Abstract
In frugivory networks, birds offer plants the advantage of dispersing their seeds away from the parent plants in exchange for macronutrients, primarily sugars, and water. The benefits for birds are clear, as they obtain food and water from fruits, and highly variable for plants, as birds may act in ways that vary from effective dispersers to seed predators. We studied two common species that interact frequently in the frugivory network of a Neotropical, periurban park: The clay-colored thrush (Turdus grayi) and a nightshade (Witheringia stramoniifolia), in order to evaluate the advantage of passing its seeds through a bird’s gut. We set up an investigation using captive thrushes that we fed with fruits of W. stramoniifolia. We had four experimental treatments: in two of them, seeds that had passed through the digestive tract of thrushes were germinated under greenhouse and controlled conditions; in the remaining two treatments, we germinated seeds that were not consumed by thrushes and were likewise germinated under greenhouse and controlled conditions. W. stramoniifolia seeds consumed by birds had a germination onset that was 1 to 2 weeks earlier than nonconsumed seeds. Mean germination rate and final germinability, however, did not differ significantly among treatments. In our study, passage through a bird’s gut accelerates the germination onset of seeds, an advantage that possibly enhances seedling establishment, but is not indispensable, for the dispersal of W. stramoniifolia. This bird–plant relationship ultimately accounts to a true mutualism.
Seed dispersal by frugivorous birds is a broadly studied subject (Janzen, 1970; Levey et al., 2002). The benefits of these relationships to both parts, plants and birds, have likewise being thoroughly documented (e.g., Bruna, 1999). Put in simple terms, birds obtain water and macronutrients from fruits, whereas plants find in birds an agent for dispersing its seeds and enhancing the germination process (Fricke et al., 2013). Numerous investigations have documented the broad variations and fine-grain specifics to this simplified scheme of mutual benefits: Birds’ nutrient intake is primarily composed of carbohydrates, with typically much smaller quantities of lipids and proteins (Jordano, 1995; Jordaan et al., 2011), whereas birds may act in a variety of ways on plants and their effects on them range from effective seed dispersers to seed predators (Bradford & Westcott, 2011; Samuels & Levey, 2005; Traveset, 1998; Traveset, Riera, & Mas, 2001a).
Our understanding of seed dispersal by birds has been building up for a while, through the study of small sets of interacting species and the analysis of its costs and benefits (e.g., Figuerola et al., 2010; Lieberman & Lieberman, 1986; Mandon-Dalger et al., 2004; Vázquez-Castillo et al., 2019) and we currently have comprehensive reviews and meta-analyses characterizing the nature of these relationships (e.g., Almeida & Mikich, 2017; Dáttilo & Rico-Gray, 2018; Fukui, 2003; Jordano, 1995, 2000; Traveset, 1998; Traveset, Riera, & Mas, 2001a).
These syntheses have some biases and limitations, since the number of studies done in ecosystems and frugivory networks in protected areas and habitats considered in well-preserved or functional condition outnumber those made in substantially altered habitats and urban environments (Barbosa et al., 2016; Costa Cruz et al., 2013; Jordano, 2000; Oliveira et al., 2015; Pejchar et al., 2008; Traveset, 1998), investigations done in the temperate region seem to outnumber those of the tropics (Almeida & Mikich, 2017; Bascompte & Jordano 2014), and the investigation of individual relationships between plants and birds, by necessity, has to be done on a case-by-case basis (e.g., Pérez-Cadavid et al., 2018).
The idea of this study arose from multiple observations of frugivory by birds that we made on a set of several dozen second-growth cloud forest plants that grow in a Neotropical, periurban park (Salazar-Rivera et al., in press; Teodosio-Faustino et al., in review). In these earlier studies, we sought to address a few questions focusing on a single relationship, that of the clay-colored thrush (Turdus grayi) and a nightshade (Witheringia stramoniifolia [Kunth], Solanaceae). Both of these species are among the most common and frequently recorded in frugivory interactions in our study site and therefore play central roles in the seed dispersal network of this area. For these reasons, we considered them a good start to elucidate whether this network in general, and this interaction in particular, account to a true mutualism.
We know from our own observations in the field and from published works (e.g., Dyrcz, 1983) that clay-colored thrushes offer the fruits in its diet a much larger dispersal distance than not having a dispersal agent at all, so we centered our work in answering whether passage through this bird’s gut (Traveset, Riera, & Mas, 2001a), and its physical/chemical effects (Samuels & Levey, 2005), would result in an enhanced, inhibited, or unaffected germination. We hypothesized that seeds that pass through a bird’s gut would have an earlier germination onset, a higher germination rate (germination speed), and germinability (the final percentage of seeds that germinate) than seeds subject to germination on their own.
The aim of this article is to understand the role of clay-colored thrushes in the germination of W. stramoniifolia seeds. We describe how we used captive birds and a simple experiment under controlled conditions to document the aforementioned germination characteristics. Our ultimate motivation was to contribute to our understanding of the role of frugivorous birds in the dispersal ecology of plants of urban and periurban greenspaces.
Methods
Study Area
The fruits we used in our study come from the Universidad Veracruzana’s Campus para la Cultura, las Artes y el Deporte (UV-CCAD), a 33-ha periurban park located in the outskirts of the city of Xalapa, Veracruz, Mexico (between 19°30’25”–19°31’11” N and 96°55’11”–96°54’48” W, at an average elevation of 1,417 m). The UV-CCAD has devoted about one half of its area to sports courts, a sports arena, a library, concert halls, and other buildings. The other half is composed of gardens, artificial impoundments, and second-growth vegetation. The latter is relatively young, as the entire campus used to be cattle pastures and soccer fields 20 years ago. For a detailed description of this study site, including its vegetation history and bird–plant frugivory network, see Salazar-Rivera et al. (in press) and Figure 1.

The Universidad Veracruzana’s Campus para la Cultura, las Artes y el Deporte in Xalapa, Veracruz, Mexico. This park is located at the interface between the ca. 600,000 people city and a large matrix of coffee fincas and second-growth vegetation that surrounds it. Both, this park and the adjacent habitat patches are part of an archipelago of protected areas that abuts the city. Figure courtesy of Sara P. Ibarra-Zavaleta, Universidad Veracruzana.
Fruits and Seeds of the Nightshade W. stramoniifolia (Solanaceae)
This nightshade is a common, Tropical Montane Cloud Forest early- to mid-successional plant at the UV-CCAD (Castillo-Campos, 1991). W. stramoniifolia, a plant whose type specimen was described from a locality less that 15 km from our study site, produces a ca. 0.5 cm racemose berry with many seeds (up to 70 in a single fruit). It is also one of the most common plants in the diet of frugivore birds at the UV-CCAD (Salazar-Rivera et al., in press).
We obtained W. stramoniifolia fruits during the fruiting season of April and May 2018. We randomly collected 60 ripe fruits in an intact condition, whose peel (exocarp) was glossy and devoid of insect damage. From the fruits collected, we split them in equal numbers for each of the experimental treatments and controls (Figure 2).

The Clay-Colored Thrush (Turdus grayi), One of the Most Frequent Birds in the Frugivory Network of the UV-CCAD in Xalapa, Veracruz, Mexico. Clay-colored thrushes feed on about two dozen plant species in this study site. In this article, we assess whether the passage of the seeds of the nightshade Witheringia stramoniifolia through this bird’s gut results in enhanced, inhibited, or unaffected germination, in its germination onset, germination rate, or final germinability. Clockwise, from the upper left corner: clay-colored thrush, W. stramoniifolia berries, a seed (the black bar scale represents 1 mm), and flower-to-fruit comparison. Photos courtesy of Magdiel Láinez, Universidad Veracruzana.
Experimental Setup
This experiment aimed to quantify the rate as well as the final percentage of germination of W. stramoniifolia seeds under four treatments (with four replicates each). Treatments 1 and 2 consisted of W. stramoniifolia seeds passing through a bird’s gut (consumed) followed by two germination conditions: (a) a greenhouse under natural light and environmental temperature (hereon greenhouse) and (b) an indoor chamber under constant light and controlled temperature (hereon controlled). Treatments 3 and 4 had W. stramoniifolia seeds that were not subjected to passage through a bird’s gut (nonconsumed) under greenhouse (Treatment 3) and controlled (Treatment 4) germination conditions. Each of the four treatments had four replicates consisting of 25 seeds each (n = 400 seeds).
We used three legally purchased individuals of clay-colored thrush to run the bird gut’s part of this experiment. These thrushes were originally captured in the wild. The clay-colored thrush is a bird common in the wild in this region (Ruelas Inzunza & Aguilar Rodríguez, 2010), considered as a species of Least Concern by BirdLife International (2018), and one of the most common species in cage bird trade (Aguilar Rodríguez, 1992). The commerce of many songbird species is a legal traditional activity in Mexico, federally regulated by the Secretaría de Medio Ambiente y Recursos Naturales, (see Acknowledgments section for additional information).
Thrushes were kept in bird cages, separate from one another, and fed the traditional diet of captive birds consisting of plantain, chopped hard-boiled eggs, and occasional regional wild fruits. The diet and feeding behavior of captive birds evidently differs from those in the wild, so birds were fed only regional wild fruits the day before the experiment in order to allow captive-bird food items out of the gut on the day of the experiment. On a given test day, thrushes were offered five randomly selected fruits of W. stramoniifolia during a single morning feeding period (with no other food items offered) and their feces collected after a few hours. Upon obtaining their feces, a few of them still containing a small proportion of partly digested fruit pulp and peel, the seeds were removed from them and mixed with those of other thrushes in order to evenly distribute any individual-related bias. The feeding of thrushes with W. stramoniifolia fruits stopped when we secured enough seeds for the experiment.
Seeds for the remaining treatments were obtained directly from the pulp of randomly selected fruits. We peeled fruits by hand in order to separate mesocarp from seeds. Following that, we washed the seeds with water to remove any visible remains of pulp and peel.
Before running experimental treatments, we made a seed viability test (TZ or tetrazolium test; International Seed Testing Association, 1996) on 50 randomly selected seeds from either consumed or not-consumed treatments, in order to determine its seedling metabolic activity as it reacts with triphenyl tetrazolium chloride before moving forward with germination treatments. Only two seeds (one of each treatment) were inviable, so we assumed initial viability was the same.
For germination, we placed 25 seeds in a Petri dish atop filter paper circles we kept damp with water. We considered that a seed had successfully germinated when the seedling radicle had emerged ≥2 mm.
We tracked the germination onset (first week with germinated seeds), germination rate (the number of seeds germinated per week beginning on the week radicles began to emerge and finishing when seeds stopped germinating), and final germinability (the number of successfully germinated seeds at the end of our observations/initial number of seeds) through weekly tallies. The weekly tallies of germinated seeds stopped when seeds in Petri dishes ceased to germinate, 9 weeks after initiation of the experiment.
Statistical Analyses
We used a generalized linear model to test for differences in germination rate and final germinability using the routine PROC GENMOD with a Poisson distribution and log-link in version 9.1 of the University Edition of the SAS statistical analysis software (The SAS Institute, 2019). In these analyses, independent variables were treatments (four levels), and time in weeks after the onset of germination (six levels), nested within treatments. The response variable was the number of germinated seeds. We estimated the dispersion parameter through maximum likelihood and fitted this output to a sequence of models using a Type 1 analysis. Finally, we fitted them to a Bonferroni post hoc test to make multiple comparisons among means. A post hoc test was necessary to identify which of the treatments were different, since the independent variable Number of days is nested within treatments. Because we are dealing with a set up composed of more than two treatments, these multiple comparisons are needed.
Results
We followed the germination onset, germination rate, and final germinability of four treatments (385 viable seeds) of the nightshade W. stramoniifolia (15 seeds, 3.7%, did not germinate).
Seeds that passed through a bird’s gut in Treatments 1 and 2 had a considerable advantage on their germination onset over the two remaining treatments. The Type 1 analysis model showed statistically significant differences in germination onset among the four treatments (χ2 = 77.7, df = 3, p < .001) as well as in the number of days that different treatments took to start germinating (χ2 = 347.2, df = 20, p < .001). This onset advantage is of 1 to 2 weeks depending on whether the seeds were subjected to greenhouse germination conditions (natural light and temperature regime) or the controlled conditions (photoperiod and temperature) chamber, respectively (Figure 3).
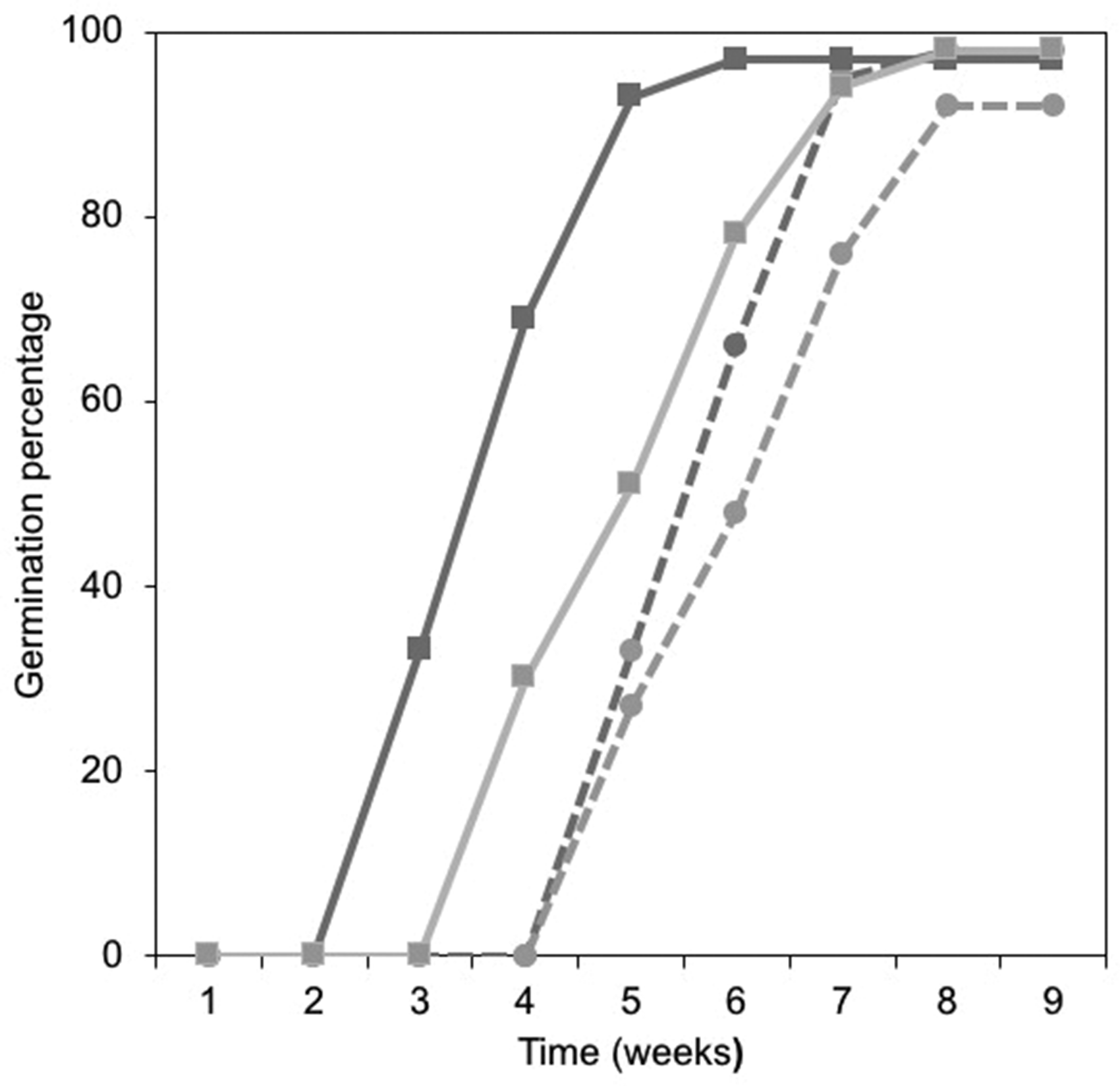
Germination Rate of Witheringia stramoniifolia, a Nightshade Found in a Neotropical, Periurban Park, Expressed in Percentage of All Seeds That Germinate Per Week. In this experiment, seeds consumed by clay-colored thrushes (Turdus grayi, depicted here in solid lines) have a germination head start over those that are not consumed by this bird (dotted lines). Dark gray lines denote consumed seeds germinated in a greenhouse under natural light and temperature conditions, whereas those in light gray are those that germinate under controlled conditions (constant light and temperature). Final germinability of all seeds is very similar.
The mean weekly germination rate among all treatments was indistinguishable, 10.6% per week (range = 10.2–10.8, SD = 0.32). All treatments followed a similarly S-shaped curve. Seeds passing through a bird’s gut germinated under greenhouse conditions (Treatment 1) experience no germination during the first 2 weeks. By Week 3, about a quarter of them germinate, and this trend continues at a similar rate until Week 5 when it levels off. Treatment 2 (consumed by bird and germinated under controlled conditions) followed a similar trend, only lagging 1 week relative to Treatment 1. In Treatments 3 and 4 (seeds not consumed by birds and germinated under greenhouse and controlled conditions, respectively), seeds followed a similar germination onset, rate, and final germinability, although they began germination at Week 5 (2 weeks after Treatment 1 and 2 weeks after Treatment 2) and reached an asymptote on Week 8.
When compared with each other, the weekly germination rates of all treatments (6 weeks with germination rate data × 4 treatments = 24 comparisons) were mostly similar. We found significant differences in three comparisons between treatments (12.5% of the total number of comparisons, Table 1).
General Linear Model (GLM) Results Comparing the Weekly Germination Rate of Witheringia stramoniifolia Seeds Under Four Treatments.

Note. The four treatments include consumed by clay-colored thrushes and germinated in a greenhouse, consumed by thrushes and germinated in a controlled-conditions chamber, nonconsumed by thrushes and germinated in a greenhouse, and nonconsumed by thrushes and germinated under controlled conditions. We only report results of weekly periods that statistically differ from the rest. See text for further details.
Last, final germinability was high regardless of treatment. The overwhelming majority of the seeds did germinate, the mean overall germinability was 96.2% (range: 92%–98%), with no significant differences among treatments (final germinability of consumed seeds was 97%–98%, and unconsumed seeds’ final germinability was 92%–98%; Figure 3).
Discussion
Seeds of the nightshade W. stramoniifolia that have been consumed by the clay-colored thrush exhibit a 1 to 2 week germination head start when compared with nonconsumed seeds. Seeds consumed by birds have an 11% to 22% faster onset than nonconsumed seed treatments. The mean germination rates of all treatments, around 10%, differ only slightly under greenhouse and controlled germination conditions (in 3 of the 24 weekly rate comparisons). The final germinability is essentially the same under all treatments and germination conditions.
The single advantage we found for this nightshade’s seeds to be ingested by clay-colored thrushes is to help accelerate the germination onset. This early onset could be of importance in the establishment of seedlings. A germination head start has been discussed in the literature as a critical advantage to avoid predators and pathogens (Traveset & Verdú, 2002). Faster germination after passing through vertebrate gut is due to the physical and/or chemical scarification of the seed coat helping radicle emergence (Barnea et al., 1990, 1991; Charalambidou et al., 2003; Reid & Armesto, 2011; Samuels & Levey, 2005; Traveset, 1998; Traveset, Riera, & Mas, 2001a).
Two other advantages of seed passage through a bird’s gut are the suppression of germination inhibitors (deinhibition happens when the pulp is separated from seeds, Robertson et al. [2006]), and the deposition of seeds mixed with fecal matter (a fertilization effect, Traveset & Verdú [2002]). These two issues warrant further investigation. Our results, however, cannot be extended simply to other species in this frugivory network: We know from the literature examining similar cases that the outcomes for birds and plants of different taxonomic groups result in differences in all three features studied here (Barnea et al., 1990; Pérez-Cadavid et al., 2018; Traveset, 1998; Traveset, Riera, & Mas, 2001a).
Clay-colored thrushes, an abundant and central species in the frugivory network of this Neotropical, periurban park, enhance the germination onset of W. stramoniifolia, but do not seem to be essential in its germination rate and final germinability. Contrary to our initial hypothesis, seeds that did not pass through this bird’s gut attain a comparable germination rate and final germinability. This means that other advantages such as seed dispersal distance or benefits associated with bird gut passage (inhibitor suppression and fertilization) should be examined in future studies. This study did not compare its two germination treatments to unpeeled, intact fruits, a test that could in the future broaden our original research question to address how seeds are released from germination inhibitors and osmotic conditions (Samuels & Levey, 2005). The services provided by birds, including endocarp scarification (Jordaan et al., 2011) and germination deinhibition via the removal of pulp from seeds (Amodeo et al., 2017), are important elements that require a different experimental setup. Such an additional treatment could help us understand what the fate of seeds in nonconsumed fruits is.
An unforeseen outcome of this investigation, at least for the nightshade under study, is that greenhouse and controlled germination experiments are capable of emulating the germination rate and final germinability observed in seeds ingested by birds. This means that in the absence of birds, W. stramoniifolia can attain a germinability that is similar although slower than bird-dispersed seeds. This fact that could be of use in designing restoration or habitat management interventions. Whether seedling establishment follows a similar trajectory in further stages remains to be studied, as multiple causes or seedling mortality may arise at a later time (e.g., Bell et al., 2006; Traveset, Riera, & Mas, 2001b).
Implications for Conservation
Birds play a key role in the dispersal of seeds of many species of plants and are often regarded as a central component of ecosystem function (Janzen, 1970). This argument is broadly applied to restoration efforts, for example, in nucleation-based projects where managers set artificial perches to enhance the arrival and establishment of target plant species (Ramírez-Soto et al., 2018a, 2018b). The outcome of our research, where the single relationship evaluated had a considerable advantage in germination onset, but not in other features of the germination process such as its rate and final germinability, represents a true mutualism. This information can be of help in informing tropical montane cloud forest restoration.
Since the germination rates are comparable, and its final germinability essentially the same, restoration of W. stramoniifolia could possibly be done in the absence of an avian dispersal agent. Moreover, treatments involving germination subject to controlled conditions underperformed those of the greenhouse, making of this nightshade a species that is very simple and inexpensive to manage. Because W. stramoniifolia is a central species in this Neotropical periurban park’s frugivory network, and the most frequently consumed native plant species in our study site, its presence fosters the existence of many other bird species besides the clay-colored thrush (Salazar-Rivera et al., in press).
Footnotes
Acknowledgments
The authors sincerely thank EIC Darren Norris, an anonymous AE, and two anonymous reviewers for their constructive and helpful criticisms. Upon completion of this study, the three individuals of clay-colored thrush (Turdus grayi) used in this study were returned to the wildlife management unit UMA Unión Estatal de Capturadores, Criaderos y Vendedores de Aves Canoras y de Ornato del Municipio de Coatepec, Veracruz (we did not, however, get our money back). G. I. S. R. thanks Mexico’s Consejo Nacional de Ciencia y Tecnología (CONACYT) for doctoral fellowship 212517 and the Instituto de Biotecnología y Ecología Aplicada’s Graduate Program in Ecology and Biotechnology (Universidad Veracruzana). E. R. I. and L. R. S. V. also thank CONACYT’s Sistema Nacional de Investigadores for fellowships 47135 and 9842, respectively. E. R. I. thanks the Secretaría de Educación Pública’s PRODEP Program for financial support (UV-PTC-868).
Declaration of Conflicting Interests
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Liliana Betancourt Trevedhan, Dean, Universidad Veracruzana’s Dirección General de Desarrollo Académico e Innovación Educativa; Dr. Ángel Triglos Landa, Dean, Universidad Veracruzana’s Dirección General de Investigaciones provided funding for the publication of this paper.
