Abstract
Background:
Large cystic osteochondral lesions of the talus (OLT) are challenging pathological conditions to treat, but particulated juvenile cartilage allografts (PJCAs) supplemented with bone grafts are a promising therapeutic option. The purpose of this project was to further elucidate the role of PJCA with concomitant bone autografts for treating large cystic OLTs with extensive subchondral bone involvement (greater than 150 mm2 in area and/or deeper than 5 mm).
Methods:
We identified 6 patients with a mean OLT area of 307.2 ± 252.4 mm2 and a mean lesion depth of 10.85 ± 6.10 mm who underwent DeNovo PJCA with bone autografting between 2013 and 2017. Postoperative outcomes were assessed with radiographs, Foot and Ankle Outcome Scores (FAOS), and visual pain scale scores.
Results:
At final follow-up (27.0 ± 12.59 weeks), all patients had symptomatic improvement and incorporation of the graft on radiographs. At an average of 62 ± 20.88 months postoperatively, no patients required a revision surgery. All patients contacted by phone in 2018 and 2020 reported they would do the procedure again in retrospect and reported an improvement in their symptoms relative to their preoperative state, especially with pain and in the FAOS activities of daily living subsection (91.93 ± 9.04 in 2018, 74.63 ± 26.86 in 2020).
Conclusion:
PJCA with concomitant bone autograft is a viable treatment option for patients with large cystic OLTs.
Levels of Evidence:
Level IV
Keywords
“Particulated juvenile cartilage allografts (PJCAs) are a promising biologic adjuvant for the treatment of large . . . osteochondral lesions of the talus (OLTs)”
Particulated juvenile cartilage allografts (PJCAs) are a promising biologic adjuvant for the treatment of large (typically over 150 mm2) osteochondral lesions of the talus (OLTs), for OLTs with a deep cystic component (>5 mm), and for OLTs that have failed prior surgical intervention. Prior to the advent of PJCA, conventional techniques used for treating large OLTs included autologous chondrocyte implantation (ACI), donor allografts, and cartilage autografts. 1 While these other modalities are effective, PJCA has been shown to have several benefits over these therapeutic interventions making it an attractive option. It is a single-stage procedure that does not require graft contouring, has no donor-site morbidity, does not elicit an allogenic immune response, and has been shown to have superior biochemical properties leading to improved integration with the surrounding host tissue.2-5
Despite PJCA having several theoretical benefits and promising early clinical results in moderate sized lesions (100-150 mm2), there has been controversy over its clinical effectiveness in the treatment of large cystic OLTs. While some authors have published successful results when using PJCA to treat large lesions,1,6 several others have shown an increased rate of failure in lesions with an area greater than 125 mm2.7,8 In the examples where PJCA had failed, the authors did not provide bone grafting for patients with a lesion depth greater than 5 mm.7,9 Meanwhile, in examples where PJCA was successfully used to treat large cystic OLTs, PJCA with concomitant bone grafting was provided for lesions deeper than 5 mm.3,10,11 We believe these variable results reported when PJCA is used to treat large OLTs may be related to the inconsistency in how the subchondral bone is treated prior to the placement of the PJCA rather than a failure of the allograft itself.6,7
Despite the importance of treating the subchondral bone when managing large cystic OLTs, there is surprisingly little literature discussing the value of replacing the subchondral bone in order to provide viability to this technique. The purpose of this study was to bring further attention to the importance of treating the subchondral bone in addition to the cartilage when using PJCA for large cystic OLTs, to emphasize the importance of lesion depth when evaluating and treating a cystic OLT, and to contribute to the literature by providing six cases with long-term follow-up showing the potential viability of this technique. Thus, in this case series, we present results from six patients who received PJCA using DeNovo Natural Tissue Allograft (DeNovo NT, Zimmer Biomet USA) with bone grafting to treat large, cystic osteochondral lesions at an average of over 62 months after surgery.
Methods
After obtaining approval from our institutional review board (PRO00032061), we conducted a retrospective chart review of patients treated with bone grafting and PJCA for osteochondral lesions of the talus between 2013 and 2017. We excluded patients with joint failure due to autoimmune disease and advanced osteoarthritis. We found a total of 6 patients who had received PJCA with bone grafting in this time frame. Patients were contacted by phone to obtain verbal consent to use their information. Preoperative computed tomography scans were used to estimate the size of the lesion prior to surgery, and preoperative and postoperative radiographs were used to assess the extent of graft incorporation (Figures 1-3).
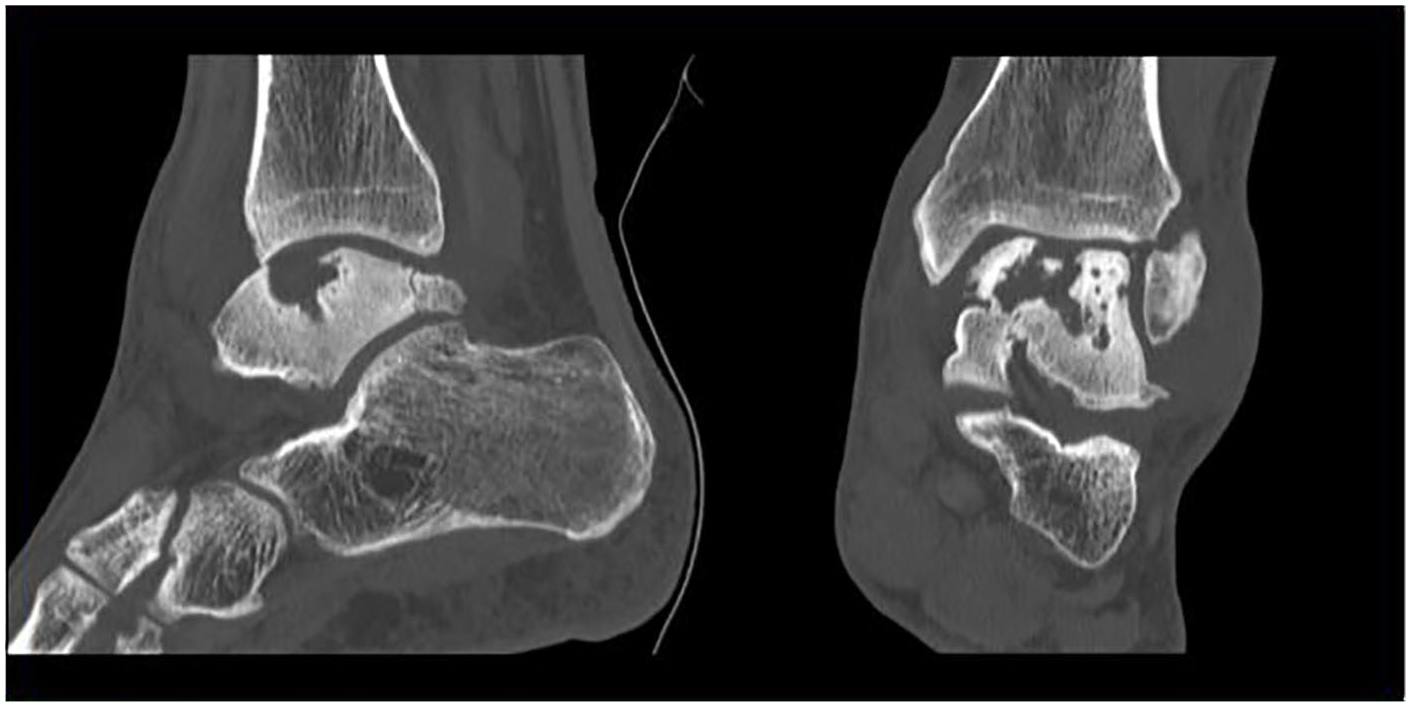
Preoperative left ankle computed tomography scan showing a sagittal (left) and coronal view (right) of patient 1’s large cystic osteochondral lesion of the talus (OLT) measuring 22.0 mm × 12.0 mm × 10.4 mm.
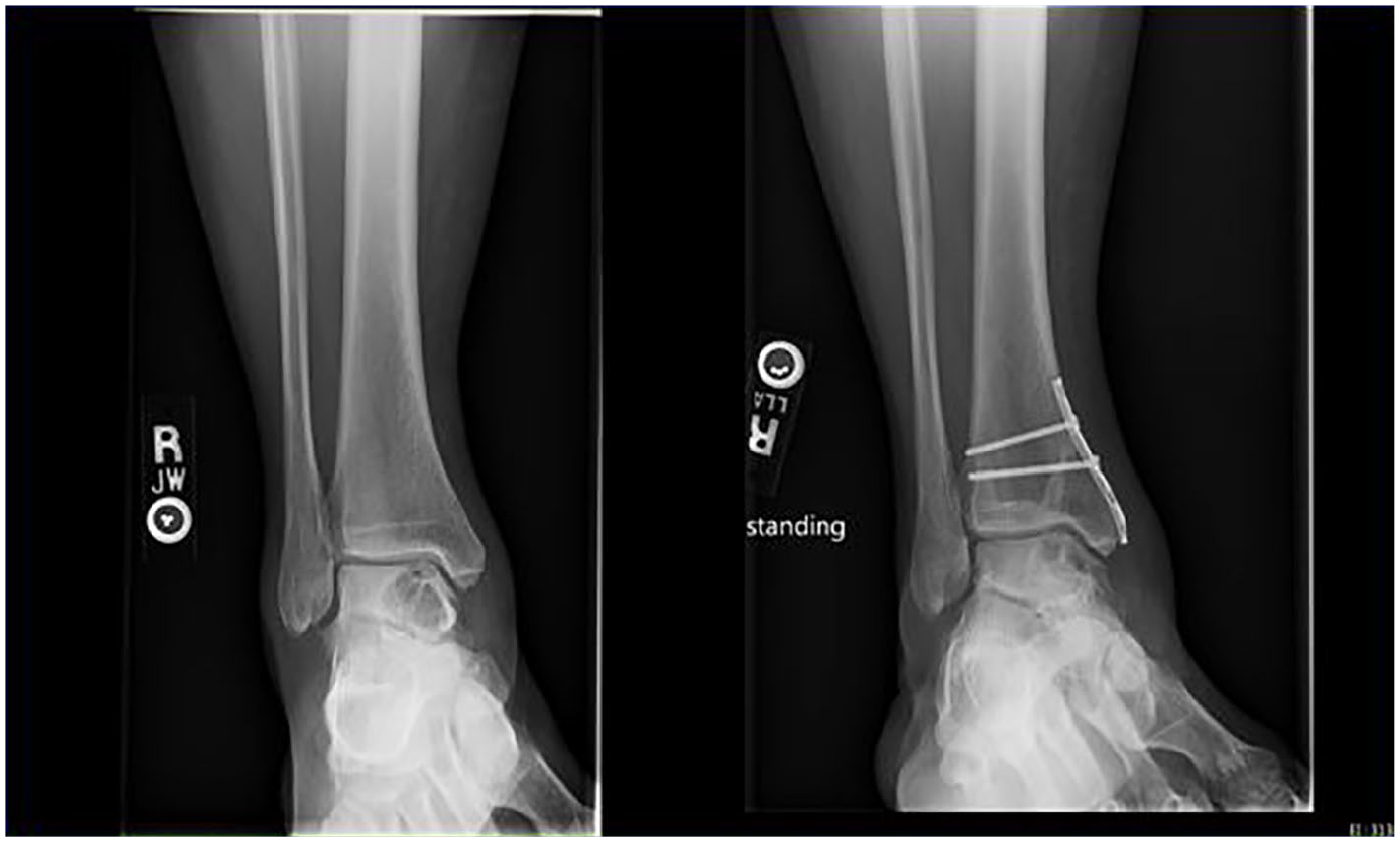
Preoperative (left) oblique ankle radiograph from patient 6 showing a large cystic osteochondral lesion of the talus (OLT) encompassing a majority of this patient’s right medial talus. One-year postoperative (right) oblique ankle radiograph showing complete incorporation of the autologous bone graft with the surrounding cancellous bone. The postoperative radiograph (right) also shows a synthes 1/3 tibial plate that was used to fix the osteotomy and was secured with 2 cortical screws in a compressive and anti-glide manner.
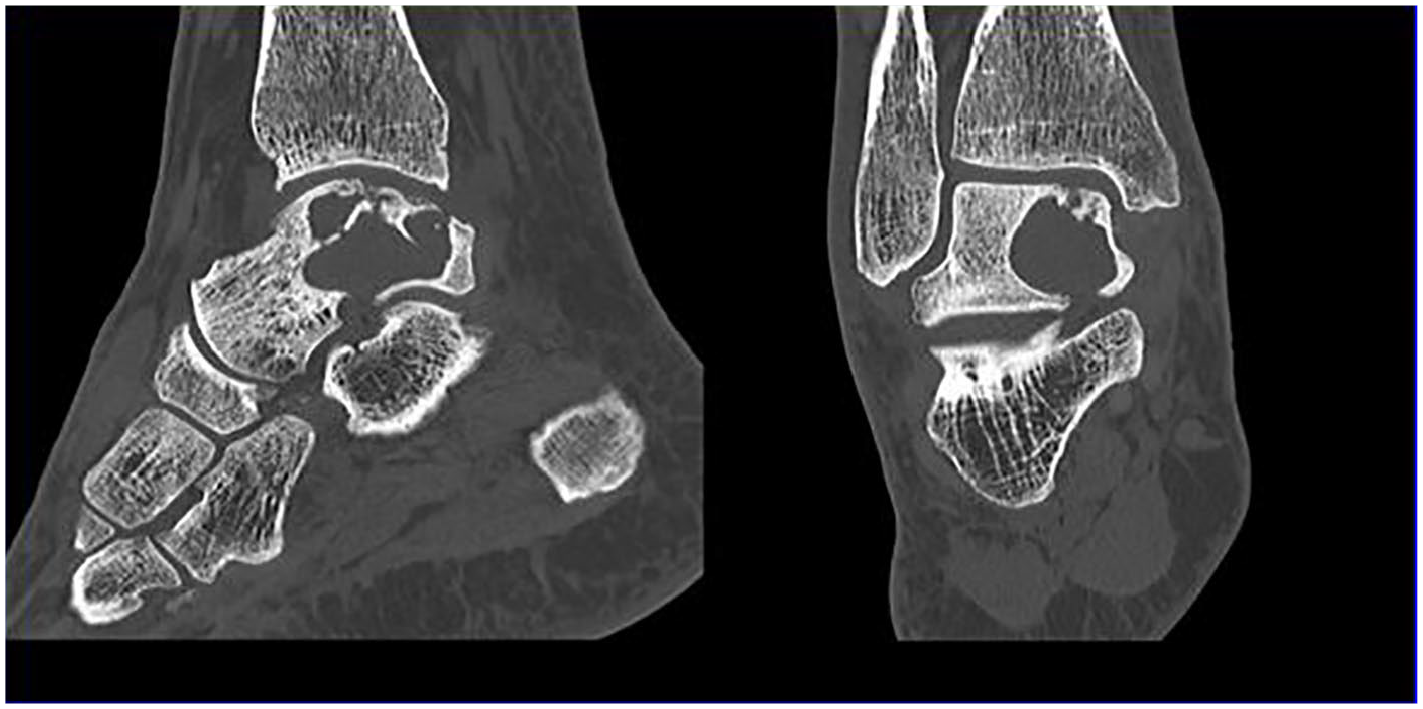
Preoperative right ankle computed tomography scan showing a sagittal (left) and coronal view (right) of patient 6’s cystic osteochondral lesion of the talus (OLT). This was the largest lesion included in our study and measured 32 mm × 23 mm × 23 mm.
Operative Technique
Preoperative imaging, history, and exam were used to determine the presence, size, and location of the osteochondral lesion. In cases where the lesion was on the medial side of the talar dome, a medial approach was performed through a medial malleolar osteotomy. In cases with a laterally located talar dome lesion, an anterior approach was made to the ankle and a Chaput osteotomy was made to provide access to the lesions. 12
The lesions were thoroughly debrided to excise all necrotic bone, loose chondral flaps, and cystic material. When a healthy, bleeding bed of cancellous bone was achieved, the defect was filled with cancellous bone autograft harvested from the distal tibia and/or calcaneal tuberosity. The bone autograft was impacted to fill the bony defect and care was taken to not over fill above the level of the native subchondral bone. A layer of fibrin glue was placed over the bone graft. Morselized DeNovo PJCA was placed onto the defect and covered with additional fibrin glue to secure it. The defect was then inspected to ensure that the allograft material remained below the level of the native cartilage and would not become prominent or risk displacement. The osteotomies were reduced and repaired with a buttress plate and screws. The patients were placed into a short leg splint and made nonweightbearing for 2 weeks in the initial post-operative period. After 2 weeks, patients were placed into a removable boot brace. At this point, they were allowed to perform range of motion exercises, but they were instructed to remain nonweightbearing for a total of 12 weeks postoperatively. After 12 weeks, physical therapy was initiated with progression of weight bearing as tolerated.
Results
Of the 6 patients, there were 5 males and 1 female with an average age at surgery of 33.0 ± 15.3 years (range: 18-58 years) and an average body mass index of 37.2 ± 7.4 kg/m2 (range: 27.89-48.5 kg/m2). Three of the patients had jobs requiring manual labor and 1 was an active smoker. Detailed patient demographic information is included in Table 1. Five patients reported a single traumatic injury or a history of multiple traumatic injuries prior to their diagnosis with an OLT, and 1 patient was uncertain of his history of trauma. All patients had large (>78 mm2) cystic OLTs with a mean lesion area of 307.2 ± 252.4 mm2 (range: 78-736 mm2). The mean lesion depth was 10.85 ± 6.10 mm (range: 6.3-23 mm). Four patients had lesions located on the posterior aspect of the medial talar dome, one had a lesion on the superior aspect of the medial talar dome, and another had a massive lesion encompassing a majority of the medial talus.
Patient Demographics and History.
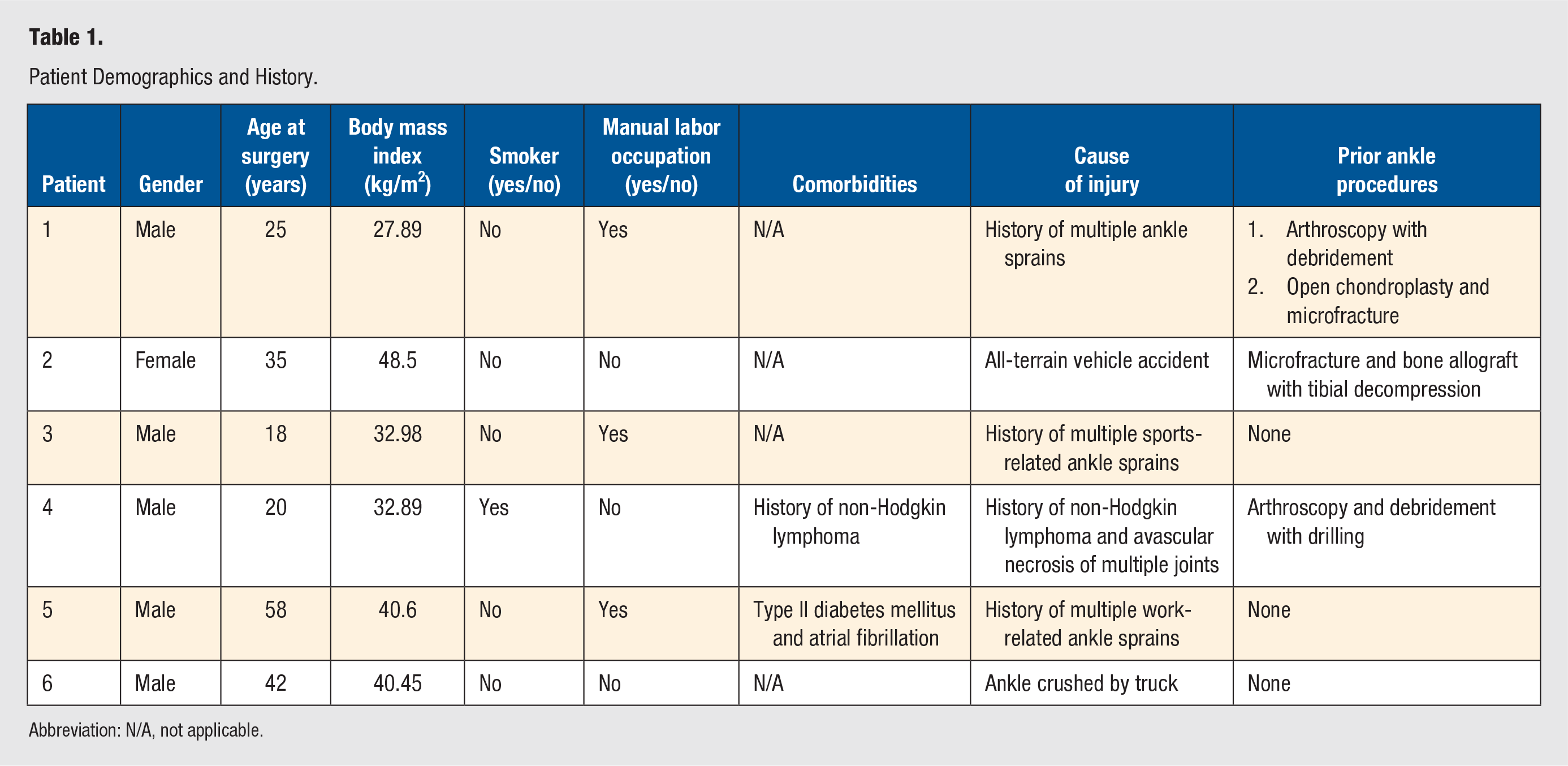
Abbreviation: N/A, not applicable.
Four patients had undergone previous procedures for treating their OLT, including arthroscopic debridement, microfracture, and drilling. The mean final postoperative follow-up appointment was 27.0 ± 12.59 weeks after surgery (range: 15-51 weeks). All six patients reported an improvement in pain following their open grafting procedures. No patients required revision surgery or returned for further treatment at an average of 62 ± 20.88 months (range: 40-85) postoperatively. Detailed individual patient data, Foot and Ankle Outcome Score (FAOS) outcomes, and visual pain scale scores are reported in Tables 2–6.
Patient Case Summary.
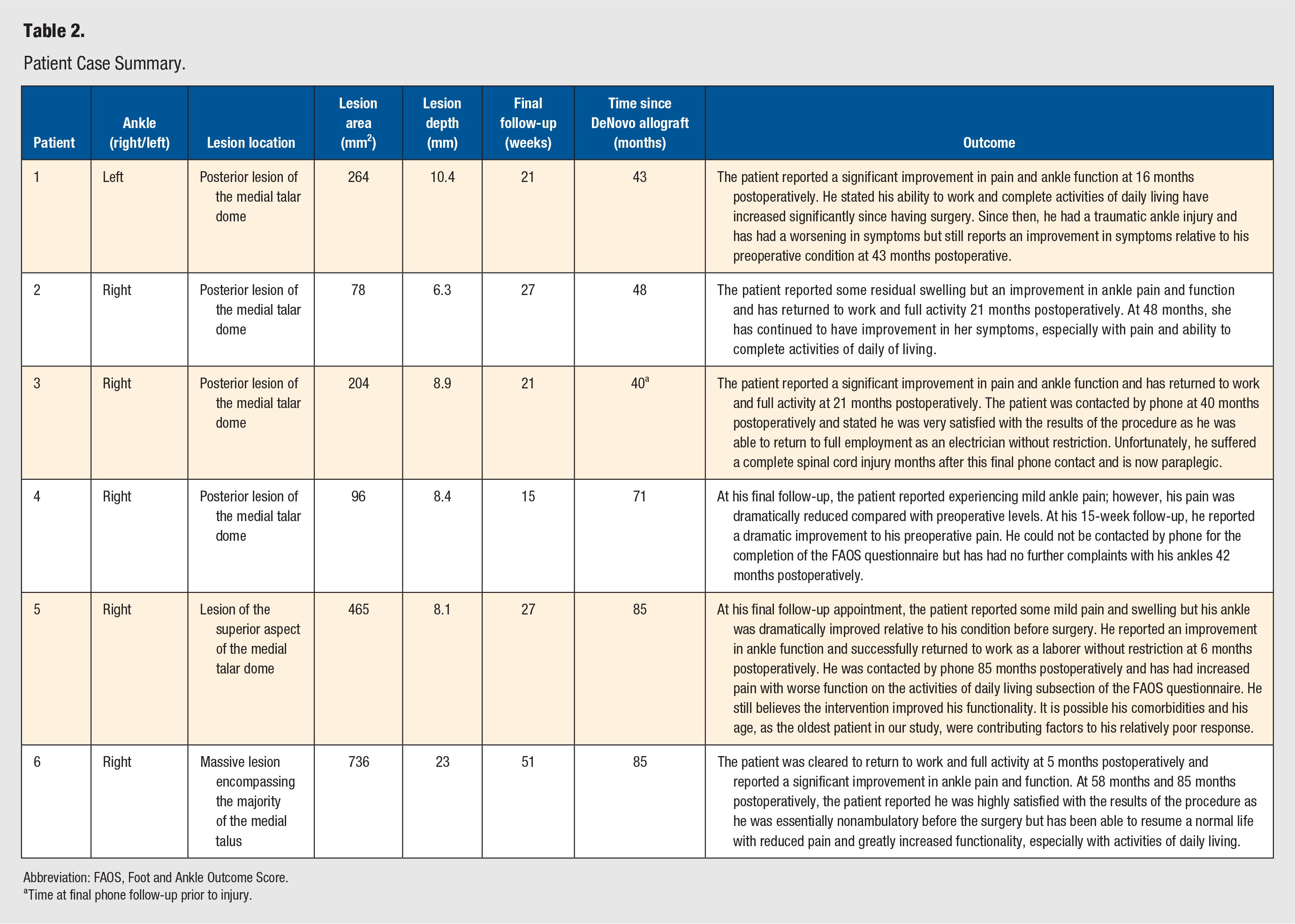
Abbreviation: FAOS, Foot and Ankle Outcome Score.
Time at final phone follow-up prior to injury.
Foot and Ankle Outcome Scores (FAOS) in 2018.
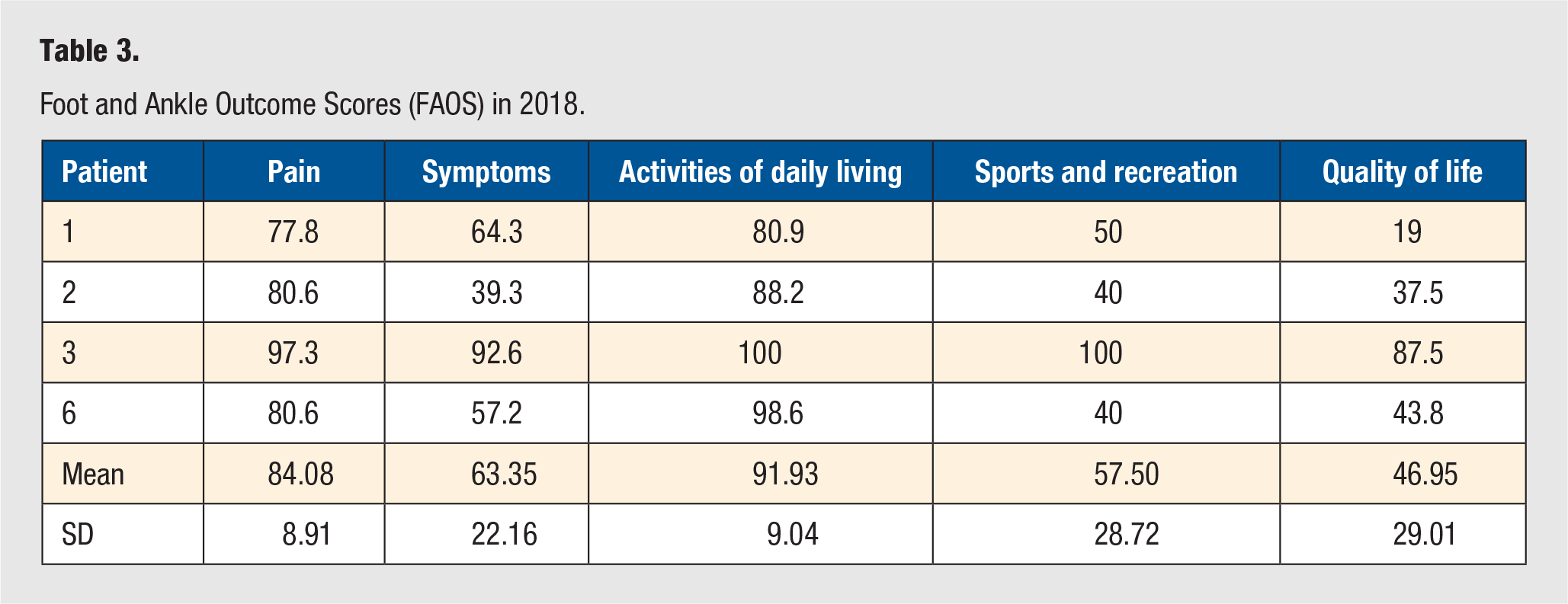
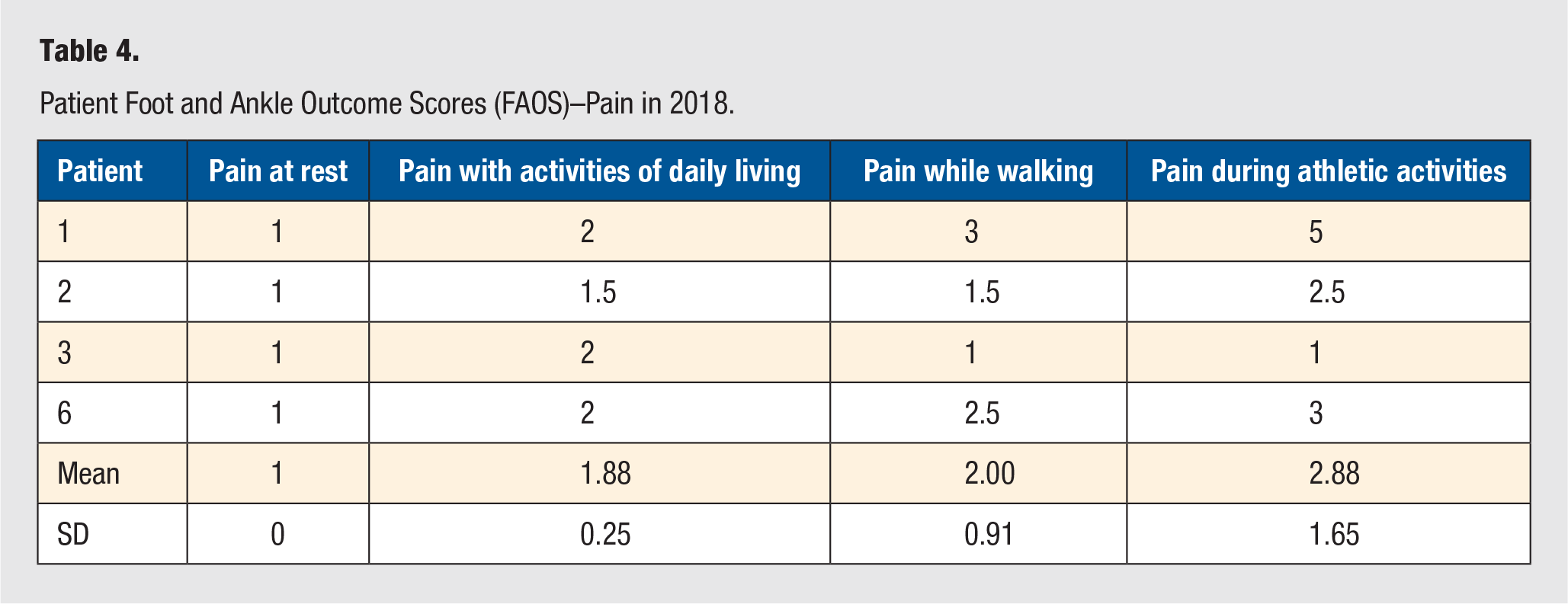
Patient Foot and Ankle Outcome Scores (FAOS)–Pain in 2018.
Foot and Ankle Outcome Scores (FAOS) in 2020.
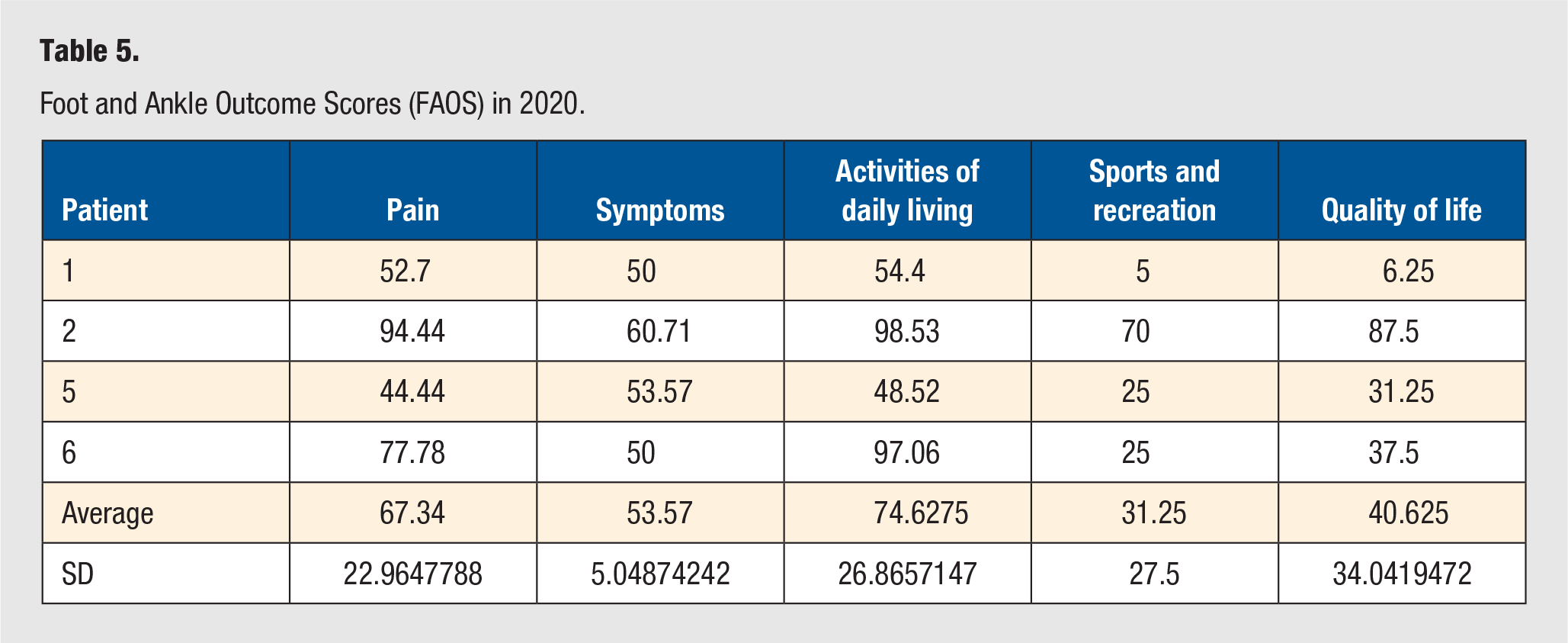
Patient Foot and Ankle Outcome Scores (FAOS)–Pain in 2020.
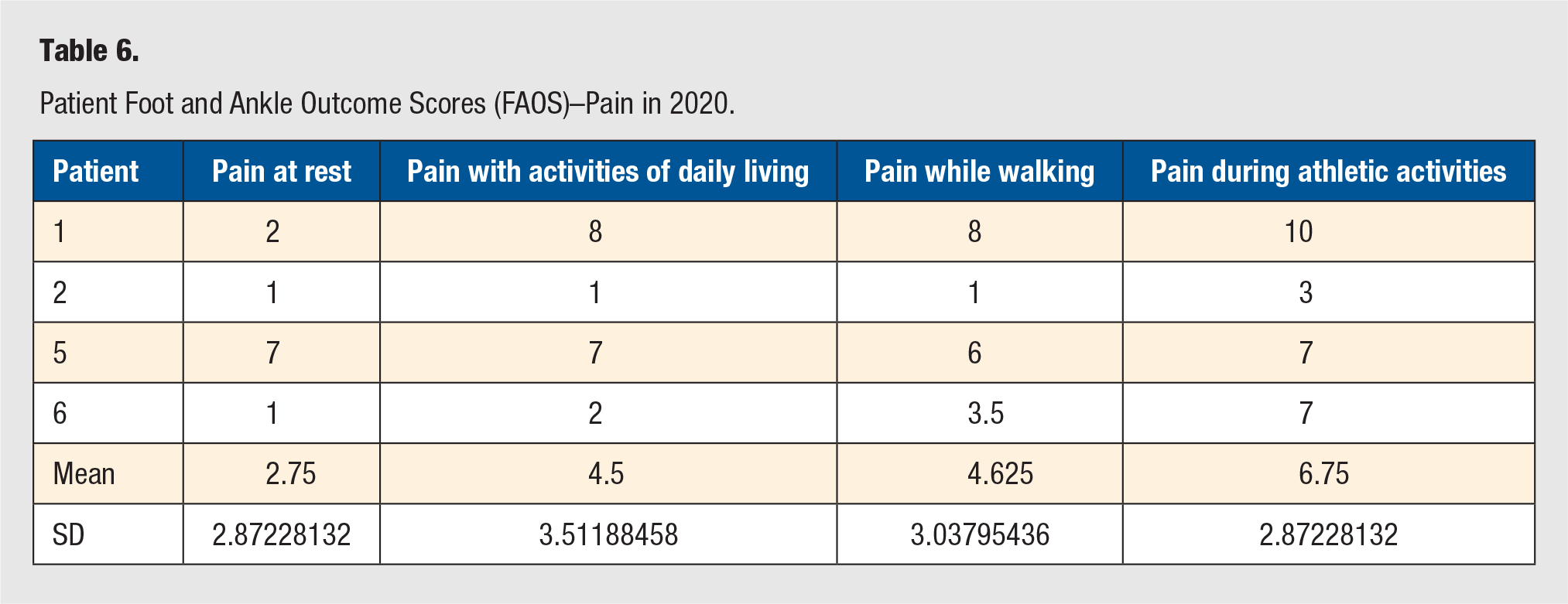
The surveys were sent to the patients at two separate time points. In 2018, 4 patients completed postoperative FAOS and visual pain scale surveys. They self-reported a mean pain score of 84.1 ± 8.91, mean symptom score of 63.4 ± 22.16, a mean activities of daily living score of 91.9 ± 9.0, a mean sports and recreation score of 57.5 ± 28.72, and a mean quality of life score of 47.0 ± 29.0 (individual patient data in Table 3). The patients also reported a pain at rest of 1.0 ± 0 (scale 1-10), a pain with activities of daily living of 1.88 ± 0.25, a pain while walking of 2.5 ± 2.0, and a pain with athletic activities of 2.88 ± 1.65 (Table 4). In 2020, 4 patients completed the postoperative FAOS and visual pain scale surveys. These 4 patients self-reported a mean pain score of 67.34 ± 22.96, a mean symptom score of 53.57 ± 5.05, a mean activities of daily living score of 74.63 ± 26.87, a mean sports and recreation score of 31.25 ± 27.5, and a mean quality of life score of 40.63 ± 34.04 (individual patient data in Table 5). The patients also reported a pain at rest of 2.75 ± 2.87, a pain with activities of daily living of 4.5 ± 3.51, a pain while walking of 4.625 ± 3.04, and a pain with athletic activities of 6.75 ± 2.87 (Table 6).
Patients 1, 2, 3, and 6 indicated that they were satisfied with the results of the procedure. While these patients reported experiencing some remaining symptoms, they also stated that the procedure had significantly improved their quality of life and allowed them to reach a level of functionality that they did not possess prior to the intervention. Patient 5 had a relatively poor self-reported outcome from the procedure but still indicated an improvement in symptoms following surgery. All 5 patients contacted by phone indicated a desire to undergo the surgery again if given the choice.
Discussion
For patients with large cystic OLTs, our study suggested that open bone grafting with PJCA tended to have good subjective patient reported outcomes as assessed with the FAOS questionnaire and the visual pain scale. The patients specifically reported an improvement in pain and an improvement in functional outcomes, especially with the activities of daily living subsection of the FAOS questionnaire. There were no reported revision surgeries. All patients but one indicated that they were satisfied with their results. The one patient who did not report satisfaction with his outcome was the oldest patient in our series and had the greatest number of medical comorbidities. Despite this, all patients reported an improvement relative to their preoperative state and would undergo surgery again if given the choice.
When treating large OLTs, the benefit with chondrocyte transplantation techniques like PJCA is that the defect is being filled with a native hyaline cartilage, consisting exclusively of type II collagen, rather than the inferior fibrocartilage, consisting of type I and type II collagen, which is produced from bone marrow stimulation techniques. 13 Transplantation techniques used to classically treat large OLTs include ACI, allografts, and autografts. ACI is an effective treatment that uses grafts consisting of cultured chondrocytes from the patient, but it has multiple drawbacks including the use of adult chondrocytes, 2-step surgeries, donor joint morbidity, and the costs associated with culturing chondrocytes.1,8,9,13 Osteochondral allografts and autografts are alternative modalities, but they also have several drawbacks including poor integration of the graft, questionable viability of the transplanted tissue, difficulty finding suitably sized donor tissues, immunological concerns for allografts, and high donor site morbidity for autografts.7,8,11
PJCA is a potentially superior therapeutic alternative to these transplantation techniques as it is a more cost-effective single-stage surgery that does not require an expensive culturing phase, does not require graft contouring, does not cause donor site morbidity, and does not elicit an allogenic immune response.2-5 The other major benefit of PJCA is derived from the biochemical and cellular superiority of juvenile chondrocytes. Juvenile chondrocytes have a more robust cellular activity, a greater volume of extracellular matrix protein synthesis, an increased rate of matrix synthesis, and an upregulation of growth promoting signaling pathways leading to increased cartilage growth, expansion, and integration with the surrounding host tissues. 14 Juvenile chondrocytes also have a proteoglycan content that is 100-fold higher relative to the neocartilage produced by adult chondrocytes. 13 Additionally, the type II collagen and type IX mRNA in fresh juvenile chondrocytes was 100- and 799-fold higher, respectively, relative to adult chondrocytes. 13 Thus, based on the in vitro data comparing PJCA to alterative treatment options for large OLTs, they would appear to be a promising solution to a difficult pathological condition.
While many studies have shown success when using PJCA to treat large lesions, several others have shown a high failure rate when managing large OLTs. We believe that failure of the PJCA technique when treating cystic OLTs is due to how the subchondral bone is treated and not due to the graft failing to incorporate with the surrounding cartilage. Currently, there is no standard procedure used for treating an OLT’s underlying subchondral bone and there has been significant heterogeneity in the methods described in the literature.6,7 In the past, the prior studies that had shown high failure rates with PJCA failed to provide bone grafting for patients with a lesion depth greater than 5 mm. For example, Dekker et al 9 found that PJCA was more likely to fail in lesions larger than 125 mm2; however, they simply performed debridement of the subchondral bone prior to placing the cartilage allograft and failed to replace the underlying subchondral bone despite having an average lesion depth of 7.25 mm. Additionally, Coetzee et al 7 reported a success rate of only 56% using PJCA to treat larger lesions; however, bone grafting was only used in less than one third of these cases with an average lesion depth of 10 mm. Interestingly, in this study, the authors had 1 revision case for a patient treated without a bone graft. During the revision, they noted that 75% of the PJCA graft had adhered to the surrounding cartilage to form a smooth articular surface and that the graft appeared proliferative and viable under histology. 7 Despite this, the patient failed the initial intervention because 25% of the graft had failed to attach over the deepest portions of the lesion, which ranged from 5 to 7 mm. 7 This led the authors to speculate on how the extent of bone loss and lesion depth influenced their poor patient outcomes and acted as a confounding variable in their study. 7
In contrast to these poor outcomes reported by Dekker et al 9 and Coetzee et al, 7 several other studies have shown success with PJCA when bone grafts were used to address the subchondral bone loss in deep cystic lesions. In a case series with 7 patients who had large cystic OLTs with an average depth of 5.85 mm, Bleazey and Brigido 10 treated each patient with PJCA and a cancellous bone sponge to fill the subchondral bone defect; they noted that all their patients were able to have significant improvements in both pain and activity scale outcome scores. Other successful examples with this technique include a study by Heida et al 11 in 2020 and a report by McDonald et al 3 in 2020. Both these studies reported significant function and pain improvements when cystic OLTs deeper than 5 mm were treated with PJCA and concomitant bone grafting.3,11 Heida et al 11 concluded that for large OLTs, PJCA that is supplemented with autologous bone grafts, when indicated, resulted in a 40% to 50% improvement in ankle pain and disability within 3.5 years. Similarly, our results using PJCA with bone grafting also suggest treating the pathology of the underlying bony lesion to provide a scaffold for the regeneration of stable subchondral bone is equally as important as repairing the damaged cartilage.1,6
To date, there have been no formal studies indicating what minimal depth PJCA requires a concomitant bone graft in order to be effective. Recent reviews by McDonald et al 3 in 2020 and Wixted et al 14 in 2020 have recommended supplementing PJCA with a bone autograft for bony defects deeper than 3 to 4 mm or 5 mm, respectively. It is important to note that these recommendations are largely based on the respective senior authors’ personal success with the technique and the success reported by Bleazey et al 10 in 2012. Other recent studies have begun to highlight the important role played by subchondral bone in joint health. 15 In addition to supporting, maintaining, and repairing the articular cartilage, the subchondral bone also aids in absorbing and distributing shock and mechanical stresses through the tibiotalar joint. 16 The subchondral bone has also been shown to communicate with the cartilage through various signaling pathways in order to better distribute stress and lesions of the bone and can contribute to the pain experienced with an OLT. 15 Thus, to best treat a large OLT, a technique should be used that restores both the bone and cartilage.
To the best of our knowledge, our study had the largest mean lesion area (307.2 ± 252.4 mm2), deepest mean lesion depth (10.85 ± 6.10 mm), and longest mean follow up (62 ± 20.88 months) where DeNovo PJCA with bone autografting was used to treat cystic OLTs. Despite this, 4 out of 5 patients contacted by phone reported satisfaction in their surgical outcome. Additionally, all 5 patients reported improvements in their functional outcomes, pain, and quality of life scores relative to their preoperative state. Moreover, none of the 6 patients in the study required a revision procedure. In particular, patient 6 had one of the best outcomes in the study despite having a lesion over 20 mm deep (Figure 3). Finally, while the cost and technical challenges must be considered when choosing a technique to treat OLT, the degree to which it improves a patient’s functional outcome is most important. Several of our patients reported experiencing very severe and debilitating symptoms before surgery that significantly impaired their ability to work and complete activities of daily living. Thus, while their FAOS scores may not be significantly higher in areas such as sports and recreation, they reported a significant improvement in pain and ability to return to work and activities of daily living.
While we believe our study contributes to the literature by outlining the long-term efficacy of treating large cystic OLTs with PJCA combined with a bone graft and highlighting the importance of the subchondral bone in managing deep lesions, there are several limitations that restrict its applicability. These limitations in our study include its retrospective nature, small sample size, lack of statistical analysis, and need for longer term follow-up. Moving forward, further studies with larger sample sizes, longer follow-up, prospective in nature, and comparative in nature are needed. Additionally, more studies are required to further elucidate the minimal depth at which a subchondral bone graft is needed to allow the PJCA technique to be viable in the treatment of large cystic OLTs. Additionally, more work is needed to understand the importance of lesion depth when selecting among the regeneration and replacement techniques used to treat large OLTs.
Conclusions
These findings suggest that PJCA with concomitant bone grafting is a viable treatment option for large, cystic osteochondral lesions of the talus. In this case series, at an average of 62 ± 20.88 months (range: 40-85) after surgery, none of the patients required revision surgery. Although the sample size of this study does not allow for detailed statistical analysis, the results are encouraging and point to the need for further research. Effective long-term management of large symptomatic cystic OLTs remains difficult thus, PJCA with a concomitant bone autograft is a promising alternative to addressing the limitations of other treatments currently available.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board (PRO00032061).
Informed Consent
Patients were contacted by phone to obtain verbal consent to use their information.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
