Abstract

A 24-year-old male with relapsing-remitting multiple sclerosis, stable on natalizumab with no relapses for nearly 10 years (Figure 1A), abruptly discontinued natalizumab, switching to teriflunomide due to loss of insurance coverage. Two months later, he developed subacute right ataxic-hemiparesis, dysarthria, and dysphagia, requiring admission to hospital for two weeks. Brainstem relapse was confirmed on magnetic resonance imaging (Figure 1B). He remained seronegative for JC virus and cerebrospinal fluid analysis was unremarkable. He was treated with intravenous corticosteroids and plasmapheresis. Deficits resolved to pre-cessation baseline with sustained radiologic improvement following resumption of natalizumab (Figure 1C).
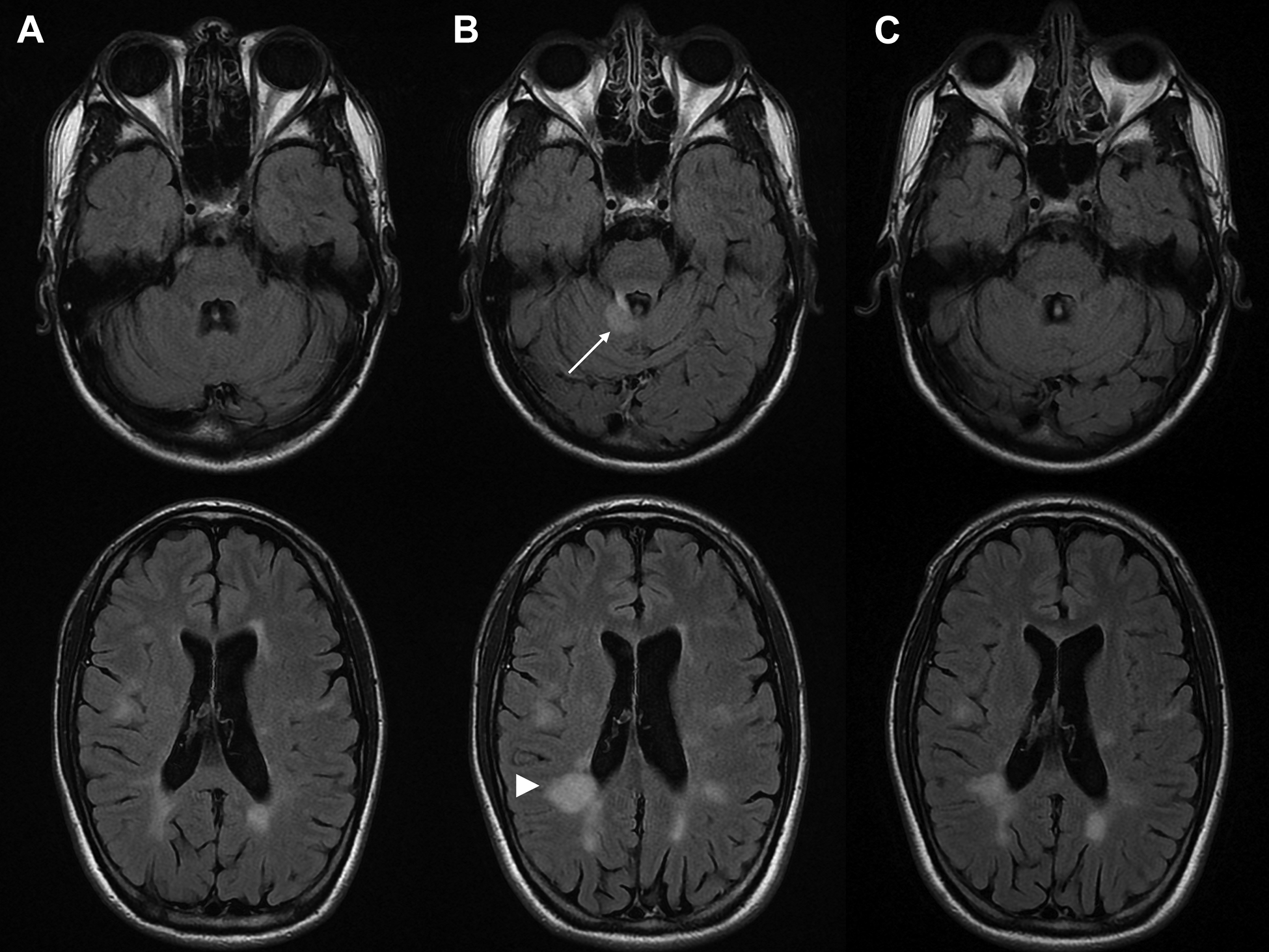
Axial T2-FLAIR MRI (A) on natalizumab, (B) 3 months after discontinuation demonstrating new T2 hyperintensities in the right superior cerebellar peduncle (arrow) and periventricular white matter, largest in right occipito-parietal region (arrowhead), and (C) 3 months after restarting therapy with improvement of T2 lesions. MRI indicates magnetic resonance imaging; FLAIR, fluid attenuated inversion recovery.
Natalizumab is a humanized monoclonal antibody against α4-integrin cell adhesion molecule, which impedes lymphocyte trafficking across the blood–brain barrier. Natalizumab is a high-efficacy disease-modifying therapy for highly active or aggressive relapsing-remitting multiple sclerosis, although long-term use may be limited by cumulative risk of progressive multifocal leukoencephalopathy in patients who are seropositive for JC virus. 1
Previous studies have reported return of clinical and radiologic disease activity to pre-treatment levels following discontinuation of natalizumab. The risk of rebound upon rapid withdrawal of natalizumab may be as high as 80%, typically occurring within 12 weeks of discontinuation. 1 Rebound may be related to progressive α4-integrin receptor desaturation and immune reconstitution, enabling circulating lymphocytes to cross the blood–brain barrier. 2 Numerous bridging strategies for natalizumab discontinuation and transition to an alternate therapeutic agent have been proposed, although consensus on the optimal approach is presently lacking. 1 Cessation of natalizumab mandates appropriate therapeutic bridging and vigilant monitoring owing to substantial risk of withdrawal-related rebound and attendant neurological sequelae.
Footnotes
Authors’ Note
The authors take full responsibility for the data collected, analysis, interpretation, and conduct of this report.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
