Abstract
Background/Objectives
There is currently no consensus regarding the optimal strategy for reversal of anticoagulation in life-threatening hemorrhage associated with factor XIa (FXIa) inhibitors.
Methods
For this clinical case report, informed consent was obtained from surrogate.
Results and Discussion
Here, we present the case of an 82-year-old female who sustained a large subdural hematoma after a fall. Her aPTT on admission was elevated at 90.4 s and remained persistently prolonged at 90.9 s 12-hour after receiving an adequate dose of 4-factor prothrombin complex concentrate (PCC). She was found to have received a factor XIa inhibitor in a clinical trial, and subsequently received recombinant activated factor VII (rFVIIa) 2 mg (45 mcg/kg) as a one-time dose, and tranexamic acid (TXA) 1 g intravenously for reversal given her intracranial bleeding in the setting of trauma complicated by recent factor XIa inhibitor use. However, given her clinical decline and high surgical risk, the patient’s family elected to withdraw care and she expired three days later. Reversal of FXIa inhibitors is challenging but may best be achieved using a combination of rFVIIa and TXA.
Practical Implications
Clinicians should consider administration of low dose recombinant activated factor VII (rFVIIa) in conjunction with an anti-fibrinolytic inhibitor such as tranexamic acid (TXA) for reversal of life-threatening hemorrhage in bleeding patients with exposure to novel factor XIa inhibitors that are currently in clinical trials.
Keywords
Introduction
Optimal reversal of anticoagulant agents that inhibit factor XIa (FXIa) is not currently known. Here we present a case of a life-threatening subdural hematoma in the setting of milvexian use for atrial fibrillation in a clinical trial.
Case Presentation
The patient was an 82-year-old female with a past medical history of hypertension, hyperlipidemia, and atrial fibrillation. She presented to an outside facility with a headache and facial droop two days after a fall. She had last taken her medications, including trial anticoagulant and daily aspirin 81 mg, the morning of outside hospital presentation. Non-contrasted CT head imaging demonstrated an acute right parietal convexity subdural hematoma with mixed hypodensity suggesting a chronic component (Figure 1). Maximum thickness was approximately 1.3 cm with 4 mm of midline shift and moderate effacement of the right lateral ventricle. She was then transferred to our tertiary care center for further evaluation and management.
Labs on admission revealed WBC 14.2, hemoglobin 11.2, platelets 252, sodium 132, hsTroponin-I 114 ng/L, PT 11.0/INR 0.98. APTT was elevated at 90.4 s after reportedly receiving 4-factor prothrombin complex concentrate (PCC), KCentra™ 50 units/kg, at the outside facility. The patient was initially conversational and oriented to person, place, time, and situation. Several hours later she developed neurological decline with progressive somnolence. Head CT was obtained showing stable right-sided subdural hematoma with cerebral edema and midline shift (Figure 1). Repeat labs drawn approximately 6 h after admission demonstrated persistently elevated aPTT of 86.1 seconds. She had rhythmic mouth and face movement, and EEG revealed several electrographic and electroclinical seizures characterized by right temporal LPDs evolving in frequency and amplitude with increasing sharply contoured theta activity, lasting for 1-3 minutes. Her seizures improved following a load of levetiracetam, but her clinical examination remained poor. CT head imaging showing a large R convexity subdural hematoma with edema and midline shift. (A) Initial axial and coronal (B) non-contrast CT head with acute mixed density 1.3 cm subdural hematoma with 4 mm of midline shift and moderate effacement of the R lateral ventricle. (C, D) Repeat non-contrast CT head with redistribution of blood products within the subdural hematoma after receiving 4-factor prothrombin complex concentrate (PCC), KCentra™ 50 units/kg.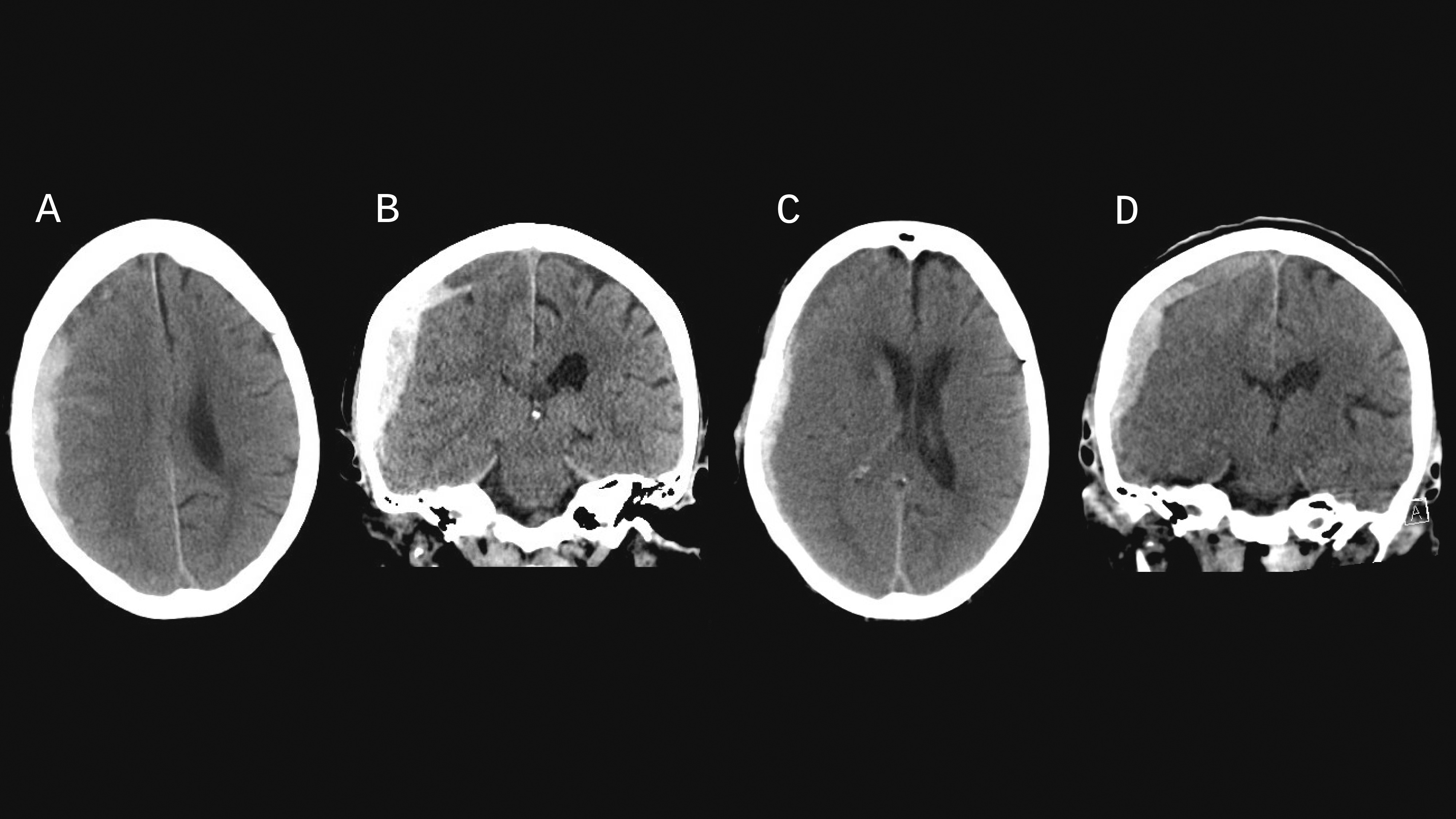
Hematology was consulted, and a laboratory evaluation revealed a persistently prolonged aPTT at 90.9 s (12 h from admission), fibrinogen 317 mg/dL, and hsTroponin I rising to 2273 ng/L. Common pathway coagulation factors II, V, X were all within normal limits. Factors VIII >70% (reference 56-186%), IX >46% (reference 58-138%) XI >36% (reference 70-159%), and XII >46% (reference 60-171%) were consistent with an inhibitory pattern due to interference with the intrinsic pathway of coagulation. A ROTEM was performed with all values within normal limits, except HEPTEM clotting time which was elevated at 243 s (reference 122-208 s).
Upon further investigation, it was discovered that the patient was enrolled in the LIBREXIA-AF double-blind randomized clinical trial to evaluate milvexian, a small molecule FXIa inhibitor, vs apixaban in patients with atrial fibrillation. The prolonged aPTT essentially unblinded the patient’s study drug, later confirmed to be milvexian.
Given potential neurosurgical intervention, the trial’s emergency hotline was contacted to determine if an immediate reversal protocol could be provided. General recommendations from the emergency hotline included consideration of tranexamic acid (TXA), recombinant activated factor VII (rFVIIa), and/or PCC. A brief literature search was conducted, and our hematologists recommended the patient be given rFVIIa 2 mg (45 mcg/kg) as a one-time dose and TXA 1 g IV every eight hours (she ultimately received only 1 dose of TXA). APTT was rechecked approximately 1 hour after administration of these agents and remained prolonged at 73.6 s.
In light of her imaging findings, seizures, and clinical decline, neurosurgical intervention was considered. A multidisciplinary discussion determined that she was high risk. This decision was predominantly based on comorbidities rather than bleeding risk, which could not be quantified with available diagnostics. Ultimately, given her age, comorbidities, and prior level of function, her family opted to pursue comfort-focused care and she expired three days later.
Discussion
Currently available anticoagulants such as heparins or direct oral anticoagulants function by directly or indirectly targeting the serine proteases thrombin or factor Xa (FXa). While inhibiting these targets is effective in preventing or treating thromboembolism, it also raises the risk of bleeding given their essential roles in hemostasis. Thus, guidelines for managing anticoagulant-associated bleeding recommend directly neutralizing or bypassing the inhibitory actions of the agent in question. 1 The need to balance anticoagulation with bleeding risk has motivated efforts to develop new antithrombotic agents with minimal impact on hemostasis. The foremost target is FXI/XIa. Animal studies and clinical experience with congenitally deficient patients suggest that FXI plays an important role in thrombosis but only participates in hemostasis in locations with pronounced fibrinolysis (e.g., oro-pharynx, GU tract). 2 FIX/XIa is thus a logical choice for targeted anti-thrombotic therapy.
While it was initially unclear whether the patient was taking milvexian or apixaban in the clinical trial, the prolonged aPTT (and failure to correct on a 1:1 mixing study) could not be explained by apixaban, regardless of PCC administration, suggesting the patient must have been on milvexian (later confirmed by unblinding). Other studies were also compatible with a circulating inhibitor, namely intrinsic pathway factor levels using one-stage aPTT-based assays (specifically, FXI, FVIII and FIX). Milvexian is primarily eliminated by the liver, with less than 20% of clearance occurring via renal excretion. 3 Though this patient did not have baseline renal disease, admission labs demonstrated CrCl <30; we would anticipate a longer half-life ∼18 hrs given this severe renal impairment compared to 13 hrs for normal renal function.
Patients with inherited FXI deficiency do not present with spontaneous bleeding, but may bleed after surgery or trauma; the severity of bleeding does not correlate with FXI levels. 4 Thus, inferring that pharmacological inhibition would similarly lead to increased bleeding after trauma, we felt that reversal was warranted. Prior to hematology consultation, reversal with PCC had been attempted. Realizing she had milvexian exposure, we recommended rFVIIa and TXA administration. Of note, although the ROTEM study was normal apart from HEPTEM, we did not consider this to be reassuring, as prior work in the setting of FXI deficiencies has found that thromboelastometry with whole blood specimens could not identify bleeder and non-bleeder phenotypes. 5 Similarly, the aPTT served as a marker of the continued presence of a FXI inhibitor in plasma in lieu of direct measurement of milvexian levels. Thus, the decision to reverse the effects of milvexian was based on clinical judgment.
This case highlights several important considerations. First, there is no evidence-based approach for the reversal of small molecule FXIa inhibitors in recipients experiencing major bleeding complications. Since it is highly protein-bound, it is assumed to be non-amenable to dialysis. A Phase 1 study (NCT04543383) investigated PCC and rFVIIa in healthy participants on milvexian although no results are available in the public domain. Secondly, as large-scale phase III trials of milvexian and other FXIa inhibitors are underway, the rate of major bleeding (while expected to be lower than with direct FXa inhibitors) remains to be established. Additionally, there is not currently a robust diagnostic strategy to identify bleeding propensity due to a FXI/FXIa inhibitor. One recent study in human plasma and whole blood in vitro found that the anticoagulant effects of milvexian as determined by aPTT clotting assays, thromboelastography, and kaolin-initiated thrombin generation assays were corrected by activated PCC or rFVIIa, but not PCC, rFIX, or rFVIII. 6 Pending further real-world data, one suggested reversal strategy is to use low‐dose rFVIIa and antifibrinolytic therapy. This recommendation is based on: a] limited pre-clinical data addressing milvexian reversal using low dose rFVIIa in a rabbit AV‐shunt model of thrombosis 7 ; b] in vitro modeling of hemostasis in the cell-based model of coagulation 8 ; and c] clinical experience derived from a small cohort of patients with inherited FXI deficiency complicated by FXI inhibitors. 9
It is especially important to note the recommended dose of rFVIIa in this scenario. 10 Consulting hematologists will be most familiar with the high doses of rFVIIa (typically 90-120 mcg/kg every 2-6 h, or up to 270 mcg/kg as single bolus) that are used to bypass the hemostatic defect in hemophilia A or B complicated by inhibitors to FVIII or FIX, respectively. Such high doses are needed to promote the direct but relatively inefficient activation of FX by FVIIa on the platelet surface in the absence of tissue factor. However, since the ‘intrinsic tenase complex’ (i.e. factors IXa, VIIIa and X) remains intact in FXI-deficient patients or patients on FXIa inhibitors, the goal is instead to modestly augment the TF-dependent conversion of FIX to FIXa which can then feed into the tenase complex to restore thrombin generation (as FIX can be activated by either TF-FVIIa or FXIa). This goal can be achieved at a much lower dose of rFVIIa (as low as 10 μg/kg), which should be administered in conjunction with an anti-fibrinolytic inhibitor such as TXA, which efficiently bypasses the inhibited antifibrinolytic role of FXIa. Of note, the higher doses of rFVIIa used for hemophilia have been associated with thrombosis in patients with FXI deficiency complicated by a FXI inhibitor. 8
Conclusion
Management of trauma-related subdural hematoma associated with administration of FXIa inhibitor therapy via clinical trial represents a clinical challenge, particularly given the lack of reliable markers for bleeding propensity. To our knowledge, this is the first published case of such a scenario. We hope that sharing our experience will assist neurohospitalists, critical care physicians, and hematologists in the management of affected patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
