Abstract
The distribution of many vertebrate species in the Amazon basin is delimited by large rivers, which are frequently regarded as geographic barriers related with speciation and are therefore of high conservation value. Rapid development in the region includes plans to dam one of its thirteen major rivers, the Xingu. Environmental impact assessment is required for large scale development within the Brazilian portion of the Amazon Basin. However, impacts on biodiversity are only considered at the species level, and taxonomic work is far from described for many groups. In particular, anuran diversity is underestimated, in part owing to the scale of the region and, for some taxa, by morphological conservatism. Here we describe genetic and phenotypic variation in the brilliant-thighed frog,
Introduction
Conservation of biodiversity in the Amazon basin is hampered by many factors, including the large size of the region, poor taxonomic knowledge, and the rapid pace of environmental change. The Amazon basin covers approximately six million km2, with predictions that 40–55% of its rainforest will be lost within the next few decades [1–2]. In the Amazon basin, the conservation of river systems and associated riparian and inter-fluvial forested areas is considered high priority by conservation biologists [1,3]. The region contains 13 major rivers sytems: Madeira, Tocantins, Negro, Xingu, Tapajós, Purus, Marañon, Ucayali, Japurá, Juruá, Putumayo, Trombetas and Napo rivers. Their respective watershed areas are modified by expansion of cattle ranching, soy bean cultivation and other agricultural activities [2, 4–5], and more recently, by widespread governmental investment in large infrastructure projects [6–7]. New dams and waterways have been recently listed among the most critical threats to freshwater-dependent ecosystems in the Amazon, their impacts ranging from large-scale alteration of biogeochemical cycles to changes in species composition of nearby biological communities [8]. In particular, the damming of rivers to generate hydroelectric power alters large areas through flooding and disruption of seasonal water flows downstream of reservoirs [7]. The value of conserving watershed ecosystems is underscored by research demonstrating that Amazonian inter-fluvial regions are known to house unique components of biodiversity [9–10].
Many amphibian species have been recorded in some Amazon localities, but as with many tropical regions, taxonomy and patterns of intraspecific diversity are generally poorly known [11]. This problem is compounded by high levels of morphological conservatism within some amphibian taxa [12–14]. Consequently, imperiled species lists, such as the International Union for Conservation of Nature (IUCN) Red List of Threatened Species, may not be directly reliable in assessing the implications of development on amphibians.
Conservation policy of Brazil, which includes the largest portion of the Amazon Basin, requires a succession of environmental studies and the evaluation of the resulting reports by analysts in a licensing institution before allowing any large developmental project and licenses to operate [15]. The first licensing steps involve compiling species inventories for an environmental impact assessment report (EIA) and subsequent environmental monitoring studies. In accordance with current legislation, surveys use species-level taxonomies to assess rarity, extinction risk, and potential value as an economic, scientific or environmental resource [15–17]. There is no explicit objective of conserving evolutionary potential, and therefore, knowledge on intraspecific variation that is currently being accrued for a range of organisms, including amphibians, is not considered when assessing the implications of development.
Globally, the focus of conservation activities in many regions has shifted over the last decade to retaining ecological and evolutionary processes [18–21]. To conserve evolutionary processes, we need to assess the degree to which different parts of a species distribution are differentiated from each other. Descriptions of genetic variation are often used to prioritize parts of a species distribution for protection, and several ways to delimit and prioritize groups of individuals in management and conservation plans have been applied [22]. These may be based not only on their relative genetic differentiation, but also on adaptive significance and ecological exchangeability, and are often referred to as evolutionary significant units (ESU's), or management units (MU's) [20, 23–24]. These approaches and recommendations have resulted in environmental legislation protecting distinct population segments (e.g. U.S. Endangered Species Act, ESA). Policy for conserving evolutionary potential has been successfully used to protect terrestrial vertebrates such as the Florida Panther [25] and the Gray Wolf [26], among others. Acknowledging intraspecific diversity would improve these regulations by providing a means of conserving evolutionary processes and cryptic species prior to formal taxonomic assignments.
In recent years, the federal government of Brazil has increased its investment in large development projects (a list of current projects is available at the Brazilian Government's
Here we argue that because major rivers are likely to drive speciation, assessment of riverine locations where development is being planned should ask whether these locations play a substantial role in generating biodiversity. Concordant phylogeographic patterns across taxa can be used to identify the geographic features responsible for genetic divergence, but such information is not available for large areas of the Amazon basin. However, because there are often concordant phylogeographic patterns for similar taxa, a fairly rapid assessment could initially be made using abundant species and proxies for intraspecific genetic divergence [27]. We illustrate this with the brilliant-thighed frog,
Here we assess the distribution of genetic variation among
Methods
From January 2007 to March 2011, we sampled 120
Geographic position of brilliant-thighed frog (


Total genomic DNA was extracted from preserved muscle tissues collected from voucher specimens using a cetyltrimethylammonium bromide protocol (CTAB) [29]. We used primers 16Sar e 16Sbr [30] to amplify a 504 bp fragment of the 16S rRNA gene via polymerase chain reactions (PCR). The 16S rRNA gene has been regarded as one of the standard markers for the study of genetic relationships among anurans because priming sites are largely conserved, and because of its high phylogenetic signal in trees based on genetic distances [11, 31]. Protocols for amplification, purification and sequencing have been described in detail previously [12, 32]. Individual sequence fragments were deposited in National Center for Biotechnology Information's GenBank (accession numbers GU017474–GU017480, JF689976, JF690007–JF690017, KF310912–KF311011).
In order to visualize the genealogical relationships among samples, as well as the distribution of haplotypes among localities, we used the 16S rDNA sequence dataset to construct an haplotype network using statistical parsimony, as implemented in TCS 1.21 [33–34], applying a 95% connection limit and considering gaps as a 5th character state. Summary genetic statistics for samples in each sampling locality were calculated in DnaSP v.5.10.1 [35]. Average genetic distances (Kimura 2-parameters) were measured between sampling localities in Mega 5.05 [36–8].
At each sampling location, we recorded advertisement calls of male
Results
We did not detect any

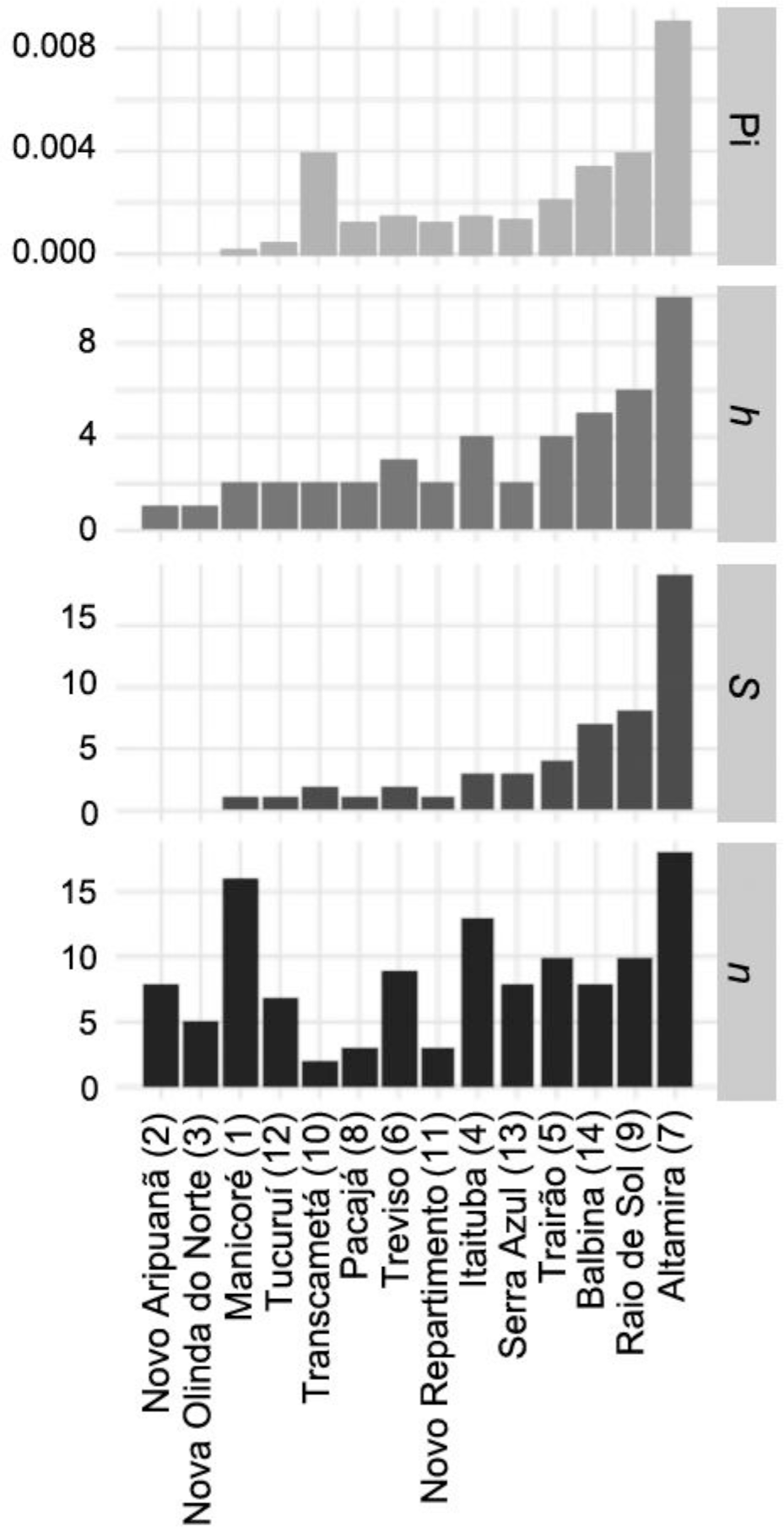
Average genetic distance among samples collected in each locality was 1.5% (Fig. 4). Lowest distances were observed between samples collected in locations within the same interfluve, notably between samples proceeding from Nova Olinda do Norte and Novo Aripuanã (western Madeira-Tapajós interfluve), and between samples from Pacajá, Anapu and Novo Repartimento (across the Xingu-Tocantins interfluve) (Table S3). Highest genetic distances (2.5–2.9%) were observed between samples collected in Itaituba (eastern Madeira-Tapajós interfluve) and locations in other interfluves (Fig. 4). Genetic distances observed between samples from Altamira and other locations were never lower than 1.1%, the lowest values being between Altamira samples and those collected in the western region of the same interfluve (Table S3). Overall, genetic distances between samples collected in different interfluves were appreciably higher than those observed between samples from the same interfluve (Fig. 5).


Calls recorded throughout the study area uncovered the existence of a distinct acoustic phenotype of

Discussion
Within the Tapajós-Xingu interfluve, construction of Belo Monte dam will potentially affect habitat occupied by a novel genetic lineage of
The genetic and vocal differences we have observed suggest that
We argue that amendments to legislation for environmental licensing of development projects in the Brazilian Amazon should include guidelines to assess intraspecific biological information. The practice of noting phenotypic characters, such as call repertoire, coloration and morphometric data, to compare with data recorded for the same species at other locations, will provide a valuable first step in evaluating whether the location where development is planned is one that generates biodiversity. If this pattern of intra-specific variation is consistent among several taxa, then these phenotypic data could be complemented with genetic data to more rigorously evaluate the importance of the location to evolutionary processes. Obtaining genetic data from widely applicable markers (e.g. regions of mtDNA) and commonplace genealogical analytical techniques will be sufficient to point out cases of pronounced intra-specific genetic divergence. Given strong evidence for the association between particular geographic features like rivers, and genetic variation for Amazonian anurans [41, 45–47], adopting surrogate measures of diversity in EIA's will reduce the risk of overlooking threatened components of diversity.
Implications for conservation
Increasingly, conservation managers are placing more importance on measures of intraspecific diversity and the conservation of evolutionary potential [27]. A central issue in preserving intraspecific diversity is the conservation of its inherent evolutionary potential in response to environmental change. Current adaptive variation in genotypes and phenotypes of a species is key to its resilience in the face of potential impacts of habitat alteration, emerging disease, or climate shifts; therefore, the focus of conservation planning has shifted to maintaining ecological processes and locations where such variation occurs [20, 23]. In addition to its evolutionary consequences, the extirpation of local amphibian populations can cause severe impacts in ecosystem functioning and ecosystem services, especially due to their biphasic life stages and their role in nutrient cycling [48–49].
We also point to the potential cost to scientific knowledge of losing particular components of intraspecific diversity. For example,
Genetic and acoustic differentiation may also correlate with differentiation in other traits of commercial significance. For example,
Recently, improvements in legislation for environmental licensing in Brazil have been suggested that make use of up-to-date ecological reasoning and techniques [53]. Considering that the Amazon is a significant reservoir of global biodiversity, further amendments to licensing procedures might require the inclusion of genetic, phenotypic and behavioural data. Ideally, the selection of groups of individuals deserving conservation or management priorities should be based on both genetic and phenotypic data [23–24]. Elsewhere the adoption of policies that include the protection of intra-specific evolutionary significant units (ESUs) or management units (MUs) have proved to be effective when applied to endangered vertebrates, for example in the U.S.A. [25,26].
Knowledge of genetic variation among interfluves for co-occurring species will be useful to the design of general conservation management strategies. At present, it is reasonable to suggest that management plans conserve evolutionary potential in geographically widespread anurans should consider, at their outset, protecting groups of individuals restricted to each one of these large interfluves. Further steps should evaluate intra-specific genetic diversity within interfluves in order to identify threatened lineages. For example, in this study, relatively high genetic divergence was detected between samples from Itaituba and those from the remaining locations between rivers Madeira and Tapajós. Itaituba is located on the west bank of the middle course of the Tapajós River, an area severely threatened by the upcoming building of at least two large reservoirs (UHE São Luiz do Tapajós and UHE Jatobá –www.pac.gov.br/energia/geracao-de-energia-eletrica).
Here we have demonstrated that a morphologically conservative frog species,
Footnotes
Acknowledgements
We thank Alejandro Estrada, William F. Laurance and three anonymous reviewers for encouragement and for many insightful comments on earlier drafts of this paper. We thank Tânia M. Sanaiotti, who first warned us of unusual ![]() ).
).
