Abstract
Background
The incidence of phantom limb pain in patients with Charcot neuroarthropathy who undergo major amputation is not well described. The purpose of this study was to determine whether patients with Charcot neuroarthropathy and diabetes who underwent either a below-knee amputation (BKA) or above-knee amputation (AKA) had an increased rate of phantom limb pain compared with those with a diagnosis of diabetes alone.
Methods
Using international classification of disease (ICD) and common procedural terminology (CPT) codes, the TriNetX research database identified 10 239 patients who underwent BKA and 6122 who underwent AKA between 2012 and 2022. Diabetic patients with and without Charcot neuroarthropathy were compared in terms of demographics and relative risk of developing phantom limb pain after AKA or BKA.
Results
Age, sex, ethnicity, and race did not significantly differ between groups. Charcot neuroarthropathy was associated with significantly increased risk of phantom limb pain following both BKA (risk ratio [RR]: 1.2, 95% confidence interval [CI]: 1.1-1.3, P < .01) and AKA (RR: 1.6, 95% CI: 1.2-2.3, P < .0068).
Conclusion
Our results indicate that patients with a coexisting diagnosis of Charcot neuroarthropathy who require BKA or AKA may have an increased risk of developing phantom limb pain.
Levels of Evidence:
Level III
Keywords
“The increased risk of developing phantom limb pain in Charcot patients suggests that prophylactic interventions for reducing phantom limb pain may be important to consider in this patient population.”
Introduction
Charcot neuroarthropathy is a devastating condition that arises as a consequence of long-standing peripheral neuropathy and repetitive micro-trauma. 1 First described as a sequela of late-stage syphilis, Charcot neuroarthropathy is now the most prevalent among patients with uncontrolled diabetes. 2 Given the continued rise in patients diagnosed with diabetes, it is likely that the number of patients who develop Charcot will continue to increase.2,3
Charcot neuroarthropathy is also associated with high rates of morbidity and mortality. 4 Sohn et al. reported a 23% lower risk of mortality in patients with diabetes alone compared with those with Charcot neuroarthropathy. This study also showed that 34% of patients with Charcot neuroarthropathy developed foot ulcers, which significantly increases the risk of amputation. The risk of amputation was 12 times higher in patients with both Charcot neuroarthropathy and foot ulcers but only 7 times higher in patients with ulceration in the absence of Charcot neuroarthropathy. 5
Phantom limb pain is a phenomenon in which patients experience painful shooting, burning, or cramping sensations in a part of their body that has been amputated. 6 The etiology of phantom limb pain is unknown, but the proposed mechanism is thought to involve reorganization and structural changes to both the peripheral and central nervous systems. 7 Possible treatments for phantom limb pain include pharmacological approaches, such as pre-emptive analgesia and anesthesia before amputation, acetaminophen and nonsteroidal anti-inflammatories, opioids, and antidepressants, as well as nonpharmacological treatments, such as transcutaneous electrical nerve stimulation (TENS), mirror therapy, spinal cord stimulation, peripheral nerve stimulation, transcranial magnetic stimulation, and regenerative peripheral nerve interface (RPNI).
Given the lack of research exploring the outcomes of Charcot patients following amputation, we sought to determine whether patients with diabetes and Charcot were more likely to develop phantom limb pain after amputation than patients with diabetes alone. We hypothesized that both groups of patients would have high rates of phantom limb pain, but Charcot patients would be more likely to develop this complication given the reorganization of their nervous system secondary to peripheral neuropathy. Understanding the relationship between Charcot neuroarthropathy and phantom limb pain may help us explore potential preventative measures to reduce the risk of phantom limb pain in these patients.
Materials and Methods
The TriNetX research database was used to establish our cohort. This network is a regularly refreshed, de-identified database that includes electronic medical record data from 83.5 million patients from over 50 health care organizations (HCOs). All information within the database is compliant with the Health Insurance Portability and Accountability Act (HIPAA) and contains only de-identified information. As such, this study was exempt from Institutional Review Board’s (IRB) approval. Retrospective data from 2012 to 2022 were collected and analyzed for this study. The database was queried using international classification of disease, tenth revision (ICD-10) and common procedural terminology (CPT) codes to identify the comparison groups (Supplemental Table 1). Group A (Charcot Group) consisted of patients with a diagnosis of both diabetes and Charcot neuroarthropathy who underwent below-knee amputation (BKA) or above-knee amputation (AKA). Group B (diabetes only) consisted of patients with a diagnosis of diabetes without Charcot neuroarthropathy who underwent BKA or AKA. Demographic information, including age at surgery, sex, ethnicity, and race were recorded (Table 1). A two-sample t-test was used to compare mean age, and chi-square t-tests were used to compare the distribution of sex, ethnicity, and race between the two cohorts. Relative risk ratio (RR), as well as the associated 95% confidence interval (CI) and P values were calculated on the TriNetX platform to determine the association between Charcot neuroarthropathy and development of phantom limb pain. For this study, significance was determined to be a P value < .05.
Demographic Data.
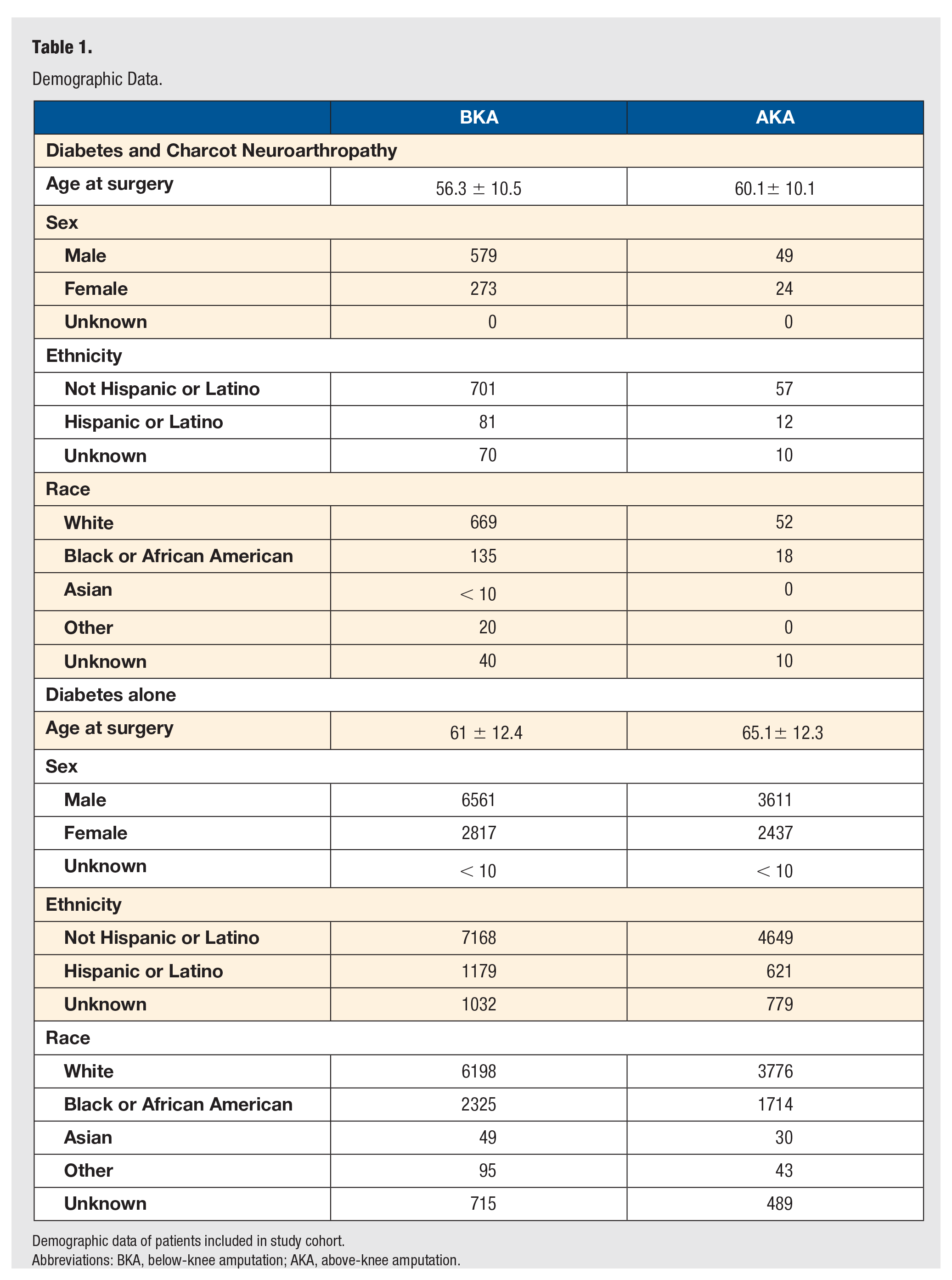
Demographic data of patients included in study cohort.
Abbreviations: BKA, below-knee amputation; AKA, above-knee amputation.
Results
There were no statistically significant differences in age, sex, ethnicity, and race between patients with diabetes and Charcot neuroarthropathy and diabetes alone in both the BKA and AKA groups (Table 2).
Demographic Differences Between Study Cohorts.
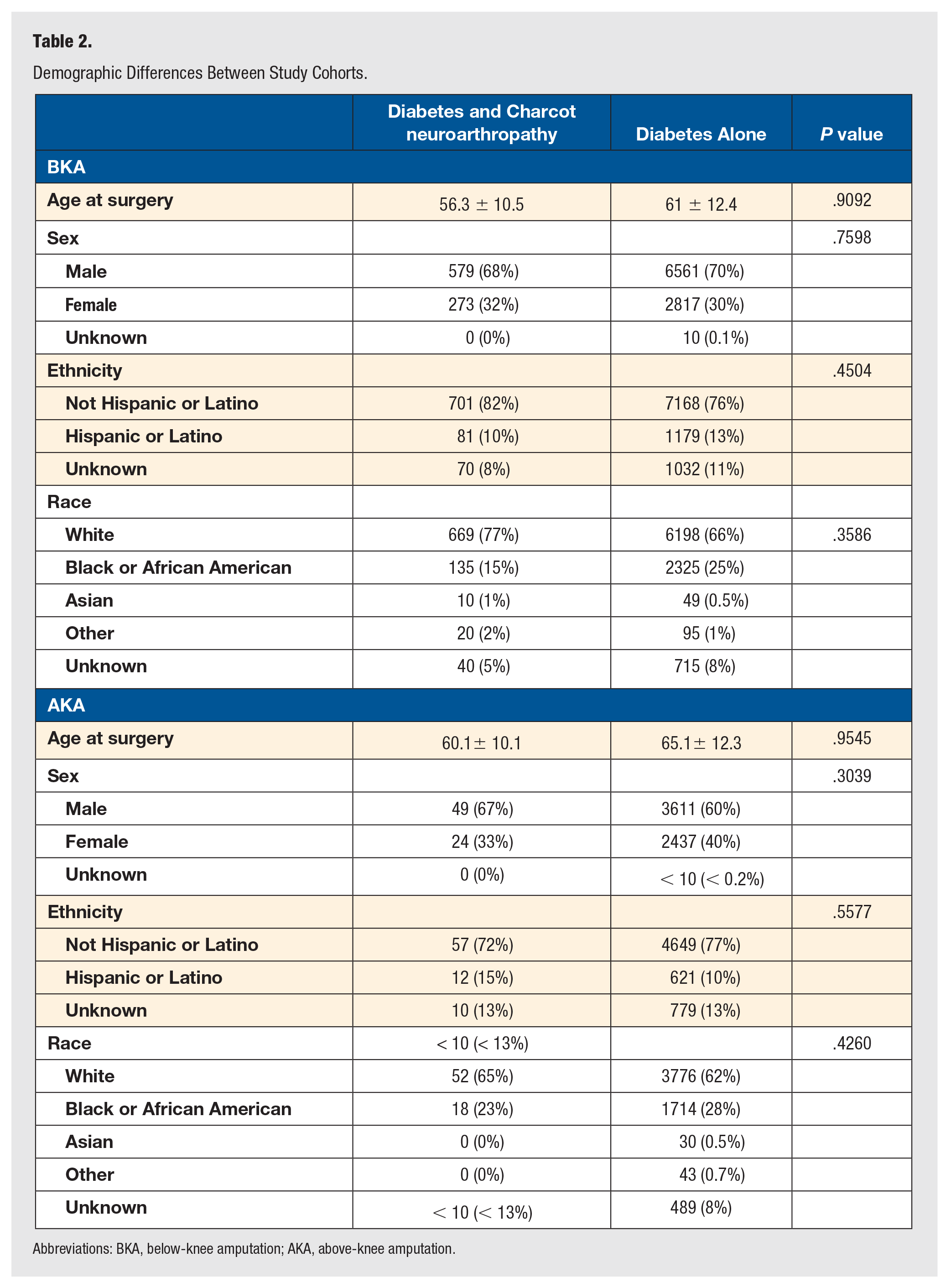
Abbreviations: BKA, below-knee amputation; AKA, above-knee amputation.
A total of 10,239 patients underwent BKA, of which 854 patients had a diagnosis of both Charcot neuroarthropathy and diabetes, and 9,379 patients had a diagnosis of diabetes alone. The rate of phantom limb pain in patients with Charcot neuroarthropathy and diabetes was 25.4%, while the rate of phantom limb pain in patients with diabetes alone was 21.4% (RR: 1.2, 95% CI: 1.1-1.3, P < .0072).
Fewer patients in this cohort underwent AKA. Of the 6,122 patients who underwent AKA, 24 had a diagnosis of Charcot neuroarthropathy and diabetes, and 1,214 had a diagnosis of diabetes alone. The rate of phantom limb pain in patients with Charcot neuroarthropathy and diabetes was 32.9% while the rate of phantom limb pain in patients with diabetes alone was 20.0%. (RR: 1.6, 95% CI: 1.2-2.3, P < .0068).
Discussion
Previous literature has discussed various risk factors that predispose patients to phantom limb pain following major limb amputation; however, the effects of Charcot neuroarthropathy on the development of phantom limb pain have not yet been described. The results of this large database study confirmed our hypothesis—the overall rates of phantom limb pain were elevated both in patients with Charcot neuroarthropathy and diabetes, and in patients with diabetes alone. However, patients with a coexisting diagnosis of Charcot neuroarthropathy and diabetes had a significantly increased relative risk for developing phantom limb pain following BKA and AKA compared with patients with a diagnosis of diabetes alone.
Pre-amputation pain has consistently been described as one of the greatest risk factors for phantom pain after limb amputation.8,9 Clinical features of Charcot neuroarthropathy include redness, swelling, pain or soreness, warmth within the foot, instability in the joints, loss of sensation in the foot, subluxation of the foot, and foot deformity. 10 The symptoms of Charcot neuroarthropathy likely contribute to pre-amputation pain, which contributes to the increased risk of developing phantom pain after amputation in this population.
There are many proposed pharmacologic and non-pharmacologic treatments for phantom limb pain; however, the increased risk of developing phantom limb pain in Charcot patients suggests that prophylactic interventions for reducing phantom limb pain may be important to consider in this patient population. Flor et al proposed a somatosensory mechanism of pain memories that are initiated following an amputation that may contribute to the formation of phantom limb pain. According to this mechanism, pre-emptive analgesia can be effectively used as an early intervention before acute phantom limb pain occurs, preventing the development of chronic phantom limb pain. 7 Karanikolas et al showed that optimized epidural analgesia or patient-controlled analgesia starting 48 hours preoperatively and continuing for 48 hours postoperatively decreased phantom limb pain in patients at 6 months compared with controls. 11 In a multicenter randomized controlled trial conducted by Ilfeld et al, patients with a 6-day continuous peripheral nerve block experienced improved post-amputation phantom pain compared with controls. 12 With the increased risk of phantom limb pain in Charcot patients, optimizing pre-emptive analgesia and continuous peripheral nerve blocks may be especially important.
RPNI, which may be performed at the time of amputation surgery is another important consideration for patients with Charcot neuroarthropathy undergoing AKA or BKA. RPNI has been previously discussed as a technique to diminish neuroma formation and post-amputation pain.13 -16 A study by Kubiak et al13,14 showed that patients who underwent major limb amputation with prophylactic RPNIs had lower rates of both symptomatic neuromas and phantom limb pain compared with control patients who underwent major limb amputation without RPNI. In this study, the major peripheral nerves of patients in the study group were isolated and sharply dissected before the RPNI construct was created while the major peripheral nerves of patients in the control group were isolated and managed with traction neurectomy, suture ligation, or burial within a nearby muscle. With the reported success of preventing phantom limb pain and the increased rates of phantom limb pain among patients with Charcot neuroarthropathy, prophylactic RPNI at the time of primary amputation is an important and worthwhile consideration for these patients.
Our study has several limitations. The online database that was used for data collection relies on accurate coding by physicians. Furthermore, the data are not representative of the entire population and are limited to participating HCOs. Consequently, the true incidence of phantom limb pain within the population may be underreported. In addition, our study did not control for other medical conditions or differences in surgical techniques that may increase or decrease the risk of developing phantom limb pain after lower extremity amputation. Finally, this is a large database study that does not differentiate between long-standing Charcot neuroarthropathy and acute flares in patients with Charcot neuroarthropathy. We did not have access to individual patient data that would be necessary to conduct these analyses.
Our work demonstrates a higher rate of phantom limb pain in patients with Charcot neuroarthropathy and diabetes, compared with those with diabetes alone. Although further research is needed, surgeons should anticipate the increased risk of phantom limb pain when operating on patients with Charcot neuroarthropathy, and consider prophylactic methods to mitigate phantom limb pain in this patient population.
Supplemental Material
sj-docx-1-fas-10.1177_19386400241230597 – Supplemental material for Charcot Neuroarthropathy Is Associated With Higher Rates of Phantom Limb After Lower Extremity Amputation
Supplemental material, sj-docx-1-fas-10.1177_19386400241230597 for Charcot Neuroarthropathy Is Associated With Higher Rates of Phantom Limb After Lower Extremity Amputation by Hannah H. Nam, Brandon J. Martinazzi, F. Jeffrey Lorenz, Gregory J. Kirchner, Vincenzo Bonaddio, Adeshina Adeyemo, Kempland C. Walley and Michael C. Aynardi in Foot & Ankle Specialist
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because all data collected was available in the public domain.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
