Abstract
Translocation is increasingly common in conservation but remains controversial due, in part, to limited information. Here, in a brief study, we consider the translocation of the southern white rhinoceros
Introduction
Translocations for conservation involve the transfer of living organisms from one area to another (see, e.g., International Union for Conservation of Nature [IUCN], 2013). The intention is to augment, establish, or reestablish a population (Schwartz & Martin, 2013; Seddon, Griffiths, Soorae, & Armstrong, 2014). Although such translocations are increasingly common their costs, risks and uncertainties make them controversial (Minteer & Collins, 2010; Müller & Eriksson, 2013; Ricciardi & Simberloff, 2009; Schwartz & Martin, 2013). These controversies result, in part, from limited information and transparency. Good information is often unavailable. Observations on survival and reproduction appear insufficient if the strange and neurotic behaviors that are sometimes seen in captive animals might occur in translocated animals (Mason & Latham, 2004; Rose, Nash, & Riley, 2017; Wolfensohn et al., 2018). Studies are an essential element to evaluate efforts and improve evidence and available information (Rout, Hauser, & Possingham, 2009; Sutherland et al., 2013).
Translocations are a key strategy for the conservation of rhinoceros species (Emslie, Amin, & Kock, 2009). The IUCN guidelines for such interventions note that many past rhino translocations have lacked monitoring and that “as a result nothing or little was learned to help guide and improve future translocations and the translocations have often ended in failure” (Emslie et al., 2009). Here, we examine white rhinos (
Uganda lost its last white rhinoceros to poaching in 1982 (Edroma, 1982). In 1997, the nongovernmental organization “Rhino Fund Uganda” was founded with the aim of establishing white rhinos in Uganda (Brett, 2002). The northern subspecies previously occurred in Uganda, but with these animals unavailable, the southern subspecies was judged a suitable replacement (Brett, 2002). The translocation aims to bolster tourism and spread the risk to this threatened species. Furthermore, rhinos can maintain habitats, including wallows and short grasslands, that benefit other species (see, e.g., Everatt, Andresen, Ripple, & Kerley, 2016): For example, wildebeest and impala have been observed to leave areas once rhinos were absent (Waldram, Bond, & Stock, 2008).
IUCN lists the white rhinoceros as “Near Threatened” but notes “the continued and increased poaching threat” (http://www.iucnredlist.org; for a more extensive review of the threats, see Taylor et al., 2017). The northern subspecies,
In 2002, an area in central Uganda was selected as suitable for white rhinos due to its “short grassland in open and wooded areas” and a predicted ability to support at least 20 animals (see Brett, 2002). A 64.2-km2 fenced ranch was established in 2004 and is now called “Ziwa Rhino Sanctuary” (hereafter “Ziwa,” Figure 1, inset). Six rhinos were introduced: four from the Solio Ranch in Kenya and two from the Disney Animal Kingdom, USA (Patton, 2009). Since 2009, each adult female has calved regularly resulting in 18 offspring (7 females and 11 males) by June 2018. Details are available in studbooks, reports, and websites (see http://rhinoresourcecenter.com and http://www.rhinofund.org). Aspects of social organization of the animals at Ziwa are described in Patton, Campbell, and Genade (2018); in brief, adult males tend to be solitary while adult females associate with their youngest offspring and sometimes with adolescent animals that may be unrelated.
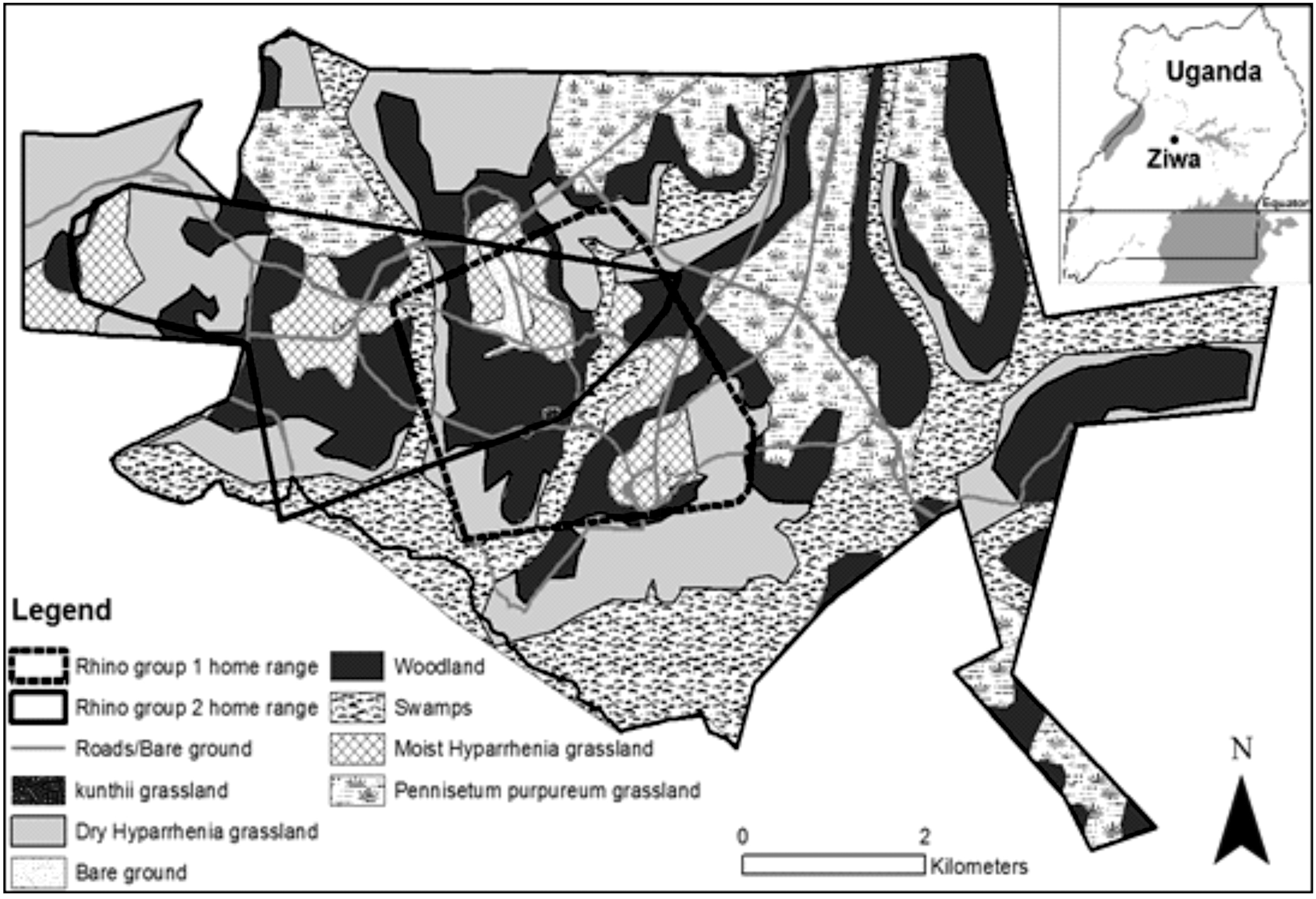
Ziwa, location, vegetation, and range of the white rhino groups. The ranch is situated in Nakasongola, in central Uganda at 1°26′53.50″N and 32°4′40.10″E (inset top right). The map was produced through a habitat survey (15 m by 15 m plots,
Translocations of white rhinos have not always had successful outcomes (McKeown, 2016; Player, 1967). For example, 21 of 59 animals translocated from Natal, South Africa, to Maputo, Mozambique, between 1967 and 1970 were dead by 1973. Five died from the translocation itself (anesthesia and stress), one was killed by another rhino prior to release, and two were found dead soon after. Seven more died after becoming trapped in swamps, three more drowned, and neighboring communities killed another three. Of the 38 remaining, many had left the reserve and some were believed to have returned to Natal (Tello, 1973 cited in McKeown, 2016). Others have noted behavioral problems in translocated animals, for example, only grazing at night (Player, 1967). It is because of such experiences that the IUCN guidelines on rhino translocation state that “Animals should be extensively monitored in the immediate post-release settling in period to determine the extent of their movements/dispersal and use of habitat (water points, feeding areas, and habitat types) at different times since release and in the different seasons. This information and maps of kernel-estimated home ranges can help refine and improve best-practice release recommendations” (Emslie et al., 2009, p. 99).
Here, we examine the state and behavior of two adult female-led groups 6 years after their introduction at Ziwa. Our goal was to contribute toward achieving these IUCN guidelines and help evaluate how the animals had adapted to local conditions.
Methods
Study Site
Ziwa is situated in Nakasongola, in central Uganda at 1°26′53.50″N and 32° 4′40.10″E (Figure 1, inset top right). The ranch is relatively flat averaging 1,045 m a.s.l., but two steeper hills rise to 1,082 and 1,080 m a.s.l. in the eastern corners. Annual rainfall averages about 1,000 mm (Brett, 2002; Patton, Campbell, Genade, Ayiko, & Lutalo, 2011). Regional vegetation includes woodlands, grasslands, and wetlands. Although tsetse (
Field Methods
Two groups (“Group 1” with two animals and “Group 2” with three animals) were monitored on alternating days, for 50 h each during the months of July and August 2011. Both groups included a 10-year-old adult female (“Bella” and “Kori” both born in 2000 in Solio Ranch, Kenya, and arrived at Ziwa together in July 2005, n.b. neither mother was pregnant at the time of our observations) with their first calves both 2-year-old males (Bella’s calf was born on June 24, 2009, and Kori’s on January 2, 2010). At the time of the study, Group 2 also included an unrelated 2-year-old male.
Rhinos were individually identified using ear notches and horn shapes (Støen, Pitlagano, & Moe, 2009). The animals are guarded and are thus habituated; nonetheless, monitoring at night was judged impractical, as the animals sometimes react aggressively to torchlight (rangers note that the animals tend to linger in specific locations at night where they generally rest and feed, see also Patton et al., 2011). At the start of the study, the body condition of the two adult females was scored (following Adcock & Emslie, 2003).
We recorded the behavior using a continuous sampling approach. Each morning, groups were located and followed for approximately 10 h from a distance of around 20 m (further if animals appeared agitated). The animals were followed from behind or to the side but never from in front. In the case of a charge, protocol was to rapidly find a safe location (this never occurred during the study but had been reported previously). We noted time, location, habitat, and temperature when behaviors changed (using a handheld GPS and a “Weathereye WEA25” located at 1.3 m). We established 397 15-m × 15-m vegetation plots while monitoring and added 24 randomly located habitat survey points across the rest of the reserve. In each plot, we measured grass height, identified the dominant species, and applied standard Food and Agriculture Organization classifications (Mwebaze, 1999).
Habitat preferences were analyzed using compositional analysis where relative habitat use (time) and availability (area) are ranked against each other (Aebischer, Marcstrom, Kenward, & Karlbom, 1993; Aitchison, 1986). The total area used by the animals was estimated from the Minimum Convex Polygon from our GPS points (
Results
Both adult females scored 4 out of 5 on the Adcock and Emslie (2003)’s condition scores—indicating above average health for females with calves.
We distinguished and mapped seven habitat types and found that three—Woodland, Dry Hyparrhenia grassland, and Swamp—comprised well over half of the total cover (with 27.6, 23.8, and 23.1%, respectively, see Figure 1).
The areas used by Groups 1 and 2 were 9 km2 and 12 km2, respectively (Figure 1). The predominant activities were grazing, resting, and moving (45%, 39%, and 10% of observation time, respectively). Episodes of resting averaged 30 min, while grazing and moving averaged 12 and 8 min each. Other behaviors comprised less than 6% of the total time (Table 1). Both groups occasionally wallowed after drinking in hot conditions (average temperature 32°C).
Summary of Observed Activities by Time and Episodes.

aThe minimum period recorded is 1 min.
bClay licking was only observed for one group at one location on one day.
Play was mainly play-fighting between juveniles or a juvenile and visiting male. The mother would watch any play-fighting and quickly intervened if the juvenile became upset—roaring and even charging to drive the other animal away (six episodes).
Most periods of movement were short (average 8 min), as animals shifted grazing locations. The proportion of time spent moving for both groups was relatively constant throughout the day (standard deviations for the percentage of observations were 12.8 and 8.9 for Groups 1 and 2, respectively; by comparison, standard deviations for resting were 32.7 and 28.7). Both groups were most active between 07:30 a.m. and 09:30 a.m., and between 3:00 p.m. and 5:30 p.m. Resting was favored from 10:00 a.m. until 02:30 p.m. (Figure 2(a)). On a typical day, temperatures rose until 01:00 p.m. (reaching about 30°C) and declined after around 04:30 p.m. Hence, rhinos generally rested during the warmest hours but resumed activity before afternoon temperatures dropped (Figure 2(a)). Group 2 was more active than Group 1, spending 35.3% versus 44.5% of total observations at rest. Temperature was positively correlated with time spent grazing and resting and negatively correlated with moving (

Activity for both rhino groups combined (a) by time of day for all observations (shaded area,
Juveniles sometimes lead activities. For example, they would often whimper at some point in the mornings, prompting the mother to stop grazing and lead the way to a resting place. The juvenile would sometimes end the resting period too by getting up, grazing, and vocalizing, until its mother also got up. Moving groups too were sometimes lead by juveniles, but we noted that adults lead more frequently in denser versus more open vegetation (Mann–Whitney
Both rhino groups spent most time in Woodland, followed by Dry, then Moist
Discussion
Our goal was to assess the degree to which southern white rhinos were adapted to conditions at Ziwa. Despite its limitations, our study complements and corroborates studies based on ranger records (Patton, 2009; Patton et al., 2018). The animals are healthy with no obvious indications of unusual or pathological behaviors—though our ability to make such assessments is constrained by available information.
At Ziwa, the animals spent most time feeding in the relatively extensive woodland and
Overall, though comparative information is limited, the animals’ behaviors did not appear out of the ordinary for the species (see also Patton et al., 2011). Overlapping ranges are normal (e.g., Rachlow, Kie, & Berger, 1999), and the area used by each group lies within the annual ranges seen in southern Africa though much larger areas are unexceptional (Pedersen, 2009; Pienaar, 1994b; Rachlow et al., 1999). Some variation likely reflects different methods, but the small area implies adequate food availability too. A short study like this cannot provide a complete picture: likely, the area used would increase over a full year of observations, as animals are reported to favor higher ground after good rains (Patton et al., 2011). There again, even a year of data cannot account for intra-annual variation or requirements following droughts or other rare events—so all such data need to be seen as provisional. Activity patterns were similar to those in southern Africa (Jordaan, 2010; Shrader et al., 2006; Støen et al., 2009). We saw no signs that the climate caused problems though the animals often rested within woodland shade through the hottest periods each day and appeared to drink and to wallow more frequently in warmer conditions.
One lesson is how our ability to judge normal behaviors is limited by suitable reference information. Despite valuable compilations on aspects of rhino biology (e.g., Ververs et al., 2017), there is no simple means to compare and judge the behavior of these animals.
Implications for Conservation
Conservation has not always learned as much from its failures as it might have, and transparency and information sharing are not always prioritized (for a more detailed discussion, see, e.g., Bower et al., 2018; Meek et al., 2015; Sutherland et al., 2013). In the case of Ziwa’s rhinos, our ability to judge “normal” behaviors was constrained by available information. These limitations also allowed us to identify apparently undocumented behaviors: while we know mother rhinos are protective (Hutchins & Kreger, 2006), we found no previous mention that they permit juveniles to lead only when visibility is good. We need more information on all aspects of behavior.
Our observations raise no concerns about the planned Ziwa translocation, though more study is clearly required (Emslie et al., 2009). Wherever the animals are ultimately released must provide water, shade, and short grasses. Monitoring will remain crucial. The threat of poaching means that the habituated animals will require guarding. In this context, any monitoring by the guards will be valuable. We encourage studies that complement these approaches. We need to know more about translocations and their outcomes.
Footnotes
Acknowledgments
We are grateful to Rhino Fund Uganda and Ziwa Ranch, specifically Angie Genade for supporting A. E. K. during his MSc research. A. E. K. thanks the rangers of Ziwa for their kindness and help and Dr. David Harper for his guidance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
