Abstract
Background and Purpose
In patients with myasthenia gravis (MG), worsening of symptoms poses a risk of respiratory failure which can be precipitated by medication use. Although purported, the risks associated with administration of certain medications are not fully elucidated. Thus, clinical decision support involving a best practice alert was executed to caution providers of drug-disease interactions when ordering a potentially harmful medication. We performed an analysis of the alert overrides with subsequent medication exposure to determine the incidence of MG exacerbations.
Methods
This retrospective chart-review evaluated adult patients with MG at 2 large academic medical centers via electronic health records between November-2019 and November-2021 who received a medication following override of the clinical decision support tool. The primary outcome was proportion of patient encounters complicated by myasthenic exacerbations after potentially harmful medication administration. Secondary outcomes included changes in motor strength, length of stay, discharge disposition, unplanned level-of-care escalations, and changes to immunosuppressant therapy following medication administration.
Results
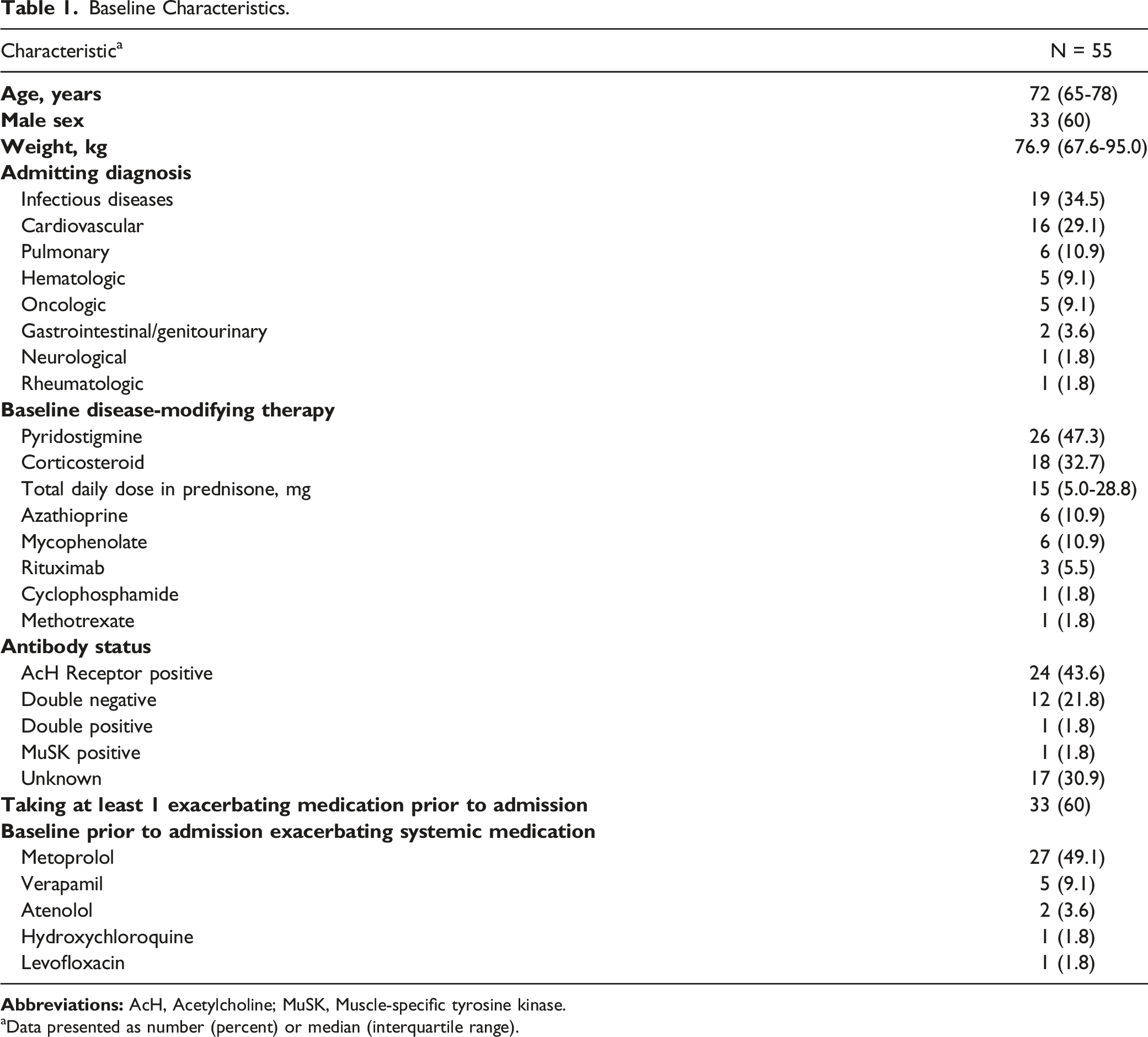
A total of 70 orders were assessed in 38 patients across 55 encounters. Medications administered during these encounters included macrolides, fluoroquinolones, β-blockers, calcium channel blockers, and magnesium sulfate. Exacerbation of disease occurred in 7 patient encounters (12.7%) and occurred after intravenous magnesium or intravenous labetalol. In 5/7 events, at least 1 other risk factor associated with a myasthenic exacerbation was present.
Conclusions
Of the medications reported to potentially worsen MG, intravenous labetalol and intravenous magnesium were the 2 agents associated with myasthenic exacerbations with a higher incidence in patients harboring additional risk factors.
Introduction
Myasthenia gravis (MG) is a chronic autoimmune disorder characterized by skeletal muscle weakness and fatigability with prevalence rates in the United States approximated at 20 per 100,000. 1 The function of ocular, limb, and respiratory muscles may fluctuate over time as antibodies can disrupt the nicotinic acetylcholine receptors throughout the neuromuscular junction. 1 Acute worsening of myasthenic symptoms poses a fatal risk of respiratory failure which can be precipitated by many factors including infections, surgery, pregnancy, and/or medications. 2 Available evidence to date examining clinical manifestations of drug-disease interactions in patients with myasthenia gravis is limited to case reports.3-7
Several classes of medications have secondary mechanisms of action at the neuromuscular junction that when administered to patients with pre-existing myasthenia gravis, can cause exacerbation of disease. 8 Common agents such as magnesium sulfate and calcium channel blockers decrease the activity of voltage gated calcium channels on presynaptic cholinergic neurons resulting in decreased vesicle fusion and acetylcholine exocytosis.6-8 Other medications such as β-blockers destabilize post-synaptic acetylcholine receptors by reducing receptor clustering; a crucial component in neuromuscular junction development. 2 Additional undiscovered mechanisms of action may be at play amongst many classes of medications contributing to worsening of MG symptoms upon administration.
In 2019, a clinical decision support tool was implemented across our health-system that alerted providers of potential drug-disease interactions upon medication ordering in patients with myasthenia gravis documented on their problem list. If overridden by the ordering provider, an inpatient pharmacist is similarly presented with the same warning upon medication verification (Supplemental Figure 1). We performed a clinical review to explore the consequences of medication exposure in patients with MG after override of the clinical decision support tool.
Methods
This retrospective chart review evaluated adult (≥18 years of age) patients with myasthenia gravis admitted to Massachusetts General Hospital (MGH) or Brigham and Women’s Hospital (BWH) between November-2019 and November-2021 who had received an inpatient oral (PO) or intravenous (IV) medication included in the clinical decision support tool (Supplemental Table 1). Medication administration events were excluded if (1) an exacerbation was present at time of admission prior to medication administration, (2) the patient was stabilized on the medication at baseline prior to hospital admission, or (3) the patient was receiving mechanical ventilatory support prior to medication administration. Patients were considered stabilized on a baseline medication if the agent was listed in their admission note as a home medication or in an insurance claim dispense record with evidence of use for 29 days or more. For patients stabilized on an identified medication at baseline, inclusion was only met if an additional exacerbating medication was administered, though the baseline medication was not analyzed for precipitation of an exacerbation.
The primary outcome was incidence of myasthenic exacerbations, defined as escalation of respiratory support following administration, initiation of intravenous immunoglobulin (IVIG) or plasma exchange for the treatment of an acute MG exacerbation within 96 hours of the administration event, negative inspiratory force (NIF) less than 20 cm H2O, and/or vital capacity (VC) less than 1 liter following administration. Respiratory status, NIF values, and VC values were analyzed the day before, day of, day after, and 2 days after the administration event. Patients were considered to have an escalation of respiratory support if their method of oxygen delivery or fraction of inspired oxygen (FiO2) increased compared to baseline within the first 48 hours following administration. The highest level of respiratory support (ie, room air, nasal cannula, positive pressure ventilation) within each 24-hour period was used for analysis. The number of doses administered was collected in all instances but only the days surrounding the initial dose administered were assessed for exacerbation. When the primary outcome was met, the Naranjo Adverse Drug Reaction Probability Scale was used to further assess likelihood of a drug-induced exacerbation. The Naranjo Scale is an instrument originally developed for use in clinical trials however, its utility has expanded beyond this to clinical practice. Probability is assigned via a score termed definite, probable, possible, or doubtful association with medication. 9 Additional risk factors that have been associated with MG exacerbations were collected including pregnancy, receipt of antibiotics for a confirmed active infection, and recent surgical procedure during the same hospitalization.
As a secondary outcome, we assessed changes in median extremity motor strength; a nursing documented score evaluated on each extremity by a 0-5 scale before and after medication administration (0: no visible muscle contractions, 1: visible muscle contraction with no or trace movement, 2: limb movement, but not against gravity, 3: movement against gravity but not resistance, 4: movement against some resistance supplied by the examiner, 5: full strength). 10 The closest set of scores within the 48 hours leading up to administration as well as the lowest set documented within 48 hours afterwards were used for analysis. Median motor strengths reported were calculated from both the median scores amongst all extremities and amongst all patients. Other secondary outcomes included length of stay, discharge disposition, unplanned level-of-care escalations (ie, transfer from an Internal Medicine service to an Intensive Care Unit [ICU]), and increased doses or new initiation of immunosuppressant therapy for MG following medication administration. Discharge summaries within the electronic health record were used to interpret discharge disposition.
Medications screened for exacerbating potential and prior to admission use included PO and IV formulations of macrolide antibiotics, fluoroquinolones, aminoglycosides, aminoquinoline agents, β-blockers, class 1a antiarrhythmics, non-dihydropyridine calcium channel blockers, and magnesium sulfate (Supplemental Table 1). Baseline MG disease-modifying therapy including pyridostigmine, corticosteroids, azathioprine, mycophenolate, cyclosporine, tacrolimus, cyclophosphamide, methotrexate, and rituximab were also collected. Patients were considered to have been exposed to rituximab if they had received an infusion within the previous 6 months. A waiver of informed consent was granted by the institutional review board.
Data Analysis
Patients evaluated for administration events across multiple hospital presentations were recorded as unique encounters. Data points were collected from patient electronic health records and were recorded using REDCap® (Research Electronic Data Capture). Analysis was performed using SPSS software version 28 (IBM SPSS Statistics, IBM Corporation, Armonk, New York).
Results
A total of 183 active orders for potentially exacerbating medications were identified. Of those, 70 (38.3%) medication administration events in 38 unique patients across 55 hospital encounters were included in the final analysis (Figure 1). The most common admitting diagnoses were for infectious disease related conditions (34.5%) followed by cardiovascular events (29.1%), (Table 1 and Supplemental Table 2). In 60% of the encounters, patients were noted to have been taking at least 1 systemic exacerbating medication prior to admission. IV magnesium sulfate was the most common inpatient medication administered (54.3%) followed by β-blockers (28.6%) (Table 2). In twelve encounters (21.8%), patients were noted to have multiple potentially harmful medications administered including combinations (2 or more) of ciprofloxacin, levofloxacin, labetalol, metoprolol, and IV magnesium sulfate (Table 3). Enrollment. Baseline Characteristics. aData presented as number (percent) or median (interquartile range). Inpatient Myasthenia Gravis Drug-Disease Medication Interactions Administered. Primary Outcome Results.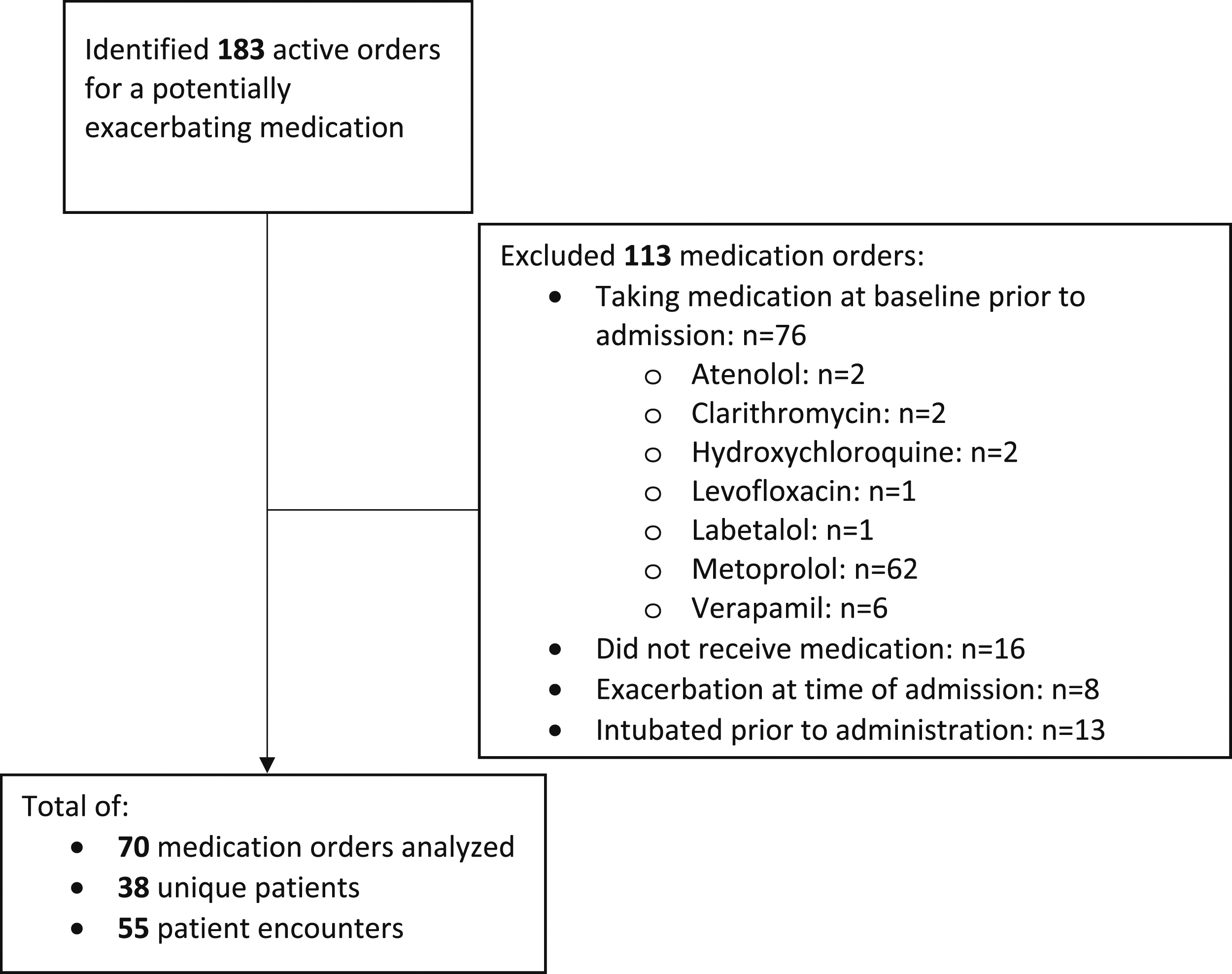
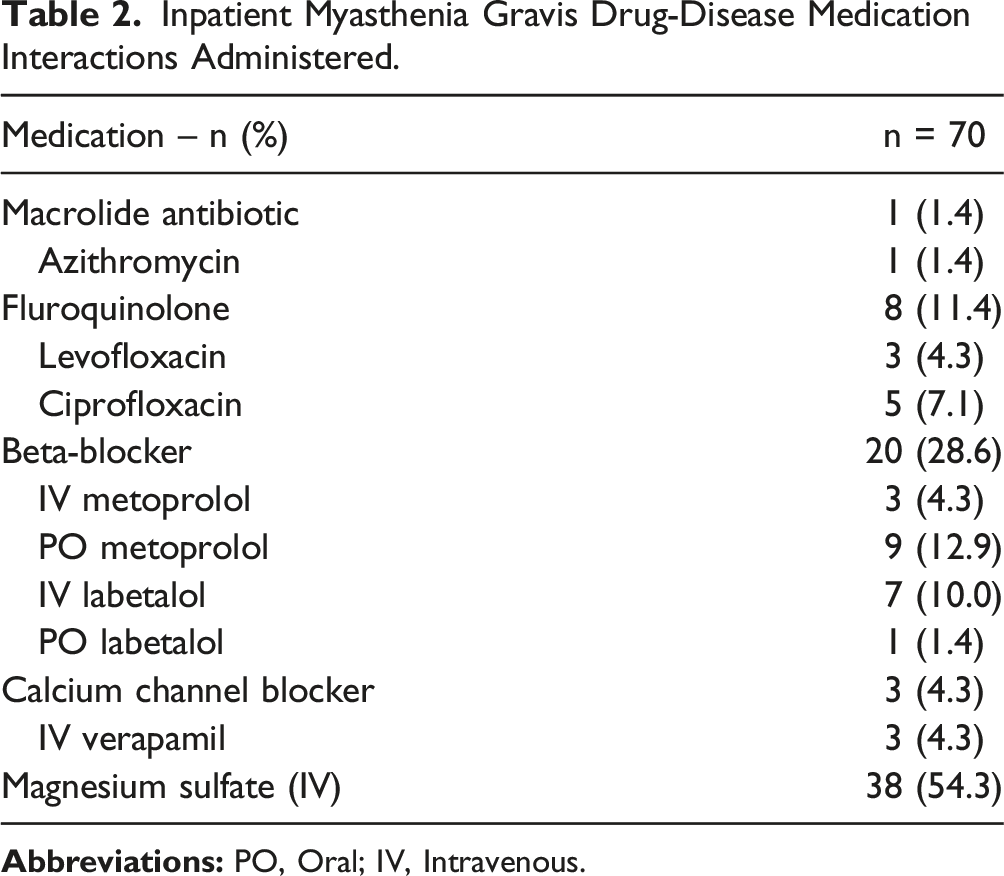

A myasthenic exacerbation occurred in 7 hospital encounters (12.7%), with all events occurring after a 2-gram infusion of IV magnesium sulfate for hypomagnesemia (median: 1.4, range: 1.0- 1.7 mg/dL) except for 1 exacerbation following 5 mg of IV labetalol. Independent confirmation of a myasthenia gravis diagnosis based on receptor positivity amongst all 7 encounters was undertaken. There were no instances of multiple exacerbations per encounter. Across all encounters, the primary outcome measure occurred following 15.8% (6/38) of IV magnesium sulfate and 5% (1/20) of β-blocker exposures. In the single patient who experienced an exacerbation after β-blocker exposure, the event occurred after administration of IV labetalol (14.3%, 1 of 7 total exposures). Three patients were observed to have an MG exacerbation after administration of potentially harmful medication combinations (2 patients after receiving a combination of fluroquinolones and magnesium and 1 patient after receiving a combination of β-blockers, fluroquinolones, and IV magnesium).
All myasthenic exacerbations were characterized by escalation of respiratory support and scored as a 3 via the Naranjo Assessment indicating a possible causal relationship. NIF and VC values were incompletely documented near the medication event in all circumstances. Of the 7 patients who experienced an exacerbation, 5 of those had at least 1 additional risk factor for exacerbation of disease documented. One patient with a documented exacerbation received 2 doses of IV magnesium 9 hours apart. While death from acute hypoxemic respiratory failure occurred 1 day after administration and their primary diagnosis was pneumonia, the cause was determined to be from an underlying metastatic sarcoma. A single patient met the primary endpoint in 2 separate encounters, both attributable to IV magnesium sulfate. Another who received IV magnesium was considered to have an exacerbation due to an ocular myasthenic event however, the patient did not meet our definition of exacerbation due to a lack of respiratory involvement. Further details of the 7 exacerbating encounters can be found in Supplemental Table 3.
Secondary Outcomes Based on Exacerbation of Disease.

Discussion
In this retrospective analysis, the incidence of a myasthenic exacerbations was 12.7% following inpatient administration of a high-risk medication. Exacerbation of disease mostly coincided with the presence of additional MG exacerbating risk factors and was solely attributed to escalation of respiratory support, mainly characterized by the requirement for intubation in those previously stable on room air. The Naranjo Scale indicated a possible cause-effect relationship in all exacerbations.
Despite the introduction of a clinical decision support tool in 2019, continued administration of high-risk medications occurred in 81% of cases and high alert-override rates were observed. Continued exposure of these agents may be attributed to a lack of familiarity of risk associated with administering potentially harmful medications in this rare disease. Previous literature evaluating clinical decision support indicates that reasons for inappropriate medication alert overrides is likely multifactorial. Reliance on clinical knowledge, medication databases, and safety nets such as pharmacist order verification combined with alert fatigue may all have a role in inappropriate medication alert overrides. Ensuring prominent alerts and/or descaling unnecessary alerts may help to reduce inappropriate overrides. 11 Of the medications that continue to be administered to patients with MG within our health-system, IV magnesium sulfate and IV labetalol were identified as potential precipitants of MG exacerbations. The apparent increased risk of IV magnesium sulfate may be due to the higher frequency of orders for which many are linked to electrolyte supplementation order sets. For this reason, optimization of order sets in the presence of MG may decrease exposure and subsequent exacerbations. Furthermore, heightened caution should be taken in patients who exhibit additional risk factors as most patients who experienced an exacerbation had at least 1 other risk factor.
Our small sample size hinders the ability to detect a meaningful association between medication exposure and exacerbation of disease particularly amongst the medications with lower administration frequencies, highlighting the need for larger scale evaluations. Additionally, as the physician was not required to document the reason for alert override, we were unable to determine the exact reason for override and eventual administration. The Naranjo score of 3 amongst all exacerbations imply a possibility of other respiratory distress etiologies and we were unable to collect all potential causes or confounders of MG exacerbations. In some instances, multiple potentially exacerbating medications were administered within close dosing proximity of 1 another making it difficult to isolate which medication was the culprit. All patients in our study were exposed to at least 1 potentially harmful medication and therefore, we were not able to compare outcomes amongst those who were exposed and those who were not. Although widely used, the motor strength score lacks specificity for myasthenic exacerbations, is limited by the large range in strength possible between grades, and relies on examiner expertise which may vary. Strength can also fluctuate throughout the day and not all patients with myasthenic exacerbations display limb weakness. Lastly, with incomplete documentation of outcome measures (motor strength, NIF, and VC values), changes in respiratory and motor strength may have been missed if they did not result in an escalation of care.
In conclusion, caution is advised when considering common inpatient medications for non-ventilated MG patients and respiratory status should be closely monitored when potentially exacerbating medications are utilized, especially concomitant ones.
Supplemental Material
Supplemental Material - Evaluation of Medication Exposure on Exacerbation of Disease in Patients With Myasthenia Gravis
Supplemental Material for Evaluation of Medication Exposure on Exacerbation of Disease in Patients With Myasthenia Gravis by Nick Petrucelli, Megan E. Barra, and Jennifer L. Koehl in The Neurohospitalist
Footnotes
Author’s Note
This article has not been submitted for publication elsewhere.
Author Contributions
All authors have contributed substantively to all parts of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
IRB Statement
The local institutional review board has approved this study (IRB: 2021P002589 – Massachusetts General Hospital).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
