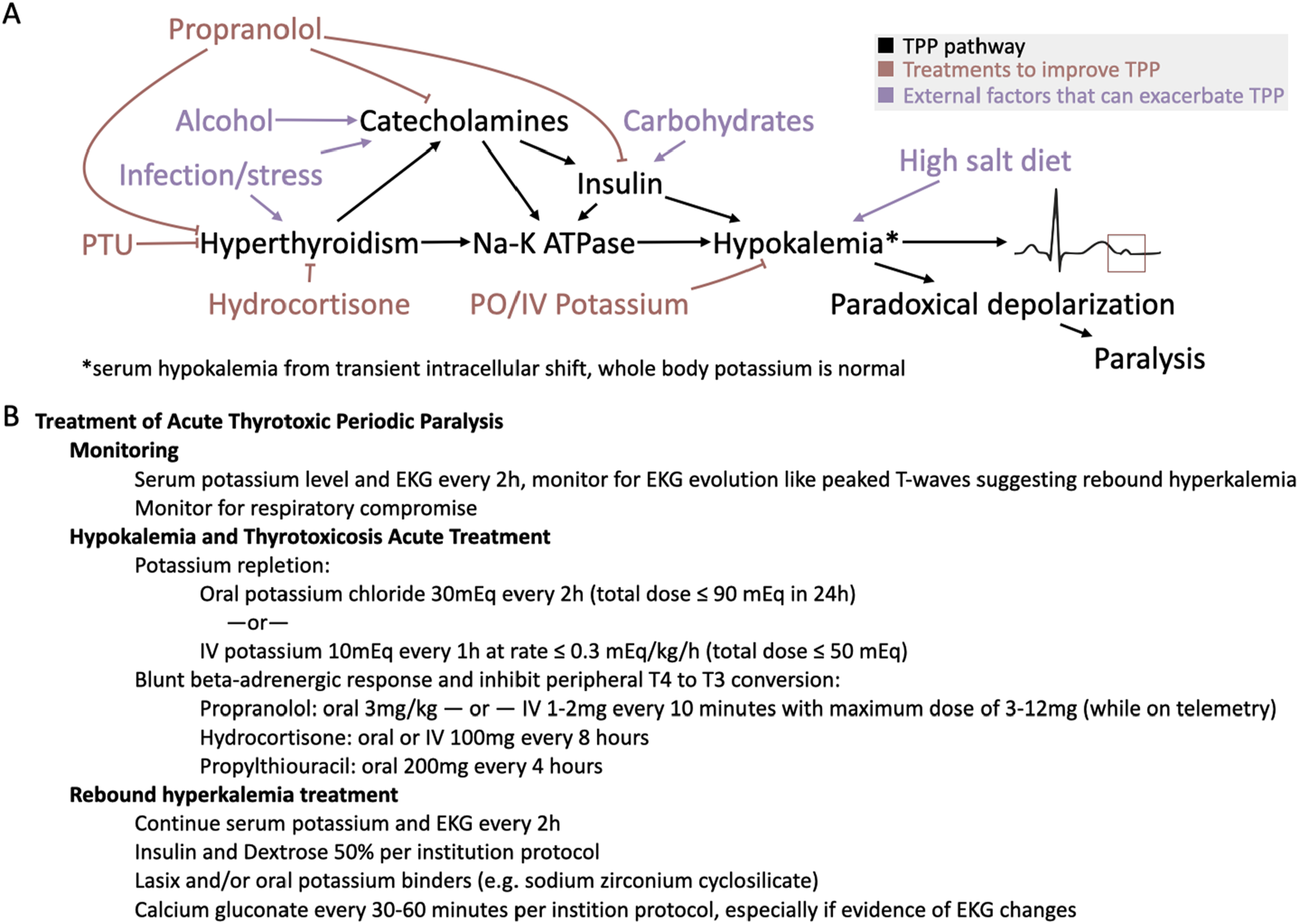
Weakness in both TPP and primary periodic paralyses are self-limited; however, attacks may be life-threatening, for example when there is severe bulbar weakness, severe respiratory muscle weakness, or when hypokalemia produces malignant cardiac arrhythmias. In these circumstances, acute TPP is a medical emergency requiring prompt diagnosis and treatment to prevent respiratory failure or cardiac arrest.1,6 If hypokalemic periodic paralysis is suspected, initial evaluation includes metabolic panels, thyroid function tests and an EKG to identify waveform changes related to electrolyte abnormalities. 10 Eventual genetic testing can be considered both for TPP and FHPP.
Management of TPP
Acute TPP management includes correcting serum potassium, treating thyrotoxicosis, and providing supportive care. Potassium replacement must be carefully titrated to prevent rebound hyperkalemia, as total body potassium is normal in TPP, with hypokalemia resulting from an intracellular shift.3,5,11 In this case, initial potassium was 2.0 mEq/L, and after receiving 100 mEq of potassium, the patient’s serum potassium rose to 6.8 mEq/L, necessitating potassium-wasting therapies and vasopressor support for diuresis-induced hypotension. This underscores the need for careful potassium repletion and diligent monitoring of potassium levels.
Absent cardiac arrythmias or respiratory compromise, potassium administration via the oral route is favored during acute attacks. Potassium chloride is thought to be best absorbed, and the chloride corrects metabolic alkalosis that is frequently present. 12 Assuming normal renal function, a suggested correction protocol is 30mEq of oral potassium every 2 hours until symptoms improve, but no more than 90 mEq of potassium in 24 hours. 13
If cardiac arrhythmias or respiratory compromise accompany the attack, then intravenous potassium is indicated with mannitol, rather than saline, as the solvent of choice. Dextrose should be avoided as it may exacerbate the attack through an exaggerated insulin response. Importantly, when administering intravenous potassium, a regimen of 10mEq every 60 minutes helps prevent dangerous overcorrection, and the total dose should not exceed 50mEq total.2,12 To minimize arrhythmia risk, the rate of IV potassium administration should not exceed 0.3 mEq/kg/h. 5
To block further intracellular shift of potassium, nonselective β-blockers blockers may be administered, to blunt the beta-adrenergic response and insulin release.9,11 Intravenous propranolol has been reported to terminate paralysis in symptomatic TPP, with dosing as high as 2 mg IV every 10 minutes, up to a total dose of 12 mg. 14 Oral propranolol at a dose of 3 mg/kg has also been used in TPP. 9
Thyrotoxicosis treatment involves medications that block the conversion of thyroxine (T4) to triiodothyronine (T3), including thionamides (methimazole, propylthiouracil), propranolol, and glucocorticoids. 15 Hydrocortisone (100 mg orally or IV every 8 hours) may improve outcomes by reversing adrenal insufficiency. 15
Acute management should include metabolic panels every 1-2 hours and continuous cardiac telemetry to monitor for arrhythmias. Serial EKGs reflect serum potassium trends, with hypokalemia causing U waves (Figure 3A) and peaked T-waves indicating hyperkalemia onset (Figure 3B). Hyperkalemia treatment involves potassium-wasting therapies with concurrent calcium gluconate to stabilize cardiac membranes. Calcium gluconate lasts 30-60 minutes and possible repeat doses might be needed until potassium-wasting therapies take effect. Electrocardiogram changes reflective of abnormal potassium levels. (A) EKG obtained with a potassium level of 2.2 mEq/L (normal, 3.6-5.1 mEq/L) showing the presence of U waves. (B) EKG obtained with potassium levels at 6.8 mEq/L showing the presence of peaked T waves. (C) EKG for comparison while potassium levels were normal at 4.1 mEq/L. The blue boxes to the right of (A-C) show magnified V2 leads for the same voltage and time windows.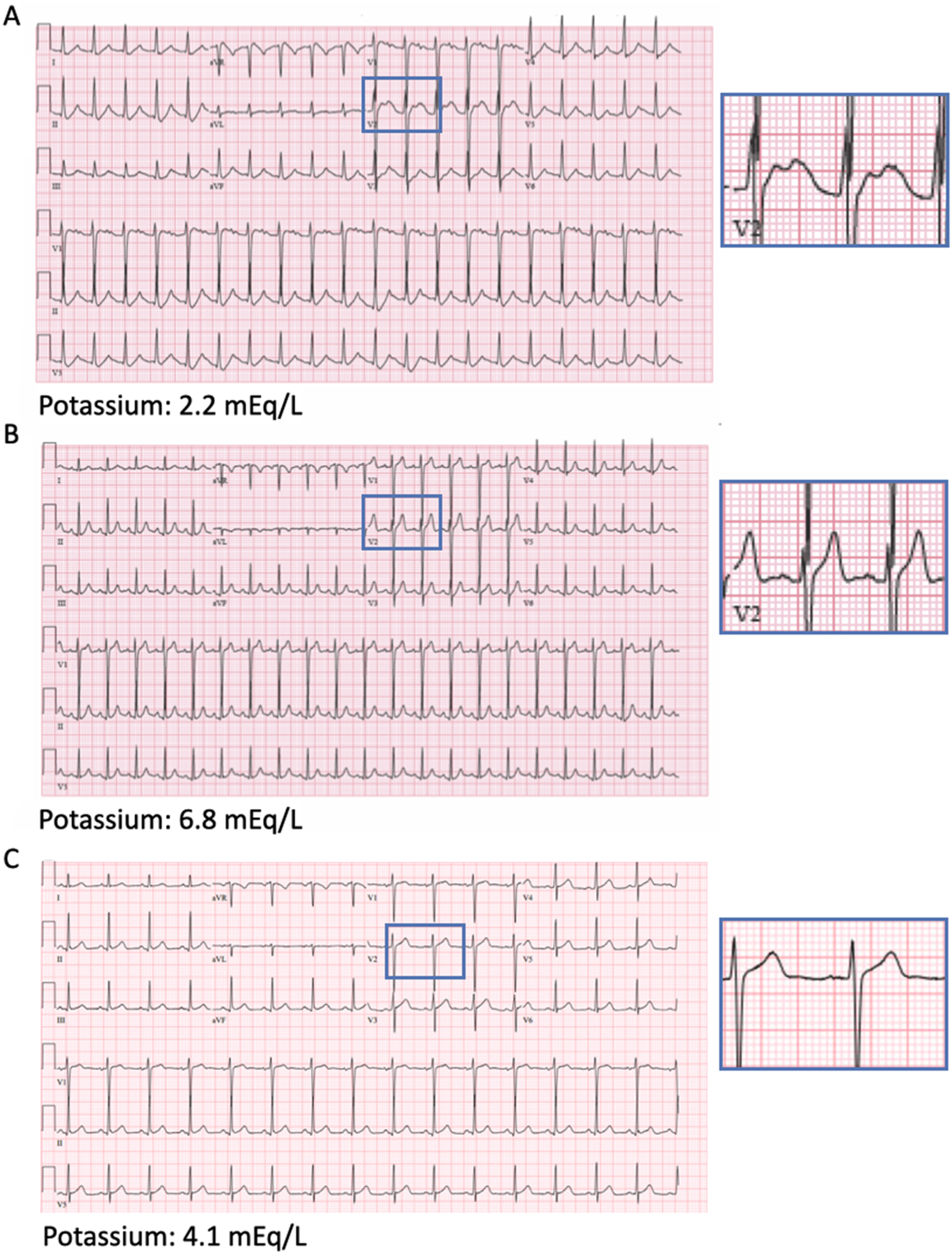
Conclusions
In periodic paralyses only a relative serum hypokalemia exists. We underscore the care necessary to titrate potassium administration and monitor potassium levels to avoid potentially life-threatening rebound hyperkalemia. Distinguishing TPP from FHPP is important given distinctive management priorities and pitfalls.
Take Home Points
• TPP and primary periodic paralyses are self-limited without lasting weakness, but urgent treatment is required if bulbar/respiratory weakness or arrhythmias occur. • Total body potassium is normal in TPP, thus post-treatment rebound hyperkalemia can occur as potassium shifts back extracellularly after an attack. • In acute TPP management, potassium correction should not typically exceed 90 mEq of potassium in 24 hours. • Use adjunctive nonselective β-blockers, thionamides, and mineralocorticoids to prevent further potassium shifts. • Peaked T-waves on EKG can be the first sign of hyperkalemia from overcorrection.
Footnotes
Author Contributions
Conceptualization: Thomas J. Pisano, Brandon Merical, Zehui Wang, Joshua M. Levine. Data curation: Thomas J. Pisano. Visualization: Thomas J. Pisano. Writing–original draft: Thomas J. Pisano. Writing–review & editing: Thomas J. Pisano, Brandon Merical, Zehui Wang, Joshua M. Levine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
