Abstract
Context:
Somatosensory dysfunction in people with chronic ankle instability (CAI) has been reported to influence not only peripheral performance but also sensory reweighting of central nervous system. Sensory constraints, such as visual or somatosensory perturbations originating from sway surroundings or sway surface, continuously occur during body movements. Dynamically reweighting somatosensory, visual and vestibular cues are crucial for postural stability. However, due to contradictory evidence, it is unclear how sensory constraints affect sensorimotor functions and sensory reweighting ability in individuals with CAI.
Objective:
To examine the impact of sensory constraints on sensorimotor functions, sensory reweighting ability, and the sensory strategy selection of people with CAI during postural control.
Data Sources:
PubMed, Web of Science, EMBASE, Cochrane, SPORTDiscus, and Medline.
Study Selection:
Two authors independently screened article titles, abstracts, and full texts to select peer-reviewed studies exploring sensory constraints on sensorimotor functions in people with CAI and healthy controls.
Study Design:
Meta-analyses of descriptive epidemiological study.
Level of Evidence:
Level 4.
Data Extraction:
Center-of-pressure, center-of-gravity, and time-to-boundary measures that represent postural stability, and muscle activity amplitude and activation onset time were extracted.
Results:
A total of 43 articles were included. Compared with healthy controls, the CAI group exhibited static (Hedges’s g = 0.53) and dynamic (Hedges’s g = 1.05) deficits with visual constraint, as well as increased medial gastrocnemius activity (Hedges’s g = 1.02) during unilateral stance with visual constraint. The CAI group showed decreased sensory reweighting during unilateral stance with visual constraint (Hedges’s g = 0.26) and bilateral stance with visual and somatosensory constraints (Hedges’s g = 0.45).
Conclusion:
Visual constraint could alter postural control and muscle activation patterns in people with CAI. Sensory reweighting ability of those with CAI may fluctuate based on task and sensory constraints, and visual dominance strategy may help people with CAI modulate posture when visual information is reliable.
Since the first epidemiology study on ankle sprain in 1977, 19 research on ankle sprains has been widespread over the past 4 decades.12,17 Epidemiology survey reported that the incidence of sport-related ankle sprains ranges from 19.0 per 1000 person-years to 26.6 per 1000 person-years, 35 and the proportion of reinjuries can be as high as 47%. 29 Repeated injuries could contribute to chronic ankle instability (CAI), featured by lifelong recurrent ankle sprains, feeling of ‘giving way,’ and instability in daily and sports activities.20,21 CAI also can lead to substantial healthcare costs. 29 Despite evaluating the efficacy of existing therapeutic strategies, numerous studies have found limited success due to the complex mechanisms underlying CAI.11,90 Hence, investigating the contributing factors of CAI remains crucial to developing effective interventions.
Previous studies supported that ankle sprain could lead to disruption of joint mechanoreceptors, thereby triggering inaccurate somatosensory feedback from the ankle-foot complex to the central nervous system and resulting in altered motor patterns.28,51 These alterations may cause sensorimotor dysfunction in people with CAI, including somatosensory impairment,51,89 muscle strength decline, 36 postural instability, 90 and inhibited motoneuron pool excitability and spinal reflex reactions. 80 Sensorimotor functions involve the sensory, motor, and central integration and processing components during body movement, which is integral for postural stability, especially under dynamic environments and complex tasks.69,70
Postural control refers to the central nervous system integrating sensory information from somatosensory, visual, and vestibular systems to achieve appropriate motor behaviors and maintain controlled and upright posture.53,64,65 The environment is changing constantly, and sensory constraints from swaying surroundings (visual constraint) or uneven surfaces (somatosensory constraint) could pose challenges to postural control.64,65 After constraining ≥1 sensations, people will rely more on unconstrained sensations to maintain balance.64,65 The most critical process is to reallocate sensory cues, called sensory reweighting, which allows people to adapt to these sensory alterations via downweighting or upweighting sensory cues. 65 For instance, healthy people without CAI generally weight 70% on somatosensory, 20% on vestibular, and 10% on visual inputs in bilateral stance with eyes open, 64 but they reweight on somatosensory and vestibular feedback during bilateral stance with eyes closed, in this situation, the somatosensory and vestibular proportions may be upweighted.
However, sensory reweighting may be diminished in people with CAI. Studies reported that individuals with CAI presented increased visual reliance during unilateral stance and those with CAI were unable to appropriately reweight available sensory cues,32,73 indicating that people with CAI exhibited different sensory strategies and reduced sensory reweighting compared with healthy controls. Sensory reweighting requires dynamic regulation of multiple sensory subsystems, 66 and investigating the visual system alone cannot reflect the interaction among different subsystems and the selection of sensory strategies. After constraining plantar-sensory and visual inputs, increased postural sway was displayed in the CAI group during unilateral stance. 75 By contrast, Sugimoto et al 78 reported that people with CAI displayed better postural control and upweighted vestibular feedback compared with healthy controls in unilateral stance with visual and somatosensory constraints, suggesting that CAI people with displayed a comparable ability to reweight sensory cues. Apparently, conflicting findings were reported regarding the effect of sensorimotor constraints on the postural control for those with CAI, and hence it is still unclear how sensory constraints affect the sensory reweighting and sensory strategy selection, and thus postural control, in people with CAI.
Apart from postural control, sensory constraints also appear to influence muscle activation and proprioceptive function.41,46,71 Kwon 41 suggested that visual constraint could increase muscle activity, whereas Rosen et al 71 indicated that visual absence may hinder muscle activity and delay onset time. Understanding the effects of sensory constraints on sensorimotor functions could help clarify health-related problems and develop targeted intervention protocols for the CAI population. Therefore, this systematic review and meta-analysis aimed to: (1) assess the effects of sensory constraints (visual, somatosensory, and vestibular constraint) on sensorimotor functions; (2) evaluate the sensory reweighting ability; and (3) clarify the characteristics of sensory strategies in people with CAI.
Methods
The current study followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guideline and was registered in the International Prospective Register of Systematic Reviews (PROSPERO) platform on October 21, 2024 (CRD 42022375855). 1
Search Strategy
Two authors conducted literature retrieval independently from inception to August 2, 2024 in the following databases: PubMed, Web of Science, EMBASE, Cochrane, SPORTDiscus (EBSCO), and Medline (EBSCO). The search strategy consisted of 3 parts: ankle-instability-related, sensory-related, and constraint-related terms. Each term in the string was combined by ‘OR’ and between each string was combined by ‘AND.’ The details of the search strategy are displayed in Appendix 1.1 (available in the online version of this article). The reference lists of included studies were also checked manually to find more potential articles.
Study Selection and Data Extraction
Inclusion criteria were as follows: (1) participants aged between 18 years and 40 years with CAI and without CAI (healthy controls); (2) sensory constraints included somatosensory (foam or unstable surface, iced water immersion), visual (sway or occluded visual surrounding), or vestibular constraints (white noise) 63 ; (3) outcome measures aimed to assess sensorimotor functions by using valid and reliable instruments 28 ; (4) case-control, cross-sectional study, or baseline data from randomized controlled trials; (5) peer-reviewed articles with full-text in English or Chinese. Exclusion criteria included: (1) reviews, case studies, animal studies, and conference proceedings; (2) studies that included only comparisons of the injured side with the uninjured side of CAI, considering the potential bilateral deficits of unilateral CAI. 31 Details of outcomes included in this meta-analysis are presented in Table 1. After removal of duplicates, 2 reviewers separately screened articles and extracted data. Demographic details, study design, sample size, and methodology (instrument, testing method, outcomes measures with means and standard deviations) were extracted. For graphical data, Engauge Digitizer Version 4.1 was used to extract values.31,86
Outcomes for meta-analysis
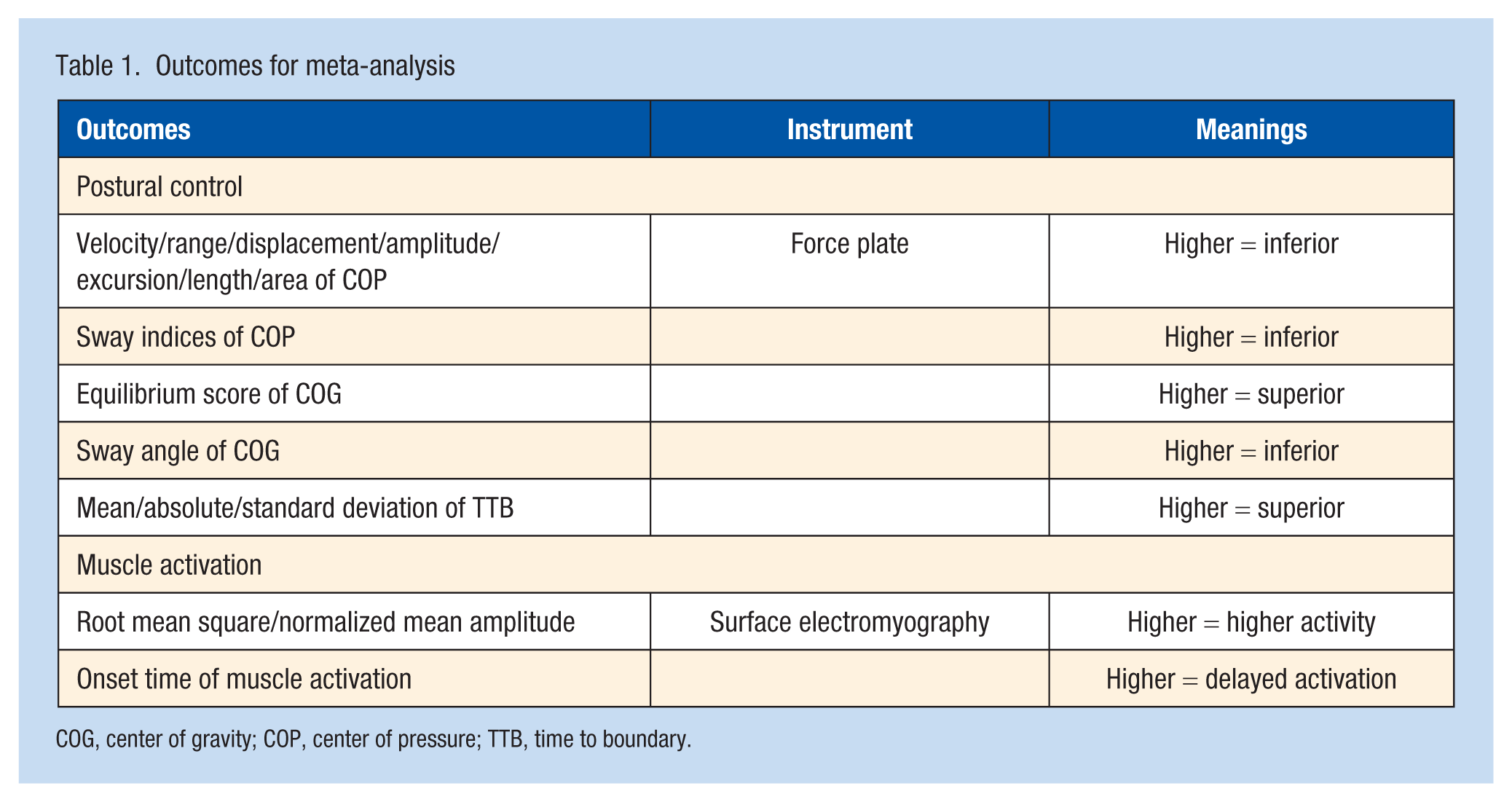
COG, center of gravity; COP, center of pressure; TTB, time to boundary.
Quality and Risk of Bias Assessment
Quality of included studies were assessed by epidemiological appraisal instrument, which was developed to evaluate ergonomics studies, and the risk of bias was evaluated by a standardized tool recommended by the Nonrandomized Studies Group of the Cochrane Collaboration.31,36,51 To evaluate the variability of CAI, the International Ankle Consortium recommendation for screening participants with CAI was applied.31,36,51 The evaluation methods and details were in accordance with previous studies.31,36,51 Disagreements were handled by consulting the third reviewer.
Statistical Analysis
Stata Version 18 (Stata Corp) with random-effect model was applied to pool data and conducted meta-analysis. Mean and 95% CI of Hedges’s g were calculated to examine the effects of sensory constraints on sensorimotor functions. To evaluate sensory reweighting, change values of postural control outcome before and after sensory constraints were used.2,73 Higher change value means reduced sensory reweighting.64,65,73 The calculation method of change value followed the Cochrane Handbook. 30 Subgroup analysis was conducted if >2 studies were included in each subgroup. The effect size was determined by Hedges’s g with small (0.2 to 0.5), moderate (0.5 to 0.8), or large (>0.8) effect. 79 Given the increasing number of studies that reported bilateral deficits in people with unilateral CAI, 31 comparisons of the uninjured side versus healthy controls were also conducted. The I2 statistic was applied to evaluate heterogeneity and I2 ≥ 75% indicated considerable heterogeneity, requiring cautious interpretation.31,51 Egger’s test was conducted to assess publication bias. 90 Sensitivity analysis was executed by removing 1 study at a time to estimate the robustness of pooled results. 90 SPSS Version 25.0 (IBM Corp) was applied to calculate the kappa value for evaluating the inter-rater consistency between the 2 reviewers.31,51,90 The inter-rater agreements were deemed almost perfect (≥0.81). 42
Results
Study Selection and Characteristics
A total of 1504 studies were identified, and 8 studies met inclusion criteria by manually checking of reference lists (Figure 1). After systematic screening, 51 studies (1229 CAI and 1072 healthy controls) were included in the systematic review and 43 articles were contained in meta-analysis. Among these 43 articles, 41 investigated postural control through static or dynamic tasks,3,4,7,8,14,16,18,22-27,33,34,37-40,47-49,54,55,57-62,67,68,72,75,78,81,82,85,87,91,92 9 explored muscle activation,10,15,27,41,44,50,71,83,84 1 assessed force steadiness and accuracy of ankle muscle, 46 1 evaluated gait variability, 74 and another investigated landing-cutting performance. 44 No study investigated vestibular constraint, 7 studies explored somatosensory constraint,3,4,10,14,18,37,78 and 48 studies assessed the effects of visual constraint on sensorimotor functions in those with CAI.3,4,7,8,15,16,18,22-27,33,34,38-41,44,46-50,54,55,57-62,67,68,71,72,74,75,78,81-85,87,91,92 Of these 48 studies, 8 investigated the effects of sway vision induced by stroboscopic glasses or swaying surroudings,18,24,25,44,46,74,78,85 and the remaining studies evaluated the influence of vision occlusion (eyes closed) on sensorimotor functions in people with CAI. Further details can be found in Appendix 1.2 (available online).
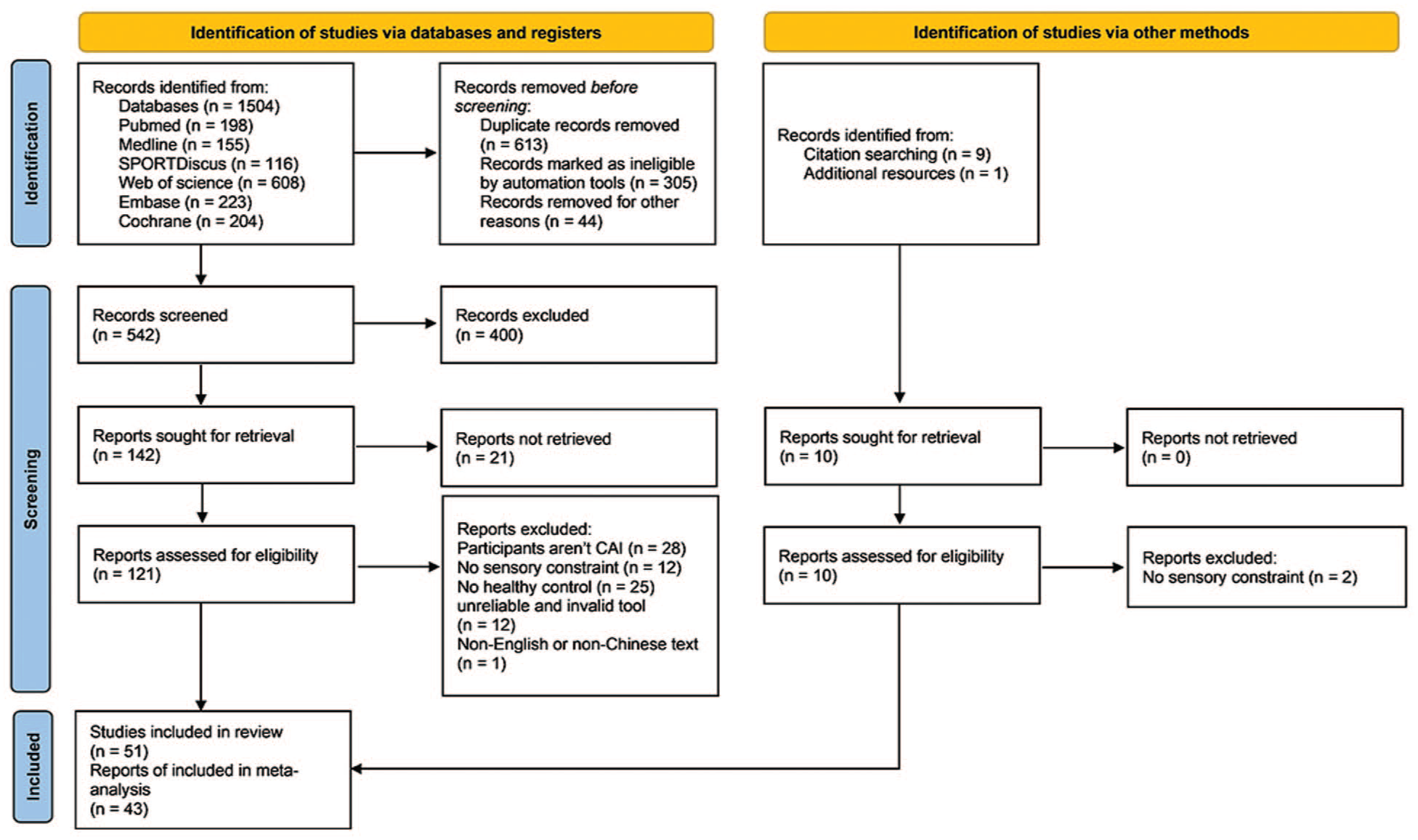
Flow chart of article screening.
Quality and Risk of Bias Assessment
For the quality of included studies (Appendix 2.1, available online), the agreement of the 2 reviewers was almost perfect (k = 0.890, P < 0.001), and the average score was 0.56. All studies provided clear purpose, main outcomes, statistical methods, and main findings, while most of the studies lack adjustment of covariates and confounders, sample size calculation, participants recruitment time and region. The risk of bias evaluation also achieved almost perfect agreements between 2 reviewers (k = 0.900, P < 0.001). All studies applied reliable and valid testing devices, normalized comparison between groups and appropriate statistical tests, but there was high risk of bias for detection (blindness) and attrition (Appendix 2.2, available online). A total of 344 agreements were achieved in the assessment of variability of CAI (k = 0.978, P < 0.001, Appendix 2.3, available online), while only 16 studies met all the recommendations of the International Ankle Consortium.
Effects of Visual Constraint on Sensorimotor Functions
For the effects of visual constraint on static bilateral stance, 5 studies assessed visual occlusion,4,18,22,47,55 and 1 study evaluated visual sway. 18 No significant differences were shown between 2 groups (Appendix 3.1, available online).
For static unilateral stance, 18 studies measured the velocity of center of pressure (COP), and quantitative synthesis presented small effects of postural deficits in the CAI group (anterioposterior [AP]: effect size [ES] = 0.29, 95% CI: 0.13 to 0.44; mediolateral [ML]: ES = 0.30, 95% CI: 0.11 to 0.49; Figure 2a). Sensitivity analysis showed higher overall velocity in the CAI group after removing the study of Zhang et al 92 (P = 0.015, ES = 0.31, 95% CI: 0.06 to 0.57), and heterogeneity also became lower (I2 changing from 97.09% to 47.13%). A total of 19 studies evaluated the amplitude, range, excursion, and displacement or length of COP, and the CAI group displayed small effects of instability (AP: ES = 0.28, 95% CI: 0.05 to 0.50; ML: ES = 0.40, 95% CI: 0.25 to 0.56; overall: ES = 0.43, 95% CI: 0.14 to 0.72; Figure 2b). A total of 11 studies assessed the sway area of COP and moderate effect of instability were presented in the CAI group (ES = 0.53, 95% CI: 0.19 to 0.88; Figure 3). The CAI group also presented small effect of lower values in AP (ES = −0.32, 95% CI: –0.59 to –0.04) and ML absolute time to boundary (TTB) (ES = –0.32, 95% CI: –0.60 to –0.05), while pooled results were unstable after removing the study of McKeon and Hertel. 54 No significant differences were found in the pooled results of mean and SD of TTB (Appendix 3.2, available online). Data from the uninjured side of 5 studies were also pooled for comparison,16,23,33,59,72 and results showed that there were no significant differences between the uninjured side of CAI and the control (Appendix 3.3, available online). Sensitivity analysis suggested that the CAI group showed postural deficits after removing the study of Mitchell et al 59 in both AP (P = 0.008, ES = 0.72, 95% CI: 0.19 to 1.26) and ML (P = 0.009, ES = 0.72, 95% CI: 0.18 to 1.25) directions. The significant differences observed in COP outcomes suggest that people with CAI could exhibit higher postural instability in both AP and ML directions after vision is restricted.
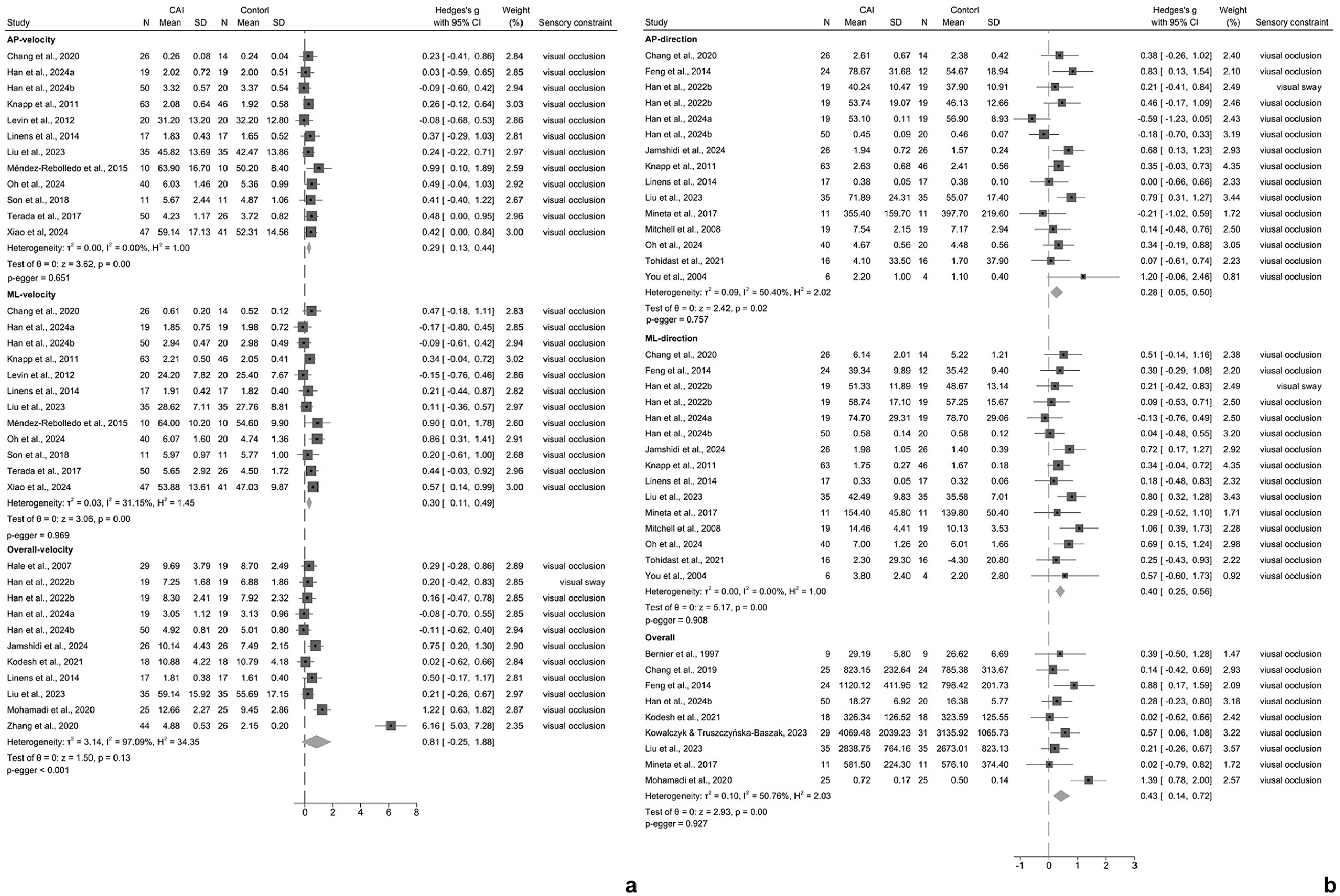
Forest plots of effects of visual constraint on static unilateral stability. (a) Velocity of COP. (b) Range/displacement/amplitude/excursion of COP. Positive Hedge’s g indicates postural deficits. AP, anteroposterior; CAI, chronic ankle instability; COP, center of pressure; ML, mediolateral.
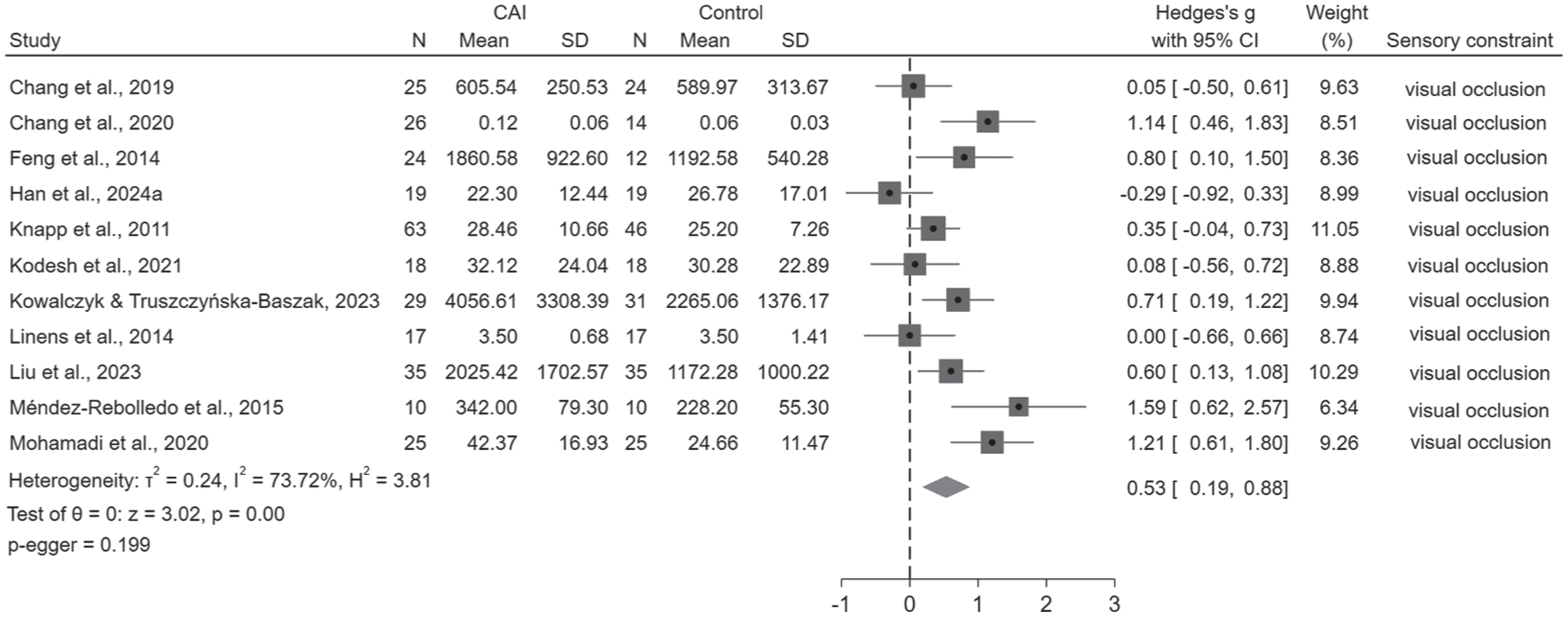
Forest plot of effects of visual constraint on sway area of COP during static unilateral stance. Positive Hedge’s g indicates postural deficits. CAI, chronic ankle instability; COP, center of pressure.
Only 2 studies assessed dynamic postural stability using force plate,24,47 pooled results showed large effect of ML instability in the CAI group (ES = 1.05, 95% CI: 0.41 to 1.69; Appendix 3.4, available online). Song et al 74 applied optical flow to produce sway vision and found higher variability of step length, step width, and ML sacrum motion, and shortened step length (Appendix 3.5a, available online). One study assessed dynamic stability using Y balance test with or without sway vision, and found that sway visual surroundings could result in postural instability in the CAI group (Appendix 3.5b, available online). 57 Lee et al 44 evaluated the kinematic and muscle activity during landing with or without visual disruption, and found that people with CAI presented more ankle inversion angle, and more peroneus longus activation from initial contact to 18% of stance phase under sway visual condition.
For muscle activation, 5 studies measured the muscle activity during static postural control and results indicated that medial gastrocnemius of the CAI group presented large effect of increased activity after visual occlusion in unilateral stance (ES = 1.02, 95% CI: 0.35 to 1.68; Appendix 3.6a, available online).15,27,41,50,58 No differences were observed in the comparison of glutes medius, peroneus longus, and tibialis anterior, while sensitivity analysis indicated that peroneus longus activity decreased after removing 1 study (P = 0.04, ES = –0.38, 95% CI: –0.75 to –0.01). 41 Two articles measured the onset time of muscle activation during transferring from bilateral stance to unilateral stance with visual occlusion (Appendix 3.6b, available online)83,84; pooled results suggested that people with CAI displayed moderate to large effects of delayed activation in gluteus medius (ES = 2.73, 95% CI: 0.32 to 5.13), vastus medialis (ES = 0.57, 95% CI: 0.12 to 1.06), peroneus longus (ES = 1.71, 95% CI: 1.10 to 2.31), and tibialis anterior (ES = 2.04, 95% CI: 1.26 to 2.83). However, no significant differences were observed in tensor fasciate latae and vastus lateralis. Rosen et al 71 reported that the time to peak activity of tibialis anterior during drop jump was delayed in the CAI group after visual occlusion (Appendix 3.7a, available online). The significant differences in muscle activation and onset time indicate that vision constraint could delay muscle activation and result in recruiting more motor units used for maintaining balance.
Only 1 study explored the effects of sway vision on force sense. 46 Compared with the controls, the CAI group showed higher force steadiness errors in 20% maximal voluntary isometric contraction of evertor and higher force accuracy errors in 20% maximal voluntary isometric contraction of evertor and abductor (Appendix 3.7b, available online).
Effects of Somatosensory and Somatosensory Plus Visual Constraints on Sensorimotor Functions
Four articles explored the effects of somatosensory constraint on static postural control (Appendix 3.8, available online).3,14,18,37 Among them, Fu and Hui-Chan 18 applied swayed surface and Kim et al 37 used iced water, while another 2 studies applied foam surface to produce somatosensory constraint.3,14 Pooled results indicated there were no significant differences between CAI and healthy control groups. Two studies measured the muscle activity of peroneus longus and tibialis anterior,10,50 and no significant differences were found (Appendix 3.9, available online).
Two studies investigated somatosensory and visual constraints on static postural control.18,78 Fu and Hui-Chan 18 measured the sway angle of center of gravity (COG) and Sugimoto et al 78 assessed the equilibrium score of COG, and they found that CAI group presented instability in swayed surface and eyes closed conditions (Appendix 3.10, available online).
Sensory Reweighting
There were no significant differences in bilateral stance, indicating CAI and healthy control presenting similar sensory reweighting ability (Appendix 3.11, available online). One study applied TTB parameters to evaluate sensory reweighting under different levels of visual occlusion and no differences were found between groups. 85 During unilateral stance with visual constraint (Figures 4 and 5), the CAI group showed small-to-moderate effects of decreased sensory reweighting (AP velocity: ES = 0.17, 95% CI: 0 to 0.34; ML velocity: ES = 0.26, 95% CI: 0.06 to 0.46; ML stability: ES = 0.26, 95% CI: 0.01 to 0.51; sway area: ES = 0.52, 95% CI: 0.18 to 0.87), which indicates that the people with CAI present higher visual reliance. Sensitivity analysis indicated that the pooled results of AP velocity lacked robustness. No differences were found in TTB parameters and the comparison of the uninjured side of CAI with healthy control (Appendices 3.12 and 3.13, available online).
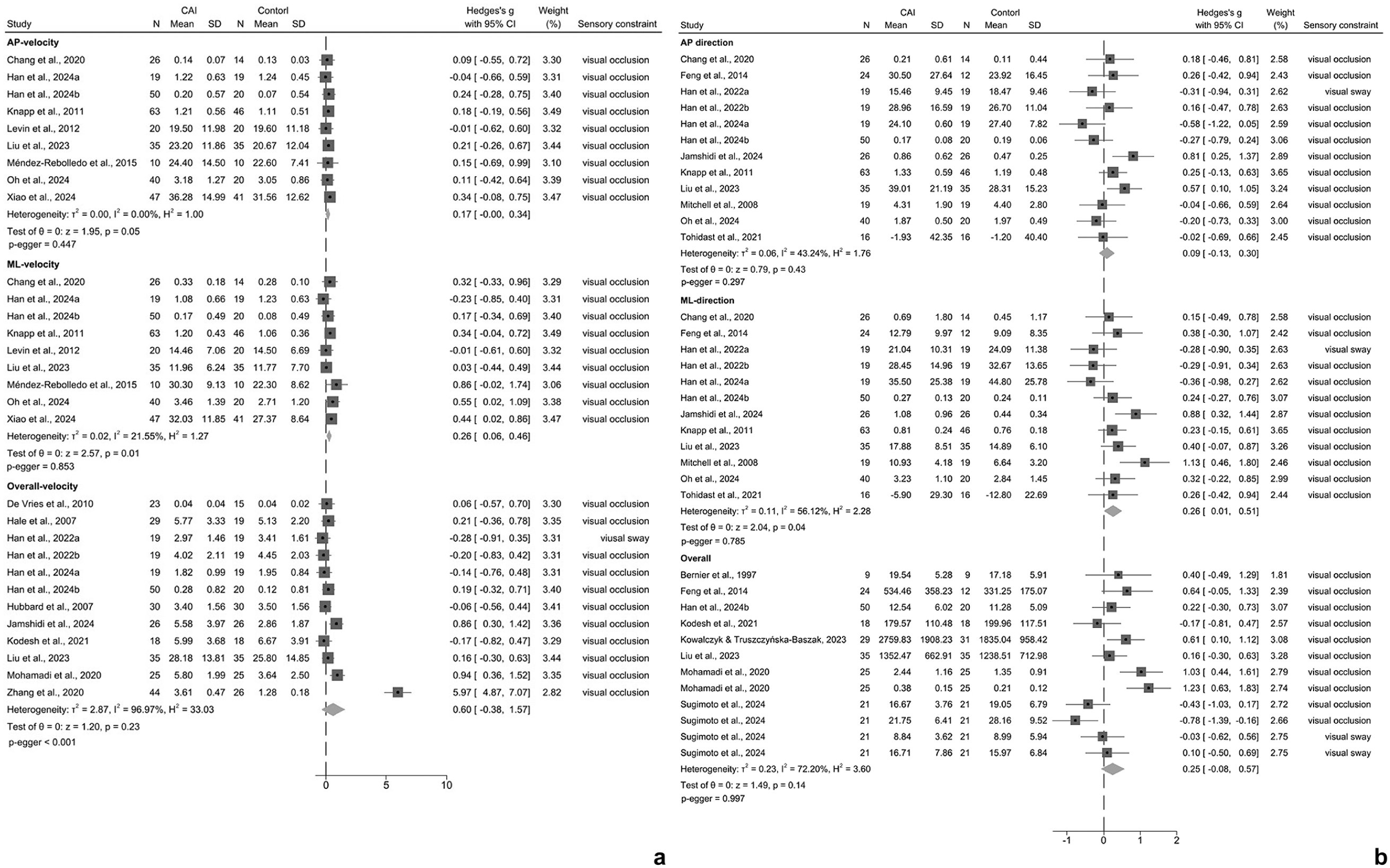
Sensory reweighting during unilateral stance with visual constraint. (a) Velocity of COP. (b) Range/displacement/amplitude/excursion of COP. Positive Hedge’s g indicates diminished sensory reweighting. AP, anteroposterior; CAI, chronic ankle instability; COP, center of pressure; ML, mediolateral.
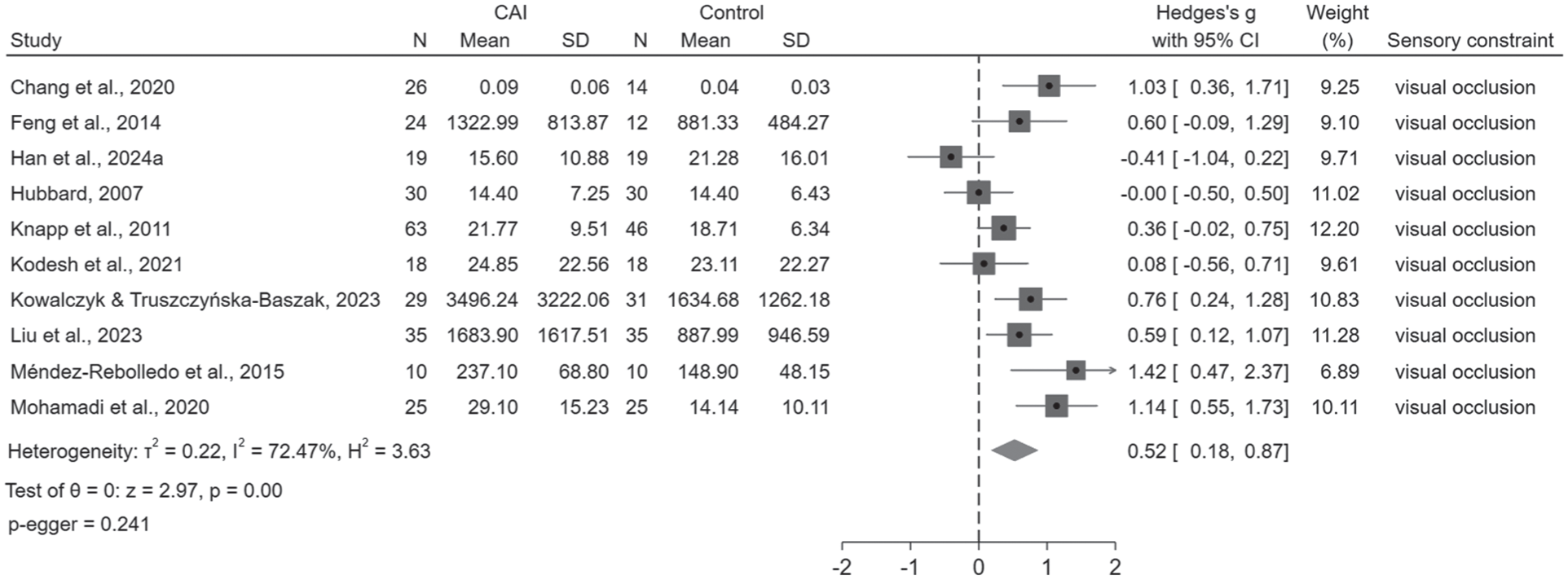
Sensory reweighting calculated by sway area of COP during unilateral stance with visual constraint. CAI, chronic ankle instability; COP, center of pressure.
Sensory reweighting with somatosensory constraint in bilateral and unilateral stances did not show significant differences between 2 groups (Appendix 3.14, available online). However, when adding visual and somatosensory constraints simultaneously (Appendix 3.15, available online), the CAI group presented small effect of decreased sensory reweighing during bilateral stance (ES = 0.45, 95% CI: 0.15 to 0.75), suggesting increased vestibular reliance. Sugimoto et al 78 also reported reduced sensory reweighting in the CAI group under sway surface and visual occlusion condition during unilateral stance.
Discussion
This systematic review evaluated the effects of sensory constraints on sensorimotor functions and sensory reweighting in people with CAI. The pooled results revealed that visual constraint could lead to postural instability in single-leg stance and dynamic control, increased medial gastrocnemius and decreased peroneus longus activity during unilateral stance, and delayed onset time of lower limb’s muscles under dynamic control in people with CAI. Specifically, the CAI group presented reduced sensory reweighting during unilateral stance with visual constraint and bilateral stance with visual and somatosensory constraint. In addition, synthesized evidence suggested that people with unilateral CAI displayed postural instability on the uninjured side during unilateral stance with eyes closure, but sensory reweighting may maintain a similar capacity to healthy controls.
Robust evidence supported the view that those with CAI displayed increased postural instability and reduced sensory reweighting during unilateral stance with visual constraint but not bilateral stance. To our knowledge, this is the first review to synthesize results from bilateral stance. Although Groters et al 22 recommended that bilateral stance could be used to evaluate postural deficits in people with CAI, our findings support the detection effect of unilateral stance on postural instability and reduced sensory reweighting. Postural stability originates from efficient integration of the abundant degrees of freedom in the sensorimotor system. 9 For people with CAI, recurrent ankle sprains imposed somatosensory constraint at the ankle joint,51,52 additional visual constraint may further limit the body’s degrees of freedom for modulating postural stability. However, for static bilateral stance, additional visual constraint may not pose challenges to the sensorimotor system in people with CAI because sufficient degrees of freedom for somatosensory feedback weakens the influence of visual restriction.9,78 With the increase in task complexity, despite the same visual constraint, limited degrees of freedom could amplify the influences of visual and ankle constraints.32,78
Moreover, this study reveals that COP measures can be used to assess diminished sensory reweighting in those with CAI. COP measures have been applied to evaluate sensory reliance and sensory reweighting in older adults. 2 Our review is the first to synthesis COP measures to quantify the sensory reweighting of CAI. A previous meta-analysis that applied TTB measures (i.e., estimated time of COP to boundary of base of support) to calculate standardized mean differences during single-leg stance with eyes closed versus eyes open and compared pooled 95% CIs between CAI and healthy groups for overlap, found that individuals with CAI exhibited greater impairments. 73 However, our study applied the change values of individual studies before and after sensory constraints to quantify sensory reweighting and pooled results did not display differences in TTB measures between 2 groups. Different synthesis approaches may cause discrepancy, and high variability in TTB boundary definitions may also lead to differences in TTB measurements. 90
In contrast, sensory reweighting and sensorimotor performance of people with CAI may be comparable with that of healthy controls when constraining somatosensory input in both bilateral and unilateral stances. Previous studies found that somatosensory cue was prioritized the most for maintaining bilateral stance, whereas visual cue was upweighted the most while maintaining unilateral stance.32,78 Meanwhile, somatosensory absence can be compensated partially by visual feedback. 70 Our study also confirmed that the CAI group presented increased visual reliance during unilateral stance, indicating that upweighted visual feedback in unilateral stance may counteract the posed somatosensory perturbation and ankle deficits. In addition, somatosensory feedback from proximal joints or other body segments may also offset potential somatosensory restriction during bilateral and unilateral stances. However, we must acknowledge the limited number of studies that explored the effects of somatosensory constraint on sensorimotor functions and the highly variable methods for inducing somatosensory perturbations, and hence this finding should be elucidated further.
Another interesting observation is that people with CAI may present vestibular reliance and a tendency of decreased sensory reweighting during bilateral stance when limiting somatosensory and visual inputs simultaneously. Functional brain imaging showed that there was enhanced functional connectivity between the thalamus and the somatosensory cortex after eye occlusion, which may help upweight available sensory cues and shift sensory processing mode away from visual dominance.5,78 As a pivotal component of the sensory system, the vestibular system acts as a gravitational receptor and provides feedback on the position of the head, trunk, and eyes in space. 56 The vestibular system usually works in conjunction with the somatosensory or visual system and serves as a reference for self-motion, particularly when sensory conflicts occur or when somatosensory and visual feedback become unreliable.64-66 However, pooled results suggested that conflicting somatosensory and visual feedback may not elevate vestibular usage in people with CAI. Compared with somatosensory constraint alone, adding visual and somatosensory constraints at the same time resulted in poorer sensory reweighting and postural control, which further supports our finding of CAI’s visual dominance strategy.
In addition, visual absence may induce increased muscle activity to resist postural sway. For instance, people with CAI present increased medial gastrocnemius activity during unilateral stance. The medial gastrocnemius plays a key role in reflex activation due to its superior torque production compared with the tibialis anterior, enabling more efficient ankle stabilization during forward movement patterns. 41 Those with CAI have been reported reduced soleus spinal reflexive excitability, and the increased medial gastrocnemius activity may compensate for the soleus deficit by trying to achieve similar stability comparable with those without CAI. 80 However, increased torque produced by medial gastrocnemius cannot modify the postural sway induced by visual absence. Furthermore, based on our findings, visual occlusion may delay muscle activation, which further increases the possibility of instability in people with CAI.
Sensitivity analysis indicates that the uninjured side of unilateral CAI displays postural instability during unilateral stance with visual occlusion after removing 1 study. 59 Previous studies also found unilateral CAI displayed bilateral sensorimotor impairments, not only in peripheral performance but also at spinal and supraspinal levels,13,31,32,77,88 indicating that somatosensory impairment at the injured side may affect the contralateral sensorimotor functions via several possible mechanisms, including “cross education” effects, interneurons, and interhemispheric interactions.31,32 We did not find sensory reweighting impairment in the uninjured side of unilateral CAI, suggesting that increased postural sway under eyes-closed condition arises from somatosensory deficits rather than visual loss. Brodoehl et al 5 found that eyes closure could enhance the somatosensory perception in healthy people, suggesting the uninjured side of CAI may retain the ability to reweight somatosensory information to compensate for visual absence. The uninjured side of unilateral CAI could present different sensory strategies compared with the injured side, with the injured side, but not the uninjured side, exhibiting visual strategy. However, a recent study reported that unilateral CAI displayed increased visual reliance on both sides, 32 which means that the bilateral sides of unilateral CAI presents visual strategy. Hence, future studies are still needed to confirm this notion in unilateral CAI.
Research Implications
First, this study found that different parameters for quantifying sensory reweighting may have potential variability, and the test reliability and sensitivity of TTB measures, such as using standardized boundary determination methods that help reduce the variability, need to be improved further. 90 Meanwhile, only small deficits were found in the CAI group during unilateral stance. More challenging postural tasks and standardized measurements should be used to evaluate the postural deficits in CAI patients, especially for clinical assessment. Second, sensory reweighting is the interaction among somatosensory, visual and vestibular systems. Isolating the contribution of a sensory modality by subtracting the condition where all sensory modalities are optimized from the condition with 1 disturbed sensory modality can measure the sensory reweighting indirectly, but cannot reflect the dynamic modulation among different sensory systems. 2 The sensory organization test, which can challenge different sensory subsystems and reports somatosensory, visual, and vestibular usage proportions, can be used to evaluate the sensory reweighting and clarify the dynamic interactions among 3 sensory systems.18,32,78 Third, due to the partial compensation effect among different sensory systems, for example, the somatosensory loss can be partially compensated by visual feedback, 69 the traditional assessment method of sensory reweighting cannot detect the compensation effect on postural stability among different systems. Further research could apply brain imaging techniques, such as electroencephalography or functional near-infrared spectroscopy, to clarify the functional connectivity among different sensory areas, which could help elucidate the mechanisms of diminished sensory reweighting in people with CAI.
Clinical Implications
This study provides evidence that people with CAI displays visual dominance strategy and sensory reweighting impairments during postural control, which highlights the importance of visual perturbation intervention in the management of CAI. Although previous studies have paid much attention to improving proprioception by balance training, the efficacy appears to be limited to enhancing sensory reweighting ability. 76 During competitive sports or complex tasks, vision is occupied by targets, and the central nervous system should rely on sensory reweighting to manage sensory allocation and achieve expected functional performance. There is emerging research on the use of stroboscopic glasses to induce visual constraint during balance training,43,45 but the efficacy for postural control and sensory reweighting enhancement still needs to be further confirmed. Moreover, increased vestibular reliance when disrupting somatosensory and visual cues indicates balance training with visual-vestibular adaptation may also benefit sensory integration and sensory reweighting in CAI patients. 6 The occurrence of bilateral deficits in unilateral CAI also urges researchers to update intervention methods that enhance sensorimotor functions on both sides.
Limitations
First, a recent study suggested sway vision may play a unique role in perturbing sensory integration compared with eyes closed. 32 Due to the limited articles and high heterogeneity of outcomes, subgroup analysis cannot be conducted based on different types of visual constraints. Our findings apply more to eyes-closed condition, and sensitivity analyses reveal the robustness of pooled results after excluding the studies with sway vision. Second, although we conducted comprehensive tests to assess the robustness of pooled results, including publication bias assessment, sensitivity analysis, and subgroup analysis, limited studies in several subgroup analyses hinder our results’ generalization. Third, due to the current study being an observational meta-analysis, confounding factors originating from cross-sectional studies cannot be controlled and sensorimotor deficits exhibiting before or after ankle sprains cannot be confirmed. Finally, varied eligibility criteria to recruit CAI and healthy controls, and different durations and sampling frequencies for postural measurements, may result in potential biases.
Conclusion
Sensory constraints could lead to extra burden on the sensorimotor system of people with CAI. Visual occlusion appears to influence postural stability by delaying muscle activation and inhibiting sensory reweighting. The sensory reweighting ability of CAI could fluctuate based on task and sensory constraints. The visual-dominant strategy of people with CAI may help maintain balance when visual feedback is reliable, but vestibular reliance during complex tasks could further hinder sensory integration. It appears that people with unilateral CAI present different sensory organization strategies between the injured side (visual reliance) and the uninjured side (upweight somatosensory cue) during unilateral stance with visual closure.
Supplemental Material
sj-docx-1-sph-10.1177_19417381261431342 – Supplemental material for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-sph-10.1177_19417381261431342 for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis by Xiaomei Hu, Ziwei Zeng, Cheuk-Yin Ho, Daniel T.P. Fong and Yijian Yang in Sports Health
Supplemental Material
sj-docx-2-sph-10.1177_19417381261431342 – Supplemental material for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-sph-10.1177_19417381261431342 for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis by Xiaomei Hu, Ziwei Zeng, Cheuk-Yin Ho, Daniel T.P. Fong and Yijian Yang in Sports Health
Supplemental Material
sj-docx-3-sph-10.1177_19417381261431342 – Supplemental material for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-3-sph-10.1177_19417381261431342 for Effects of Sensory Constraints on Sensorimotor Functions in People With Chronic Ankle Instability: A Systematic Review and Meta-analysis by Xiaomei Hu, Ziwei Zeng, Cheuk-Yin Ho, Daniel T.P. Fong and Yijian Yang in Sports Health
