Abstract
Objective
Seizures in COVID-19 patients continue to be a common reason for consulting the neurology service in the inpatient setting. This paper assesses the frequency of new onset seizures in adult hospitalized COVID-19 patients.
Method
PubMed and EMBASE were searched, with fifteen cohort studies identified to calculate the primary outcome, which was the frequency of new onset seizures in hospitalized COVID-19 patients. An inverse variance meta-analysis of single proportions with a random effects model was applied to these cohort studies to calculate the primary outcome. Risk of bias in individual studies was assessed using the 10-item risk of bias tool for prevalence studies.
Results
The meta-analysis revealed a frequency of .71% (95% confidential interval: .32-1.25, I2 = 89%, 147/28242 patients) for acute symptomatic seizures in patients with COVID-19. For secondary outcomes, the risk of seizures in patients who had EEG completed was 8.49% (95% confidential interval: .62-24.07, I2 = 14%, 44/535 patients). Slightly less than half of patients with COVID-19 and seizures were reported to have acute imaging abnormalities (45.7%) with acute vascular insults being commonly reported. Only a small percentage of COVID-19 patients with seizures (2.8%) met the criteria for COVID-19 encephalitis as determined by the international encephalitis consortium.
Conclusion
The frequency of seizures in COVID-19 was .71% (95% confidential interval: .32-1.25). Slightly less than half of COVID-19 patients had head imaging abnormalities as a complication of COVID-19 infection. Only a small percentage of patients with seizures and COVID-19 met the criteria for COVID-19 encephalitis.
Key Points
• The frequency of new onset seizures in COVID-19 was .71% ([95% confidential interval]: [.32-1.25] • Slightly less than half of COVID-19 patients with seizures had evidence of acute structural abnormalities on head imaging. • Only a small percentage of patients with COVID-19 had seizure due to acute viral encephalitis.
Introduction
Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV2 is a novel coronavirus that originated in Wuhan, China in December 2019 (COVID-19), and was the cause of the 2019-2022 COVID-19 pandemic.1-3 Primarily a respiratory pathogen, clinical features range from asymptomatic infection to fevers, cough, and shortness of breath.1,3 Infection with SARS-CoV2 is now known to be implicated in neurological complications such as neuropathy, stroke, and seizures.4-12 The existing data on new onset seizures in patients with COVID-19 is heterogenous and thus it is unclear what this association of seizures in COVID-19 is.13-15
We conducted this systematic review and meta-analysis to explore the relationship of new onset seizures in patients with COVID-19, since this can be a reason for consulting neurology in the inpatient setting. Since an EEG is often obtained in the inpatient setting for patients presenting with seizures and/or prolonged unexplained encephalopathy, we also wanted to evaluate how common it was to find subclinical seizures on EEG in this subset of COVID-19 patients that presented with new onset seizures. The patient population of interest were hospitalized patients with COVID-19 who experienced first time seizures, without a prior history of epilepsy. Patients with pre-existing and chronic medical conditions are most vulnerable to COVID-19 and have a higher likelihood of hospital admission. Therefore, to improve the generalizability of this meta-analysis, we included patients with and without a prior central nervous system (CNS) insult. Exacerbation of seizures in COVID-19 patients with a preexisting history of epilepsy was outside the scope of this meta-analysis.
The primary outcome of interest was the frequency of new onset seizures in adult hospitalized patients with COVID-19. Secondary outcome measures aimed to address the following questions that could help guide management: (1) What was the frequency of seizures in hospitalized patients with COVID-19 who had EEG completed? (2) Amongst the COVID-19 patients with seizures, how often were structural abnormalities found on head imaging and (3) How often was the cerebrospinal fluid (CSF) abnormal in patients with COVID-19 and seizures?
Methods
Protocol and Registration
We developed a review protocol before the conduction of this systematic review and meta-analysis, but it was not registered.
Institutional Review Board (IRB) Approval
An IRB approval was not applicable to this study since this was a systematic review and meta-analysis.
Eligibility Criteria
We included studies that met the following criteria: • The study design was an observational study, a case series, or a case report. • The study population consisted of hospitalized patients with COVID-19 that had a new onset seizure within 1 week of being tested positive for COVID-19 via a nasopharyngeal swab, lower respiratory, or cerebrospinal fluid (CSF) RT-PCR test.
We excluded the following: • Patients with a history of epilepsy were excluded. • Articles that did not contain original data of the patients (eg, guideline, editorial, review, and letter) were excluded from the secondary review. • Studies were removed by consensus if there was no clear suspicion for a clinical or electrographic seizure based on the information provided in the articles.
Information Sources and Search
All observational studies, case series, and case reports which included patients with COVID-19 and seizure were identified using a 2-level strategy. We utilized 2 databases, PubMed and EMBASE which were searched through a period of 15 months, from the beginning of the pandemic (12-01-2019) through (03-01-2021). We did not apply language limitations.
We searched these 2 databases using the advanced MeSH search terms: ((Seizure) OR (Electroencephalography) OR (Status Epilepticus)) AND (COVID-19).
Study Selection and Data Collection Process
We identified relevant studies through a manual search of secondary sources including references of initially identified articles, reviews, and commentaries. All references were downloaded for consolidation, and elimination of duplicates. Two independent authors (D.S and F.G.) reviewed the search results separately to select studies based on the above inclusion and exclusion criteria. Disagreements were resolved by consensus.
Data Items
Primary outcome was the presence of new onset seizures in hospitalized patients with COVID-19. In studies that evaluated both new onset seizures and breakthrough seizures in patients with preexisting epilepsy, we extracted the relevant data only. Secondary outcomes included whether EEG was obtained, presence and type of head imaging abnormalities, and the results of CSF testing when these latter 2 tests were obtained.
Risk of Bias in Individual Studies
Risk of bias in individual studies was assessed using the 10 item risk of bias assessment in prevalence studies assessment tool. 16
Summary Measures and Synthesis of Results
Hospital based cohort studies were utilized for the meta-analysis of the frequency of seizures in COVID-19 patients. We performed an inverse variance meta-analysis of single proportions with a random effects model using the double arcsine method to calculate the frequency of seizures in patients with COVID-19. A random effects model was chosen to address the uncertainty of the effect size, which is related to the different exclusion and inclusion criteria used in the included observational studies.
For the secondary outcomes, we analyzed individual patient data from the cohort studies, case series and case reports. Other variables such as ICU admission, patient demographics, seizure as presenting feature to the hospital, and mortality were calculated by summation of events divided by the number of total patients from all studies whose information was available for each value. We conducted this systematic review and meta-analysis according to PRISMSA 2020 guidelines, and used the AMSTAR-2 criteria for systematic reviews when possible.
Results
Study Selection and Study Characteristics
The database search term yielded a total of 308 articles that we reviewed based on title and abstract. Of these, we excluded 193 articles based on article type (clinical guidelines, consensus documents, reviews, systematic reviews, conference proceedings, and conference abstracts). We also excluded articles with irrelevant topics, articles reporting only breakthrough seizures in COVID-19 patients with prior diagnosis of epilepsy, and articles without seizures reported in patients with COVID-19. One hundred and one articles were identified for potential inclusion in this study. Fourteen articles were removed due to duplicates.
Eighty-five articles met the inclusion criteria and were assessed for the systematic review and meta-analysis as summarized in the PRISMA flow diagram (Figure 1). The PRISMA checklist (based on 2020 update) can be found under supplementary materials (Table S1). PRISMA 2020 flow diagram for study selection. Reason 1: Reports looking at only breakthrough seizures in COVID-19 patients with established diagnosis of epilepsy. Reason 2: No clear suspicion for seizure based on information provided; or not evaluated by neurologist. Reason 3: Primary outcome not reported.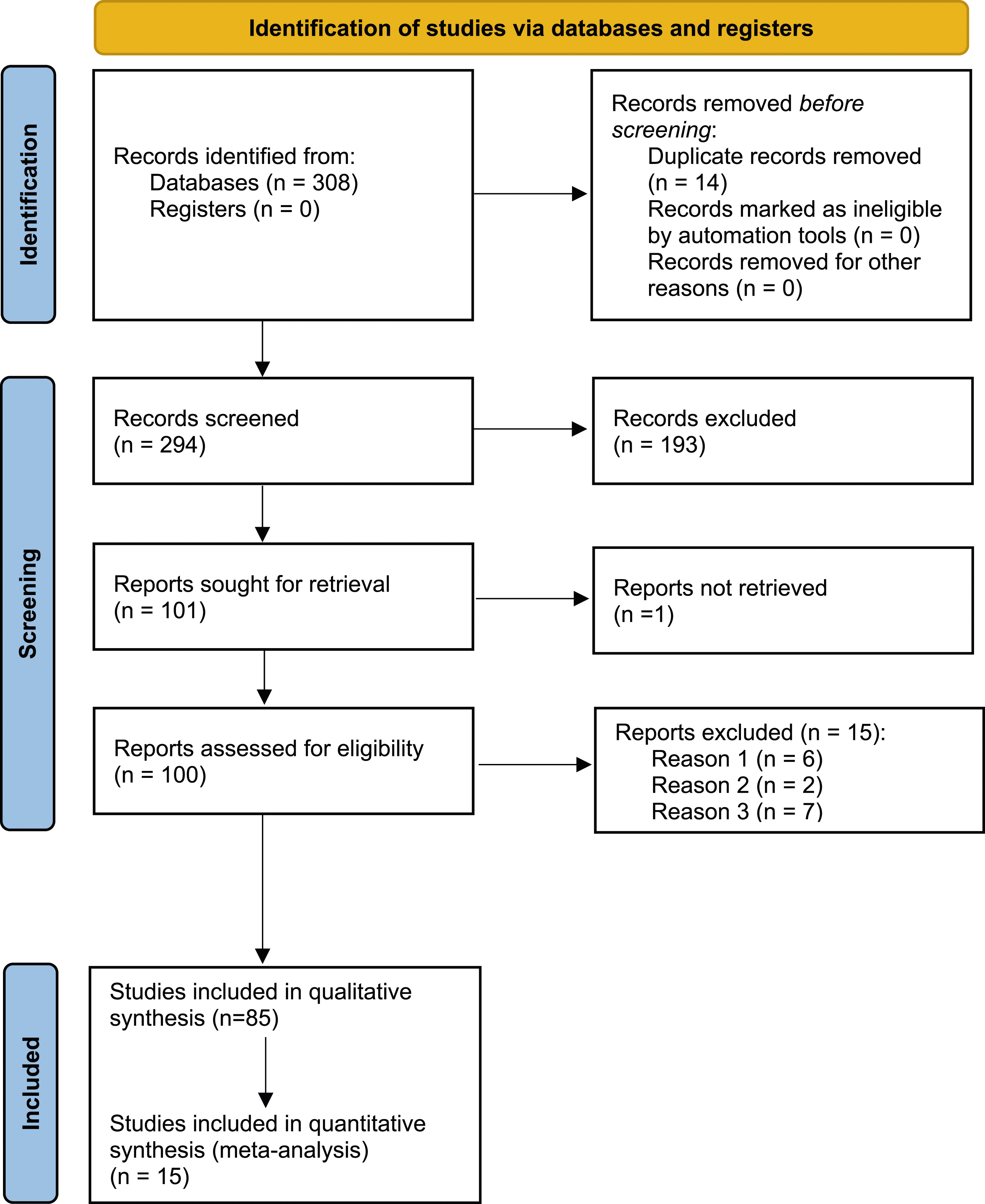
Primary Outcomes
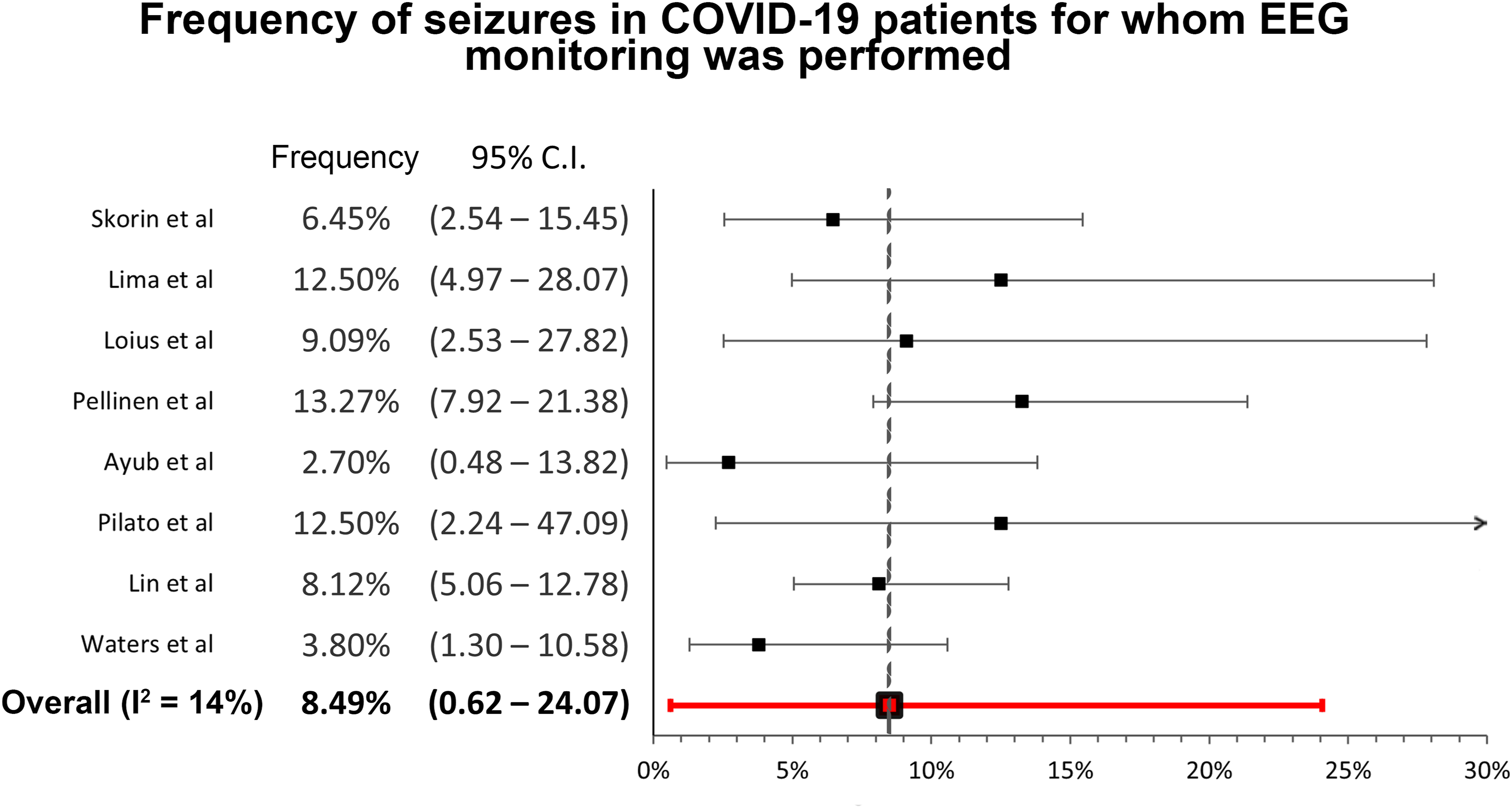
This meta-analysis revealed the frequency of new onset seizures in hospitalized COVID-19 patients is0.71% (95% confidential interval: .32-1.25, I2 = 89%, 147/28242 patients) (Figure 2).10,12,17-30 The frequency of new onset seizures in hospitalized COVID-19 patients who had EEG performed was 8.49% (95% confidential interval: .62-24.07, I2 = 14%, 44/535 patients) (Figure 3).31–38 The detailed description of studies utilized for the meta-analysis can be found in supplemental Table S2, which also includes clinical characteristics and diagnostic findings for these subjects. Frequency of new onset seizures in hospitalized COVID-19 patients. Frequency of seizures in COVID-19 patients for whom EEG monitoring was performed.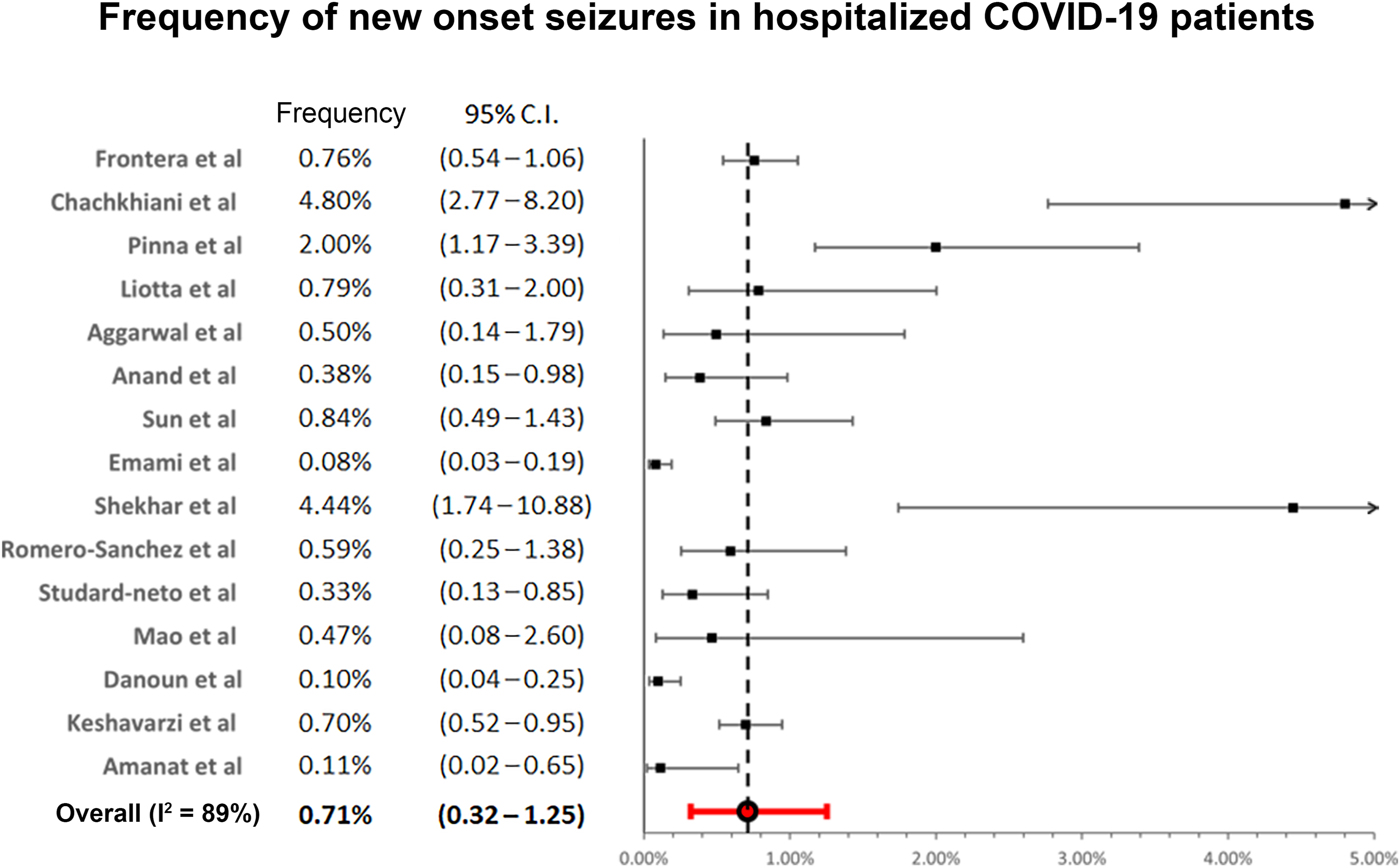
Secondary Outcomes
Imaging and CSF Work-Up by Diagnosis in COVID-19 Patients With Seizures.
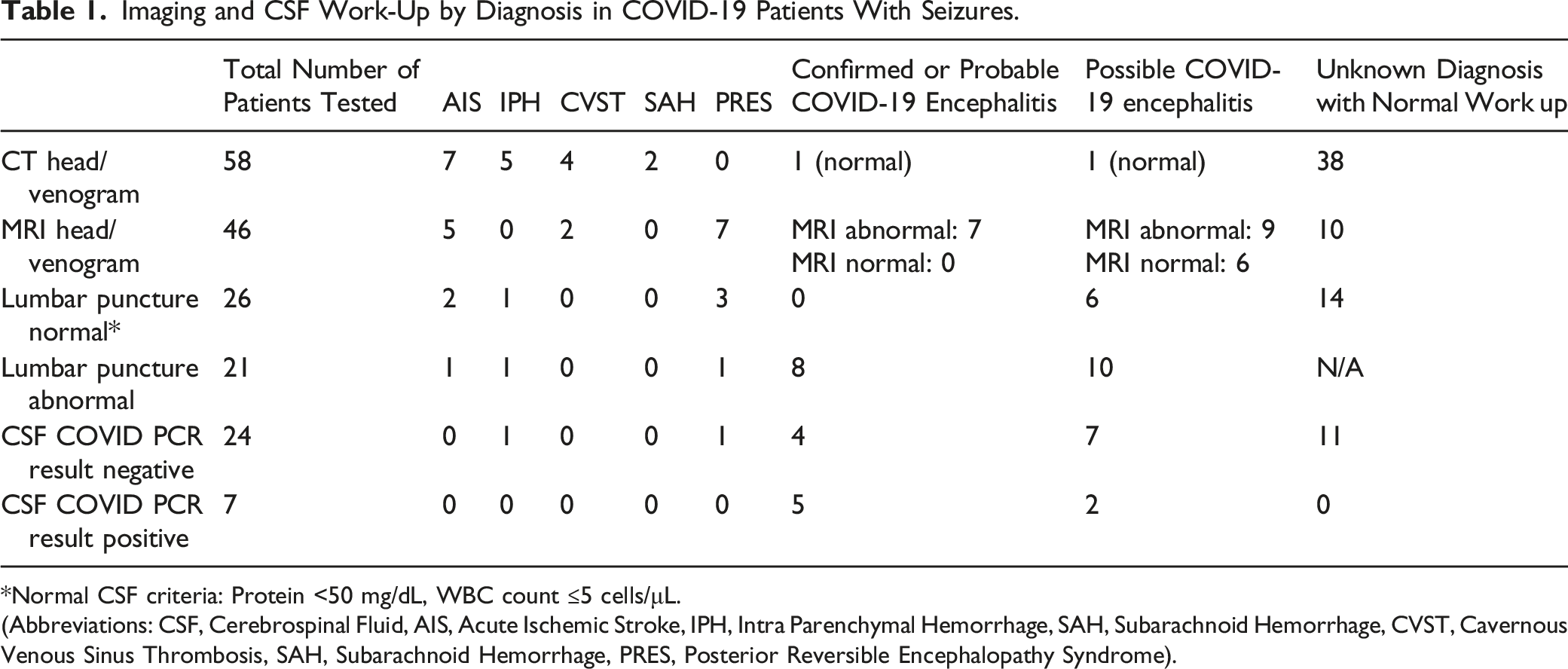
*Normal CSF criteria: Protein <50 mg/dL, WBC count ≤5 cells/μL.
(Abbreviations: CSF, Cerebrospinal Fluid, AIS, Acute Ischemic Stroke, IPH, Intra Parenchymal Hemorrhage, SAH, Subarachnoid Hemorrhage, CVST, Cavernous Venous Sinus Thrombosis, SAH, Subarachnoid Hemorrhage, PRES, Posterior Reversible Encephalopathy Syndrome).
For patients who underwent a lumbar puncture (LP), 44.7% (n = 21/47) reported an abnormal CSF with either an elevated protein, WBC, or both. CSF glucose was not reported in most of the studies, but was within normal limits in the few studies that reported it. Seven patients (22.6%) out of the 24 tested positive for CSF SARS-CoV2 by PCR.
Characteristics of Work up in Patients With Possible, Probable or Confirmed COVID-19 Encephalitis Based on International Encephalitis Consortium Criteria.
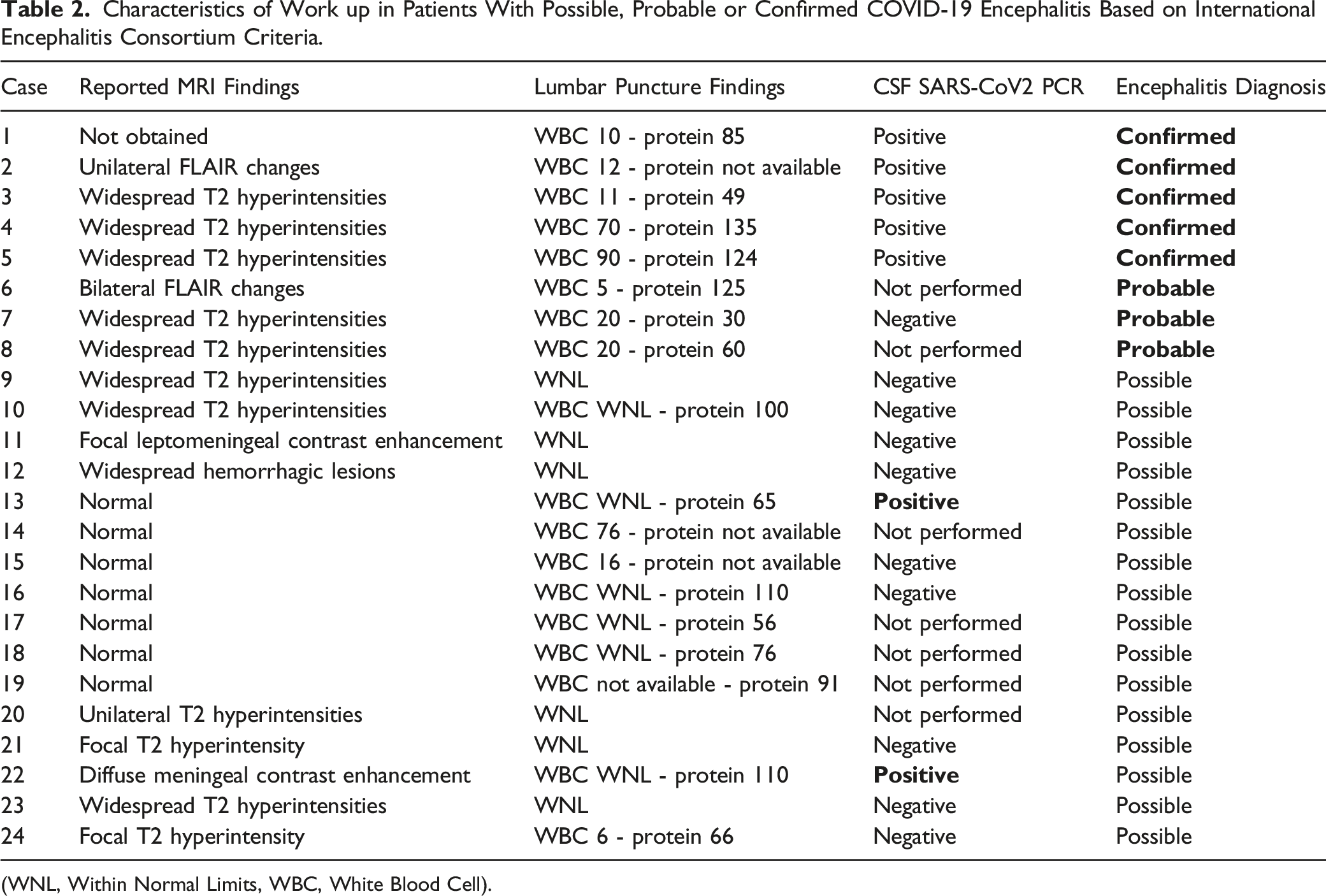
(WNL, Within Normal Limits, WBC, White Blood Cell).
Of note, the larger studies used in the above meta-analysis contained limited individual patient data. Thus, the findings shown in Table 1 are obtained mainly from the case series and case reports of seizures in COVID-19, and are thus subject to publication bias, potentially overestimating the occurrence of the observed abnormalities
Risk of Bias in Individual Studies
Risk of bias for all individual studies was low, and is summarized in supplemental Table 2S.
Patient Demographics and Characteristics
Of the data available, there were 53.6% (n = 60) males and 46.4% females (n = 52) with new onset seizures and infection with COVID-19. The mean age in the adult population was 52.2 years and the mean age of the pediatric population was 7.3 years. Twenty-one patients were pediatric patients, and 259 were adults >18 years of age.
Seizure Characteristics, ICU Admission and Mortality
Of the total COVID-19 patients with new onset seizures (n = 280), 107 patients (38.4%) experienced seizure as a presenting symptom for seeking acute medical attention while the rest experienced a seizure after being diagnosed and starting treatment for COVID-19. Forty-six patients (16.4%) were reported to have status epilepticus. Seventy-seven (27.5%) patients with seizures were reported to require ICU level care. For studies that reported mortality outcomes for COVID-19 patients with new onset seizures, the mortality rate was 22.9% (n = 19).
The seizure semiology was reported to be bilateral tonic-clonic, myoclonic, non-convulsive, and focal motor, with generalized tonic-clonic seizure being reported as 64.0% (n = 55/86). Encephalopathy was the most common reason reported for obtaining an EEG, however, exact numbers of patients that received an EEG for encephalopathy were not explicitly stated in the studies.
Management/Treatment
Longer term maintenance anti-seizure drug (ASD) data for seizure control was not available. All adult patients required acute and short-term maintenance treatment with an ASD. The most commonly used ASD was levetiracetam in the adult population (67.6%, n = 48/71) and phenobarbital in the pediatric population (38.5%, n = 5/13) with the remaining cases unreported. Only 1 pediatric patient did not require further maintenance with ASD after the loading dose. Twelve patients (14.3%) were treated with IV immunoglobulins (IVIG). One patient was treated with plasma exchange (PLEX) prior to receiving IVIG.
Discussion
Seizures associated with COVID-19 have been reported as a rare complication of SARS-CoV2 infection. This systematic review and meta-analysis were conducted to determine the overall frequency of new onset seizures in hospitalized COVID-19 patients without a history of prior epilepsy. An additional objective was to investigate the potential etiology of seizures in order to guide diagnostic assessment and subsequent management.
The important findings of this study can be summarized as follows: (1): The frequency of new onset seizures in COVID-19 patients was .71% (95% confidential interval: .32-1.25, I2 = 89%, 147/28242 patients), (2): Acute vascular insults were the most common MRI abnormality demonstrated in COVID-19 patients with new-onset seizures, and (3) The clinical criterion for encephalitis was rarely met in COVID-19 patients with new-onset seizures. The high heterogeneity in the studies was expected given that these were observational studies with differences in how the population parameters were set.
New onset seizures can be a first manifestation of epilepsy or it may result from a provoking event such as metabolic derangements, adverse medication effects, and acute CNS insult such as a vascular insult or encephalitis. 40 In hospitalized patients, the incidence of new onset seizures is estimated to be around 5.2%, 41 while in the critically ill hospitalized population, this incidence can be high as 34%. 42 Seizures in critically ill patients can be multifactorial, with metabolic derangements, medication side effects or acute CNS injury playing a role.
It is recognized that seizures can be a presenting system of viral encephalitis, with population studies approximating CNS infections to account for 15% of new onset seizures.43,44 With known neurotropic viruses like HSV, seizures may occur in as many as 40%–60% of patients during the acute stage of infection.45,46 Coronaviruses have previously demonstrated neuro-invasive properties, 47 and are theorized to gain access to the CNS via the ACE-2 receptor. 48
Despite these concerns for a special neurotropism, a recent meta-analysis only reported a .215% (95% confidence interval: .056 - .441) incidence of SARS-CoV2 encephalitis in COVID-19 pateints. 49 Due to safety regulations, neuropathological data from autopsies is limited in comparison to the entity of the pandemic, 50 but the few available autopsy studies demonstrate direct neural invasion by SARS-CoV2 to be a rare mechanism of neuronal injury. Conversely, thromboembolic vascular changes were the most frequently encountered. 51
This meta-analysis demonstrated seizures to be an uncommon complication from COVID-19, and analysis of the individual patient data revealed seizures due to COVID-19 encephalitis to be an even rarer occurrence (Table 1). The definitive diagnosis of COVID-19 encephalitis appeared challenging with some studies basing this diagnosis on a partial assessment (missing LP or MRI or both). Furthermore, objective diagnostic criteria were not reported by any study. This paper applied the International Encephalitis Consortium criteria in an attempt to categorize the patients into a group with possible, probable, or confirmed COVID-19 encephalitis (Table 2).
In the group with possible COVID-19 associated encephalitis, nine patients had non-specific MRI abnormalities. Six of these patients with non-specific MRI findings had a normal CSF, and 3 had an abnormal CSF. Of note, there were 2 patients that we categorized into the possible COVID-19 encephalitis group who had a positive SARS-CoV2 CSF PCR test. Both these patients had a normal WBC and an elevated protein only as well as imaging findings that were not consistent with encephalitis (Table 2). This is presently of unclear clinical significance. One possible explanation is that these 2 patients were early in their disease process and thus did not manifest the typical CSF or MRI findings of encephalitis.
There were 10 patients categorized into the possible COVID-19 encephalitis group (five with normal imaging and 5 with non-specific MRI findings) who had an abnormal CSF. The CSF albumin index, which is 1 way to assess for an intact blood-brain barrier 52 was not reported in any of the studies which made it difficult to assess for the clinical significance of these findings. It is known that seizures can cause mild CSF abnormalities, 53 thus it is plausible that this abnormality was due to the seizures themselves.
Acute vascular events were commonly encountered in patients with seizures and COVID-19 (Table 2), but for most patients with COVID-19 and seizures, it was unclear what the etiology for the seizure was. In many cases, it was assumed in the reports to be related to critical illness, and this was noted especially when assessment with MRI and LP was unrevealing. Given the limited details on seizure semiology and imaging data, it was difficult to delineate 1 cause from the other, when reasoning was not explicitly stated in the text. It should also be considered that the cause of the seizure could be multifactorial from a combination of factors such as acute vascular injury, metabolic disturbances, hyperthermia and medication toxicity.54-56
This review and meta-analysis have some limitations. First, this covers a brief period through the first peak of the pandemic and before vaccinations were available. Therefore the sample size may still be limited. Second, there was substantial heterogeneity in the patient population, as noted in the high I2 obtained in the results of the meta-analysis, given that these were all observational population-based studies. The effect of this heterogeneity on the primary outcome was limited by using a random effects model. While it should be recognized that this does not resolve the issue completely, it mitigates it significantly. Additionally, the risk of bias for all individual studies was low, further strengthening the results of the meta-analysis. Also, the evaluation of secondary outcomes was subject to publication bias since information about individual patient characteristics was lacking in the larger studies and obtained mainly from case series and case reports.
It may be argued that patients with prior CNS insults are at higher risk for new onset seizure and possibly should have been excluded. However, the intent of this review was to capture all-comers to the hospital with COVID-19 and new onset seizures, regardless of a history of prior CNS insult. This would be truly representative of the COVID-19 population since medical comorbidities predispose patients to severe COVID-19 infection needing acute inpatient care.
Future studies could evaluate for new onset seizures in patients with COVID-19 without a prior CNS insult as well as the incidence of seizures in vaccinated individuals with COVID-19. Other studies could better evaluate the underlying mechanism of seizures in patients with COVID-19. Longer term studies could investigate if COVID-19 patients with acute symptomatic seizures are at higher risk of subsequent epilepsy overall, as well as evaluating for the risk of later epilepsy in the patients with COVID-19 encephalitis.
Conclusion
This systematic review and meta-analysis assessed the frequency of new onset seizures in hospitalized patients with COVID-19 and found it to be .71% (95% confidential interval: .32-1.25), and the risk of seizures in patients with completed EEG was 8.49% (95% confidential interval: .62-24.07). Head imaging and/or CSF abnormalities were encountered in almost half the patients with COVID-19 and seizures, however, only a small percentage (2.85%) of these patients met objective clinical criteria for COVID-19 encephalitis (n = 8).
Supplemental Material
Supplemental Material - New Onset Seizures in Hospitalized COVID-19 Patients; A Systematic Review and Meta-Analysis
Supplemental Material for New Onset Seizures in Hospitalized COVID-19 Patients; A Systematic Review and Meta-Analysis by Daniah Shamim, Fatima Ganatra, Murtaza Khan and Gary Gronseth in The Neurohospitalist
Footnotes
Author Contributions
Daniah Shamim: Conceptualization (lead), Writing – original draft (lead), review and editing, formal analysis (supporting), Methodology (supporting)
Fatima Ganatra: Writing – original draft (supporting), review and editing, formal analysis (supporting), methodology (supporting)
Murtaza Khan: Methodology (supporting), formal analysis (supporting), review and editing (supporting)
Gary Gronseth: Methodology (lead), formal analysis (lead), conceptualization (supporting)
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
