Abstract
Adipose tissue is an abundant and clinically accessible source of stromal cells. Stromal vascular fraction (SVF) and nanofat have been widely investigated for their regenerative potential; however, commercial systems vary considerably in yield, viability, and regulatory oversight. Most devices report fresh results only, with limited validation following cryopreservation. Mesenchymal stromal cells derived from adipose tissue have also attracted attention due to their accessibility, immunomodulatory effects, and multipotent differentiation capacity. Uvence has developed a proprietary workflow for adipose tissue processing that integrates washing, cryopreservation, thawing, and emulsification within a Human Tissue Authority-regulated laboratory. The process includes Good Manufacturing Practices (GMP) Annex 1-aligned environmental monitoring and independent quality control (QC) testing. Critically, this workflow validates postthaw cell viability, addressing a gap in current SVF/nanofat approaches. Three cryopreserved donor samples demonstrated a mean postthaw viability of ∼91% (range 90.5–92%), consistently exceeding the International Federation for Adipose Therapeutics and Science (IFATS)/ International Society for Cell and Gene Therapy (ISCT) 70% threshold. Benchmarking against global systems showed Uvence postthaw viability to be equivalent to or higher than fresh outcomes reported for enzymatic platforms (Celution, 85–91%; InGeneron, 86%) and mechanical platforms (Lipocube, Tulip, ∼96%). Unlike competitor devices, Uvence has validated freeze—thaw performance, providing a stable and compliant platform. This study also presents in vitro culture and characterization of stromal cells expanded from Uvence nanofat-derived SVF samples, including flow cytometry, morphology, and trilineage differentiation. Flow cytometry confirmed high expression of CD73, CD90, and CD105, with minimal expression of CD34/CD45, consistent with the ISCT criteria. While these findings are limited to research characterization and do not constitute approval for therapeutic use, they demonstrate that the Uvence workflow delivers a quality-focused approach to adipose tissue processing.
Impact Statement
This study defines a standardized workflow for the preparation and postthaw validation of adipose-derived stromal cells using the Uvence nanofat workflow. By integrating washing, cryopreservation, thawing, and emulsification within a controlled research framework, the method delivers consistently high cell viability and quality after storage. It addresses a critical gap in current tissue-engineering practice, where postthaw validation of stromal vascular fraction products is rarely reported. The results highlight how standardized, compliance-aligned adipose-tissue processing can support robust in vitro characterization of stromal cell populations and provide a foundation for future translational research in regenerative medicine.
Introduction
Adipose tissue is a widely studied source of regenerative cells due to its abundance, ease of harvest, and richness in multipotent stromal/stem cells. Since the first landmark description of adipose-derived stem cells by Zuk et al. in 2001, 1 the field has rapidly evolved, with numerous commercial systems offering “nanofat” or stromal vascular fraction (SVF) preparations for regenerative use. Mesenchymal stromal/stem cells (MSCs) are multipotent progenitors present in various tissues, including bone marrow, adipose tissue, and umbilical cord. They are defined by the International Society for Cell and Gene Therapy (ISCT) as plastic-adherent cells that express CD73, CD90, and CD105 and lack expression of CD34 and CD45, with the ability to differentiate into adipogenic, osteogenic, and chondrogenic lineages.2,3 Adipose-derived MSCs (AD-MSCs), particularly from nanofat-derived SVF, offer a practical and minimally invasive source with regenerative promise.4–6
Nanofat techniques have gained popularity in esthetics and reconstructive medicine,7,8 yet the quality and reproducibility of stromal cell preparations remain inconsistent. Moreover, the viability and consistency of these preparations vary significantly. Freshly isolated SVF can contain endothelial, immune, and stromal subpopulations, with reported viabilities ranging from 46% to 97%.9–12 SVF obtained either by enzymatic digestion or mechanical emulsification (nanofat) contains heterogeneous cell populations including stromal cells with MSC features, pericytes, endothelial progenitors, and immune cells. 13
In addition, marketed devices often focus on mechanical yield without embedding rigorous regulatory or quality control measures. 14 Recent studies have shown that many nanofat devices and commercial SVF prioritize mechanical processing but lack harmonized sterility assurance, quality control measures, and traceability. Head-to-head comparisons across devices demonstrate substantial variability in yield, viability, and stromal composition. 14 Similarly, findings from a GMP-compliant manufacturing study suggest that most systems do not employ consistent process controls and validated workflows, undermining the need for standardization in adipose tissue processing. 12
Uvence addresses these limitations through a validated, standardized sequence of saline washing, controlled-rate cryopreservation, cryostorage, thawing, and mechanical emulsification, in accordance with HTA regulatory requirements. This process incorporates QC testing and EU GMP environmental monitoring to ensure the safety, compliance, and integrity of the adipose tissue samples.
Commercial systems demonstrate variable results: enzymatic systems such as Celution® and InGeneron Transpose® RT report 85–91% fresh viabilities,15–18 mechanical systems such as Lipocube Nano and Tulip NanoTransfer report ∼96%,10,11 while other methods like Lipogems® (microfragmented fat) report >95% viability but without SVF isolation.13,19 Notably, most platforms lack postthaw validation, which limits their utility in tissue banking and standardized workflows.20,21
Most nanofat and SVF systems report fresh viability but lack validated evidence of viability after cryopreservation, despite growing clinical demand for sample banking and staged procedures. Research shows that commercial SVF/nanofat platforms can differ significantly in yield, cell viability, and the composition of downstream cells. However, there is limited documentation regarding sterility, bioburden testing, environmental monitoring, or traceability frameworks.6,22,23 This study aims to fill this gap by validating postthaw cell viability within a regulated workflow.
The distinguishing feature of the Uvence workflow is its validation of postthaw viability, integration of HTA-governed quality controls with traceability, and consistency across processing batches; these elements are not consistently demonstrated in conventional nanofat/SVF systems such as Celution,16,17 InGeneron, 18 Lipocube, 10 and Tulip, 11 which only generate fresh outputs without assessing postthaw integrity.
International frameworks, including ISCT/IFATS guidelines, recommend that stromal cells exhibiting MSC features in vitro meet minimal criteria (plastic adherence, fibroblast-like morphology, expression of CD73/CD90/CD105, and trilineage differentiation).2,13 In this study, we characterized MSCs from culture-expanded SVF cells isolated from Uvence nanofat obtained from three donors, comparing them against a commercial Promocell MSC control. This research was conducted strictly for characterization purposes, and the MSCs described here are not approved for clinical application. This article also compiles peer-reviewed benchmarks to collate reported viability data from enzymatic and mechanical SVF/nanofat systems, including Lipocube, Tulip, Celution, InGeneron, Lipogems, Hy-Tissue, Nanofat Converter, ARAT/MEST, and SVF-gel (Table 1).
Global Viability Benchmarks (Fresh vs Post-Thaw)
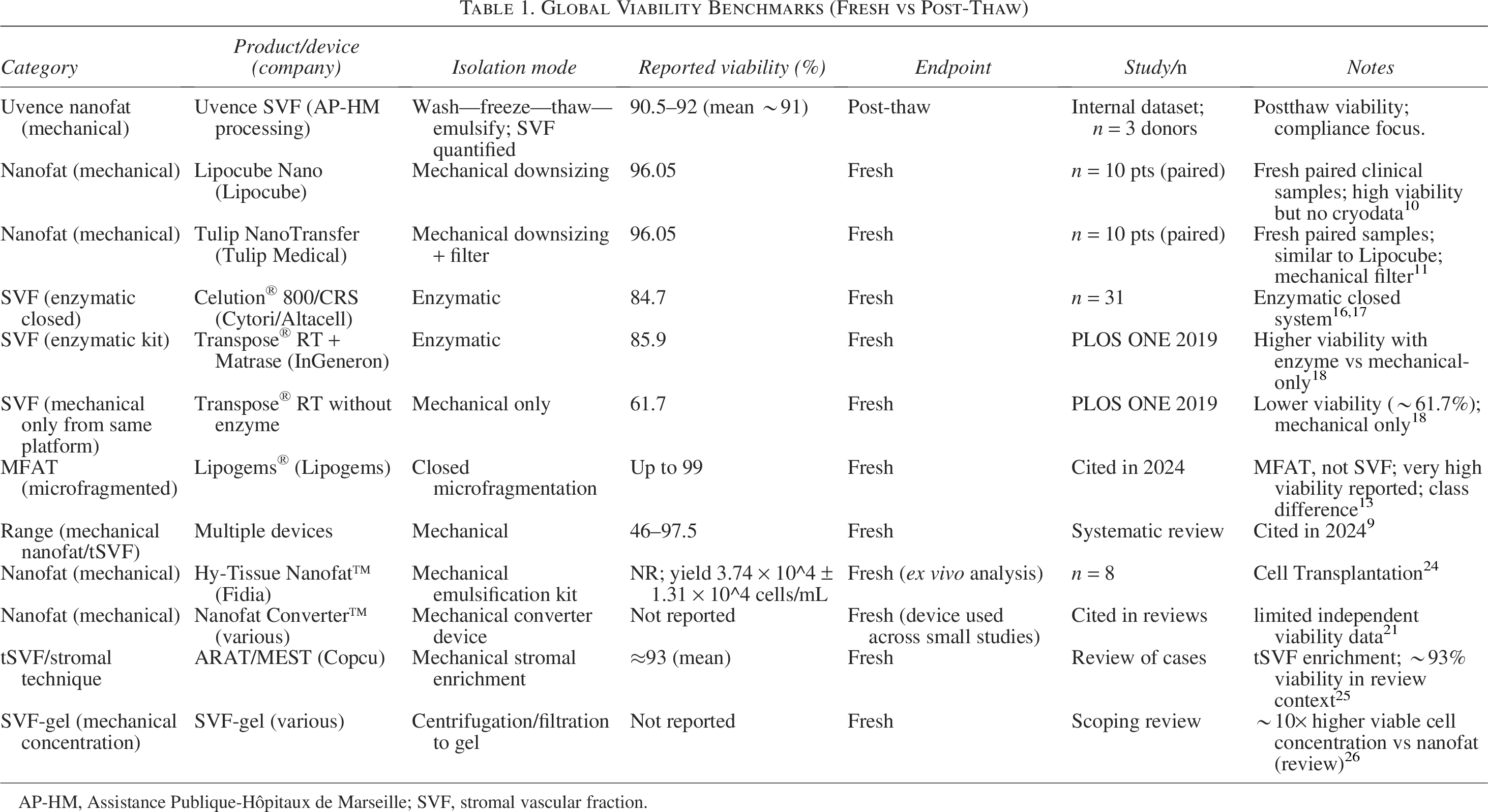
AP-HM, Assistance Publique-Hôpitaux de Marseille; SVF, stromal vascular fraction.
Methods
Tissue procurement and initial processing
Human adipose tissue was procured under Human Tissue Authority (HTA) License [22719] at the Uvence facility in accordance with donor consent, exclusively for research and validation purposes and not for therapeutic use. Adipose tissues were processed using the proprietary Uvence method. Although the workflow is proprietary as an integrated system, it follows a standardized sequence based on established adipose tissue handling principles.4,9,27 The validated workflow incorporates saline washing, controlled-rate freezing using CryoMACS bags, followed by cryopreservation, 28 standardized thawing conditions, and a defined mechanical shear-based emulsification step. Initial washing and cryopreservation steps were performed within the licensed Uvence facility.
Adipose tissue processing
Adipose tissue was initially collected using a closed-bag filtration system and washed using 0.9% sodium chloride employing gravity-driven filtration to remove blood residues, lipids, and other contaminants while maintaining tissue integrity. After washing, the microfat was transferred into sterile CryoMACS® freezing bags, specifically designed for cell and tissue cryopreservation.
Cryoprotection was achieved by mixing the microfat with equal parts of freezing solution (in the same CryoMACS bags) consisting of 80% 4% human albumin and 20% dimethyl sulfoxide (DMSO), a method consistent with recognized adipose-derived cell cryopreservation protocols.28,29 The CryoMACS bags were then sealed, frozen using a validated controlled-rate freezing method at −1°C/min until they reached −80°C, and subsequently stored at −150°C in a deep freezer. 30
All washing and cryopreservation activities were conducted within the Uvence laboratory using an ISO Class 5 biosafety cabinet (Grade A) and monitored according to EU GMP Annex 1 guidance. All processed samples subsequently passed bioburden testing.
All donor samples were cryopreserved for 3 to 4 months before thawing. Cryopreserved donor samples were shipped under temperature-controlled conditions to Assistance Publique-Hôpitaux de Marseille (AP-HM, France), a university research facility, for subsequent thawing, emulsification, and cell culture procedures under institutional oversight for nonclinical laboratory investigations.
Thawing and emulsification steps at AP-HM were performed following the Uvence protocol. Frozen CryoMACS bags were rapidly thawed in a dry bath at 37°C for 3–5 mins (agitation) until residual ice remained, after which thawing buffer (1:3, 5% human albumin: 0.9% NaCl) was added and the tissue was gently agitated. This washing cycle with thawing buffer was repeated twice, followed by a saline wash. The thawed microfat was transferred into a 10 mL syringe, then emulsified (mechanically sheared) using a proprietary device, for a total of 30 passes, to generate a nanofat or stromal vascular tissue product, in alignment with the principles described in contemporary mechanical SVF/nanofat literature.6,20,21
This integrated workflow, incorporating washing, closed or sealed bag-based cryopreservation, standardized thawing, and controlled mechanical emulsification, ensures consistency and traceability, minimizes contamination risk, and preserves stromal cell viability for downstream analysis.
Experiment
SVF isolation and viability quantification
For cell viability, Uvence nanofat samples were enzymatically digested by dilution 1:1 with clinical-grade Ringer’s lactate, preincubation at 37°C, followed by NB4 collagenase (0.25 Wünsch units/mL) for 45 min at 37°C with agitation. These conditions mirror established SVF isolation protocols reporting >80% viability.31–33 Digestion was stopped with 0.9% NaCl + 5% human serum albumin (HSA; Octopharma Ltd), centrifuged at 400 g for 5 min, and the pellet resuspended in 0.9% NaCl + 5% HSA. Viability was assessed using the NucleoCounter® NC-202™ (Chemometec).
The Uvence workflow itself is entirely mechanical. Collagenase digestion was used only for laboratory viability quantification and MSC culture isolation, as it remains the standard method for assessing viable nucleated cells (VNCs) and isolating adherent stromal cells for in vitro assays. 33
Cell culture and cryostorage
For the three donor samples, 0.2–0.4 × 106 VNCs were seeded into Corning T25 cm2 flasks and maintained in a humidified incubator at 37°C and 5% CO2. Cells were cultured in Dulbecco’s Modified Eagle’s Medium/Nutrient Mixture F-12 supplemented with 10% fetal bovine serum (FBS) and an antibiotic solution.32–34 Media were changed three times per week. Upon reaching confluence, adherent cells were enzymatically detached using trypsin-EDTA (passage 1) and expanded into T75 cm2 flasks for further growth. At the second confluence, cells were again detached (passage 2) and cryopreserved in 90% FBS and 10% DMSO, using a controlled-rate freezing process, and stored in the vapor phase of liquid nitrogen. This protocol allowed for consistent expansion and preservation of adipose-derived stromal cells.
MSC morphology was confirmed using an EVOS M5000 microscope (3,150 µm field width). The MSC-cultured cryovials were then shipped under temperature-controlled conditions to Alcyomics Ltd. (Newcastle upon Tyne, UK) for MSC characterization.
MSC characterization strategy
Characterization was performed independently by Alcyomics Ltd., using an in-house, validated study protocol on the three donor samples. Cryopreserved MSC vials were thawed and allowed to recover for 48 h in adherent culture prior to any downstream analysis. Characterization included morphological assessment, flow cytometry (FACS) immunophenotyping, and trilineage differentiation assays.
Morphology analysis
Postthaw MSCs were seeded at a density of 2 × 105 cells per well in 12-well plates. Phase contrast microscopy was used at 24 h and 48 h to assess cellular morphology. Cells demonstrated a fibroblastic, spindle-shaped morphology consistent with mesenchymal stromal cell phenotype, forming adherent monolayers with colony formation, as described in previous literature.35–37
Flow cytometry
Data acquisition and analysis were performed in SpectroFlo (Cytek Biosciences). Cells were collected using TrypLE Select (a gentler enzyme designed to preserve surface markers), washed in PBS, and stained with a validated panel of fluorochrome-conjugated monoclonal antibodies against CD73, CD90, CD105, CD34, and CD45 (BioLegend). Staining included viability exclusion using LIVE/DEAD™ Red dye. Analysis was conducted on a Cytek Aurora full-spectrum flow cytometer, with gating performed on single, live cells using FSC-A × FSC-H and Live/Dead exclusion. A total of >10,000 live singlet events were recorded per sample. The same antibody master mix and lot were used across all donor and control samples to maintain consistency. Gating strategy was optimized using single-stain and unstained controls. Isotype and single-stain controls were used to establish gates. Promocell MSCs were included as a control.
Trilineage differentiation assays
Cells were seeded into differentiation media at 2 × 105 per well for adipogenic and osteogenic assays and 9 × 105 cells in micromass culture for chondrogenic differentiation. Media and reagents were sourced from STEMCELL Technologies. Uninduced control wells were included for each lineage.38–41
Adipogenic differentiation
MSCs were cultured to 80–90% confluence, then induced with adipogenic medium (MSC Adipogenic Differentiation Medium 2). Cells were maintained for 14 days with medium changes every 3 days. Lipid droplet accumulation was visualized by Oil Red O staining after fixation with 10% formalin. Counterstaining was done using hematoxylin.
Osteogenic differentiation
MSCs at ∼100% confluence were induced using Osteogenic Differentiation Medium 2 for 13 days with medium changes every 3 days. Calcium deposition was detected by Alizarin Red S staining. Cells were fixed in 10% formalin and counterstained with hematoxylin.
Chondrogenic differentiation
Spheroids were formed in AggreWell™800 plates and cultured in chondrogenic differentiation medium for 21 days, with media changes every 2 to 3 days. Spheroids were harvested, fixed in 4% PFA, and embedded in low-melt agarose. Paraffin sections (3 µm) were stained with Alcian Blue and Hematoxylin or H&E for glycosaminoglycan detection and general histology, respectively.
Experimental Results
Cell viability
Across three donors, postthaw SVF viability ranged 90.5–92% (mean ∼91.1%), with VNC yields of 1.8–2.6 × 106 (Table 2). All three donors achieved more than 90% viability, indicating methodological consistency. These findings are consistent with prior systematic reviews and comparative reports describing variability in viability outcomes across enzymatic and mechanical methods.9–12,16–18 All donor samples were cryopreserved for 3 to 4 months before thawing; no differences in viability were observed across this interval, and postthaw viability remained consistent (90.5–92%). However, long-term stability beyond this duration was not evaluated and represents an area for future study.
Uvence Donor Results (AP-HM Viability)
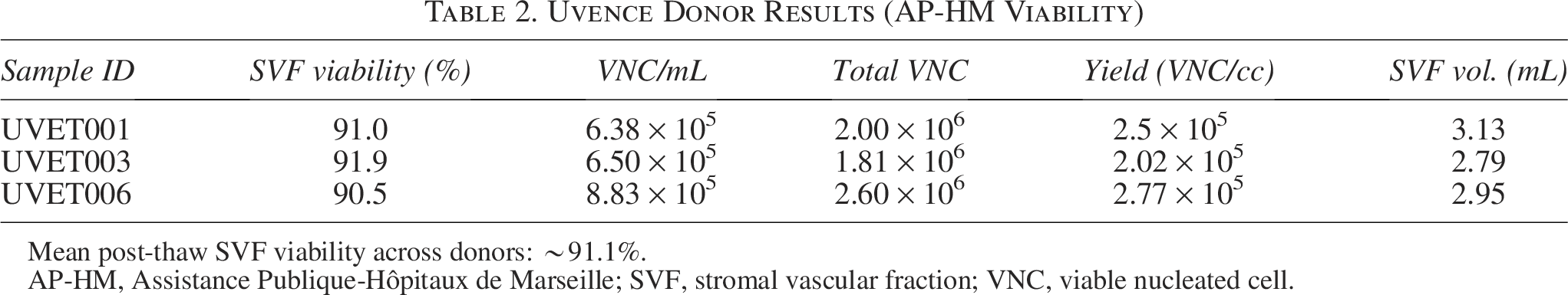
Mean post-thaw SVF viability across donors: ∼91.1%.
AP-HM, Assistance Publique-Hôpitaux de Marseille; SVF, stromal vascular fraction; VNC, viable nucleated cell.
Microscopy analysis
The MSC morphology was confirmed using an EVOS M5000 microscope (3,150 µm field width) following the cell culture by AP-HM. Representative phase-contrast microscopy images (EVOS M5000, ThermoFisher Scientific) of cultured MSCs at passage 2 (P2) clearly illustrate typical MSC morphology, characterized by elongated, spindle-shaped, adherent cells forming confluent monolayers. Images were captured at a magnification of ×4, with a visual field length of 3,150 µm, demonstrating consistent morphology (Fig. 1) across three donor samples. MSCs were also imaged at 24 h and 48 h (Figs. 2 and 3) postthaw by Alcyomics Ltd., where morphology was evaluated based on spindle shape, adherence, and colony formation, in line with previous reports.35–37

Representative MSC morphology at P2 from donors [UVET001 (Donor 1), UVET003 (Donor 2), and UVET006 (Donor 3)]. MSC, mesenchymal stromal/stem cell.
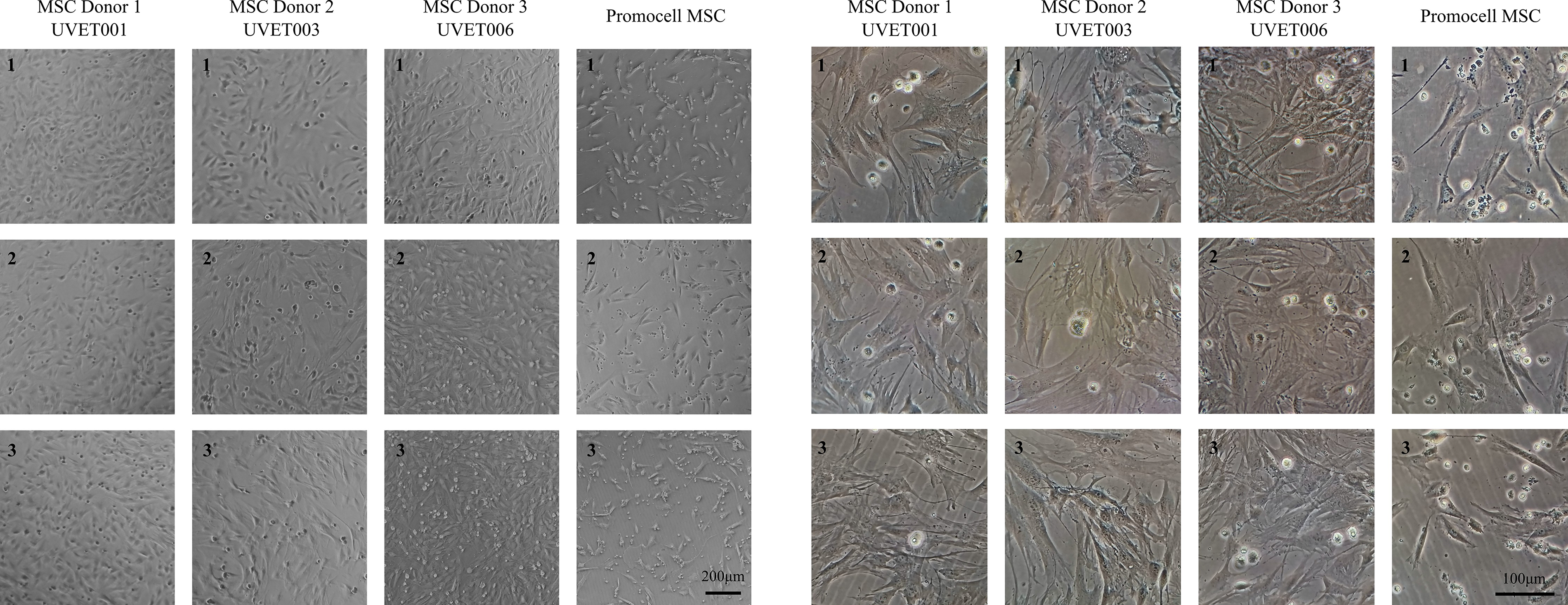
Morphology 24 h postthaw at two magnifications, 120× (left) and 600× (right), illustrating spindle-shaped, adherent cell morphology typical of MSCs in all three Uvence samples. MSC, mesenchymal stromal/stem cell.
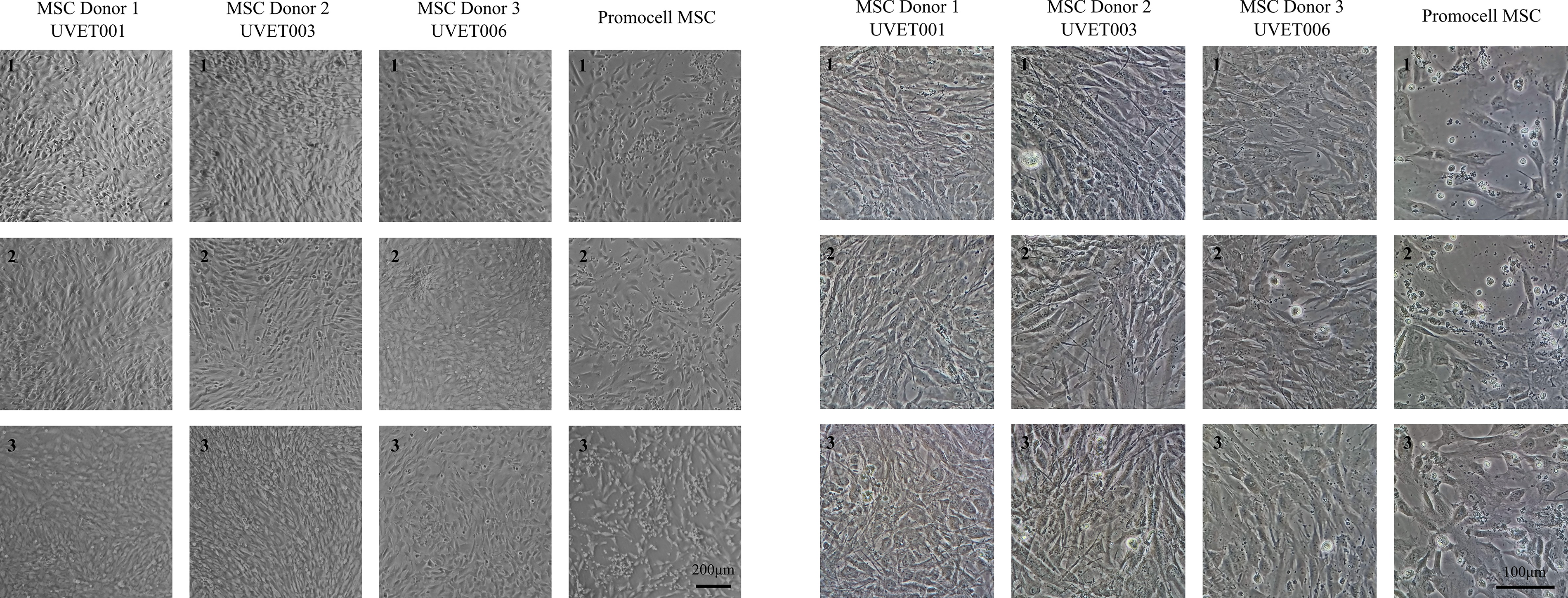
Morphology after 48 h of culture at two magnifications, 120× (left) and 600× (right), showing further adherence and colony expansion characteristic of healthy MSCs in all three Uvence samples. MSC, mesenchymal stromal/stem cell.
Flow cytometry
The three donor-derived MSC populations were assessed by FACS, and cells were confirmed to have an immunophenotypic profile consistent with ISCT MSC criteria in vitro, showing robust expression of CD105, CD73, and CD90, with absence of CD34 and CD452,13 (Table 3 and Figs. 4, 5). The observed viability across donor samples ranged from 79.9% to 85.1%. Triple-positive (CD73+CD90+CD105+) populations were 87.1% (Donor 1), 95% (Donor 2), and 88.2% (Donor 3), compared with 91.7% for Promocell MSCs. CD34−CD45− identity exceeded 99% across all samples, and CD34−CD45−CD73+CD90+CD105+ ranged 86.3–94.4%.
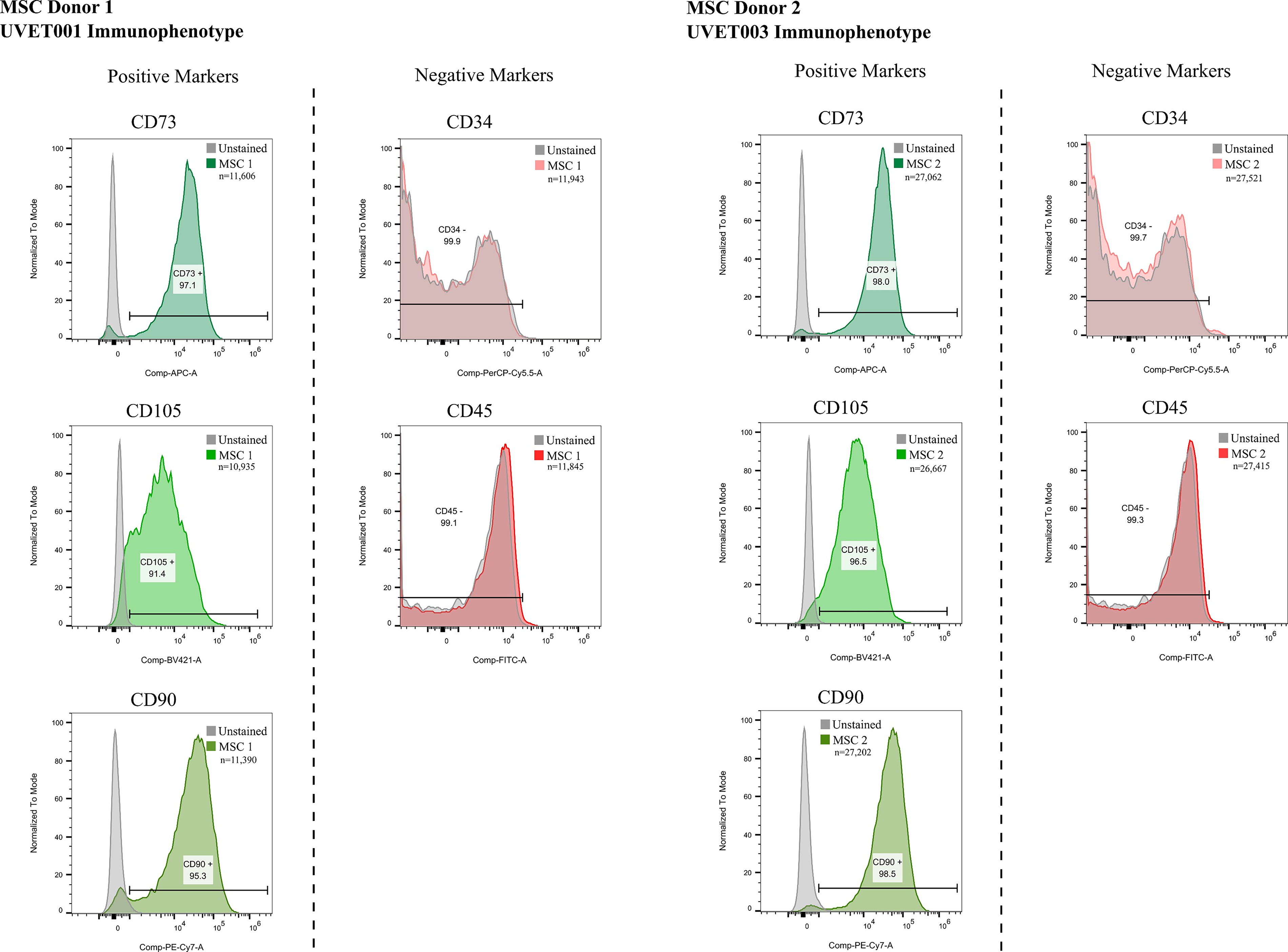
FACS Histograms of MSC Immunophenotypic Markers for Uvence Donors 1 and 2. FACS, flow cytometry; MSC, mesenchymal stromal/stem cell.
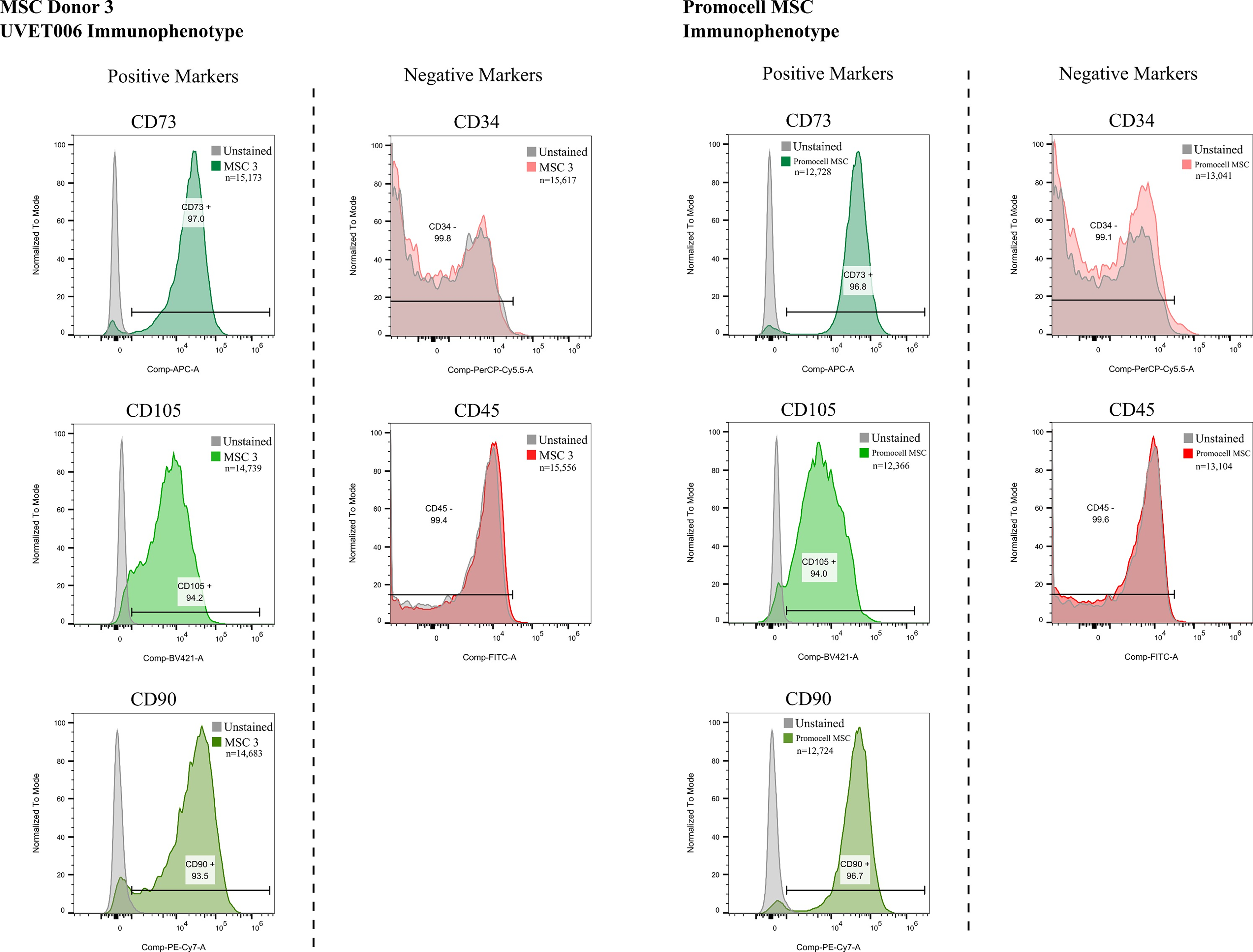
FACS Histograms of MSC Immunophenotypic Markers for Uvence Donors 3 and Promocell MSC control. FACS, flow cytometry; MSC, mesenchymal stromal/stem cell.
Immunophenotype—Percentage of Live Single Cells
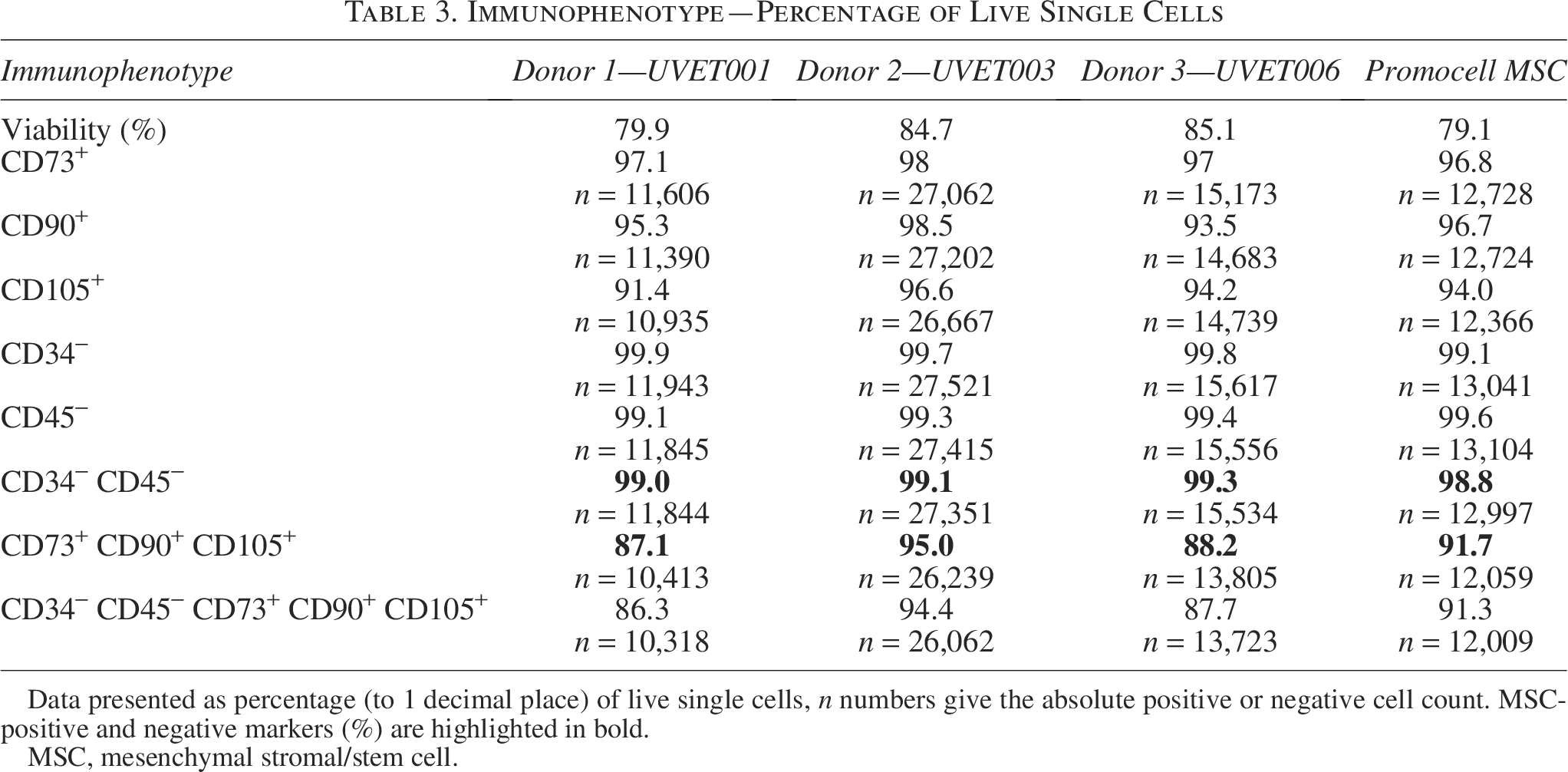
Data presented as percentage (to 1 decimal place) of live single cells, n numbers give the absolute positive or negative cell count. MSC-positive and negative markers (%) are highlighted in bold.
MSC, mesenchymal stromal/stem cell.
The histograms (Figs. 4, 5) show unstained and stained modal cell counts for MSC-positive markers—CD73, CD105, and CD90, and negative markers—CD34 and CD45. Positive and negative gates are indicated and marked by the black bracketed line. The percentage of live single cells (number below immunophenotype) and absolute cell counts (n) are included in the images.
Adipogenic differentiation
Following 14 days in adipogenic differentiation media, all donor and control MSCs differentiated into adipocytes. A clear presence of internal lipid droplets within the cells grown in differentiation media was detected by Oil Red O staining (Fig. 6), consistent with adipogenic differentiation reports in the literature.38–41 The Oil Red O staining of Donor 1 is comparable with that of Promocell MSCs but shows fewer extensive lipid droplets than donors 2 and 3.
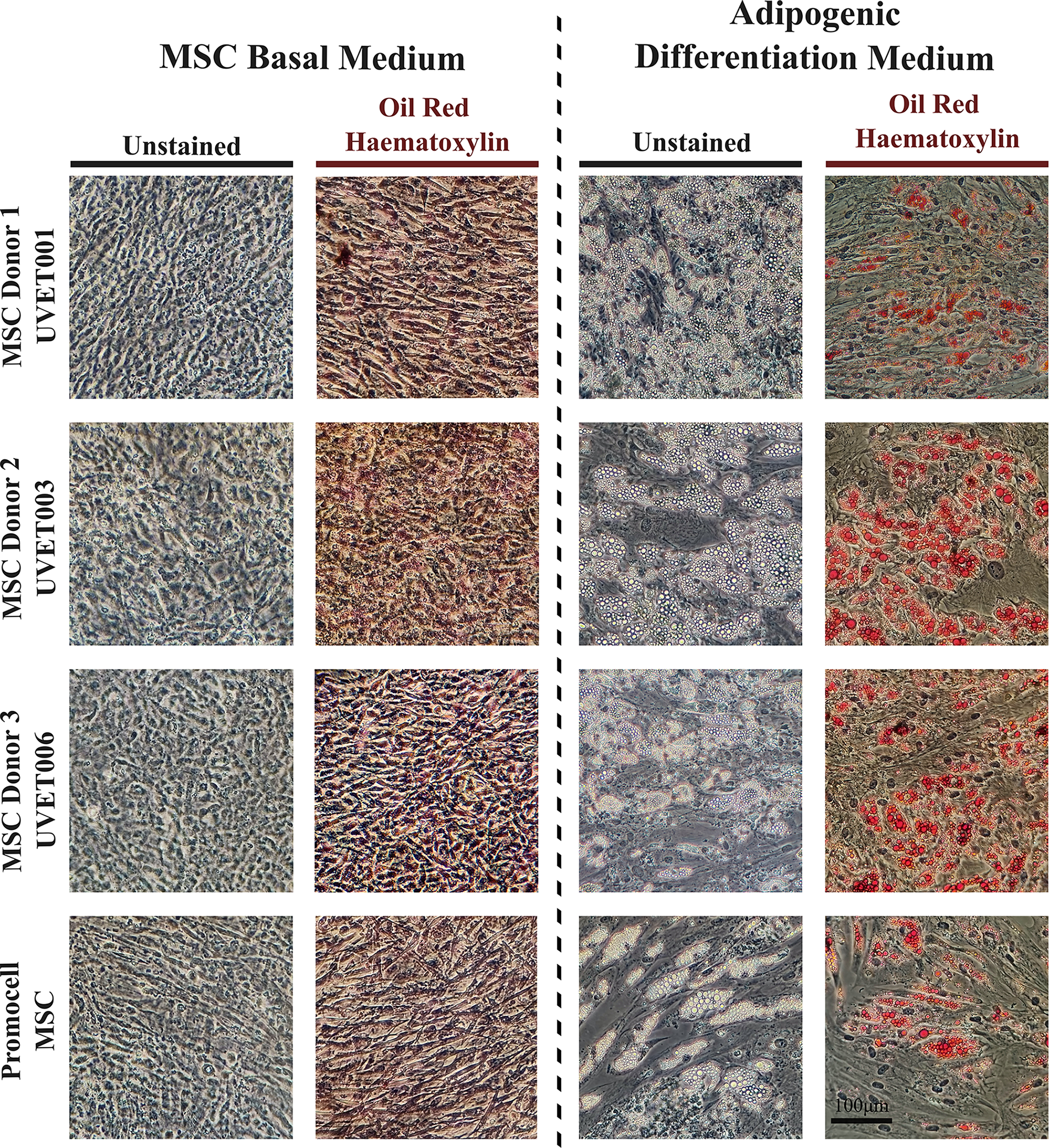
Adipogenic Differentiation of Uvence Donors with Oil Red O stain (Duplicate Oil Red O/Hematoxylin stained and unstained wells of cells grown in both basal medium and adipogenic medium. Images taken at 600× magnification).
Osteogenic differentiation
After 13 days in osteogenic differentiation medium, all donor and control MSCs differentiated into osteocytes. Calcium deposition was detected using Alizarin Red S staining (Fig. 7). Donor 1 showed more condensed osteogenic sheets, producing intense staining, in line with the variability described in previous osteogenic differentiation studies.38–41 In UVET001, the dense appearance is due to delamination of the osteogenic cell sheet during extended culture. This led to contraction and condensation of the cell sheet, resulting in reduced visibility of individual cell morphology in the final image.

Osteogenic Differentiation of Uvence Donors with Alizarin Red stain (Duplicate Alizarin Red/Hematoxylin-stained and unstained wells of cells grown in both medium and osteogenic medium. Images taken at 600× magnification. The cell morphology of UVET001 in osteogenic medium is difficult to observe due to cell sheet delamination and extended culture time. The cells have condensed, making it challenging to see their morphology.
Chondrogenic differentiation
MSC spheroids from all Uvence donors and the Promocell control were initially cultured in basal media before chondrogenic differentiation was induced after 48 h. Spheroids in basal media became adherent between days 3–9 and diminished in size and number, while those in differentiation media remained in suspension and grew consistently within each donor group. Donor 2 exhibited the largest spheroids (∼600 µm) and signs of spheroid fusion by day 14. Upon fixation and Alcian blue staining, which marks glycosaminoglycans in chondrocytes, differentiated spheroids from Donor 1 (∼300 µm), Donor 2 (large and fused), and Donor 3 (∼500 µm) stained consistently (Fig. 8), confirming chondrogenesis as described in established MSC characterization protocols.38–41 In contrast, undifferentiated spheroids were smaller, poorly structured, and lacked Alcian staining. Promocell MSC spheroids were smaller than Uvence donors but showed positive staining (Fig. 8). No spheroids persisted in the undifferentiated condition for Donor 1 and Promocell controls. Figure 8 shows Alcian blue and hematoxylin staining of chondrocyte spheroids, with matched nonsequential sections stained with hematoxylin and eosin. Each image pair shows a different spheroid. Images taken at 200× magnification; 250 µm scale bar in the bottom-right frame. Donor 1 and Promocell MSCs did not form spheroids in basal D10 medium, and therefore, no interpretable images could be generated.
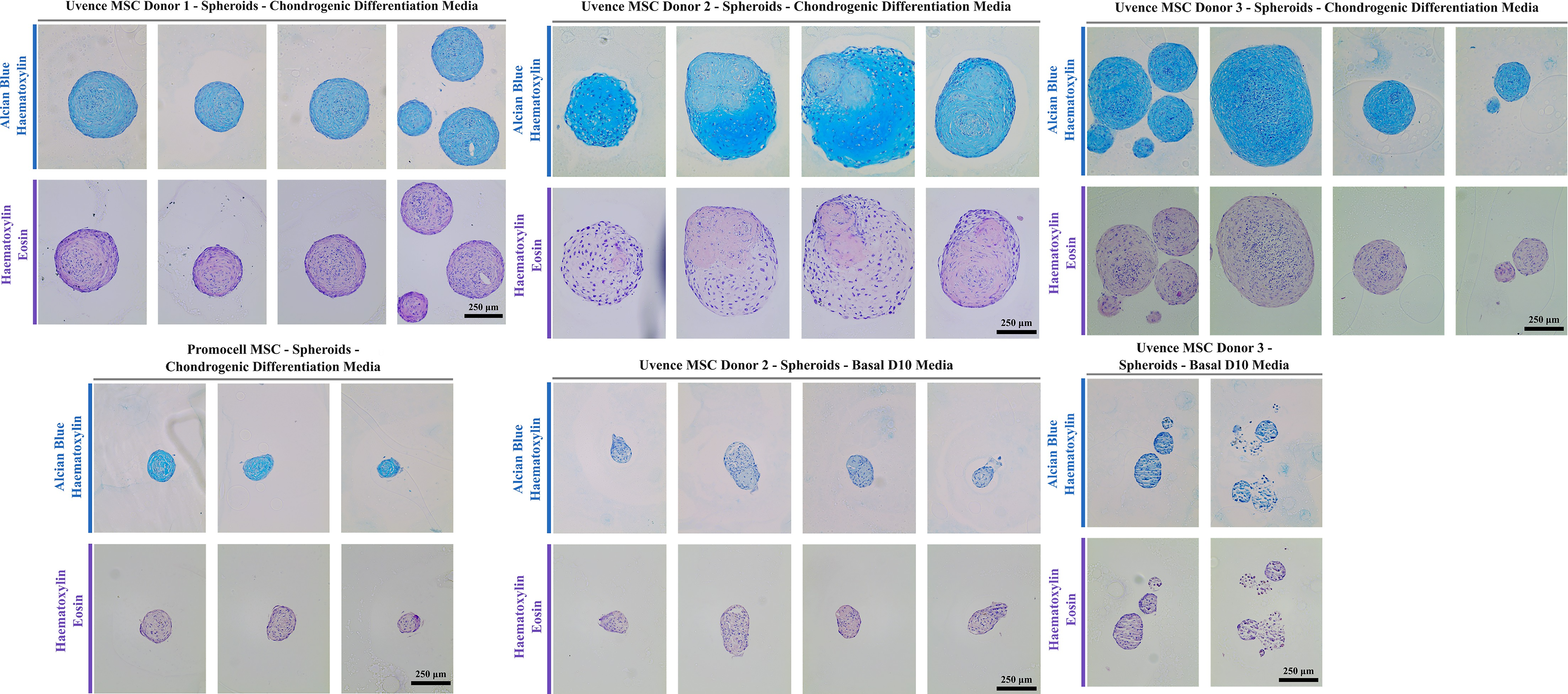
Uvence Donor 1, 2, and 3 and Promocell MSC chondrocyte spheroids (Donor 1 and Promocell MSCs did not form spheroids in basal D10 medium, and therefore, no interpretable images could be generated). MSC, mesenchymal stromal/stem cell.
Discussion
Uvence postthaw SVF viability aligns with or exceeds many enzymatic SVF preparations reported in the literature,16–18 despite the more stringent postthaw endpoint. Even compared with fresh competitor systems such as Lipocube, Tulip, and Celution, which report ∼90% to 96% fresh viability,10,11,16,17 Uvence postthaw viability remains highly competitive. Across the field, the IFATS/ISCT consensus suggests that ≥70% viability is a commonly cited threshold for SVF release, 13 which Uvence consistently meets under HTA-regulated workflows.
All samples in this study were cryopreserved for 3 to 4 months before thawing. Published cryobiology data show that once SVF or adipose-derived stromal cells are stored below the glass-transition temperature (≤–135°C), long-term storage has minimal impact on cell viability or phenotype, as most cryodamage occurs during freezing and thawing rather than during storage.42–45 Long-term MSC studies support this principle, with adipose-derived and other MSC types showing preserved viability, identity and function after 1–10 years of cryostorage.46–49 Therefore, the 3 to 4-month storage period used in this study falls well within the range demonstrated to maintain stromal cell integrity, and comparable postthaw viabilities would also be expected with more extended storage under stable cryogenic conditions.
These findings are restricted to in vitro research characterization and do not imply regulatory approval or therapeutic use under HTA frameworks. Numerous reviews have examined fat-grafting methodologies, delivery routes, and regenerative applications,50–52 while additional analyses have evaluated translational aspects of mesenchymal stromal-cell–based interventions.53,54 Plastic and reconstructive outcomes, including wound healing and burn reconstruction, have been extensively reported using adipose-derived cell therapies.55–57
The consistency in MSC phenotype and differentiation capacity observed across the three donor samples supports the stable performance of the validated Uvence method for nanofat processing and cell isolation. The processing pipeline utilized in this study followed rigorously validated laboratory protocols designed to preserve cellular viability and phenotype. While the experimental outcomes fulfill the ISCT-defined criteria for MSC characterization,2,13 procedural refinements could further optimize marker-expression uniformity and yield.
Overall, the study demonstrates that cryopreserved, mechanically processed adipose tissue can serve as a reliable source of viable stromal cells for in vitro characterization and method development. These findings align with current ISCT standards2,3,13 and recent reviews highlighting the translational potential of adipose-derived stromal cells in regenerative and reconstructive medicine.54–58
A limitation of this study is that in-depth MSC characterization was performed on three donor samples. While this reflects a common approach in methodological optimization studies, the results should be interpreted as demonstrating technical feasibility rather than population-wide biological reproducibility. Donor-to-donor variability is well recognized in adipose-derived cell research, and future work with larger cohorts may help further contextualize these findings.
Conclusion
Adipose tissue processed via the Uvence method can yield SVF suitable for MSC culture and phenotypic characterization. When performed under controlled and validated conditions, this workflow consistently produces viable stromal populations meeting ISCT/IFATS in vitro standards. The findings highlight the potential of Uvence-processed adipose tissue as a standardized source for isolating and expanding MSCs from nanofat-derived SVF for research and analytical applications. The process provides a framework for future studies evaluating process consistency, viability, and functional comparability across donors. Continued optimization of isolation, culture, and marker-preservation techniques will further enhance the robustness of this workflow for downstream in vitro applications. These findings are limited to in vitro research characterization and do not imply regulatory authorization or therapeutic use within HTA frameworks.
Process advantages of the Uvence workflow
Most marketed nanofat or SVF systems report fresh cell viability immediately after processing. Uvence demonstrates ∼91.1% postthaw viability following cryopreservation, which is equivalent to or better than that of leading fresh nanofat and enzymatic SVF devices. Unlike other marketed platforms, the Uvence workflow ensures cryopreserved stability, providing a consistent, high-quality starting material for future use. Within the Uvence framework, adipose tissue harvested at the clinic is transported under controlled conditions to the licensed Uvence laboratory for processing and cryostorage. Prepared Nanofat can be returned to clinics on demand for subsequent use. This controlled, traceable workflow operates under HTA oversight and GMP Annexe 1–aligned environmental monitoring and independent bioburden testing, ensuring tissue integrity, consistency, and compliance.
Ethical Approval
Work was conducted under an HTA license [no. 22719]; donor consent allowed research/characterization only, excluding human application.
Authors’ Contributions
A.A.K. and O.D.A. conceived the study, coordinated the experimental design and tissue processing, and prepared the article. O.D.A. also contributed to tissue harvesting and data interpretation. S.R.M. assisted with data validation and article review. C.D. and J.M. (AP-HM) performed SVF processing, cell culture, and viability testing. J.P., A.I., and A.M.D. (Alcyomics) conducted the MSC characterization work and verified related data. All authors reviewed and approved the sections relevant to their contribution and consented to the submission of the final article.
Footnotes
Acknowledgments
The authors thank the technical staff who assisted with sample handling and data collection and acknowledge the donors for providing tissue for research purposes.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
