Abstract
Bagassa guianensis Aubl. is a tree of high timber value (Moraceae family) native to the Amazon. In this study, we characterized the morphoanatomical and biochemical properties of Bagassa guianensis seeds at different levels of fruit control based on biometric, morphoanatomical, color and biochemical analyses. Four stages of seed development have been identified in the fruit of Bagassa guianensis. The seeds presented endosperm, being classified as albuminous. Seeds from fruits with 5 GY 4/4 colors and green pigmentation (stage I) presented fully formed embryos. According to PCA (principal component analysis) data, it was found that all stages of seed dominance had a significant effect and highly correlated with the biochemical properties of the seed. Despite the comparatively low biometric values, high levels of Total Soluble Sugars (3.58%), Total Soluble Proteins (2.12%) and Reducing Sugars (1.43%) were observed in seeds at this stage. On the other hand, the content of amino acids (0.87%) and starch (3.30%) was higher at late maturity. The highest amounts of starch were detected from stage II onwards (31.29 mg/g DM), when the seeds reached physiological maturity. Biochemical and morphological information from seeds are relevant for planning conservation, observation and restoration strategies, and this study on the species B. guianensis is essential.
Introduction
The family Moraceae comprises about 63 genera and 1500 plant species distributed throughout tropical and subtropical regions, including a few species from temperate areas (Souza & Lorenzi, 2012). In Brazil, this family is represented by 21 genera and 231 species, being 71 of them regarded as endemic and 206 referred to as synonyms (Pederneiras et al., 2020). Based on phylogenetic inferences, Moraceae was recovered as one of the nine families presently recognized in the order Rosales (APG IV, 2016).
Bagassa guianensis Aubl. is a member of the family Moraceae, considered an endemic species to the Amazon forest where it is locally known as “tatajuba”. This is a dioecious tree, reaching 15 to 30 m in height, and characterized by an elongate canopy and a thick and fibrous bark (Rios et al., 2011). The leaves of this species are simple, opposite, three-lobed in juveniles but entire in mature specimens, while the fruit is globose with a fleshy pulp and numerous flattened seeds (Embrapa, 2004). In Pará, northern Brazil, the flowering of Bagassa guianensis takes place from August to September, and the production and dispersal of fruit usually occurs between November and February (Rios et al., 2011). This is a valuable timber species, particularly because of the workability and resistance of wood to diseases and parasites, being widely used for construction, carpentry, high-quality furniture, and boat-building (Lima et al., 2018).
As a result of their high-quality timber and intensive exploitation, natural populations of Bagassa guianensis have been decreasing (Rios et al., 2011), representing a major concern particularly because this species presents low rates of germination and survival (Miranda & Carvalho, 1998). Even though studies about seeds of tropical tree species have been intensified over the last years (Ribeiro-Oliveira & Ranal, 2014), most species lack basic information about the morphoanatomy, biochemistry and maturation of seeds, such as the native trees from the Amazon region.
The maturation and the development of seeds encompass several physical, physiological and biochemical changes from their fecundation up to the formation of mature seeds (Marcos Filho, 2018). Therefore, the identification of morphoanatomical and biochemical alterations in seeds along their developmental stages is essential to understand this process (Trancoso et al., 2021).
In addition, the analyses of biochemical aspects of seeds during maturation allow inferring the levels of reserve compounds in cotyledons (Henning et al., 2010). This information is helpful to design proper procedures related to seed technology, since vigor and storage abilities of seeds are influenced by their compounds (Carvalho & Nakagawa, 2012), particularly in relation to carbohydrates, proteins, and lipids (Zhao et al., 2018). These stored compounds are consumed during germination and development of embryos up to the formation of autotrophic seedlings (Buckeridge et al., 2004).
Moreover, the knowledge about the biology and the structure of native plants is fundamental to in situ or ex situ conservation programs, as well as to the identification of species and physiological, biochemical, and evolutionary inferences. Therefore, the goal of this study was to characterize the morphoanatomical and biochemical properties of Bagassa guianensis seeds at different levels of fruit control based on biometric, morphoanatomical, color and biochemical analyses.
Methods
The fruit samples of Bagassa guianensis were collected randomized in March 2021 form from five plant matrices of annual dispersal along the sampled area, which comprised agroforestry systems located on private properties, in the municipality of Altamira (03°22'56.87” S; 52°26'02.89” W). The collected material was transported to the Laboratory of Biotechnology (BIOTEC-ATM), at the Federal University of Pará-UFPA, campus of Altamira, for further selection of undamaged fruit. Both fertile and sterile branches were collected for preparation of exsiccates and deposited in the Herbarium Padre José Maria Albuquerque (HATM) from the Faculty of Biological Sciences, Campus of Altamira (code identification numbers 2323 and 2324).
Morphological and Anatomic Analyses
To determine the different stages of development of the fruits, 100 samples of fruits of different colors from the canopy and the ground were collected. The classification of fruit coloration was carried out according to the catalog of colors developed by Munsell (1976). The morphological identification of fruit and seeds followed Barroso et al. (1999).
For the analysis of the morphology of the fruit of Bagassa guianensis (length, width, and thickness), 25 fruits were analyzed for each stage (I, II, III, IV), after the Munsell classification. For the morphological analysis of the seeds (length, width, and thickness) 100 sample units of each stage were considered. For biometric analyses, we measured the width, length, and weight of fruit and seeds using a digital pachymeter (Mtx) and an analytical scale (AUY220, Shimadzu). The humidity content of seeds was determined after drying them in over at 105 ± 3°C for 24 h (Brasil, 2009).
The anatomic studies were carried out in seed samples from each stage fixed in FAE (formaldehyde, glacial acetic acid, and ethanol 50% at a ratio of 1:1:18, v/v) for 48 h and then stored in ethanol 70%, according to Kauss & Arduin (1997). Afterwards, the seeds were cut into longitudinal and transversal sections using slides followed by clarification in sodium hypochlorite and distilled water (1:1). The final cuts were washed in distilled water, stained with toluidine blue and mounted onto glass slides and coverslips using glycerin.
Biochemical Analyses of Seeds
The seeds were dried in a bench oven (Solab – SL-100) at 65°C for 48 h. Subsequently, the material was milled for further biochemical analyses. The dried material was stored and sent to the Experimental Forestry Unit at the State University of Feira de Santana (UEFS), in Bahia, northeastern Brazil, in 2021.
The extraction for the quantification of Total Soluble Amino Acids (AA), Total Soluble Sugars (TSS), and Reducing Sugars (RS) was performed using 0.2 g of dried material homogenized in 10 mL of 0.1 phosphate buffer (pH = 7). The ninhydrin test (Yemm & Cocking, 1955) was used to quantify amino acids, using glycine as a standard sample. The reading was performed in a UV spectrophotometer (UV-M51) at 570 nm.
For the extraction of starch, 0.2 g of dried material was placed into a centrifuge tube, homogenized in 10 mL of 80% ethanol, and incubated at 40°C for 10 min. The amount of TSS and starch was determined based on the anthrone test as described by Yemm & Willis (1954), using glucose as a standard carbohydrate and reading parameter at 620 nm in a UV spectrophotometer. The quantification of RS was accomplished based on the procedure reported by Miller (1959), using 3,5-dinitrosalicylic acid (DNS) and glucose as a standard sample. The reading was performed at 540 nm in a UV spectrophotometer.
The protein fractions from seeds at different stages of maturation were extracted according to their solubility (albumins, globulins, prolamins, and glutelins), according to the methodology proposed and adapted by Osborne (1924). The protein fractions were determined according to Bradford (1976) using bovine serum albumin (BSA) as a standard sample in a UV spectrophotometer at 595 nm.
The extraction and the quantification of lipids were carried out in a Soxhlet-type apparatus using the cold extraction method with petroleum ether for 4 h, adapted from the method reported by Silva (1990).
All biochemical assays were performed in triplicate, with samples obtained after harvesting the fruits from the matrices that were mixed to obtain the amount of material necessary for the analyses.
Statistical Analyses
For the analysis of the morphology of fruits and seeds, and quantification of lipids, descriptive statistics were performed, showing the mean and standard deviation. To analyze the relationship between the morphological (length, width, and thickness) and biochemical Total Soluble Amino Acids (AA), Total Soluble Sugars (TSS), Reducing Sugars (RS), Proteins and protein fractionation parameters, the PCA (principal component analysis), with a Biplot sorting chart, followed by the ENVIFIT analysis, with an error probability considered less than 5%. This function seeks the maximum correlation between the vectors of the corresponding variables (morphology and biochemistry) of the Bagassa guianensis fruits with the ordination points (stage of fruit development). The correlation can vary between 0 and 1, that is, the closer to 0, the lower the correlation. The analyzes were performed in the R language (R CORE TEAM, 2020).
Results
Morphological, Biometric and Anatomic Analyses
Description of epicarp coloration according to Munsell (1976).

The fruit of Bagassa guianensis is recovered by a granular surface, formed by female flowers clustered into globose inflorescences (capitula composed of thousands of flowers). The infructescence is composed of several flattened seeds. According to the data published by Embrapa (2004), the fruit of Bagassa guianensis comprises a seed protected by the pericarp. The epicarp is thick and characterized by a granular surface, with a rough texture, varying from green (immature forms) to brown or dark brown in mature and highly mature fruit, respectively. The length and width of fruit ranged from 27.39 mm to 30.04 mm long and from 68.76 mm to 63.26 mm wide. In immature samples (Figure 1A), the pulp derived from a developed fleshy receptacle presents a greenish color. In mature fruit (Figure 1B), the pulp becomes orangish, representing the edible portion, being larger than in immature samples and encompassing seeds with a greater amount of mucilage in their tegument as a result advanced maturation stage. Longitudinal cuts of immature (A) and mature (B) fruit of B. guianensis.
Descriptive statistics of fruit traits at different stages of maturation (Min. V = minimum value. Max. V = maximum value, S = standard deviation, and CV = coefficient of variation). n = 25 fruits/ stage.

The diameter of the fruit samples ranged from 30.04 mm to 52.77 mm (average of 43.60 mm) at stage I; from 31.85 mm to 59.88 mm (average of 44.13 mm) at stage II; from 41.13 mm to 63.26 mm (average of 51.42 mm) at stage III; and ranged from 41.77 to 60.19 mm (average of 48.49 mm) at stage IV. The weight of fruit samples ranged from 48.80 g to 72.70 g. A variation in fruit size was observed throughout their development, particularly in relation to weight as indicated by the highest coefficient of variation (CV = 37%) of this trait in fruit samples at stage II. On average, the fruit samples between 12.60 g and 71.6 g encompassed 97 seeds. On the other hand, fruit with weight values between 15.9 g and 80.90 g presented a mean number of 296 seeds, while samples between 38.23 g and 116.22 g had a mean number of 420 seeds per fruit.
The seeds of Bagassa guianensis are flattened and slightly oval, presenting a brownish coloration and a conical embryo filling most of the seed (Figure 2). A large amount of mucilage is observed on epicarp of seeds in mature fruit when compared to that observed in green fruit samples. Seeds of B. guianensis seeds: (A) visible part of the mucilage; (B) trichomes, (C) longitudinal section showing the cotyledon (Co), endosperm (En), and hypocotyl-radicle axis (Ex).
Descriptive statistics of seed traits at different stages of maturation (Min. V = minimum value. Max. V = maximum value, S = standard deviation and CV = coefficient of variation). n = 100 seeds/stage.

Because of their small size, a total of 100 seeds were weighed to determine their average weight at each stage. The seeds at stages I and II presented the highest weight, while those from stages III and IV had the lowest average weight values (0.36 g and 0.34 g, respectively). A remarkable variation was observed in the seed size, “width” the most variable trait, as represented by high CV values at stages I and II (CV = 13.76% and 13.19%, respectively), followed by stages III and IV (CV = 12.33 % and 10.70%, respectively).
The highest humidity values were reported in seeds at stage I (76.9%), followed by those obtained at stages II (75.1%) and III (72.1%). The seeds with the lowest humidity content were observed at stage IV (71.5%).
The main structures in the seeds of Bagassa guianensis are shown in Figure 3. The seeds are egg-shaped with a more acuminate end at one side and a truncated extremity on the other side (Figure 3A), being composed of tegument, endosperm, embryo, and hypocotyl-radicle axis. A layer of trichomes covered by mucilaginous substances was present over the external portion of the pericarps, which can help in the absorption and retention of water around the seeds. Below, we observed an envelope protecting both the cotyledons and the embryo (Figure 3B). This envelope is divided into three layers: the outer layer is relatively thin, while the middle layer is thick and composed of sclereids. The inner layer is thin and comprises a single row of cells. The hypocotyl-radicle axis is short and slightly flattened (Figures 3C and D). The plumule is undifferentiated, without differentiation of the leaf primordia. The embryo has two fleshy and asymmetrical cotyledons (in both shape and size), corroborating the report by Barroso et al. (1999), who mentioned the presence of irregular cotyledons in representatives of Brossimum. Main structures observed in transversal sections of seeds of Bagassa guianensis stained with toluidine blue (A, B) transversal sections, (C, D) transversal section showing the embryonic axis (Co: cotyledon; En: Endosperm; Ex: Hypocotyl-radicle axis; Tg: integument, Tr: Trichome).
Biochemical Analyses
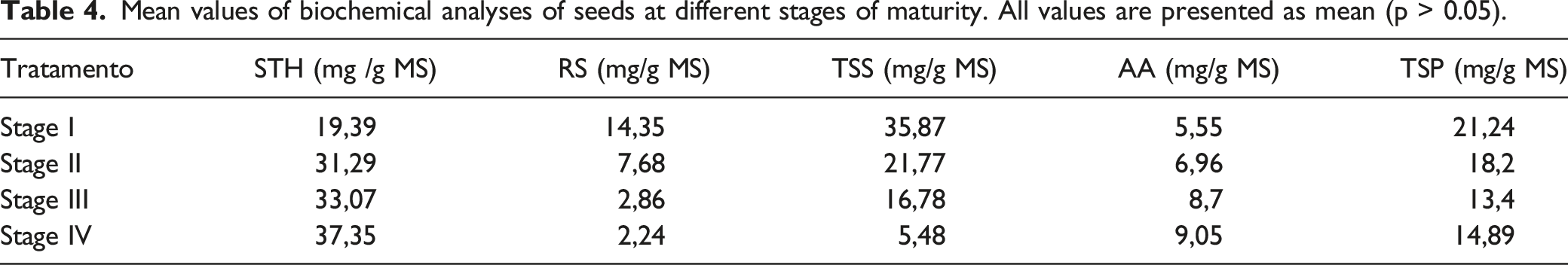
Mean values of biochemical analyses of seeds at different stages of maturity. All values are presented as mean (p > 0.05).
The PCA result showed that the first two axes were explained very well with 95% of the relationship between maturation stages and biochemical properties of the seed of Bagassa guianensis (Figura 4). As a result of ENVIFIT analysis at 0.01% probability of error, it was found that all stages of seed maturation had a significant effect and are strongly correlated with seed biochemical properties, except for stage III, which correlated strongly with biometric characteristics (length, r2 = 0,9; diameter, r2 = 0,81 e weight, r2 = 0,74). In summary stage I is associated with total soluble sugars and Reducing sugars (r2 = 0,82 e r2 = 0,94 respectively), stage II was associated with total soluble proteins (r2 = 0.94), while stage IV was associated with amino acids (r2 = 0,91) e Starch (r2 = 0,68).
The content of total soluble (TSS) and reducing (RS) sugars during the development of seeds of Bagassa guianensis are shown in Table 4. At stage I, the seeds presented 35.87 mg TSS/g MS, followed by 21.77 mg TSS/g MS at stage II. An exponential reduction of sugars was observed at stages III and IV, with values of 16.78 and 5.48 mg TSS/g MS, respectively, resulting in significant differences among stages.
About RS, the seeds from stage I showed 14.35 mg RS/g MS, while those from stage II presented 7.68 mg RS/g MS, followed by a remarkable drop at stages III and IV, reaching values of 2.86 and 2.24 mg RS/g MS, respectively, when the intensive histodifferentiation of seeds is completed (Souza et al., 2018). Differently from TSS and RS, the starch content was higher at the later stages of development, being equal to 37.35 mg /g MS at stage IV, 33.07 mg /g MS at stage III, and 31.29 mg/g MS at stage II, without significant differences among such stages. The lowest starch content was reported in seeds at stage I (19.39 mg/g MS).
Table 4 shows the values of aminoacids present at distinct stages of seed maturation. The lowest value was observed at stage I (5.55 mg of AA/g DM). The highest amount of aminoacids was observed at stages IV and III (9.05 mg of AA/g DM and 8.70 mg of AA/g DM, respectively). No significant differences were reported among seeds at mature stages.
Average values of protein fractionation of seeds at different stages of maturity. All values are presented as mean (p > 0.05).

Regarding the protein fraction and the maturation stages of the seed of the species Bagassa guianensis the PCA result showed that the two first axes explained very well with 93% of the variation of the data (Figure 4). The ENVIFIT analysis was highly significant except for Albumin, which showed no relationship with the stages evaluated (r2 = 0,12 e p = 0,27). Stage I and II, on the other hand, are associated with Globulins and Glutelins (r2 = 0,91 e r2 = 0,87 respectively). Stage III was more closely associated with the biometric parameters Length (r2 = 0,97), Diameter (r2 = 0,97) e Weight (r2 = 0,95). And lastly, stage IV is associated with Prolamine (r2 = 0,68). PCA plots of the morphological relationship and biochemical properties of B. guianensis seeds according to maturity stage: TSS: Total soluble sugars; STH: Starch; AA: Amino acids; RS. Reducing sugars; TSP. Total soluble proteins. **a significant test result ( p ≤ 0.05).
The evaluation of the biochemical composition in mature seeds (stage III) of Bagassa guianensis showed that lipids are the main reserves of seeds with a content value of 34%, followed by starch (3.33%), Total Soluble Proteins (1.34%), Total Soluble Sugars (1.67%), Total Soluble Amino Acids (0.87%) and Reducing Sugars (0.29%).
Discussion
Previous studies indicated that coloration is an efficient and feasible indicator to determine the physiological maturation of several fruit species (Silva et al. 2009; Rubio et al. 2013). In the case of Jatropha curca, Rubio et al. (2013) recommended that fruit should be harvested at stage A, characterized by 5 YR 3/2 coloration, because of their high rates of vigor and germination in this period.
The coloration of fruit has also been used to identify the period of physiological maturation in seeds, as reported in Anadenanthera colubrina (Vera Cruz et al., 2021), Albizia hasslerii (Ristau et al., 2020) and Amburana cearenses (Allem.) A. C. Smith. (Lopes et al., 2014). According to Srimathi et al. (2013), establishing a standard evaluation of fruit coloration in forest trees might represent a suitable alternative to improve the collection of viable seeds.
The fruit of Bagassa guianensis at different maturation stages were very similar in structure and shape. On the other hand, their biometric traits and weight values varied according to each stage, particularly in relation to length and diameter. Similar results were observed by Schulz et al. (2014) in Inga laurina, who reported increased diameter and length in yellow fruit samples at stage III of maturation.
Likewise, the highest values in length, width and weight were identified in samples at stage III, regarded as mature fruit, while the lowest values were observed in immature fruit at stage I. This pattern results from hormone-regulated processes that increase the size and the weight of fruit along maturation stages (McAtee et al., 2013). In addition, we observed progressive weight gains in fruit of Bagassa guianensis at each maturation stage, as also reported in Poincianella pyramidalis (Lima et al., 2012), Jatropha curcas (Silva et al., 2012) and Spondias sp. (Dantas et al., 2016).
As for the biometric parameters of seeds, the highest values in length, width and weight were observed in seeds at stage III, whereas the lowest values were observed in samples at stage I. The seed morphology in Bagassa guianensis followed the anatomical distribution pattern reported in Brosimum gaudichaudii, another member of the family Moraceae (Jacomassi et al., 2010). Furthermore, no anatomical differences were observed among seeds of Bagassa guianensis at distinct maturation stages, as also described by Jacomassi et al. (2010) in seeds of mature and immature fruit of Brosimum gaudichaudii. The presence of mucilage in the seed testa probably contributes to water retention, even though mucilaginous substances might be harmful to the plant development because they are regarded as germination inhibitors (Bezerra et al., 2015). The presence of endosperm in seeds of Bagassa guianensis, typical of albuminous seeds, should be pointed out because this is a rare trait in eudicotyledons. For instance, previous reports identified exalbuminous seeds in species of the family Moraceae, such as Brosimum gaudichaudii (Jacomassi et al., 2010) and Helicostylis tomentosa (Arruda et al., 2017).
The humidity content in seeds of Bagassa guianensis decreased along their development, reaching very low values at stage IV, when the fruit were fully mature. According to Barros Neto et al. (2014), seeds present high contents of water right after their formation and these values decrease at distinct rates according to each species, climatic conditions, and development stage. Bewley et al. (2013) stated that the highest moisture content at the beginning of maturation is essential for cell expansion and for the transference of metabolites from plants to seeds, besides being helpful to the accumulation of reserves. Fortado and Vera Cruz et al. (2021) also reported a similar behavior in seeds of Anadenanthera colubrina at different stages of maturation, identifying high humidity values during the first stages followed by a drastic reduction as the seeds developed.
The concentration values of total soluble sugars (TSS) and reducing sugars (RS) at the beginning of seed development were higher than those observed in subsequent stages. These data agree with the pattern reported by Caccere et al. (2013) in seeds of Inga vera, a native species to Brazil, characterized by a remarkable reduction of RS in the end of seed maturation. The highest TSS content at initial stages is expected since these compounds represent the main reserve supplies of cotyledons and provide energy to seed development. Smeekens et al. (2010) reinforced that sugars are major drivers of embryonic growth, development and germination, playing a key role in plant metabolism, as intermediate substrata. In general, the accumulation of carbohydrates is higher during the first stages of embryo formation when compared to other reserves such as lipids and proteins, but it decreases as the embryonic development continues (Souza et al., 2018). Similarly, the seeds of B. guianensis showed the highest levels of TSS and RS at the initial stages of development.
The highest accumulation of TSS and RS during the beginning of maturation in seeds is usually opposed to that observed in starch, since sugars, like glucose, are the precursors of starch biosynthesis (Beltrão & Oliveira, 2007), being essential for cellular metabolism. As a matter of fact, we observed that TSS has decreased simultaneously as the starch content increased along the maturation of seeds in Bagassa guianensis. Similar results were reported by Caccere et al. (2013) in Inga vera and by Souza et al. (2018) in Hevea brasiliensis L., who described an increased accumulation of starch through the development of seeds. This behavior in the starch content along the maturation process is likely to be related to the preparation of seeds for germination, when they rely on readily available sources of energy. In fact, the breakdown of lipids takes longer periods and demands higher energetic costs when compared to starch (Souza et al., 2018). Even though starch is not the main reserve in seeds of B. guianensis, the accumulation of this carbohydrate from stage II on indicates that seeds are physiologically mature at this stage.
The contents of total soluble aminoacids revealed an inverse relationship to those observed in proteins (Figure 5). Many authors reported that aminoacids undergo variation throughout the embryonic development and germination since these biomolecules are associated with germinative events. Accordingly, Azevedo Neto et al. (2009) showed that increased levels of aminoacids are associated with the degradation of proteins. Differently from the present results, Silva (2015) identified higher values of aminoacids at the initial stages of maturation in seeds of Acacia mangium when compared to the final stages. Most likely, the increased content of aminoacids at the advanced stages in seeds of Bagassa guianensis refer to a period of accumulation in reserves, as the content of proteins decreased along their maturation process. According to Pieruzzi (2009), high contents of aminoacids are related to the role of these compounds as osmotic agents required during the germination of seeds. PCA plots of the morphological relationship and Protein fractioning in seeds of B. guianensis during distinct stages of maturation. **a significant test result (p ≤ 0.05).
The highest values in protein content of Bagassa guianensis were observed at the initial stage of seed development. This accumulation of proteins in seeds during the beginning of their maturation is usually explained by the high demand of distinct proteins and enzymes to control metabolic processes, cell division and the formation of new substances (Duarte et al., 2020). Similarly, reduced amounts of proteins in mature seeds were also reported in seeds of Jatropha curcas L. (Silva et al., 2018), who described high values of protein content in yellow fruit right after harvesting. Nonetheless, divergent results from those herein described were identified during the development of Tylosema esculentum, a native species to Africa (Mosele et al., 2011) and in Pongamia pinnata (L.), an Indian species (Pavithra Gowda & Shivanna, 2014), since both species were characterized by increased protein content at final stages of seed maturation.
The use of the PCA applied for data evaluation proved to be an excellent tool for the study of the vectors of the variable morphology and biochemistry of the seeds of Bagassa guianensis with the ordering points of the development stage of the fruit, according to PCA data, it was found that all seed maturation stages had significant effect and highly correlated with seed biochemical properties. This is because, as the seed maturation process progresses, the biochemical activity increases, due to the production of enzymes in the cells (Carvalho; Nakagawa, 2012; Marcos-Filho, 2015. This biochemical activity causes great variation in the composition of the main substances in seed reserves, such as carbohydrates, proteins, and lipids (Buckeridge et al., 2004).
Glutelins, prolamins, albumins and globulins are reserve proteins that store macronutrients, such as nitrogen, carbon and sulfur, for the formation of new tissues in the embryo, thus playing a major role in seed development and germination (Marcos-Filho, 2015; Silva., 2019). Prolamins are more abundant in grasses, glutelins are usually found in cereals, while globulins and albumins are predominant in dicotyledons (Souza et al., 2012). The glutelins, which represent the main reserve proteins in monocotyledonous species but not in dicotyledonous plants, have predominated in seeds of Bagassa guianensis. A similar pattern was also reported in seeds of other native forest species from different families, like Mizilaurus itauba (Lauraceae), Byrsonima lancifolia (Malpighiaceae) and Eschweilera ovata (Lecythidaceae) (Souza et al., 2012) and Myracrodruon urundeuva (Anacardiaceae) (Pupin et al., 2017). As pointed out by Zhao et al. (2018), the mobilization of reserve proteins might take place during the maturation of seeds and, to some extent, during the germination and development of seedlings, which can account for their abundance in the studied species.
According to Pupin et al. (2017) glutelins are proteins with good nutritional quality and have higher levels of essential amino acids such as lysine and tryptophan, the more essential amino acids the protein has, the more noble this food is considered. For Oliveira (2012), the use of biochemical maturation indices can detect an increase or decrease in the contents of substances that, from an ecological point of view, would have the function of attracting or repelling the disperser or the predator. In the forest, the fruits of Bagassa guianensis are consumed by several animals due to their light and sweet taste (EMBRAPA, 2002), thus it can be suggested that there may be a relationship between the chemical composition of the fruits and the behavior of the animals that consume them. consume.
The seeds of Bagassa guianensis were classified as oleaginous because of the high proportion of ether extract (34%). Lopes et al. (1999) reported lower values in fruits of Pourouma cecropiifolia, another member of the family Moraceae and native to Amazon. A biochemical screening of seeds from some forest species was also carried out by Baleroni et al. (2002), including Brosimum gaudichaudii, reporting high levels of proteins, lipids and carbohydrates in their seeds. Similar values of total soluble sugars were also observed by Solanki et al. (2020) in mature seeds of Moraceae species, reaching up to 2.2 g of TSS/100g in seeds of Artocarpus heterophyllus. On the other hand, lower values of proteins and starch were observed in Bagassa guianensis when compared to the seeds of Artocarpus heterophyllus, Artocarpus altilis and Artocarpus integer (a mean value of 7.9 g in protein and 13.2 g in starch in 100g of seed flour) analyzed by these authors.
The seeds of Bagassa guianensis from fruit samples characterized by 5 GY 4/4 coloration and green pigmentation (stage I) presented fully formed embryos. In spite of their low biometric values, high amounts of Total Soluble Sugars (3.59%), Total Soluble Proteins (2.13%) and Reducing Sugars (1.42%) were observed at this stage. On contrary, greater amounts of other reserve compounds, such as aminoacids (0.87%) and starch (3.31%), were observed during the final stages of maturation. The highest accumulation of starch started at stage II (31.29 mg/g DM), when the seeds reach physiological maturity.
Conclusion
Bagassa guianensis is an infructescence made up of numerous seeds, which during the maturation stages go fully green, greenish-yellow, brown, and dark brown colors, with a thick epicarp and sizes ranging from 27.39 mm to 68.76 mm. The seed was classified as albuminous, where the presence of endosperm was observed, with a conical embryo that fills practically the entire seed. The seeds of B. guianensis showed high levels of glutelin as one of the main reserve proteins. The species was classified as an oilseed due to the higher proportion of ether extract (34%) found in the seeds. The second highest content was starch, with the greatest accumulation in the final stages of seed maturation. The highest levels of TSP, TSS, and RS were observed for the early stages of seed development, while amino acids and starch showed high contents in the final stages of seed development. The results found in this study on seeds at different stages of ripeness showed a correlation between biometry, morphology, and biochemistry.
Implications for Conservation
Additional scientific studies are needed to obtain knowledge on the behaviour of native species, biochemical composition of seeds during maturation and the reserve contents of the cotyledons to assist in seed and seedling establishment. There are few studies and information related to biochemistry, anatomy and morphology of native species of the Amazon region. This article provided important information on anatomy and biochemistry of Bagassa guianensis seeds at different stages of maturation. Biochemical and morphological information of seeds is relevant for planning conservation, propagation, and restoration strategies (Peraza-Villarreal et al., 2018). The species has great economic and social importance, due to the use of its wood widely used in civil construction, but due to intense exploitation, it can lead to cases of scarcity, mainly due to its long regeneration process. Therefore, this study contributes to document the scientific knowledge about the species, as well as the understanding of morphological, anatomical and biochemical processes, besides making it possible to understand and conserve plant biodiversity, providing through this initial study, the sequence of studies on the species.
Footnotes
Acknowledgments
The authors would like to thank the farmers who provided their areas for fruit collection, the Postgraduate Programme in Biodiversity and Conservation (PPGBC - UFPA/Altamira), and the Biotechnology Laboratory of the Federal University of Pará (BIOTEC-Altamira), who contributed to the research.
Declaration of Conflicting Interests
The authors declare that the research was carried out in the absence of any commercial or financial relationships that could represent a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported: National Programme for Academic Cooperation in the Amazon - PROCAD/Amazon, call for applications 21/2018 - Coordination for the Improvement of Higher Education Personnel (CAPES); Pro-Rectory of Research and Postgraduate Studies (PROPESP); and Postgraduate Programme in Biodiversity and Conservation (PPGBC – UFPA/Altamira).
