Abstract
Susac Syndrome was first described as an inflammatory microangiopathy of the brain and retina. Since then, multiple articles have been published in attempts to improve the understanding of this rare disease. Clinically Susac Syndrome is known to present with triad of encephalopathy, sensorineural hearing loss and branch of retinal artery occlusion (BRAO), along with characteristic “snowball” or “spoke” appearing white matter lesions of the corpus callosum. It has been characterized by vast heterogeneity in terms of its presenting symptoms, severity, and clinical course. Although subset of patients present with severe forms of Susac Syndrome and can develop prominent residual neurologic deficits, it has been reported to be mostly non-life-threatening and only few fatal cases have been described in the literature. Based on the available case reports with fatal outcome, mortality has been related to the systemic complications either during acute disease flare or during chronic-progressive phase. We describe a case of fulminant Susac Syndrome complicated by the sudden and rapid progression of diffuse cerebral edema leading to brain herniation and ultimate brain death, in order to increase awareness of this rare and catastrophic complication.
Keywords
Introduction
Susac syndrome is a rare form of neuroinflammatory microvascular arteriopathy, specifically affecting the precapillary arterioles of the eyes, ears, and brain, which was first described in 1979. 1 Historically Susac syndrome was believed to be a form of autoimmune endotheliopathy, given presence of anti-endothelial cell antibodies (AECA) in the subset of patients with the diagnosis, though the cause of the observed clinical disease predilection for cerebral, ocular, and cochlear endothelial cells remained unclear. 2 Recently this hypothesis has been challenged and CD8+ T-cell mediated inflammation and damage of the endothelial cells now proposed as the main inciting pathologic process, though the specific endothelial cell antigen that is the target of the cytolytic immune reaction remains elusive. 3
Clinically Susac Syndrome presents with triad of encephalopathy, acute sensorineural hearing loss and branch of retinal artery occlusion (BRAO). However, concomitant presentation of all three characteristic symptoms is rare and has only been described in 13% of the patients, making the diagnosis challenging. 4 This requires high degree of suspicion as Susac Syndrome remains to be a clinical diagnosis in conjunction with characteristic radiographic findings from brain magnetic resonance imaging (MRI) and retinal fluorescein angiography (FA). Central nervous system (CNS) manifestation makes up the majority of symptoms at the disease onset, headache being the most commonly reported symptom occurring in up to 80% of patients. 5 The clinical severity of the disease has been reported to be variable, though the majority of patients present with mild to moderate forms according to CNS involvement. There have been only few fatal cases described in the literature, none of which have been associated with diffuse cerebral edema or herniation.6–9
Case Report
A 33-year-old man with a pertinent past medical history of congenital sensorineural deafness status post left cochlear implant placement at the age of 3 with a strong family history of deafness in both the patient’s sister and maternal aunt, though without genetic workup, presented to the Mayo Clinic Florida (MCF) as a transfer from outside facility (OSH) for acute onset encephalopathy of unknown etiology. Prior to presentation, the patient lived independently, worked at the grocery store and was able to communicate with speech and sign language.
The patient first presented to OSH with complaints of rhinorrhea, congestion, cough, headache, and nausea/vomiting of three-day duration. A viral syndrome was suspected as the cause of his symptoms and he was discharged home from the emergency department (ED). Following his initial evaluation, the patient missed two consecutive days of work that resulted in a wellness check by the family, and he found him to be lethargic and febrile with associated generalized weakness. On this second presentation to ED, the patient was febrile to 101F and minimally responsive, only withdrawing to painful stimulation, with intact cranial reflexes. Prominent hyperreflexia with sustained clonus was noted on initial neurologic examination. Due to the progressive decline in mental status and concern for airway protection he was intubated.
During this second ED admission, it was revealed that the patient had actually been complaining of new onset headaches for three months along with ill-defined visual changes, including “blurry” and “double” vision that were intermittent and transient. Work-up, including computed tomography (CT) of the head and temporal bone to evaluate for cochlear implant failure, was unrevealing. He had been diagnosed with migraines and was placed on topiramate and rizatriptan without improvement in his symptoms.
After intubation at the OSH, the patient was started on empiric antibiotics for working diagnosis of encephalitis/meningitis. Head CT along with computed tomography angiography (CTA) were unremarkable. Cerebrospinal fluid (CSF) analysis revealed opening pressure (OP) 19 mm H2O, white-blood cell (WBC) 23 (75% neutrophils, 13% lymphocytes), red-blood cells (RBC) 21,225, protein 186 and glucose 56. CSF gram stain and culture, along with encephalitis/meningitis PCR panel were negative. Remaining systemic infective workup was unrevealing except respiratory culture grew Stenotrophomonas maltophilia and he was treated with Bactrim. At this point in time, given low suspicion for CNS infection, antibiotics were discontinued, and possibility of autoimmune encephalitis was considered. The patient underwent repeat CSF analysis on the 5th day of hospitalization that revealed an OP 25, WBC 0, RBC 4700, protein 268, and glucose 68. CSF was sent for oligoclonal bands and autoimmune/paraneoplastic encephalitis panel, both of which eventually resulted negative. CT chest/abdomen and pelvis as well as scrotal ultrasound was negative for occult malignancy. Serum screening tests for common autoimmune disorders/vasculitis (ESR, CRP, ANA, ANCA, SSA, SSB, Scl-70, Sm-ab, RNP-ab, Jo1-ab, NMO, MOG) were unremarkable. Patient was treated with high dose methylprednisone (IVMP) followed by 5-day course of intravenous immune globulin (IVIG). Electroencephalography (EEG) revealed diffuse background slowing with predominant theta and admixed 2-3 Hz delta frequencies. Periods of frontal intermittent rhythmic delta activity (FIRDA) was noted that was state independent Figure 1. The OSH was unable to obtain brain MRI due to cochlear implant, hence, patient was transferred to MCF on day 7th for further management and workup. Snapshot from continuous EEG monitoring [Bipolar montage; LFF 1 Hz; HFF 70 Hz; Notch 60 Hz; Sensitivity 7mV/mm; Timebase 15 mm/sec] A. Admixed theta and delta slowing B. Frontal intermittent rhythmic delta activity (FIRDA).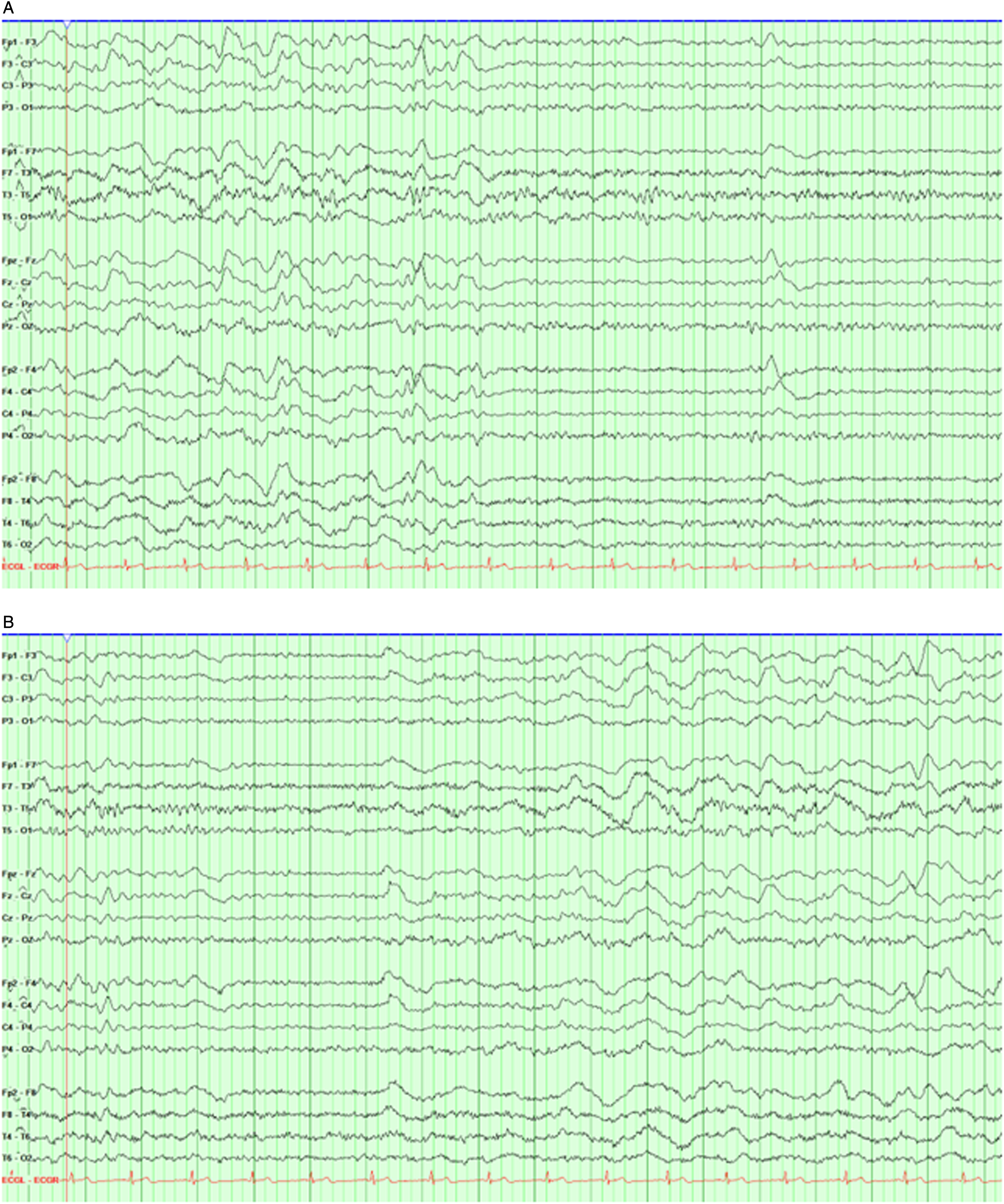
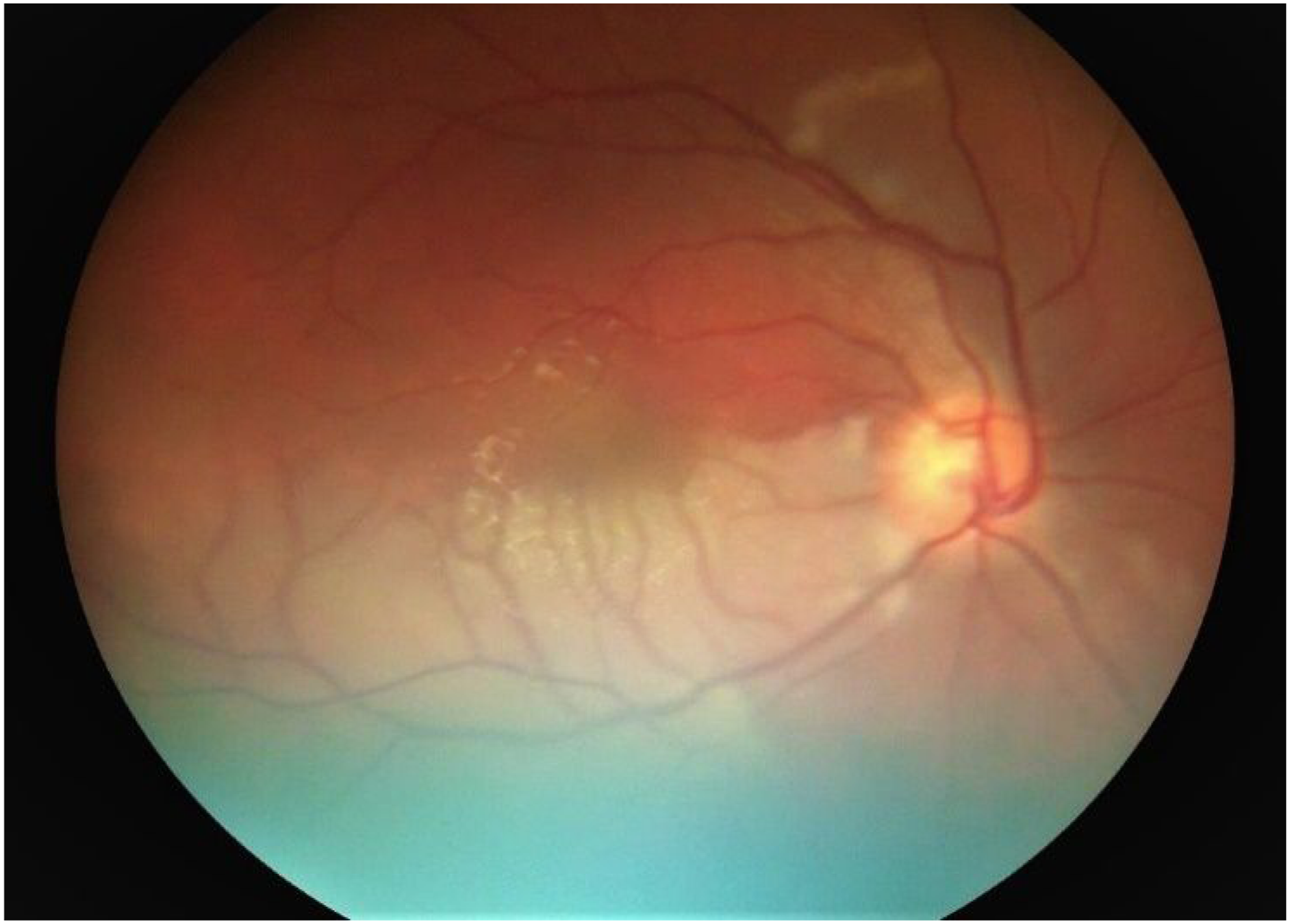
Upon presentation to MCF, patient was intubated and minimally responsive off sedation with a Glasgow Coma Scale (GCS) of 7T (E4, V1T, M2). Cranial nerve examination was remarkable for disconjugate gaze with left esotropia. Diffusely increased tone (right > left) and hyperreflexia, bilaterally upgoing toes and positive Hoffman’s sign were also present. Explantation of left cochlear implant was performed on day 4th from presentation to MCF to allow for MRI of the whole neuroaxis with and without contrast. Brain MRI was remarkable for extensive multifocal T2 hyperintense lesions involving brainstem, cerebellar hemispheres, cerebellar peduncles, subcortical white matter, bilateral thalami, and corpus callosum. Patchy enhancement and diffusion restriction accompanied some of the lesions Figure 2. Given pattern of distribution of T2 hyperintense lesions, particularly involving the corpus callosum, Susac Syndrome was suspected as potential etiology. The patient was evaluated by ophthalmology and was confirmed to have right superotemporal branch retinal artery occlusion with small inferior occlusion via fundoscopy Figure 3, further supporting diagnosis of Susac syndrome. The 1 mg/kg prednisone equivalent maintenance steroid dose was continued and given the severity of the patient’s clinical presentation combined with a probable diagnosis of Susac Syndrome as a result of the aforementioned workup, immunosuppressive therapy with mycophenolate mofetil and rituximab were initiated with plans to repeat brain MRI after second dose of rituximab. Cyclophosphamide was deferred given patients youthful age and substantial risk of infertility and long-term hematologic malignancy. Brain MRI with and without contrast. A-C. Axial diffusion weighted (DWI) MR image at the level of basal ganglia shows numerous areas of restricted diffusion without apparent diffusion coefficient (ADC) correlate but corresponding hyperintense lesions on fluid-attenuated inversion recovery (FLAIR) MR sequence, consistent with subacute strokes. Patchy enhancement of some of the lesions (not shown). D-F. DWI image at the level of corona radiata again demonstrates numerous areas of restricted diffusion with ADC correlate and corresponding FLAIR changes, suggestive of acute to subacute strokes. G. Sagittal T1-weighted MR image demonstrates multiple hypointense lesions in the body of corpus callosum. Fundoscopic examination with ophthalmoscope demonstrates regional retinal ischemia due to superotemporal branch retinal artery occlusion. Small inferior occlusion at the edge of the optic disc.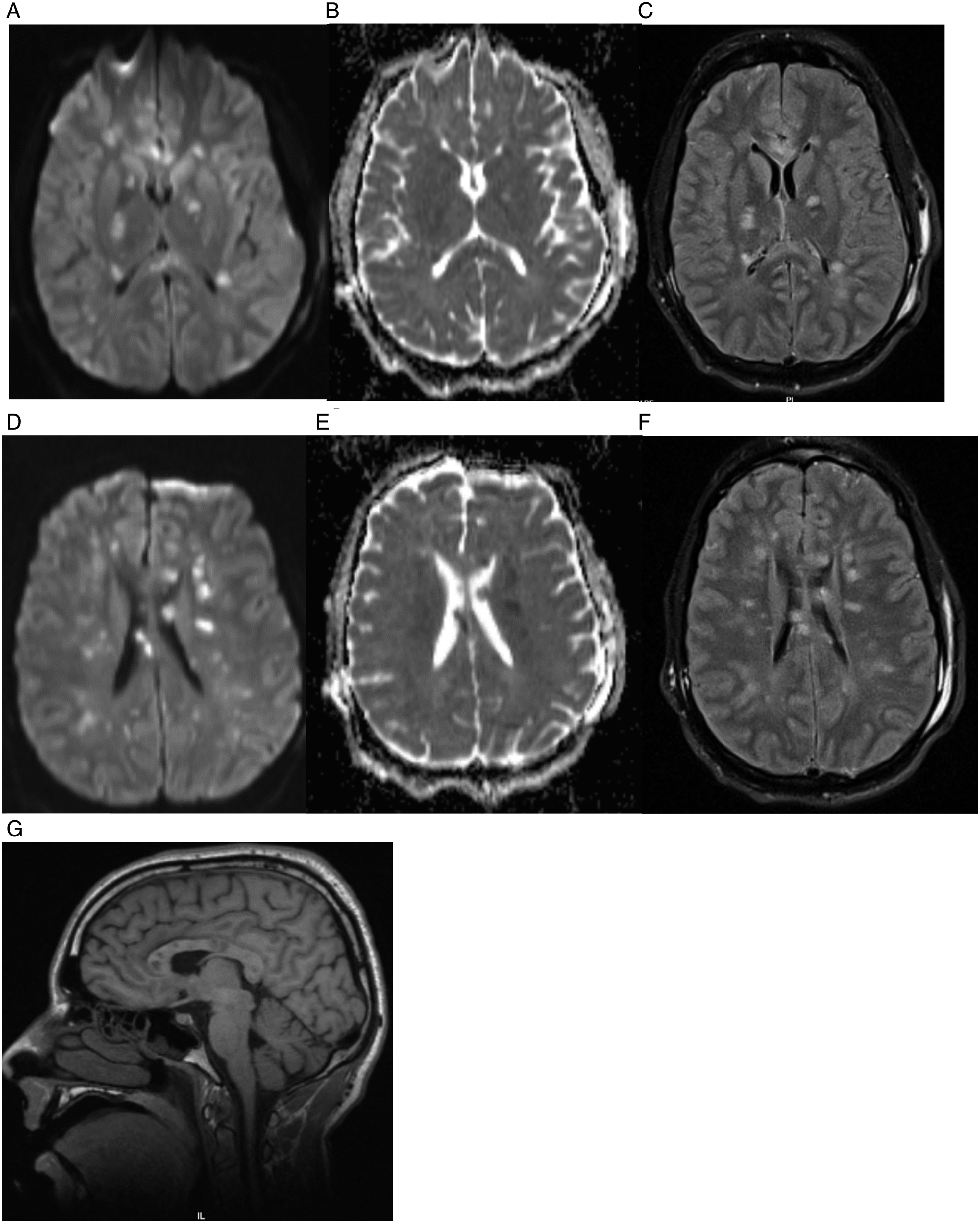
Patient showed mild improvement after initial therapy with GCS improving to 9T (E4, V1T, M5), though subsequently his progress plateaued. He underwent tracheostomy and percutaneous gastrostomy tube placement with plans to transition to a neurorehabilitation center. However, on the 20th day of his hospitalization at MCF, the patient suddenly decompensated with acute onset hypotension with dilated and unreactive pupils on examination. Two hours prior to acute decline he was reported to be at his baseline of GCS 8-9 with intact cranial nerve reflexes. Telemetry review leading up to this point revealed no episodes of hypotension, severe hypertension, or hypoxemia. He has been afebrile. No interruption in immunosupressive therapy or steroid dose adjustment occured. No potentially causative metabolic derangementswere identified either, such as hyponatremia, liver failure with hyperammonemia, uremia, diabetic ketoacidosis or hypercarbia. He was started on vasopressors for hemodynamic support and received multiple doses of hyperosmolar therapy with Mannitol; Unfortunately, emergent CT scan was consistent with diffuse cerebral edema, sulcal effacement and uncal herniation Figure 4. The patient was pronounced brain dead on the same day per institutional brain death examination protocol. No autopsy was pursued per family’s wishes. Computed tomography (CT) scan without contrast. A. Axial CT scan demonstrates uncal herniation. B. Diffuse cerebral edema with sulcal effacement.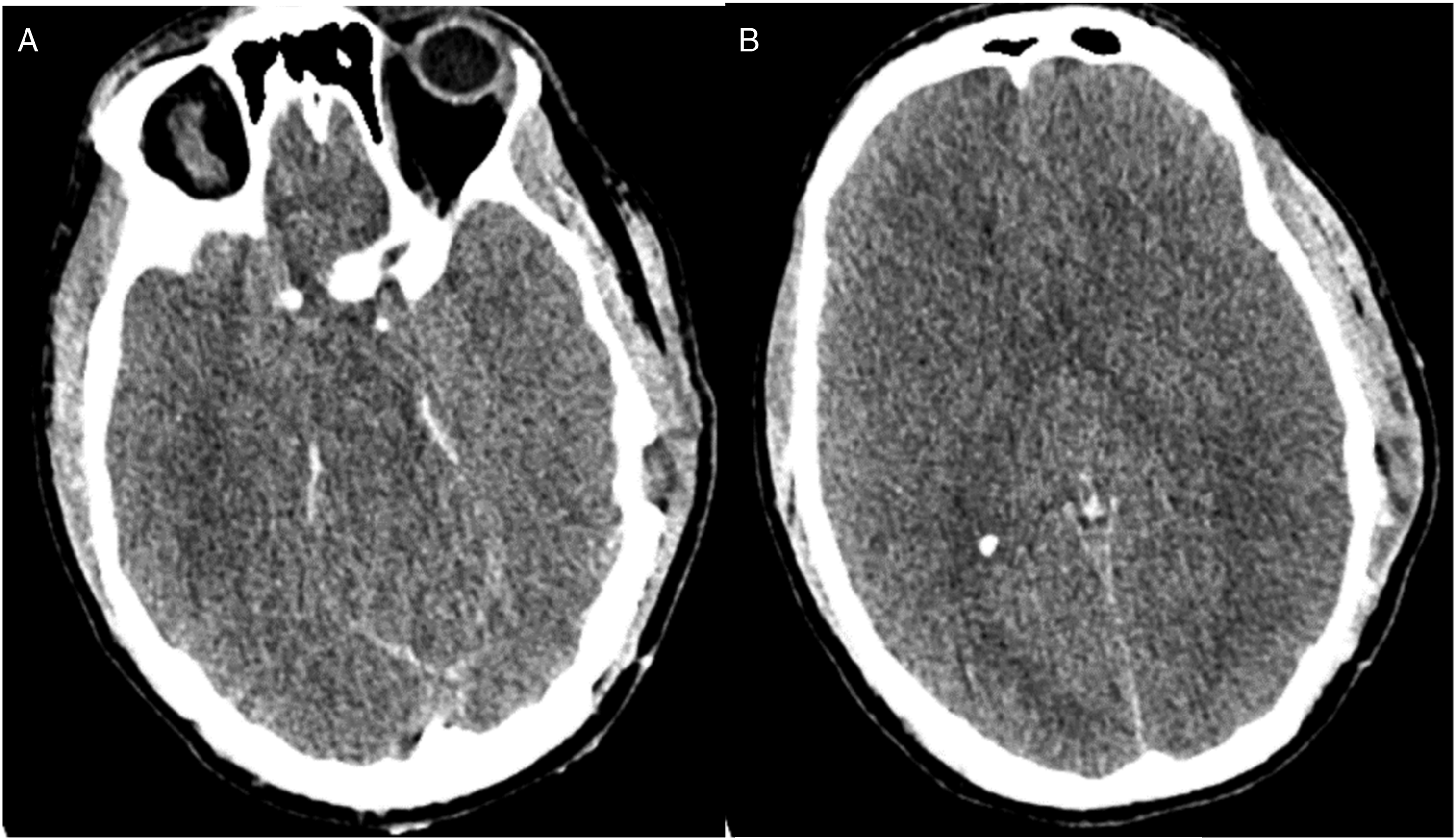
Discussion
Syndrome is a rare neuroinflammatory disease characterized by triad of encephalopathy, sensorineural hearing, and vision loss. Ever since its first recognition in the late 1970s as an immune-mediated microangiopathy, selectively affecting small caliber cerebral, retinal, and cochlear blood vessels, over 400 cases have been published to increase the awareness of this rare disorder. Most of our current knowledge regarding this condition is based on the available case reports and small case series, hence, its true global incidence and prevalence remains unknown. 10
Susac Syndrome is more commonly found in young females (20-40 years) with female-to-male ratio of 3.5:111. Predilection for female sex further supports its autoimmune nature. Historically Susac Syndrome has been recognized as autoimmune endotheliopathy, especially after discovery of AECA in the subset of patients. However, later it was reported that AECA is not sensitive nor specific for Susac Syndrome and is found in less than 30% of patients with definite diagnosis. 12 Recently it was discovered that cytotoxic CD8+ T-cells might have more prominent role in the disease pathogenesis, based on the autopsy findings and animal comparison models. 3 Terminally differentiated effector memory CD8+ T-cell number was noted to be higher both in CSF and serum of patients with Susac syndrome compared to healthy controls or multiple sclerosis patients. 13 The end result of the maladaptive immune response is damage of the endothelial cells, leading to the blood brain barrier (BBB) breakdown and occlusion of small caliber arterioles. This explains characteristic radiographic findings of leptomeningeal enhancement and microvascular strokes on the brain MRI, respectively. Given Susac Syndrome doesn’t affect large vessels, most of the T2 hyperintense lesions are limited to the subcortical white and gray matter.14,15 This makes development of catastrophic diffuse cerebral edema very atypical. In our case there were no apparent inciting factors for cerebral edema, such as worsening neurologic exam to suggest development of new extensive lesions, metabolic derangements, infections, prolonged hypotension, severe hypertension, or hypoxemia. However, given extensive CNS involvement the patient could have experienced transient cerebral circulatory arrest from diffuse inflammatory or neurotoxic effects of underlying disease process or due to abnormal autoregulation from dysfunction of vasomotor centers in the brainstem.
The clinical course of Susac syndrome is variable. It has been divided into three major clinical courses – monocyclic (active phase lasting ≤2 years), polycyclic and chronic-progressive.5,16 The majority of patients have a monocyclic course, defined as fluctuating disease within first two years with no further recurrence, while polycyclic continues to recur even after initial 2-year period. Only around 4% of patients develop chronic-progressive course, which typically correlated with more severe acute clinical manifestations and significant residual deficits.5,14 The most common clinical manifestation at disease onset are CNS symptoms (80%), followed by ocular (50%) and auricular (30%) involvement.17,18 All three components of the classic triad are rarely present on presentation. CNS symptoms vary from headaches to confusion, behavioral disturbance, progressive cognitive decline and memory difficulties, aphasia, ataxia with gait difficulties, long tract signs, seizures, and coma. Headaches are the most prevalent symptom and are commonly described to be migrainous in nature. Typically, headaches proceed other neurologic manifestations but not more than 6 months. 12 Visual symptoms can include scintillating scotomas, blurry vision, photopsias and visual field cuts from recurrent BRAOs. Visual field cuts are typically altitudinal and respect the horizontal meridian. 19 Fundoscopic examination commonly reveals areas of retinal pallor suggestive of focal ischemia from multifocal BRAOs. Arterio-arterial collaterals can also be seen later on in more severe cases.10,14 Gass plaques represent lipid-laden deposits in the arterial wall from periarteriolar inflammation, which are typically located away from arterial bifurcation sites or BRAOs and been described in patients with Susac Syndrome, though they are not specific to the disease process. 20 From a vestibule-cochlear standpoint sensorineural hearing loss is the most common clinical manifestation and is thought to be secondary to cochlear apex ischemia. 11 Hearing loss can be sudden, bilateral, and accompanied by roaring tinnitus. 21 Other rare manifestations outside of the classic triad can include livedo reticularis, arthralgias, myalgias and bowel ischemia.22,23
On account of significant clinical heterogeneity few diagnostic criteria have been proposed over the years to aid in identification of possible, probable, and definite Susac Syndrome. 4 Susac syndrome remains to be a clinical diagnosis with ancillary tests providing additional supportive information when clinical triad is incomplete. Brain MRI offers invaluable information and helps differentiate Susac Syndrome from other disorders such as multiple sclerosis (MS), acute disseminated encephalomyelitis (ADEM) or other vasculitides. 24 Typically, three principal areas are affected – subcortical white matter, especially corpus callosum, deep gray nuclei and leptomeninges. 15 The spinal cord is rarely involved with only few cases being described in the literature.25-27 “Snowball” or “Spoke” appearing T2 hyperintense central corpus callosum lesions are pathognomic and are invariably present in Susac Syndrome.15,28 A newer MRI finding described in patients with long tract signs is the “string of pearls” like involvement of the internal capsule. 11 Some patients with peripheral retinal involvement may not have clinical visual complaints, hence, full ophthalmologic evaluation with dilated fundoscopic examination is essential. Fundoscopic exam may not always demonstrate characteristic findings of retinal pallor or attenuated branch arteries. In such cases retinal fluorescein angiography (FA) can confirm presence of BRAO or arterial wall hyperfluorescence (AWH). Some authors even suggested including presence of BRAO and AWH on FA and callosal lesions on MRI as an independent diagnostic factor for definite Susac Syndrome. 10 Given broad differential diagnosis most patients with suspected Susac Syndrome undergo lumbar puncture (LP) with CSF analysis, even though it is not included as required diagnostic criteria. CSF profile in patients with Susac Syndrome is usually benign with mild lymphocytic pleocytosis and elevated protein. IgG synthesis index and oligoclonal bands (OCBs) are rarely found. 5
There are no randomized control trials (RCTs) to guide management of Susac Syndrome. Most of the treatment recommendations are based on the expert opinion and the need for immunosuppression given underlying neuroinflammatory process. The most recent guidelines proposed by Rennebohm et al 16 divided treatment recommendations based on the disease severity. Extremely severe cases require high-dose IVMP followed by steroid taper, IVIG, Cyclophosphamide (Mycophenolate mofetil if unable to use Cyclophosphamide) and Rituximab. There has been controversy over use of antiplatelets for stroke secondary prevention with some papers recommending it use, while others citing its ineffectiveness and recurrence of symptoms.13,14
In summary, there is still lot to learn about Susac Syndrome and its pathogenesis, which can contribute to the development of more targeted therapies. Fortunately, vast majority of cases end up in remission, some patients with minimal to no residual deficits. However, there are more severe forms of the disease that require increased vigilance especially during first few months following disease onset. With this case report we aim to increase awareness of this rare but possible complication and debate if intensifying therapy or increasing frequency of imaging surveillance within first few weeks might have role in such fulminant forms of Susac Syndrome.
Footnotes
Acknowledgements
We wish to express our sincere gratitude for patient’s family for granting consent for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
