Abstract
Background:
Ankle sprain causes proprioceptor injuries and prolonged joint deafferentation, which might lead to maladaptive neuroplasticity in patients with chronic ankle instability (CAI), especially in the cerebellum. Previous studies have indicated the impairment of superior cerebellar peduncle (SCP), but the inferior cerebellar peduncle (ICP) and middle cerebellar peduncle (MCP) have not been fully analyzed.
Hypothesis:
The cerebellar peduncles of participants with CAI would have altered fractional anisotropy (FA) and orientation dispersion index (ODI) in comparison with healthy controls without ankle injury history. In addition, FA and ODI would be correlated with the duration or severity of the sensorimotor deficits in CAI.
Study Design:
Cross-sectional study.
Level of Evidence:
Level 3.
Methods:
A group of 27 participants with CAI and 26 healthy controls underwent diffusion-weighted imaging scanning, with the cerebellar peduncles as the regions of interest. The measures obtained by single-shell diffusion tensor imaging and the multishell neurite orientation dispersion and density imaging were used. Correlation analyses were performed to examine the potential relationship between the FA/ODI and both the normalized Y-balance scores and the durations of ankle instability.
Results:
The ipsilateral ICP of the injured ankle in participants with CAI showed significantly lower FA (Cohen d 95% CI, –1.33 to -0.21; P = 0.04) and marginally significant higher ODI (Cohen d 95% CI, 0.10 to 1.20, P = 0.08) when compared with the same measures in the control group, with the ODI being positively correlated with the duration of ankle instability (r = 0.42, P = 0.03).
Conclusion:
The ICP in participants with CAI exhibited impaired integrity and a trend of abnormally organized neurites in comparison with a healthy control group.
Clinical Relevance:
The impairments of ICP might be an ongoing part of the pathological process of CAI, having the potential to become a target for the diagnostic evaluation of this clinical entity.
Keywords
Lateral ankle sprain (LAS) is among the most prevalent sport-related pathologies, and up to 70% of the general population will experience a LAS during their lifetime.10,16 Subsequent residual symptoms, including the self-reported perception of ankle instability and recurrences of sprains, known as chronic ankle instability (CAI), affect >40% of the patients after the initial acute LAS. 10 Patients with CAI are significantly limited concerning their participation in athletic sports and even physical activities of daily living. 10 Numerous treatments for CAI have been developed, such as external supports and even surgical restoration of the torn ligaments to restore ankle laxity.5,28 However, even after these or other therapeutic attempts, some patients will still have persistent ankle instability.5,28 Thus, the mechanical laxity of the ankle might not be the main mechanism of CAI, and efforts to explore other possible underlying factors of CAI are necessary.
The sensorimotor factors were suggested to play an important role in the development of CAI.15,31,36 Freeman et al9,19 first proposed that the proprioceptive afferent nerve endings would be injured along with the rupture of ligaments’ collagenous fibers during the occurrence of an ankle sprain. Proprioceptive signals provide the central nervous system (CNS) with information regarding the static spatial location and dynamic attributes (such as velocity and acceleration) of body parts in space, which is critical for the motor system to maintain balance and coordinate movements.13,23 This suggests that impaired ankle proprioception could give rise to increased joint instability and sprain rate.13,34 Recently, some researchers further suggested that the prolonged reflexive inhibition and activity-dependent plasticity that occur after joint injury might lead to maladaptive neuroplasticity of the CNS and cause persistent joint dysfunctions, which was also supported by several lower extremity injuries (eg, CAI, anterior cruciate ligament injury).14,19,20
With the development of MRI, the noninvasive in vivo quantification of the structure of CNS became accessible, and the cerebellum came to be known as an important site of sensorimotor deficits after ankle injuries in recent MRI studies.29,37 The cerebellum serves as proprioceptive sensory processing and balance control, consisting of 3 lobules (anterior lobules, posterior lobules, and vermis) and communicating with the brainstem and within cerebellar lobules through 3 pairs of cerebellar peduncles: inferior cerebellar peduncle (ICP), middle cerebellar peduncle (MCP), and superior cerebellar peduncle (SCP).21,30 By means of voxel-based morphometry of gray matter, Xue et al 37 reported atrophy of the cerebellar vermis in patients with lateral ankle instability. Regarding white matter, Terada et al 29 applied diffusion-weighted imaging (DWI) and observed posture-related SCP impairments in persons with LAS history, but the ICP and MCP were not analyzed in their study. As a result, a comprehensive analysis of the 3 cerebellar peduncles might benefit us in understanding the sensorimotor deficits in CAI.
DWI, which evaluates water diffusion, could be used to evaluate the structure of white matter: fluid diffuses most quickly along the major axis, so classical diffusion tensor imaging (DTI) analysis can differentiate the directionality and magnitude of water diffusion in neural tissue. 18 The aforementioned study by Terada et al 29 used DTI analysis and observed the lower fractional anisotropy (FA) values of SCP in patients. However widely used, classical DTI analysis can evaluate only the gross integrity of white matter, offering inherently low physiological specificity. 39 Neurite orientation dispersion and density imaging (NODDI) analysis based on the multishell DWI is a novel approach in the sport injury literature, and includes the neurite density index (NDI), which reveals the density of neurites; the orientation dispersion index (ODI), which reveals the angular variation of neurite; and the volume fraction of isotropic water (FISO), which reveals the isotropic extraneurite compartment (Figure 1).7,39 Recently, Xue et al 35 also performed an initial attempt of DTI and NODDI analysis on the corticospinal tract in patients with CAI and observed significantly lower FA and higher ODI in the affected tracts. Therefore, the application of classical DTI as well as the novel NODDI measures to the 3 cerebellar peduncles could provide more insight into cerebellar abnormality in patients with CAI.7,39
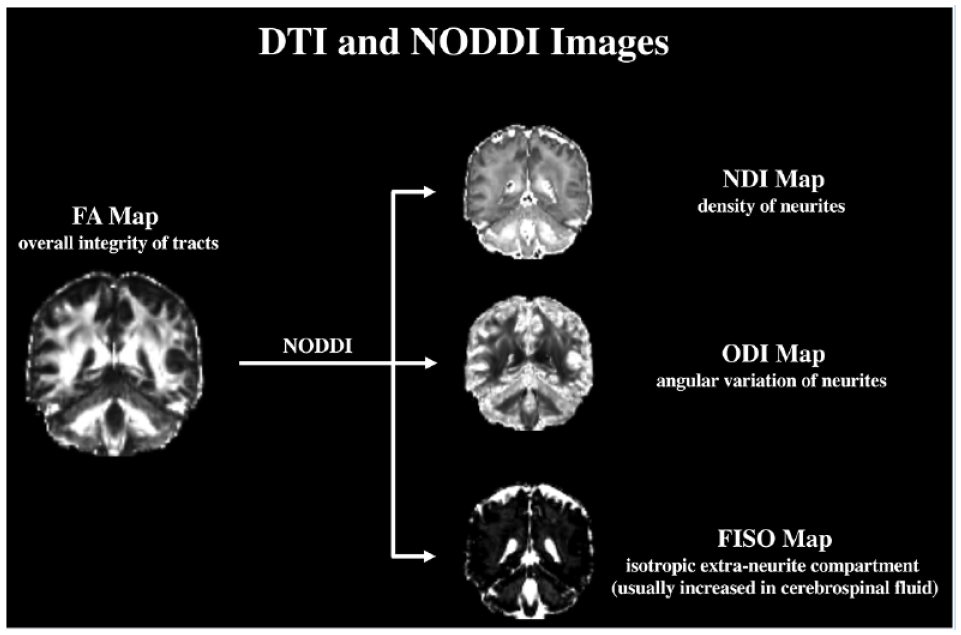
Visualization of FA maps as the primary outcome and the NODDI maps as exploratory outcomes with physiological meaning. FA, fractional anisotropy; FISO, volume fraction of isotropic water; NDI, neurite density index; NODDI, neurite orientation dispersion and density imaging; ODI, orientation dispersion index.
The current study aimed to conduct exploratory research on whether the properties of the 3 cerebellar peduncles were altered in participants with CAI when compared with healthy controls. In addition, the possible correlations between the potentially abnormal DTI/NODDI measures and the clinical features of CAI were investigated. Focusing on FA and ODI measures as the primary outcomes, it was hypothesized that the 3 cerebellar peduncles of participants with CAI would be impaired in comparison with those of healthy volunteers and that possible DTI/NODDI impairments would be correlated with the duration or severity of sensorimotor deficits.
Methods
Study Design
This was a cross-sectional study on the microstructural differences of cerebellar peduncles between participants with CAI and healthy controls without ankle sprain history. All the research protocols were approved by the Ethics Committee of Huashan Hospital (No. 2016-413), and all participants signed informed consent forms before enrollment. A single clinical assistant was responsible for recruiting participants and performing data acquisition and another blinded magnetic resonance technologist performed the image scanning and analysis. The report of this study fully followed the Strengthened Reporting of Observational Studies in Epidemiology Statement. 32
Participants
A total of 56 college students from the local university were recruited, including 28 participants with CAI and 28 healthy controls. G*power Version 3.1 (https://stats.oarc.ucla.edu/other/gpower/) was used to perform the prior sample size estimation, with FA values of SCP being estimated from the only previous DTI research on ankle instability (Cohen d effect size, 0.80). 29 Setting the statistical power and significance level at 0.80 and 0.05, respectively, the final sample size was the sum of its estimated minimum size (26 participants per group) and potential exclusions (2 participants per group).
The enrollment criteria of the CAI volunteers followed the guidelines of the International Ankle Consortium, which included the following: (1) ≥1 “significant ankle sprain” that resulted in pain, swelling, and ≥1 interrupted day of desired physical activity; (2) significant sprain that occurred ≥12 months before enrollment; and (3) persistent symptoms of ankle instability assessed by means of the Cumberland Ankle Instability Tool (CAIT) questionnaires (CAIT score <24).11,17 Healthy controls did not have any history of “significant ankle sprains.” Exclusion criteria for both groups were as follows: (1) history of previous surgeries to the musculoskeletal structures or a fracture requiring realignment in either lower extremity; (2) acute injury to musculoskeletal structures of lower extremity joints in the period of 3 months before enrollment; (3) history of major medical illnesses (eg, cardiovascular, respiratory, neurological, autoimmune, mental disorders); (4) current usage of psychotropics, excitants, or beta-blockers. 11 In addition, the right-footedness of all participants was also required, which was based on their preference when kicking a ball.
Data Acquisition
Demographic and clinical information, including sex, age, body mass, height, CAIT scores for self-reported instability, duration of ankle instability (measured from occurrence of first significant sprain), and other ankle injury features, were collected before scanning. For participants with bilateral injury history, the ankle with the lower CAIT scores was defined as the unstable ankle and tested in this study. Sensorimotor deficits was assessed by the Y-balance test (YBT) and only normalized posteromedial reach distances were used, which have greatest effect sizes and small variability to measure deficits in CAI.12,27 Each participant performed 3 formal trials after 6 practice trials, and the average distances were divided by the entire lower limb length (from the anterior superior iliac spine to the most distal portion of the medial malleolus) to give YBT scores. 12 Afterward, the brain was scanned using a 3.0-T MRI scanner (Magnetom Prisma, Siemens Healthineers) with the following acquisition parameters: (1) DWI with simultaneous multislice acquisition 38 : resolution, 2.00 mm isotropic; acquisition matrix, 110 × 110; flip angle, 90 deg; echo time, 72 ms; repetition time, 4100 ms; 30 diffusion gradient directions, multishell protocol with b values of 6 × 0 s/mm2, 30 × 1000 s/mm2, 30 × 2000 s/mm2, 30 × 3000 s/mm2 in 1 scan; scan times, 7 minutes and 4 seconds; (2) T1-weighted magnetization prepared 2 rapid acquisition gradient echo images 8 : resolution, 1.00 mm isotropic; acquisition matrix, 176 × 239; flip angle, 0 deg; echo time, 2.98 ms; repetition time, 5000 ms; scan times, 8 minutes and 16 seconds.
Data Analysis of DWI Images
Before processing, a blinded radiologist manually checked all images to exclude any potential abnormalities. Next, the eddy currents and head motions were corrected for all diffusion images through an affine alignment to the averaged b = 0 image using the linear image registration tool in functional MRI of the brain (FMRIB) software library Version 6.0 (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki). Also, individual brain masks were generated from the b = 0 image using the brain extraction tool of FMRIB to exclude nonbrain tissues. Then, maps of the primarily focused FA, and the mean diffusivity (MD) and radial diffusivity (RD) were derived based on the single-shell images with a b value of 1000 s/mm2 using the diffusion toolbox of FMRIB; the NODDI metrics of NDI, ODIm, and FISO were derived based on the multishell images with b values of 1000, 2000, and 3000 s/mm2 using NODDI toolbox Version 1.05 (https://www.nitrc.org/projects/noddi_toolbox) (Figure 1). 39 After that, the probability density maps of white matter that derived from T1 images were estimated using the unified segmentation algorithm implemented in Statistical Parametric Mapping Version 12 (https://www.fil.ion.ucl.ac.uk/spm/software/spm12/), and the FA maps were registered to the MNI152 space through the white matter maps using Advanced Normalization Tools Version 2.1 (https://picsl.upenn.edu/software/ants/).1,2 Finally, based on the saved FA-to-MNI registration parameters, all non-FA outcomes were also normalized to the MNI152 space. 33
In this study, region of interest analysis was confined to the cerebellar peduncles (ICP, MCP, and SCP). Binarized masks of cerebellar peduncles were segmented anatomically by Johns Hopkins University International Consortium for Brain Mapping DTI tracts and label atlases (Figure 2). 33 Voxels with FA values <0.2 were also excluded from the masks to reduce partial volume effect. Since the cerebellar peduncles are mainly ipsilaterally dominant, the ipsilateral cerebellum of the unstable ankle (ie, left side of the participants with left ankle instability) was measured in the CAI group, whereas the cerebellar side in the control group was randomly selected (Microsoft Excel Version 2021, Microsoft Inc). 29 The averaged DTI and NODDI measures within the individual masks of cerebellar peduncles were extracted from the MNI-normalized maps for further analysis.
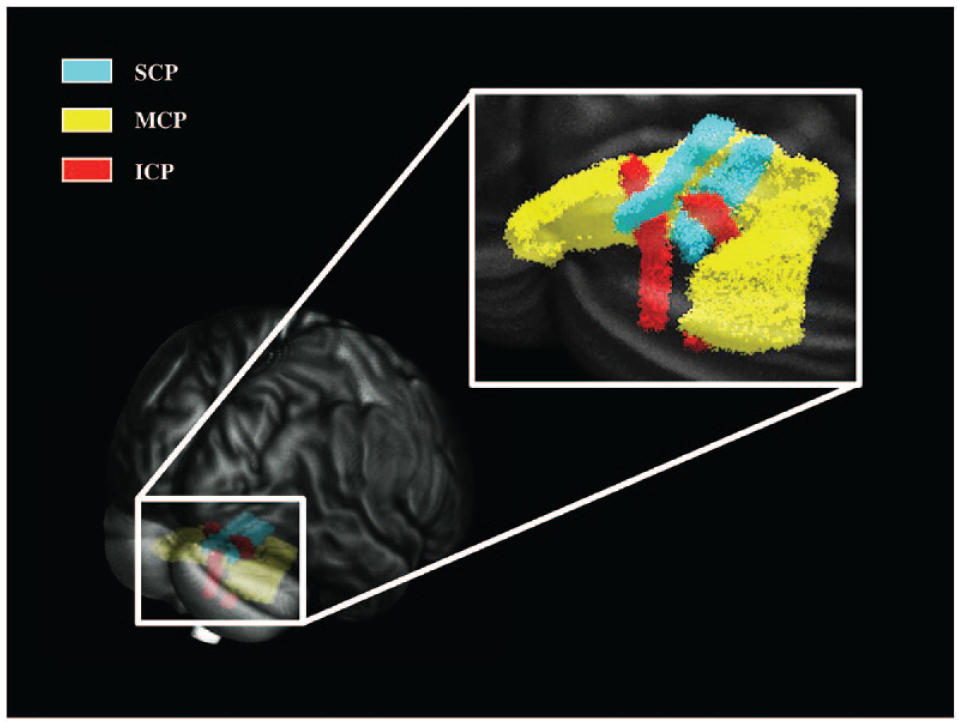
Visualization of cerebellar peduncles. ICP, inferior cerebellar peduncles; MCP, middle cerebellar peduncles; SCP, superior cerebellar peduncles.
Statistical Analysis
Statistical analyses were carried out on GraphPad Prism Version 9.0. (GraphPad Software). Data are expressed as means and standard deviations or medians [interquartile range]. Chi-square tests, Mann-Whitney U-tests, and independent Student t tests were applied to examine the equivalence of demographic variables and the differences of clinical features between groups. Independent t tests were also employed to compare the 2 volunteer groups for both DTI and NODDI outcomes. The magnitude of between-group differences were estimated by Cohen d effect sizes with 95% CIs, classified as small (0.2-0.5), moderate (0.5-0.8), or large (>0.8) effect size. 4 False discovery rates correction was performed among the P values of the FA and ODI values as the primary outcomes, 3 while uncorrected exploratory analyses were performed for MD, RD, NDI, and FISO. The cerebellar peduncles with significant outcomes in between-group comparisons were used in the following correlation analysis within the CAI group. Pearson’s correlation tests were employed to examine the relationship between DTI/NODDI outcomes and the YBT scores, whereas Spearman’s correlation tests assessed the relationship between DTI/NODDI outcomes and the duration of ankle instability. The significance level was set at α = 0.05, and any P < 0.1 was considered marginally significant.
Results
In all, 27 participants with CAI and 26 healthy controls were formally included in the analysis. One participant with CAI and 2 controls were excluded due to image artifacts. The duration of ankle instability in the CAI group was 8.0 [3.0, 10.0] years in this study. For the analyzed ankle, 14 left and 13 right ankles were defined as the unstable side of the participants with CAI. Among them, 4 participants have bilateral injury history, with 2 left and 2 right ankle defined as unstable. Based on random matching, 13 left and 13 right ankles were selected as controls in participants without ankle sprain history. No significant difference between the 2 groups was observed in terms of demographic features, including sex (P ≥ 0.99), age (P = 0.99), body mass (P = 0.69), height (P = 0.88), and body mass index (P = 0.52) . The CAI group had significantly lower CAIT scores (19.00 [13.50, 22.00] vs 29.00 [28.00, 30.00], P < 0.01) as well as YBT scores (1.01 ± 0.10 vs 1.08 ± 0.12, P = 0.04) when compared with healthy controls. The detailed demographic and clinical features of all participants are presented in Table 1.
Demographic and ankle injury characteristics for 2 groups
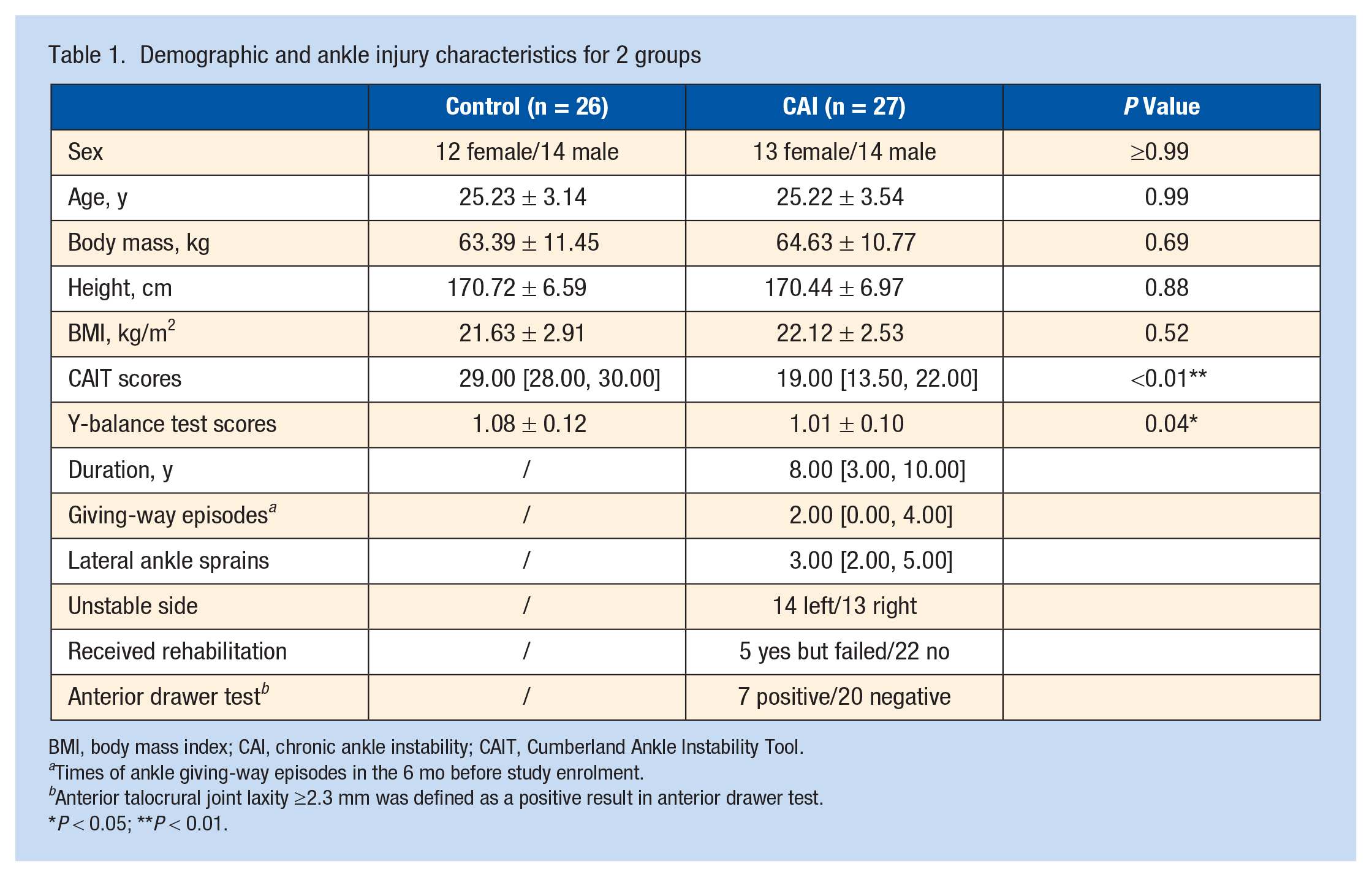
BMI, body mass index; CAI, chronic ankle instability; CAIT, Cumberland Ankle Instability Tool.
Times of ankle giving-way episodes in the 6 mo before study enrolment.
Anterior talocrural joint laxity ≥2.3 mm was defined as a positive result in anterior drawer test.
P < 0.05; **P < 0.01.
For the between-group comparison of DWI outcomes, the ICP of the CAI group showed significantly lower FA values (0.43 ± 0.03 vs 0.45 ± 0.02, Pcorrected < 0.01) and marginally significant higher ODI values (0.25 ± 0.02 vs 0.24 ± 0.02, Pcorrected = 0.08) with moderate effect size when compared with healthy controls. As for the FA of SCP, the difference between the CAI and the control group was only marginally significant, with moderate effect size before correction (0.47 ± 0.02 vs 0.49 ± 0.03, Pcorrected = 0.12) (Table 2). No significant differences between the CAI and control group were found for the FA values of MCP, or for any other DWI outcomes in all peduncles (P ranged from 0.12 to 0.99) (Table 2 and Table 3). For the correlation analyses, the ODI values of ICP were significantly inversely correlated with the duration of ankle instability (r = -0.53; P = 0.03), but no significant correlations were observed between the FA values and the duration or between the FA/ODI values and YBT scores (P = 0.16-0.24) (Figure 3).
Primarily analyzed DWI outcomes of cerebellar peduncles
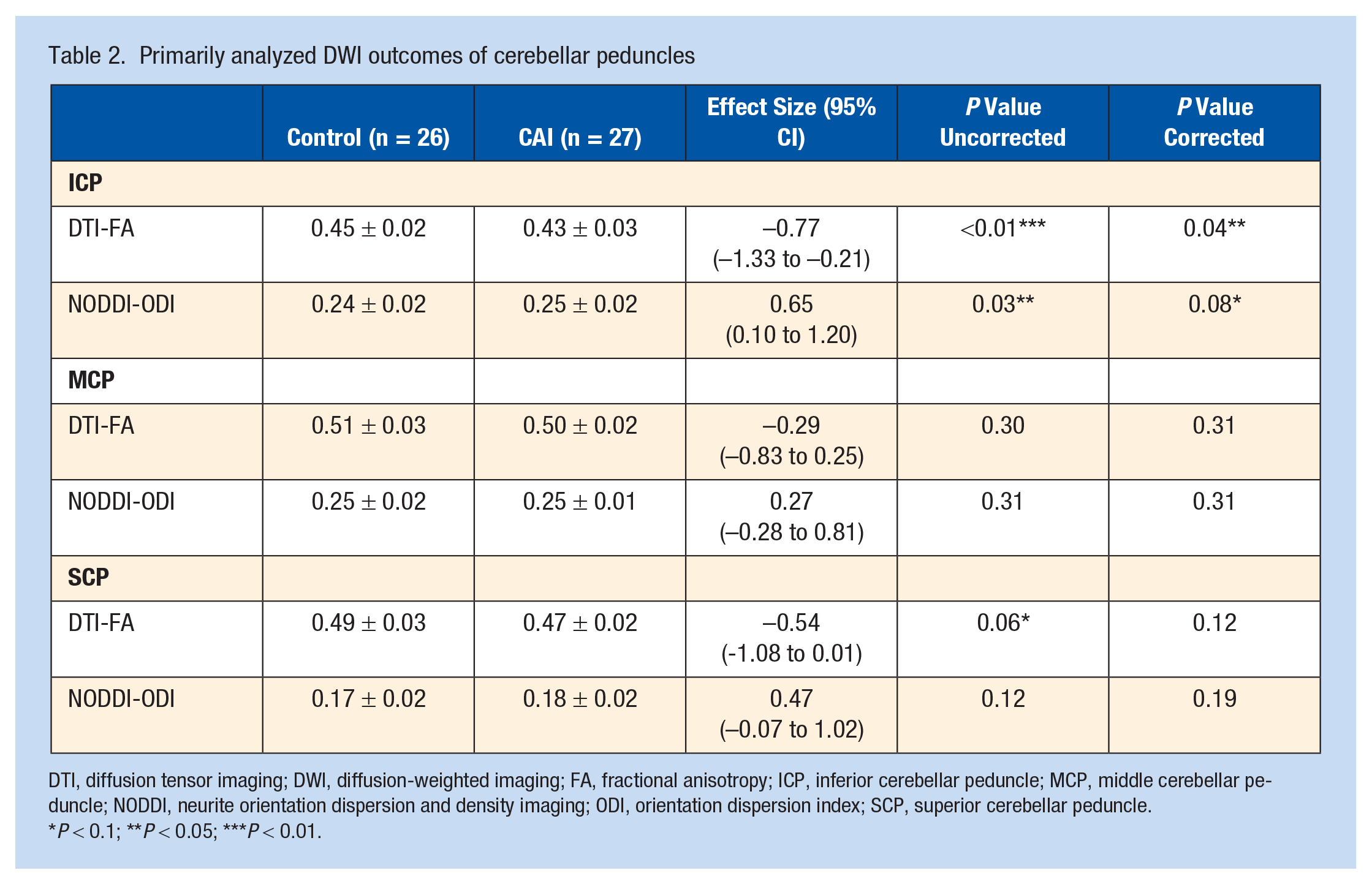
DTI, diffusion tensor imaging; DWI, diffusion-weighted imaging; FA, fractional anisotropy; ICP, inferior cerebellar peduncle; MCP, middle cerebellar peduncle; NODDI, neurite orientation dispersion and density imaging; ODI, orientation dispersion index; SCP, superior cerebellar peduncle.
P < 0.1; **P < 0.05; ***P < 0.01.
Exploratory DWI outcomes of cerebellar peduncles
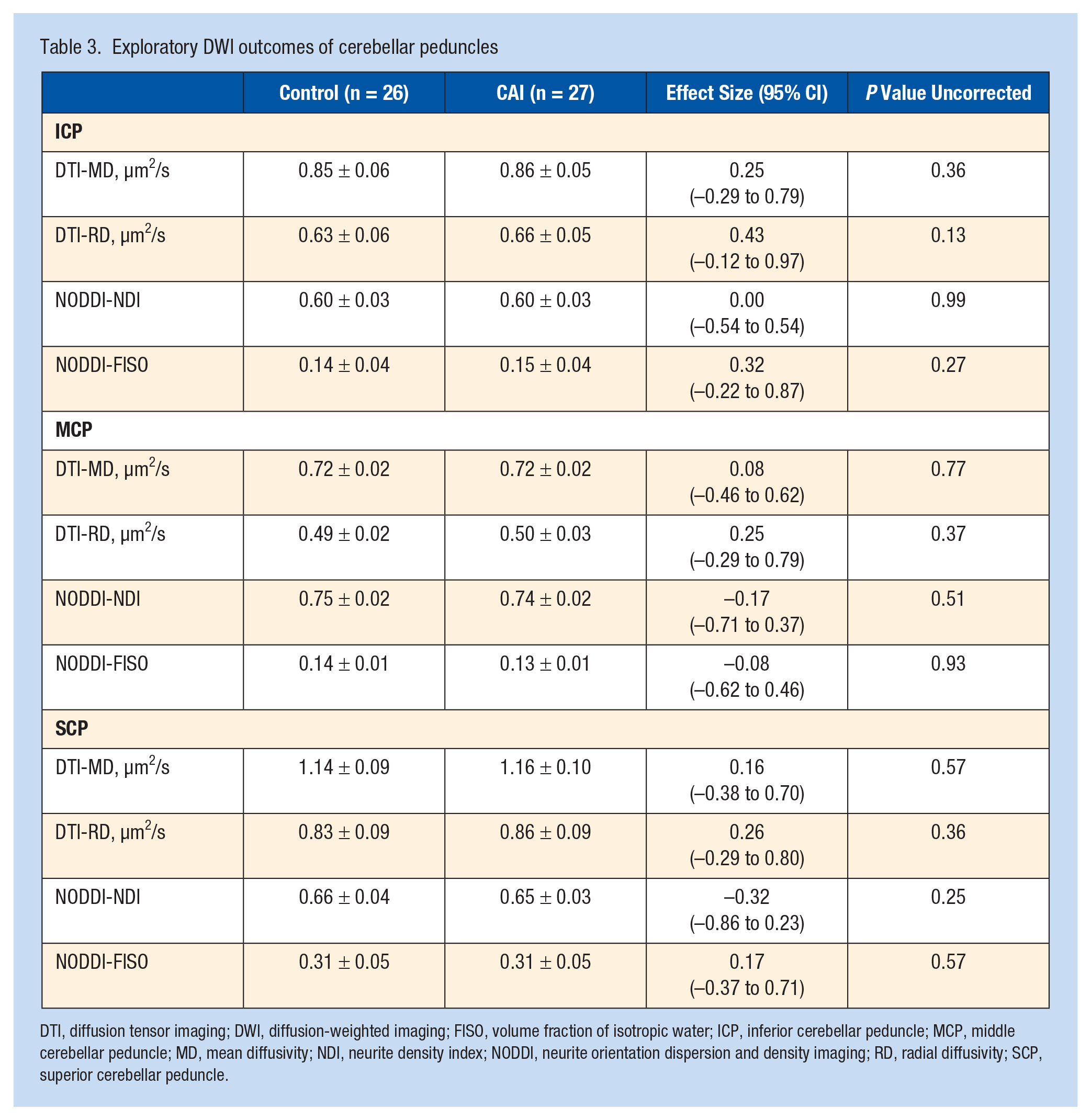
DTI, diffusion tensor imaging; DWI, diffusion-weighted imaging; FISO, volume fraction of isotropic water; ICP, inferior cerebellar peduncle; MCP, middle cerebellar peduncle; MD, mean diffusivity; NDI, neurite density index; NODDI, neurite orientation dispersion and density imaging; RD, radial diffusivity; SCP, superior cerebellar peduncle.
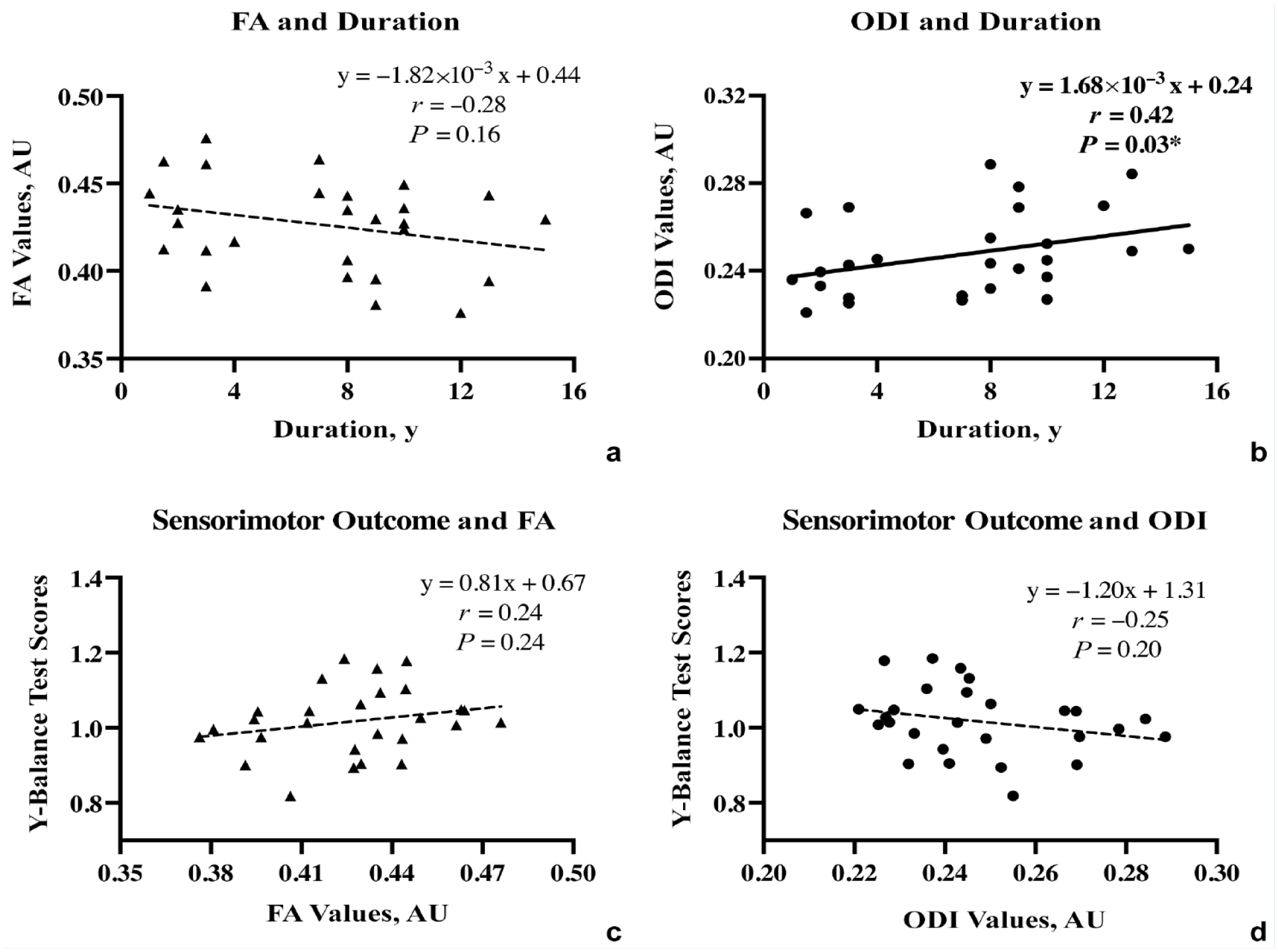
Scatter plot between ODI values/FA values of ICP and the duration or severity of the sensorimotor deficits (Y-balance test scores) in the CAI group. Statistically significant results with P < 0.05 indicated (*). CAI, chronic ankle instability; FA, fractional anisotropy; ICP, inferior cerebellar peduncle; ODI, orientation dispersion index.
Discussion
To the best of the authors’ knowledge, this is the first study on CAI to comprehensively explore the 3 cerebellar pathways utilizing both the DTI and the NODDI strategies. The most important findings of this study were that (1) the ipsilateral ICP of the unstable ankle in participants with CAI showed impaired integrity (lower FA) and (2) tended to have an abnormal angular variation of neurite (higher ODI) when compared with persons without ankle injury history. In addition, the abnormality of ODI was significantly associated (negatively correlated) with the duration of ankle instability.
Sensorimotor Pathway and Cerebellar Peduncles
The proprioceptive information originating in joints ascends through the posterior and anterior spinocerebellar tract and reaches the spinocerebellum via the ICP and SCP, respectively.23,25 The proprioceptive information (along with vestibular and visual inputs) and the corresponding motor commands will be repeatedly estimated by a circuitry that consists of (1) the ascending cerebello-thalamo-cortical tract from the cerebellar nuclei to the sensorimotor cortex via the SCP, and (2) the descending cortico-ponto-cerebellar tract from the cortex to the cerebro-cerebellum via the MCP.23,25 Finally, the encoded motor commands descend from the primary motor cortex through the corticospinal tracts and, through alfa motoneurons, reaches the peripheral muscles to maintain the balance.23,25 Certainly, all of these 3 cerebellar peduncles are essential for the proprioceptive sensory processing, and balance would be significantly disrupted if any of them were impaired.24,25 Therefore, taking into account the consistent association between CAI and proprioception deficits, the present study suggests that the impairment of the cerebellar peduncles, especially the ICP, might be a relevant underlying factor.
Impaired ICP and DTI/NODDI Outcomes
The structure of cerebellar peduncles could reveal the basic mechanistic changes in various clinical situations of sensorimotor deficits (eg, ageing, traumas, and multiple sclerosis),19,29,33 and the properties of the cerebellar peduncles could be improved by repeated training or impaired by prolonged disuse.24,25 The aforementioned theory of Freeman et al9,19 highlighted the interruption of sensory inputs caused by injuries to proprioceptors: a scenario supported by subsequent findings indicating that long-lasting regional inflammatory environments (eg, effusion, pain, and high capsule tension) could also contribute to the proprioceptive deafferentation in the sprained joint. As for the previous ankle instability-related study on the cerebellum, the atrophy of the cerebellar vermis (spinocerebellum) might also be indicative of potential maladaptive neuroplasticity of the proprioceptive input pathway. 37 Although the impairment of SCP was observed in persons with ankle sprain history also by Terada et al, 29 the findings of this study were not able to undoubtedly confirm their observation in patients with CAI. Regarding potential reasons, the additional CAI symptoms and the younger participants (mean age 27/28 years vs 58/63 years) might be the main difference between the 2 studies. The inclusion of a group of volunteers who had an ankle sprain but did not develop CAI is very much needed in future research to explore the role of the cerebellar features in central mechanisms that, despite having similar initial sprain histories, lead to different prognoses. In addition, more controlled studies that compare young/old participants with/without CAI might be needed to fully explain how the interaction of aging and ankle injuries influences cerebellar features. In the present study, the results of impaired ICP provided further evidence of white matter abnormalities in the proprioceptive input site.
The significantly lower FA values (DTI outcome) of ICP found in this study demonstrate a greater loss of structural integrity of the tracts. Providing a physiological explanation for this impairment, the marginally higher ODI values of ICP in participants with CAI also revealed a possible association between the disturbed coherence of neurites and the CAI-related deficits in the proprioceptive input pathway. As in a recent CAI study on the corticospinal tract, this study also did not observe significant differences beyond FA and ODI, which might hint that the structural integrity and coherence of neurites might be the specific pathological features of the affected white matter in CAI. 35 In this study, no significant correlation was observed between objective YBT scores and cerebellar features, which is inconsistent with the initial hypothesis of this study. The potential reason might be the complexity of the YBT (integrating proprioception, strength, and flexibility) that reduces its accuracy in reflecting the sensorimotor alternation in CAI.12,29,31 On other hand, a significant association was observed between the duration of ankle instability and ODI values, which points to ICP impairment being caused by CAI, while the reverse may not be ruled out by this cross-sectional study.
Clinical Implications
The mechanistic results of this study will be of especial interest to both patients and clinicians. Detailed assessments were necessary before, during, and after treatment. Although there exist several test protocols to measure proprioception deficits and predict the risk of reinjury in CAI, their accuracy and clinical feasibility are far from satisfactory. 31 Although not cost effective now, the popularization of 3.0-T MRI scanners in clinics and batched software designed for both single-shell DTI and multishell NODDI analysis might make the FA and ODI values of ICP a convenient biomarker of the severity of CAI, and assist as a specific indicator in treatment planning and prognosis assessment in the future. 39 Concerning intervention, the findings of an impaired ICP might guide the development of more effective rehabilitation strategies for proprioception deficits in CAI. Since some maladaptive neuroplasticity factor is hypothesized to be an underlying mechanism of persistent ankle instability, interventions attempting to restore the proprioception pathway of the ICP might provide better outcomes.22,26 For example, augmented, repetitive, and task-specific balance training was proved to increase the fiber integrity of cerebellar peduncles in patients with neuropathy, and some supplementary enhancing strategies (eg, neuromuscular electrical stimulation) might also be able to activate blocked sensory pathways.5,6,24,25 Certainly, more prospective studies are still needed to determine whether the results of this study could help optimize the clinical management of CAI.
Limitations
Major limitations of this study should be noted. First, the cross-sectional design could not fully explain the causal relationship between the CNS difference and the CAI. A prospective study of ankle sprains with baseline imaging may help validate these results. Second, this was an exploratory study that used only region of interest analysis to motivate future research on the established CAI-related region (ie, cerebellar peduncles). A whole brain, voxel-wise analysis supported by a larger sample size would be needed to provide more comprehensive evidence of white matter alternations in CAI. Third, the region of interest analysis was based on the whole cerebellar peduncles, which did reduce its sensitivity in detecting between-group differences because the proprioceptive afferent fibers originating from the ankle are only a part of the cerebellar peduncles (merged with visual and vestibular information, as well as proprioceptive afferent fibers from other parts of the body). Fourth, despite the full theoretical basis of the maladaptive neuroplasticity and proprioception deficits, the lack of significant correlation between the ICP measures and the behavior scores limited the quality of this study. Fifth, the duration of ankle instability is based on self-reported events. Although an injury incident leading to serious consequences may be impressive, recalling that event years later may require an effort inevitably biased. Notably, about half of the patients may never seek any professional healthcare for their initial ankle sprains, so the lack of accurate medical records might be a common problem in the retrospective analysis of CAI. 10 Sixth, the sample size estimation referred only to the FA values of SCP in previous studies, which may lead to a sample size insufficient to detect significant between-group differences for other outcomes.
Conclusion
This study revealed that the ICPs of persons with CAI exhibited impaired integrity and a trend of abnormally organized neurites, indicated by lower FA and higher ODI when compared with healthy controls. The degree of abnormality in neurite organization was correlated significantly with the duration of CAI, which indicates that the impairment of ICPs could be a structural maladaptive process of neuroplasticity induced by CAI.
Footnotes
Acknowledgements
The authors report no potential conflicts of interest in the development and publication of this article.
