Abstract
Anthropogenic impact has been heavy in remote oceanic islands, including the introduction of alien species, having negative effects on native seabirds. The isolated and subtropical Rapa Nui (Easter Island) is one of the few known breeding sites of the red-tailed tropicbird,
Keywords
Introduction
Human colonization in remote oceanic islands caused dramatic landscape changes and had negative consequences for their fauna, especially the avifauna (Anderson, 2002). An indirect but long-lasting anthropogenic disturbance was the widespread introduction of mammalian predators, including domestic animals such as cats, dogs, pigs, and goats (Krajick, 2005), as well as pests such as house mice (Bolton, Stanbury, Baylis, & Cuthbert, 2014) and rats (Jones et al., 2008). Anthropogenic impact has been reduced in some previously impacted oceanic islands (e.g., Gaskin, 2011; Hatfield, Reynolds, Seavy, & Krause, 2012). In contrast, the isolated and subtropical Rapa Nui (Easter Island) is inhabited by around 6,000 people and annually receives around 80,000 tourists. One of the inherent human-induced threats for seabirds breeding at Rapa Nui is past and current presence of introduced predators. The red-tailed tropicbird,
The red-tailed tropicbird is widely distributed in tropical regions of the Pacific and Indian Oceans (del Hoyo, Elliott, & Sargatal, 1992). It is globally listed as “least concern” by the International Union for Conservation of Nature (BirdLife International, 2017); however, the Australian breeding population of the red-tailed tropicbird is currently listed as near threatened due to predation by feral cats and Black rats (Garnett, Szabo, & Dutson, 2010). Similarly, the breeding colonies at southeastern Hawaiian Islands are threatened by introduced predators (VanderWerf & Young, 2014). In Chile, in the southeastern Pacific Ocean, breeding colonies are only found on the subtropical ecoregion of Rapa Nui and Salas y Gomez island and on the Desventuradas Islands (Aguirre, Johow, Seeger, Johow, & Rubio, 2009; Schlatter, 1987). The species is currently listed as vulnerable by the Ministry of the Environment, Chile (2017). Adults are monogamous, have biparental care (Boland, Double, & Baker, 2004), and lay only one egg each season (E. A. Schreiber, 1996). The chicks are semialtricial, they hatch with their eyes closed but are covered in down (Nice, 1962). At Rapa Nui, the red-tailed tropicbird breed mainly on the cliffs of Poike (Jaramillo et al., 2008; Marin & Caceres, 2010) and on the inner and outer slope of the Rano Raraku volcano (Flores, Lazo, Campbell, & Simeone, 2017; Marin & Caceres, 2010). Rano Raraku is one of the most visited sites of the Rapa Nui National Park. At the entrance of the park, at this site, there are several facilities (coffee shop, stores, a picnic area, and trash bins) with high tourist affluence and trash accumulation. Consequently, there is a permanent presence of mammalian predators, such as dogs and cats (usually fed by tourists), and pests, such as
There are two breeding seasons for the red-tailed tropicbird at Rapa Nui, a “winter season” starting around April to June and a “spring season” starting around September to November each year. Flores et al. (2017) registered 53 nests at Rano Raraku considering the spring seasons of 2014 and 2015 and reported a breeding success of 38% and 27%, respectively. We surveyed part of the colony on September 2016 and registered 13 active nests with red-tailed tropicbirds incubating an egg. After a visit on October 2016, we found that all those 13 nests were empty. The eggs were lost for unknown causes, but predation by introduced species is one of the possibilities. The red-tailed tropicbird colony at Rano Raraku is relatively new, being regularly used since 2007 (Flores et al., 2017), and it may contain a high proportion of new or young breeders, as suggested for the red-tailed tropicbird colony at O’ahu, Hawaii, which started around 2005 (VanderWerf & Young, 2014). Inexperienced breeders (i.e., young or first breeders) may leave eggs unattended because of deficient coordination of incubation shifts (B. A. Schreiber & Schreiber, 1993). Unattended eggs have been observed in Rano Raraku, and predation was assumed after the loss of eggs (Flores et al., 2017) but without any evidence of predators attacking the eggs. Likewise, the impact of eggs consumption by introduced predators on the breeding success is unknown. The goal of this study was to assess the potential predation risk on unattended red-tailed tropicbird eggs at Rano Raraku, Rapa Nui. We performed a simple experiment using hen eggs as a proxy for red-tailed tropicbird eggs placed in previously used nests. We evaluated (a) how fast unattended eggs are visited by a potential predator, (b) the most frequent predatory species visiting unattended eggs, (c) for how long each predatory species try to depredate an unattended egg and perform other activities at nest, (d) the successful predatory events by species, (e) which type of nest is most frequently visited by predators (exposed vs. protected), and (f) whether the distance of the nests to the entrance of the park correlates with the frequency of mammalian predators encounters at nests (considering that the entrance of the park is a source of organic waste).
Methods
Study Site and Experimental Design
Rano Raraku is a broad volcanic cone rising from an open plain in the south east of Rapa Nui (27°07′S, 109°17′W). It was the main source of the volcanic tuff used for moai (anthropomorphic statues) carving (Richards et al., 2011). Most red-tailed tropicbird nests are associated with moais or rocks (considered to be protected), others are situated on the slopes dominated by the introduced molasses grass (Flores et al., 2017; considered to be exposed).
On November 18, 2016, we surveyed the most active breeding sector in the outer slope of the Rano Raraku volcano (Flores et al., 2017; Figure 1). We covered a large area of this sector, and we found one red-tailed tropicbird incubating an egg, all the other nests that we observed were empty but recently used as confirmed by the presence of feces, down, and feathers (personal observation). On November 20, 2016, three hen eggs of free-living birds raised at Rano Raraku were placed in three of the empty nests (one egg per nest). Each experimental egg was monitored by a Bushnell Camera Trap settled in each nest to record the visits and behavior of potential predators (Figure 2). The following day, nests were checked in the morning and evening to attempt to determine diurnal and nocturnal predators. None of the eggs were depredated. After each inspection, eggs and cameras were changed to other nests. On the second day, the same procedure was performed. Again, none of the eggs were depredated. Therefore, on the third day, eggs and cameras were placed in other nests and were left with no further change during 4 days. Since the third day, the nests were checked every day at 20:00 until the end of the experiment (6 days in total). A total of 15 nests were used and categorized in terms of the potential protection for the breeding birds. Ten nests were categorized as “protected” (totally or partially covered by rocks or moais or inside crevices) and five as “exposed” (covered by grass only).
Rano Raraku volcano at Rapa Nui. The square indicates part of the breeding area that was surveyed and where experimental eggs were placed at empty red-tailed tropicbird nests. One of the empty red-tailed tropicbird nests at Rano Raraku used in this study showing an experimental egg and a Bushnell Camera Trap.

Video Analyses
All videos were analyzed using the Behavioral Observation Research Interactive Software (Friard, Gamba, & Fitzjohn, 2016). We registered the predatory species that visited each nest. We constructed an ethogram for each predatory species recorded, registering the cumulative time they spent in activities of interaction and no-interaction with the experimental eggs. The activities considered as interaction were handling, biting, sniffing, kicking, pecking, and eating the eggs. The activities we considered as no-interaction were grooming, walking, standing, sniffing around the area ignoring the egg, hunting insects, and eating anything else than the eggs.
Potential differences in predation threat between the two nests categories were analyzed by standardizing the frequency of predator’s visit to the survey time for each replicate. Then, we employed a Generalized Linear Model (GLM) procedure with a binomial probability structure because the response variable is a ratio (number of visits/survey time). The distance of the nests to the entrance of the park was calculated using the Q-Gis software (2017) using GPS data. We used the Pearson’s product moment correlation coefficient to test whether the distance of the nests to the entrance of the park correlates with the frequency of mammalian predators encounters at nests. To analyze the data, we used R (R Core Team, 2012).
Results
Records of the Visits by Potential Predators to Simulated Unattended Eggs in Nests of the Red-Tailed Tropicbird,

aOne of the visits was only captured by a picture and, therefore, there was not record of the duration of the visit. bDistance not recorded. cTwo of the visits were only captured by pictures and, therefore, there was not record of the duration of that visits.

Potential predators recorded visiting simulated unattended eggs in nests of the red-tailed tropicbird,
Number of Video Records and Time Spent by Potential Predators Visiting Simulated Unattended Eggs in Nests of the Red-Tailed Tropicbird,
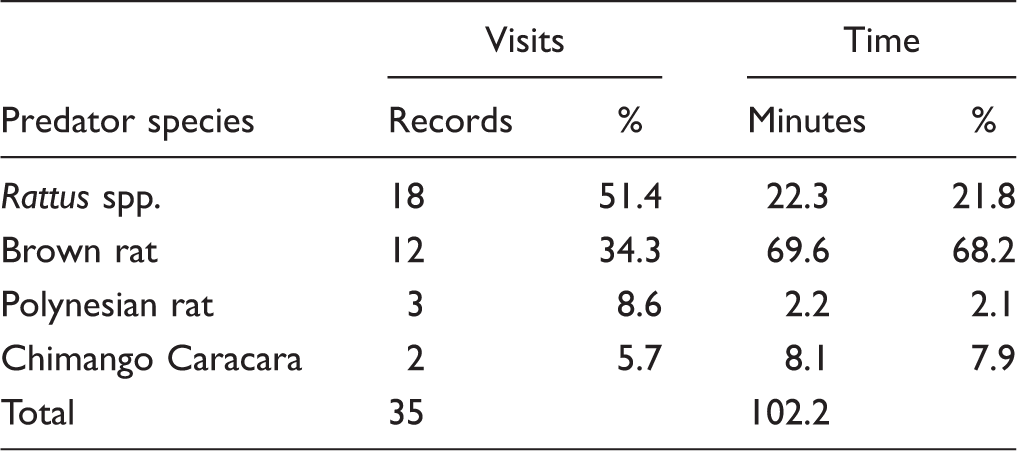
Most rat specimens were recorded at night hours, with only three visits at daylight. The raptor Chimango Caracara was only recorded at the Nest 12 (categorized as exposed). The two visits of this raptor occurred during daylight on 2 consecutive days. It was not possible to determine whether these visits were from the same individual or not. The first visit lasted less than 2 min and the second visit lasted 6 min. In both encounters, the avian predator attempted repeatedly to break the experimental egg by kicking and pecking it without success.
Meantime Spent at Nests of the Red-Tailed Tropicbird,

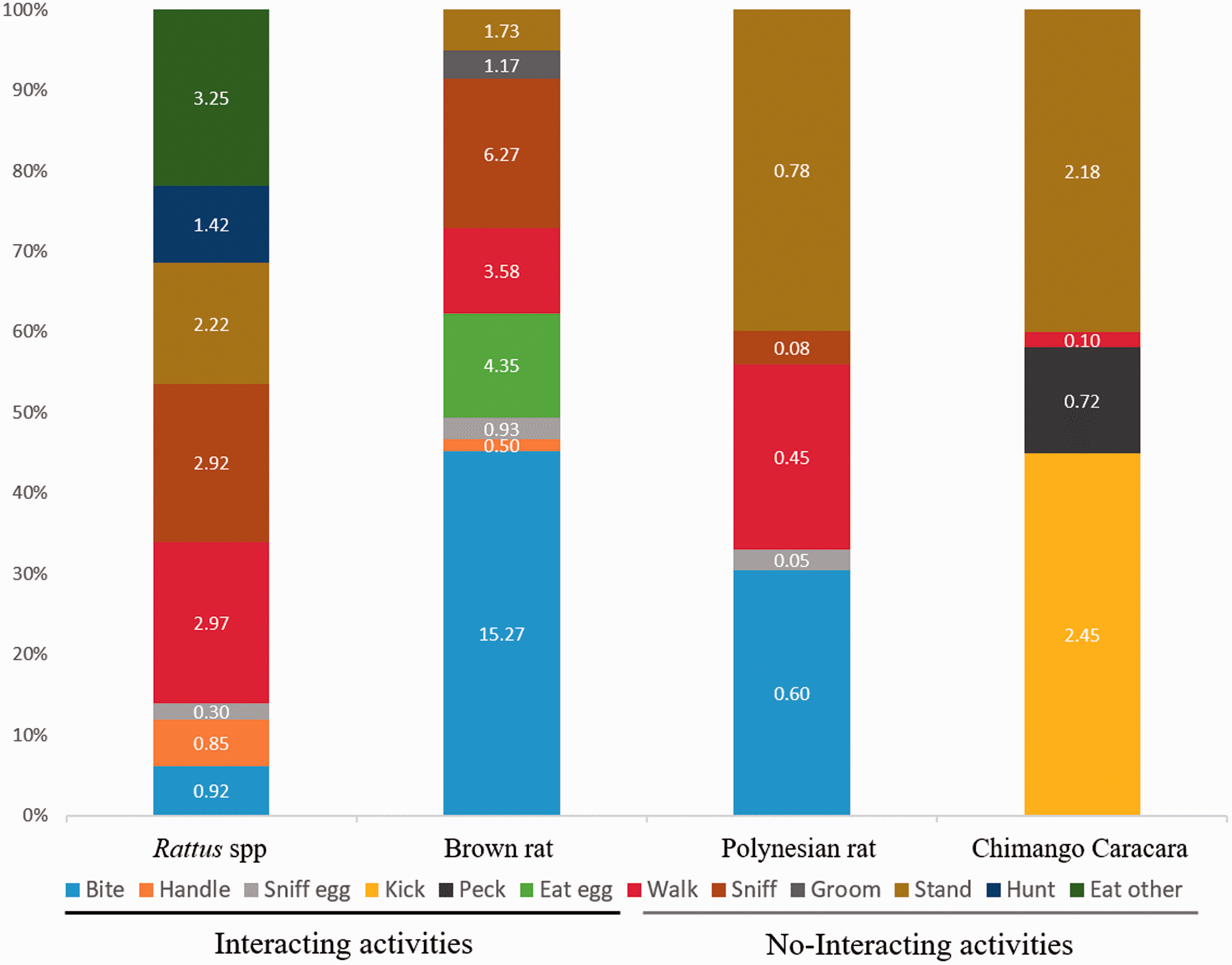
Proportion (%) of the total time and total time (min) spent at nests of the red-tailed tropicbird,
Only one egg was successfully broken and depredated. This egg was settled at Nest 10, and the predator was identified as a Brown rat. The visit lasted a total of 23 min and was the longest predatory visit recorded in this experiment. The rat broke the experimental egg after 6 min of interaction with the egg and was recorded eating the egg for more than 4 min.
There was no significant difference in the frequency of predatory visits between protected and exposed nests (deviance test = 0.1402, Pr(>Chi) = 0.7081). Finally, there was no correlation between the distance of the nests to the entrance of the park and the frequency of mammalian predators encounters at nests (Pearson’s correlation = −.316;
Discussion
Invasive species are a severe threat for island ecosystems (Krajick, 2005). Native seabirds of oceanic islands are especially vulnerable because they have not evolved defense against introduced mammalian predators (Furness & Monaghan, 1987). The Black rat
The other invasive species that tried to depredate on one of the experimental eggs was the raptor Chimango Caracara
Although the size of red-tailed tropicbird eggs (mean length = 5.6 cm, mean width = 5.0 cm) is similar to the size of the hen eggs used in this study (mean length = 5.4 cm, mean width = 4.0 cm), an analysis with scanning electron microscopy (Hitachi SU-3500) showed that the egg shell of the red-tailed tropicbird is significantly thicker than the egg shell of the hen eggs used in this study (Wilcoxon test,
Thus, considering that predators visits lasted only the 0.3% of the total experimental time, that only one egg was successfully broken and preyed, and that red-tailed tropicbird eggs are thicker than the hen eggs used in this study, we suggest that rats and Chimango Caracara are a low threat for unattended red-tailed tropicbird eggs at Rano Raraku, Rapa Nui (Easter Island).
Nevertheless, it is worth noting that our experiment was conducted in the absence of breeding birds in the area. It may be possible than in an active colony predatory attempts to unattended eggs increase if the presence of breeding birds acts as a cue for these generalist’s predators. Similarly, the absence of breeding birds during our experiment may also explain the lack of camera records of other potential predators (i.e., dogs and cats; Flores et al., 2017), which are probably attracted by the birds.
Implications for Conservation
The presence of human-introduced predators on this relatively new colony at Rano Raraku on Rapa Nui should raise concern and motivate research regarding the potential pervasive effects on unattended red-tailed tropicbird nestlings. It is possible that this area is one of the few available sites in the South Pacific Ecoregion for new or young breeders born in Rapa Nui, Salas y Gomez and Desventuradas Islands, the only relatively close breeding areas for the species.
Footnotes
Acknowledgments
We are grateful to the indigenous community of Ma'u Henua and to the National Forest Corporation-Rapa Nui for their support and for allowing access to Rano Raraku at the Rapa Nui National Park.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by a postdoctoral research grant awarded to A. I. Varela (FONDECYT N° 3160324), by a MSc scholarship (CONICYT N° 22161894) awarded to N. Luna, and by the Millenium Nucleus of Ecology and Sustainable Management of Oceanic Islands (ESMOI), a Scientific Initiative supported by the Ministry of Economy, Development and Tourism (Chile). Funding for publication was provided by Universidad Católica del Norte, Coquimbo, Chile.
