Abstract
Introduction
Metal mining activities have an essential role in the economic development of modern societies but often have significant negative long-term impacts on natural ecosystems (Chen et al., 2019; Xiao et al., 2020) due to the generation of mine tailings rich in heavy metals and metalloids that are not biodegradable and accumulate in the environment. Metals bioaccumulate in different organisms and incorporate into trophic chains (Mussali-Galante et al., 2013a). Thus, not only are metals a risk for organisms in direct contact with a source (i.e., via inhalation or by skin contact), but also to others who are indirectly exposed via their food sources (Soliman et al., 2022).
Mexico has a long history of mining activities; mining accounts for approximately 4% of national gross domestic product (SE, 2020). Unfortunately, mining in Mexico has led to environmental deterioration throughout the country. For example, the state of Morelos in south-central Mexico has a long history of metal mining (e.g., silver [Ag], copper [Cu], iron [Fe], lead [Pb]). While mining activity has declined in the state (SGM, 2020), abandoned mine tailings continue to threaten natural ecosystems. Old mining operations have left a legacy of numerous solid mine tailings from which metals leach out into streams and groundwater or are dispersed by wind (CONANP, 2005).
Huautla, a small town in southern Morelos located inside the MAB-UNESCO Sierra de Huautla Biosphere Reserve (REBIOSH) is surrounded by approximately 780,000 tons of silver, lead, and zinc (Zn) mine solid tailings that have gone without proper environmental treatment (Márquez-Huitzil et al. 2022). Metals are responsible for numerous documented negative effects on local biota and humans (Mussali-Galante et al., 2013a; Mussali-Galante et al., 2013b; Tovar-Sánchez et al., 2016; Muro-González et al. 2020, Santoyo-Martínez et al., 2020; De la Cruz-Guarneros et al., 2021) (Figure 1). Study sites in the Huautla and Ajuchitlán streams in the southern portion of the State of Morelos, Mexico where anurans were collected. Sites (AU1, HU1, HU2, T1 and DW), towns and the approximate location of mine tailings are shown. The grey shading indicates the area inside the Sierra de Huautla Biosphere Reserve (REBIOSH). The orange shading indicates the location of unnamed tailings within the Las Presas tailings, according to data 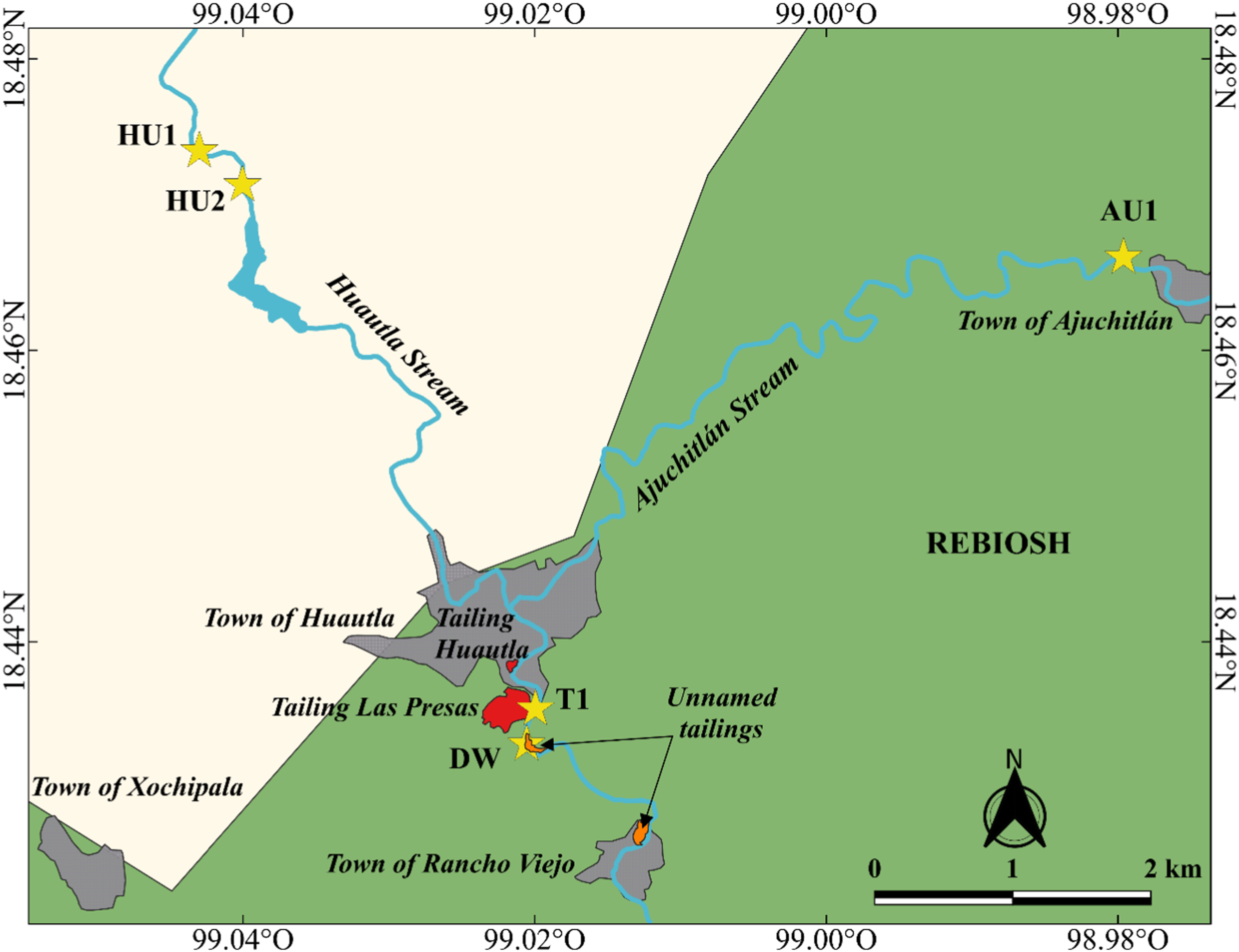
Anurans are particularly sensitive to metal pollution (Egusquiza-Morínigo, 2016; Kiesecker et al., 2001; Venne et al., 2006). They incorporate metals into their bodies primarily via respiration, ingestion and skin absorption (Hopkins, 2007; Ilizaliturri, 2010). Exposure to metals is one of many anthropogenic stressors threatening amphibians in tropical environments and has led to extirpations (Hopkins, 2007; Jofré et al., 2012) and alteration of community structure (Ficken & Byrne, 2013; Calderon et al., 2019). While a growing body of literature has reported on anthropogenic effects on anurans in Mexico (Hernández-Ordóñez et al., 2015, 2019), few have directly documented the effects resulting from metal exposure and bioaccumulation on their community structure (but see Suárez et al., 2016).
At least 38 species of anurans have been described in the state of Morelos (Castro-Franco et al., 2006; Ramírez-Bautista et al. 2023). Tropical dry forests in the southern portion of the state are home to at least 29 species (Castro-Franco et al., 2006). While most anurans require freshwater environments in early stages of their ontogeny, as adults they adopt life strategies that lead to differential use of areas throughout a stream ecotone. Species such as Van Vliet´s Frog (Smilisca baudinii), Mexican Leaf Tree Frog (Agalychnis dacnicolor) and Dwarf Mexican Treefrog (Tlalocohyla smithii) are tree-living species; Zweifel´s Frog (Lithobates zweifeli) and Showy Leopard Frog (L. spectabilis) live in areas surrounding permanent waterbodies; and Giant Toad (Rhinella horribilis) and Confusing Toad (Incilius perplexus) live in terrestrial environments but do not move far away from bodies of water (Cortés-Suárez, 2017). Differential habitat use has been suggested to lead to varying potential metal bioaccumulation among species (Stolyar et al., 2008; Zhou et al., 2008; Jofré et al., 2012; Severtsova & Aguillón-Gutiérrez, 2013; Severtsova et al., 2013; Gastelum et al., 2019; Thanomsangad et al., 2019). Similarly, some evidence exists that anuran community structure can be affected by mining activities; species sensitive to metal bioaccumulation may disappear from communities close to mining operations (Ficken & Byrne, 2013; De Lucca et al., 2018; Calderon et al., 2019).
Here we document metal concentrations in water and sediment from stream habitats, and report metal bioaccumulation in Smilisca baudinii, Rhinella horribilis, Lithobates zweifeli and L. spectabilis, from sites located along a stream near Huautla. Sites were located at different positions upstream and downstream from mine tailings; we expected higher metal concentrations and bioaccumulation in areas closer to these structures. Further, we report on anuran community structure in these sites; where we expected differences in composition and diversity related to the proximity to mine tailings. This is the first report of metal bioaccumulation in anurans of the state and region, and one of few accounts of the anuran fauna in the REBIOSH.
Methods
Study Sites
The Sierra de Huautla Biosphere Reserve (REBIOSH) is a natural protected area in the southern State of Morelos (Figure. 1). REBIOSH was established in 1996, but mining (for Ag, Pb, Zn) activity had been present in the area since 1774 (Ruíz de Velasco, 1890; Volke and Velasco, 2005) and until 1991 (CONANP, 2005), when it ceased. Named mine tailings Huautla and Las Presas, located approximately at 18° 26' N-99° 01' W and 18° 27' N-99° 01 'W (at an altitude of 995 m above sea level), remained at less than 500 m from streams once mining activity ceased (Mussali-Galante et al., 2013b
The Huautla and Ajuchitlán streams are small (maximum width = 10.5 m), intermittent, medium gradient systems that drain a semi-arid mountainous area of REBIOSH dominated by dry tropical forest. Once the Ajuchitlán joins the Huautla, the stream runs S and E to join the Amacuzac river (Balsas Basin). The study sites were located along the Huautla and Ajuchitlán streams. The Huautla runs generally N to S from the town of Quilamula, feeds the Lorenzo Vázquez (also known as Cruz Pintada) reservoir (≥ 300,000 m3) (CEAGUA, 2017), continues S towards Huautla where it meets Ajuchitlán Stream and crosses the town of Huautla, to continue south adjacent to the tailings. The Ajuchitlán runs generally NE to SW for about 8 km after crossing the town of Ajuchitlán and entering a relatively small gorge where a paved road transiently crosses the stream before it joins the Huautla (Figure 2). The Ajuchitlán is also dammed upstream from the town. In the dry season (November-June) both streams typically loose longitudinal connectivity with pools remaining in some areas; during the rainy season (July-October), both streams regain connectivity. Both systems have a relatively quick response in discharge after precipitation events and erosion from surrounding mountains and tailings is common. Prevailing winds in the area are from the North (Ortíz, 2019). Sampling sites. The physical structure of the study sites is shown. The letters indicate: A) AU1, B) HU1, C) HU2, D) T1 and E) DW. All photos by Eduardo Aarón Chávez Ramírez.
For this study, we sampled five sites located on Ajuchitlán (n = 1, site AU1, approximately 5.5 km upstream from mine tailing Huautla) and Huautla streams (n = 4). Sites in the Huautla were located 8.4 (HU1) and 6.2 (HU2) km upstream from the tailing Las Presas. Two other sites were located 0.4 (T1) and 1.6 (DW) km downstream from tailing Las Presas (Figure. 1). Sites were chosen given their ease of access and their position relative to the mine tailings.
Sample Collection
We sampled water and sediment from each site in January 2019. Three water and sediment samples were obtained in each site. Three - 500 mL water samples were obtained from each site following methods in NMX-AA-051-2016 (NMX, 2016). All samples were preserved in the field by adding suprapure nitric acid (NHO3) to attain pH < 2. Water samples were transported to the laboratory in clean polypropylene bottles and refrigerated at 4.0 °C (± 2.0 °C) until further processing. Three superficial sediment samples were obtained following procedures in NMX-AA-112-SCFI-2017 (NMX, 2017) and NMX-AA-014-1980 (NMX 1980) from pools in each stream. Sediment samples were obtained with a shovel and kept in heavy duty polyethylene 500 mL bags (Whirl-Pak®) which were then transported to the laboratory at 4°C. In all, 30 (15 water and 15 sediment) samples were obtained and processed.
We sampled the anuran community and obtained liver samples for metal concentration measurements at different times of the year in 2018-2019. The anuran community was sampled in sites HU1, HU2, T1 and DW once monthly between June and September 2018; sites AU1, HU1, HU2, T1 and DW (Figure 1) were sampled once monthly in June and July 2019. Sampling periods were restricted by budgetary and logistical constraints. Six 20 x 2 m (total area = 20m2) transects were established within each site alongside the wetted portion of the stream (Scott & Woodward, 2001). We sampled for anurans within these transects. To avoid double counting of individuals, transects were separated by at least 50 m. All transects (total transects = 156) were surveyed once during each field trip by three observers at nighttime (20:00-24:00). Within each transect we carried out visual encounter surveys (VES) (Crump & Scott, 2001), in which we slowly walked along the transect looking into all possible microhabitats (to a height of 2 m above ground) where anurans could be located. Detected anurans were captured manually using nitrile gloves; each individual was placed in its own clean plastic bag until it was identified (Casas-Andreu et al., 1991).
Individuals were identified following Canseco-Márquez & Gutiérrez-Mayén (2010), Flores-Villela et al. (1995) and Frost (2021). Once identified and counted, most individuals were released in the vicinity of the collection site, but some were kept for further processing (see below). All individuals were collected under permit SGPA/DGVS/008320/18 granted to EACR (author).
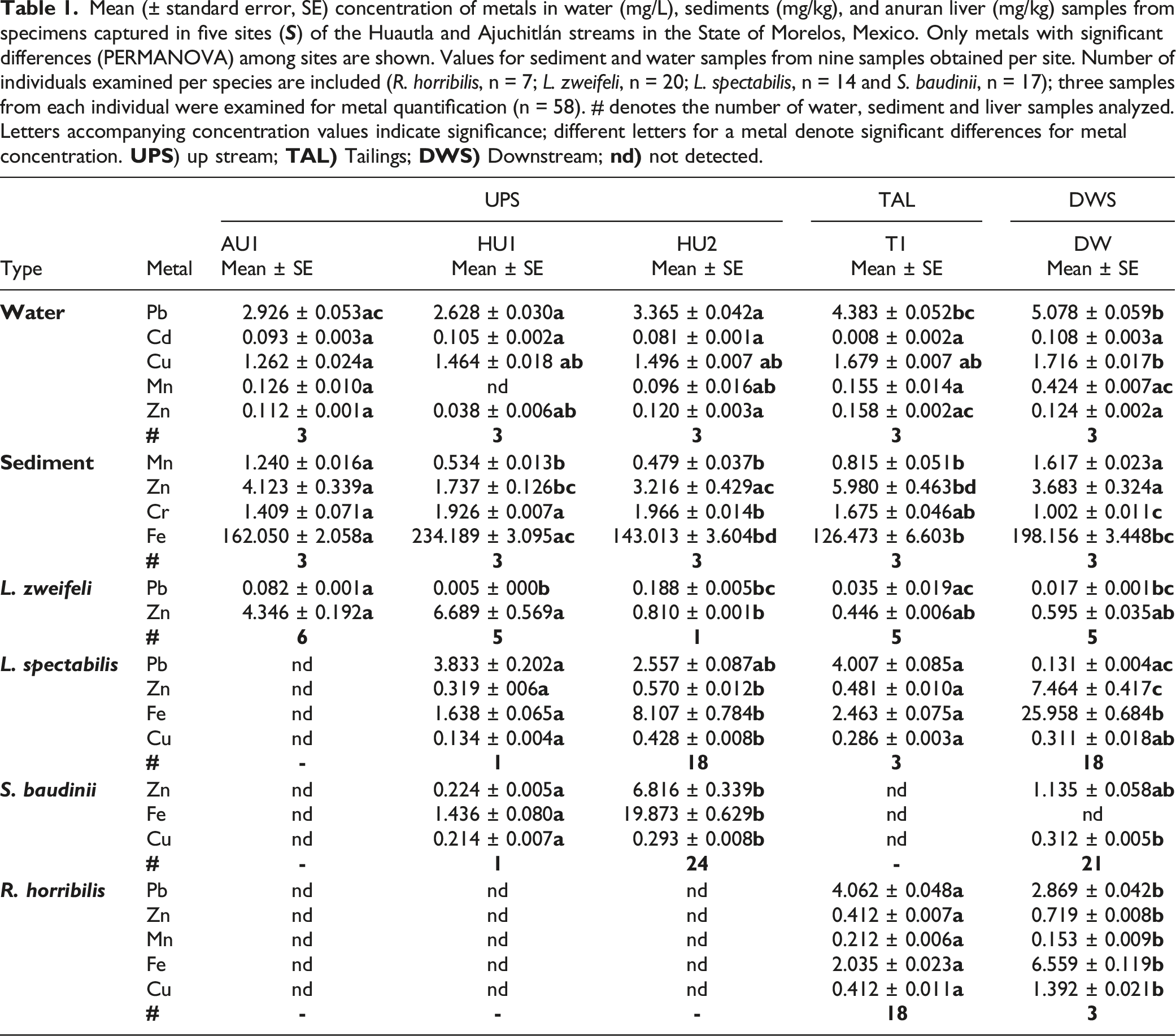
Mean (± standard error, SE) concentration of metals in water (mg/L), sediments (mg/kg), and anuran liver (mg/kg) samples from specimens captured in five sites (
Concentration of Metals in Water, Sediments and Anurans
Water and sediment samples were processed to quantify metal (Fe, Mn, Zn, Cu, chromium [Cr], Cd and Pb) concentrations. Prior to metal concentration analyses, water samples were filtered with a 45-µm membrane prewashed with nitric acid (1%) (see NMX-AA-051-SCFI-2001 [NMX, 2001]).
A total of 58 livers (7, 20, 14 and 17 for R. horribilis, L. zweifeli, L. spectabilis, and S. baudinii, respectively) were analyzed to determine the concentration of metals (Cd, Cr, Cu, Fe, Mn, Pb and Zn) following previous studies in the area (Mussali-Galante et al., 2013a; De la Cruz-Guarneros et al., 2021). Samples were individually dried (electric stove at 70°C, until they reached a constant weight) and sieved following the method established by the Mexican standard NMX-AA-132-SCFI-2006 (NMX, 2006). Each dehydrated liver was placed in a polyethylene terephthalate (PET) container previously washed with a 10% nitric acid solution. For metal analysis, a 0.25 g (dry weight) porcelain-pulverized portion of each liver was used. Samples were subjected to acid digestion using an accelerated reaction system microwave (CEM®MARS-5), using 10 mL of 70% HNO3 in closed Teflon pumps. Samples were dissolved to a final volume of 50 mL for analysis. A sample without tissue was processed concurrently and was utilized as a control. Metals were then analyzed by atomic absorption spectrophotometry (GBC-908-AA, Scientific Equipment Ltd.), a technique that determines the concentration of a metallic element in a sample, calibrating the spectrophotometer with standard solutions containing known concentrations of each analyzed item. The standard calibration curves showed correlation coefficients (r2) between 0.99 and 1. For each metal analyzed the average value of three repeated quantifications for a sample is reported. Concentration values for water are reported in milligrams/liter. Concentration values for sediment and liver samples are reported in milligrams/kilogram (mg/kg) dry weight. The minimum detection limits (mg/L) according to the manufacturer of the spectrophotometer were 0.0004 for Cd, 0.003 for Cr, 0.001 for Cu, 0.005 for Fe, 0.0015 for Mn, 0.01 for Pb and 0.0005 for Zn. When metal concentrations were below detection limits, we used 0.5 of detection limit for statistical analyses.
Statistical Analyses
We calculated the average and standard deviation for each metal concentration for each species per site. We compared metal concentration (for each metal) among sites and per species among sites using Bray-Curtis distances from a triangular matrix using permutational multivariate analysis of variance PERMANOVA (Anderson et al., 2008). Metal concentration data used in PERMANOVA were fourth root-transformed based on type 1 sequential sums of squares (Legendre & Gallagher, 2001). In these analyses, species or sites were used as fixed factors. Each was analyzed using pairwise analyses via PERMANOVA t with 9999 permutations. P-values were calculated using Monte Carlo simulations following Anderson et al. (2008). PERMANOVA draws statistical inferences in a distribution-free process based upon geometric partitioning of distance measures. We used a principal component analysis (PCA) to explore metal concentration variability among sites (for water, sediment, and liver samples) and species. All analyses were carried out using Primer 7.0.13 (Clarke & Gorley, 2015).
We explored species richness, capture rates (number of specimens captured per sampling hour), total and relative abundance, sampling completeness and species diversity from anuran community data. Only data from replicated transects (AU1 n = 12; HU1-DW n = 36) in each site were used for these calculations and comparisons. Since temporal differences in diversity were not significant (tHutchenson =0.585, df = 4212.9, P = 0.558), data from transects in each site taken on different dates were grouped for analysis. We analyzed sample completeness in all sites to assess richness variation in relation to Chao 1 and Bootstrap estimators (Chao & Jost, 2012; Gotelli & Colwell, 2011; Moreno et al., 2011). We estimated three diversity dimensions based on Hill’s numbers: Species richness or Chao 1 (q0), the number of common species or Shannon-Wiener´s exponential (q1), and the number of dominant species or inverse Simpson (q2) (Chao, 1984; Jost, 2006; Moreno et al., 2011). For each measure of diversity, we calculated confidence intervals (CI) at 95% which were then used to estimate among-site differences. These analyses were carried out in R (iNEXT package, V 2.0.17) (Hsieh et al., 2016). Comparisons among diversity indexes were based on Euclidean distance matrices via PERMANOVA (Anderson, 2001). We also evaluated anuran assemblage similarity using Sørensen and Jaccard indexes (Magurran, 1988; Moreno, 2001). Rank-abundance curves were generated for the assemblages in each site. These curves depict the abundance of each taxon relative to the total number of individuals in the assemblage (Whittaker, 1965).
Results
We found significant differences in metal concentrations among sites for water (Fpseudo = 3.6725, Pperm = 0.0005) and sediment (Fpseudo = 172.27, Pperm = 0.0001). The first two axes of the PCA on metals in water samples explained 79.1 % of variability, driven mainly by Pb, Cu and Zn concentrations, with Mn and Cd being relatively less important drivers (Figure 3A). The similarity percentage analysis showed differences (at P < 0.05) between AU1, HU1 and HU2 with T1 and DW, but not between AU1, HU1 and HU2 or between T1 and DW. Cu, Pb, Zn and Mn were mostly responsible for these differences. T1 and DW had higher Cu, Pb and Mn, compared to AU1, HU1 and HU2 (Table 1, Figure 3A). PCO triplot of variability in the levels of metals in A) water and B) sediment from five sites in the Huautla and Ajuchitlán streams in Morelos, Mexico. The centroids of the groups are indicated by the site label and the contribution of each metal in a site is indicated by the relative size of the pie section.
The first two axes of the PCA analysis on metal concentrations in sediment explained 98.2% of variability among sites (Figure 3B). This variability was driven by Fe, Cr and Mn concentrations, with Zn being relatively less important. Similarity percentage analysis on metals in sediment concentrations showed differences (at P < 0.05 and P < 0.001) among all sites, with Fe, Zn, Mn, or Cr being important in explaining among site similarity depending on the sites being contrasted; no consistent differences were found among sites located close or far from mine tailings (Table 1, Figure 3B).
Metal Concentrations in Anurans
Pb, Zn, Cu, Mn, Fe and Cd bioaccumulated in livers of the examined species (Table 1). Detectable Zn and Cu concentrations were found on specimens from all sites and species. Cr was not detected in any sample. Pb was not detected in L. zweifeli from sites HU1 or DW. Mn was not detected in S. baudinii from DW, L. spectabilis from DW or in any L. zweifeli. Fe was not detected in S. baudinii from DW. Cd was not detected in S. baudinii from HU1, on L. spectabilis from HU1 or T1, or any R. horribilis (Table 1).
For liver we found significant differences among sites (Fpseudo = 4.1874, Pperm = 0.0002), species (Fpseudo = 6.6511, Pperm = 0.0003) and their interaction (Fpseudo = 4.3124, Pperm = 0.0002). For L. zweifeli, among site Fe,Cu and Cd concentrations were similar (F = 2.07, df = 4, P-perm = 0.12; F = 1.68, df = 4, P-perm = 0.18; and F = 2.38, df = 4, P-perm = 0.096; respectively). Pb concentrations in L. zweifeli were significantly higher in site AU1 than in sites HU1, HU2, T1 and DW (F = 8.44, df = 4, P-perm = 0.002). Zn concentrations for this species were significantly higher in site AU1 than in site HU2 and site T1 (F = 3.09, df = 4, P-perm = 0.018). For L. spectabilis, among-site Pb concentrations were similar (F = 2.87, df = 3, P-perm = 0.06); but Zn concentrations were significantly higher in sites HU2 and DW, than in sites HU1 and T1 (F = 7.90, df = 3, P-perm = 0.006). In L. spectabilis, Mn concentrations were significantly higher in site HU2 than in sites HU1, T1 and DW (F = 4.03, df = 3, P-perm = 0.03); concentrations from site DW were also higher than those in sites HU1 and T1 (F = 4.03, df = 3, P-perm = 0.03). Concentrations of Fe in L. spectabilis were much higher in site DW than in site HU2 and sites HU1 and T1, respectively (F = 6.58, df = 3, P-perm = 0.007). Cu concentrations in L. spectabilis were higher in site HU2 than in sites DW and sites HU1 and T1 (F = 5.10, df = 3, P-perm = 0.01). For S. baudinii Pb and Mn concentrations were similar among sites (F = 0.92, df = 2, P-perm = 0.42 and F = 0.55, df = 2, P-perm = 0.612, respectively). For R. horribilis median Pb (F = 392.27, df = 1, P-perm = 0.0001) and Mn (F = 54.31, df = 1, P-perm = 0.0004) concentrations were significantly higher in site T1 than site DW. In R. horribilis, median Zn (F = 317.92, df = 1, P-perm = 0002), Fe (F = 868.13, df = 1, P-perm = 0001) and Cu (F = 76.30, df = 1, P-perm = 0003) concentrations were significantly higher in site DW that in site T1.
Considering metal concentrations in liver (irrespective of species), the first and second axes of the PCA analysis explained 70.1% of variability among sites. Variability was driven by Pb, Zn, Cu and Fe, with Mn and Cd being relatively less important (Figure 4A). Sites AU1, HU1, HU2 and T1 were significantly different (at p<0.05 or p<0.001) from DW, but no differences were found among AU1, HU1 or HU2. AU1 was significantly different from T1 (p<0.001) (Table 1). These differences were mostly driven by Cu and Pb concentrations, with Mn, Zn and Fe also having some contribution. PCO triplot of variability in the levels of metals in amphibian livers from five sites in the Huautla and Ajuchitlán streams in Morelos, Mexico. A) Includes the contribution of each metal in amphibian’s livers by site. B) Includes the contribution of each metal in amphibian livers for a species. C) Includes the contribution of each metal in amphibian livers in a species x site interaction. Centroids of the groups (either site and/or species) are presented.
Lithobates spectabilis, L. zweifelli and S. baudini differed in the concentration of metals in liver, irrespective of site (Figure 4B). First and second components of a PCA on this data explained all among-species variability, and this was driven by changes in Pb, Fe and Zn, with other metals playing a relatively smaller role. Nearly all metals were found at higher concentrations in L. spectabilis, except Cd (higher in L. zweifeli) and Pb (higher in S. baudini).
Generally, site x species interaction on similarity percentages analyses for L. spectabilis, L. zweifeli and S. baudinii indicated significant differences in liver concentrations between sites far from (AU1, HU1 and HU2) and close to the mine tailings (T1 and DW). A PCA of such interaction explained close to 70% variability on first and second axes (Figure 4C), and was most influenced by Fe, Pb and Zn concentrations, with other metals playing a relatively smaller role. For L. spectabilis, significant differences were found between HU1, HU2 and T1 with DW, but not with T1. These changes were primarily driven by Pb, Mn and Cd (Table 1). For L. zweifeli, significant differences were found between AU1, HU1 and HU2 with T1, but not DW (Table 1); Zn and Cu concentrations were responsible for these differences. For S. baudinii, significant differences were found between HU2 and T1 with DW and between T1 and DW; other sites did not show differences. Differences in concentrations of Pb, Fe, Zn, Cu and Mn guided these differences (Table 1).
Anuran Communities
Anuran community composition, community parameters and similarity indexes from five sites (AU1, HU1, HU2, T1 and DW) sampled in the Huautla and Ajuchitlán streams in the southern portion of the state of Morelos, Mexico during 2018-2019. For species data we present the number of individuals captured in each site; the number not in parenthesis is the total number of individuals captured, the number in parenthesis is relative abundance (%) for the species in a site across both years of study. For each species, next to its name we present its conservation status according to IUCN (2023). Community parameters and similarity indexes are as described in methods section.

DW (n = 55) and T1 (n = 53) had the highest anuran abundance and richness (S = 8 and S = 7, respectively) (Table 2). HU1 and HU2 had intermediate richness (both with S = 5) and AU1 had the lowest species richness (S = 3). The number of common and equally abundant species were significantly lower in AU1 than in HU1, HU2, T1 and DW (F = 6.383; df = 4, P-perm = 0.0001). Paired comparisons PERMANOVA showed differences between AU1 and HU2, T1 and DW (P-perm < 0.01); HU1 and HU2, T1 (P-perm < 0.01); HU2 and AU1-DW (P-perm < 0.01); T1 and AU1, HU1, HU2 (P-perm < 0.01); and DW and AU1, HU2 (P-perm < 0.01).
The most abundant and prevalent species was Lithobates zweifeli (n = 74, 5 sites) followed by Rhinella marina (33, 5) and Smilisca baudinii (31, 4); least abundant species were Hypopachus variolosus (2, 1) and Agalychnis dacnicolor (2,1) (Table 2, Figure 5). Considering all sampling events, in AU1 the most and least abundant species were L. zweifeli (n = 36) and D. arenicolor (n = 1) respectively (Table 2). Most and least abundant species in HU1 were L. zweifeli (n = 15) and R. horribilis (n = 1) (Figure 5). Lithobates spectabilis was the most abundant species in HU2 (n = 13) while the least abundant species (n = 2) were L. zweifeli and R. horribilis. The most abundant species in T1 were R. horribilis (n = 21) and L. zweifeli (n = 11) and the least abundant species was H. variolosus (n = 2). Smilisca baudinii and L. zweifeli were the most abundant species (n = 15 and n = 10, respectively) in DW; D. arenicolor was the least abundant (n = 1) species (Table 2, Figure 5). Rank abundance curves for anurans from five sites in the Huautla and Ajuchitlán streams, Morelos. Curves are presented independently for each site (AU1, HU1, HU2, T1 and DW). The nine species that occurred at the sampled sites are shown with codes: Ip) I. perplexus; Rh) R. horribilis; Ad) A. dacnicolor; Sb) S. baudinii; Ts) T. smithii; Da) D. arenicolor; H. variolosus; Ls) L. spectabilis; Lz) L. zweifeli.
Community composition was most similar (using Jaccard´s index) between sites AU1 and HU1, and T1 and HU2 (Table 2). Sites HU2 and HU1, and HU2 and T1 were also similar; and HU2 and AU1 were most dissimilar (Table 2). HU2 and T1 were most similar based on Sorensen´s index and HU2 and AU1 were again, most dissimilar. Generally speaking, AU1 was most dissimilar to other sites using either similarity index (Table 2) and species composition (Figure 3).
Discussion
Metals are known to have important consequences for the individual health of anurans and are a known factor for the decline of several anuran populations worldwide (Lee, 2000; Hernández et al., 2013; Severtsova & Aguillón-Gutiérrez, 2013; Aguillón-Gutiérrez & Ramírez-Bautista, 2015; Suárez et al., 2016). However, metal effects on anurans in the wild and at higher levels of biological organization have been poorly studied in Mexico (Ilizaliturri, 2010; Aguillón-Gutiérrez & Ramírez-Bautista, 2015). In addition to presenting updated information for the anuran community at the REBIOSH (Herrera-Balcázar, 2014; Roa-Mata, 2017), our work constitutes the first record of metal bioaccumulation for anurans in the protected area and the State of Morelos. Our findings should alert regional managers on potential threats on the anuran communities from metal exposure.
Metal concentrations in water differed significantly among all samples and sites closest to mine tailings (T1, DW) typically had the highest values. Cu, Zn and Pb in site T1, and Cd and Mn in DW, had the highest concentrations among sites. Generally, mean metal concentrations in water from the Huautla Stream were higher for Pb than Cu, Mn, Zn and Cd, in decreasing order. Metal concentrations in sediments on the other hand, showed among-site significant differences for Fe, Zn, Mn and Cr, with Mn, Fe and Zn relatively higher in T1 and DW, and Cr higher in HU2 and HU1. Zn and Mn were higher in site AU1 than in sites HU2 y HU1; Mn was also more concentrated in DW than in AU1. Sediment metal concentrations in the Huautla Stream were higher for Fe than Zn, Cr and Mn, in decreasing order. We suggest elevated metal concentrations in both matrices for T1 and DW are due to their relative proximity to the Las Presas mine tailings. Sediment samples taken from different soil levels in these mine tailings (Solís, 2016) show higher concentrations for Fe than Zn, Pb, Mn, Cu, and Cd, in decreasing order; although all were at higher concentrations than we found in our samples. Additionally, tailings have low stability and are highly susceptible to wind and water erosion (Volke and Velesco, 2005), which could lead to metal bioaccumulation via lixiviation (Márquez-Huitzil et al., 2022) in sites T1 and DW. Pb, Cd, Cu and Zn concentrations in water samples from the Huautla Stream (0.082, 0.0018, 0.009 and 0.118 mg/L, respectively) exceeded toxicity reference values (TRV) suggested by EPA (2019). This renders water from the Huautla not recommended for human consumption. However, we note that our samples were taken during the dry season, when metal concentrations usually are higher than those in the wet season given lack of dilution of potential contaminants (Islam et al., 2017; Pandey et al., 2019; Proshad et al., 2019; 2020). Water hardness and its specific ionic properties, as well as chemical composition of substrates can lead to among-matrix metal concentration variability and bioavailability and should be studied further in the area (Miranda et al., 2022).
Contrary to our expectations, upstream sites showed elevated metal concentrations. While relatively distant from studied mine tailings, we recognize that the entire area could be subject to relatively elevated natural metal concentrations. What today is the REBIOSH was for decades one of the most important mining districts in Morelos state and many mines and other tailings exist in the area (Volke and Velesco, 2005, Mussali-Galante et al., 2013b
We found Pb, Zn, Cu, Mn, Fe and Cd concentrations in the livers from analyzed anurans. While some of these can be considered essential (e.g., Zn and Fe) and were expected in livers, others (e.g., Pb and Cd) are not and their presence suggests exposure to anthropogenic contaminants. Our findings agree with other studies that report liver as one (but not the only) important target organ for metal bioaccumulation in several organisms, including anurans (de la Cruz et al., 2021; Curi et al., 2021). Diet and potential exposure to metals in water and sediment could explain this finding (EPA, 2019; Esteller et al., 2015; Rodríguez-Rodríguez, 2020; Solís, 2016). Lepidopterans, Coleopterans, Hymenopterans and even larval anurans are known food items for R. horribilis, L. spectabilis, L. zweifeli and S. baudinii (Barrios-Damián, 2006; Mendoza-Estrada et al., 2008). Many of those are primary consumers that may incorporate metals adhered to organic or inorganic material and sediment in streams. Once bioavailable, metals are known to bioaccumulate and biomagnify along the food chain (Penicaud et al., 2017). An additional route for metals exposure in anurans, includes direct contact with contaminated water (Calderon et al., 2019; Jofré et al., 2012). Highly permeable skin, typically involved in respiration, is a well-known route for metal bioaccumulation in amphibians (Sparling, 2017).
While our focus was not to detect anomalies or other effects of metal accumulations in Huautla anurans, we note that Pb, Fe, Cd, Mn, Zn and Cu are known to cause effects at the individual, population and community levels in anurans. Elevated concentrations of Pb and Fe can cause morphological abnormalities in developing anurans (Severtsova et al., 2013; Severtsova & Aguillón-Gutiérrez, 2013; Aguillón-Gutiérrez & Ramírez-Bautista, 2015; Aguillón-Gutiérrez, 2018) which could lead to declines in recruitment to adult populations. Cd accumulation can lead to decrease in larval anuran survival, especially for species highly dependent on plant material (Gastelum et al., 2019), but another effect can be skin detachment (Severtsova et al., 2013; Aguillón-Gutiérrez & Ramírez-Bautista, 2015). Mn, Zn and Cu are essential micronutrients that at high concentrations can be highly toxic, as they accumulate in different organs in adult anurans and induce oxidative stress (via oxygen reactive species [ROS]). This in turn can alter the antioxidant defense system (AOS) (Londoño-Franco et al., 2016; Prokić et al., 2016; Stolyar et al., 2008) and ultimately cause genotoxic damage, which has been related to detrimental effects at higher levels of biological organization (Mussali-Galante et al., 2014).
We found several metals accumulating in livers of anurans in the Huautla river, but the concentrations they attained are in general lower than those found in similar studies. For example, concentrations of 0.09 mg/kg for Cd, 2.78 mg/kg for Cr, 19.70 mg/kg for Zn and 46.40 mg/kg for Cu, have been reported in muscle and liver from anurans (Thanomsangad et al., 2019, Zhelev et al., 2020). A lower concentration in the Huautla versus other systems could result from slow but steady mine tailing leaching into the environment as mining activities ceased more than 30 years ago.
Anuran metal bioacummulation was not consistent among species or sites. Given the location of sites relative to the position of mine tailings, we expected T1 and DW to have higher concentrations of all metals examined. However, some metals were found at higher concentrations in sites located at greater distance from mine tailings. Almost all metals analyzed, except for Cd, were found at higher concentrations in L. spectabilis. Cd was found at higher concentrations in L. zweifeli and Pb in S. baudinii. These three species have a close relationship to water bodies for reproduction, feeding and thermoregulation. They primarily feed upon aquatic invertebrates and are also cannibalistic. These could be the reasons for having elevated metal concentrations in liver versus other species. However, lack of consistency in results among species suggest multifactorial influences in metal bioaccumulation for anurans in the region. High variability in metal concentrations in anurans may stem from differential tissue bioacummulation rates for each metal, differential exposure and excretion rates at the individual or species level and generally differences in metal toxicokinetics and toxicodynamics (Thanomsangad et al., 2019).
Anuran Communities
In our study we found approximately 45% of all species reported for the REBIOSH (Herrera-Balcazar, 2014) and 75 % of all species previously reported for the study area (Roa-Mata, 2017). The highest richness, diversity and abundance of anurans was obtained from T1 and DW (sites also closest to existing mine tailings). Anuran species distribution and abundance in a landscape depend on a variety of factors influencing individuals, populations and communities (Álvarez-Grzybowska et al., 2020; Muths et al., 2020). Among these, anurans are highly susceptible to humidity conditions. Tropical dry forests are subject to seasonally dry conditions which can be stressful for anurans; many streams in such areas can dry up for extended periods. Under such conditions, we hypothesize that a richer community can be found in areas near mine tailings given increased water retention capacity. Areas with inactive mine tailings (i.e., areas where not new materials are being dumped) tend to have highly porous soils which can retain more water, leading to increased vegetation cover and organic matter (Ramos-Arroyo & Siebe-Grabach, 2006; Ryzhenko & Bokhonov, 2020). This in turn could provide increased humidity conditions for anurans and facilitate their burrowing-in during the dry season (Mora et al., 2012).
Anuran communities in the study area were dominated by species in the Hylidae family, which is common for neotropical regions (Both et al., 2008; Pansonato et al., 2011). Families Bufonidae and Ranidae had two species each and Microhylidae only one (Table 2). That semi-aquatic L. zweifeli and L. spectabilis were the most abundant species may follow their close association with riparian areas (Canseco-Márquez & Gutiérrez-Mayén, 2010) throughout the year, even during dry seasons (Chávez-Ramírez, 2017). Rhinella horribilis, a species with relatively high tolerance to desiccation given a thick and impermeable skin (Cruz-Piedrahita et al., 2018) was also abundant, especially in T1. Similar abundance was observed for S. baudinii, a species inhabiting trees but with aquatic larvae. The least abundant species were Hypopachus variolosus and Agalychnis dacnicolor. This last species, larger than other hylids in the region and sometimes considered a generalist, can often be found in areas with anthropogenic activity (Mendenhall et. al., 2014, Soto-Sandoval et al., 2017).
While our study provides important information on community composition and potential threats from mining pollution in the REBIOSH, we acknowledge that our period of study was short, that we were not able to explore communities during the dry season, that our study area and sample number for some species was small and that we assumed no anuran movement among sites. We studied the anuran community only during the wet season as this is when previous studies (Herrera-Balcazar, 2014; Roa-Mata, 2017) had identified the highest abundance and diversity for the group. Prior to this study we carried out pilot sampling trips during the dry season (when only isolated pools were present in the streams) and we were unable to capture any anurans (applying a similar sampling effort). Our study only encompassed three small streams in the REBIOSH; several other lotic systems in the REBIOSH exist which remain relatively unexplored (i.e., the riparian area of the Amacuzac River) and where the anuran communities require further study. However, our assumption that individuals cannot move among sites is generally supported by existing literature, as generally anurans do not move >1km away from their breeding site (Jeliazkov et al., 2019; Lemckert 2004). For metal analyses in some sites we were only able to obtain and process one individual of a given species. We caution that metal concentrations from such samples, absent measures of variability, should be used simply as a reference to compare against other species but not considered representative of the entire species in the site. We set our study area to explore if metals from inactive mine tailings were present in anuran communities. Having found some pollutants in livers for anurans in the Huautla, we propose that similar studies are needed in the more than ten active or dormant mining areas in Morelos (SGM, 2020).
Much remains unknown about the biology, life history, population dynamics and community structure of anurans in southern central Mexico. Descriptive studies such as the one we present here are an initial but important step toward a better understanding of anuran biology and distribution. Our work joins several other documents from the area (Castro-Franco et al., 2006; Herrera-Balcazar, 2014; Roa-Mata, 2017) to aid in our understanding of the distribution of the various anuran species in this biologically diverse area of the country. While all species we encountered are regarded of least concern by IUCN (IUCN, 2023), we caution that other species or evolutionary significant units for some species could be facing greater threats from a variety of stressors than acknowledged in large-scale conservation assessments.
Implications for Conservation
Mining will continue to be an important economic activity worldwide (SE, 2020). It will also continue to be an important environmental problem as it contributes contaminants and dangerous residues (Xiao et al., 2020) and alters entire ecosystems (Ali & Khan, 2018; Chen et al., 2019). The effects from metal pollution will persist at all levels of biological organization (Mussali-Galante et al., 2013b) as they move via food webs (Fuentes et al., 2020). While the heaviest mining activity in the State of Morelos has ceased for the moment (Téllez-Ramírez & Sánchez-Salazar, 2018), its polluting legacy remains a threat for numerous socio-ecosystems in this area.
Anurans play an important role in riverine food webs and link various components in riparian ecotones (Hecnar and M’Closkey et al., 1996; Cortés-Gomez et al., 2015). Given their exposure to metals in the study area, they could serve as a route of exposure for fish, birds and mammals in the region (Ilizaliturri, 2010; Hernández-Plata et al., 2020). While our results did not show an impact of metals on anuran richness, abundance, or diversity among the sampled sites, we cannot discard possible effects at lower levels of organization. It is possible that future studies might find DNA adducts, oxidative damage or metallothionein induction at the molecular level (Hu et al., 2021). In addition, sex proportion and age structure alterations, low reproductive success, loss of genetic diversity, lowered fitness (Sasaki et al., 2016; Girotto et al., 2020), or population declines should be explored. These and higher level effects, such as alterations in energy and nutrient cycles and food webs at the ecosystem level need to also be addressed (Mussali-Galante et al., 2013a, 2014). Identifying these effects will require level-specific biomarkers able to detect biological changes that might be obscured by natural or other anthropogenic influences. Additionally, future studies shall consider phenotypic plasticity in anurans facing stressors such as metal exposure and include other potential target tissues (i.e., muscle, bone).
Despite the difficulty in carrying out above mentioned studies, they are key to conservation efforts in the REBIOSH. Here we have presented the first lines of evidence to understand the potential effects of metals to anurans in the area. It is upon building blocks like this study that better conservation strategies for protected areas can be achieved.
Supplemental Material
Supplemental Material - Metal Bioaccumulation and Assemblage Attributes for Anurans in the Sierra de Huautla Biosphere Reserve, Mexico
Supplemental Material for Metal Bioaccumulation and Assemblage Attributes for Anurans in the Sierra de Huautla Biosphere Reserve, Mexico by Eduardo Aarón Chávez-Ramírez, Patricia Mussali-Galante, Efraín Tovar-Sánchez, Leticia M. Ochoa-Ochoa and Norman Mercado-Silva in Tropical Conservation Science
Footnotes
Acknowledgements
Partial support for this project was provided by PRODEP project DSA/103.5/15/3073 awarded to NMS (author). M. Adams reviewed earlier versions of this manuscript. ![]() ) of the Centro de Investigación en Biodiversidad y Conservación, Universidad Autónoma del Estado de Morelos, México. EACR´s MS degree was supported by CONACYT scholarship number 705953. PA Eguía-Lis, OC Bello and C Ilizaliturri Hernández assisted with field sampling, sample processing or data analyses.
) of the Centro de Investigación en Biodiversidad y Conservación, Universidad Autónoma del Estado de Morelos, México. EACR´s MS degree was supported by CONACYT scholarship number 705953. PA Eguía-Lis, OC Bello and C Ilizaliturri Hernández assisted with field sampling, sample processing or data analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the PRODEP, DSA/103.5/15/3073, CONAHCYT; 705953.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
