Abstract
Background
Transcatheter closure of patent ductus arteriosus (PDA) in small, premature infants is increasingly performed in the contemporary practice. Data regarding optimal timing of PDA closure are scarce. We therefore decided to compare morbidity and mortality of early and late device closure in preterm infants.
Methods
Hospitalizations of 166,583 newborn infants diagnosed with PDA were identified from the Kids’ Inpatient Database (KID) in 2009, 2012, 2016, and 2019. Of these, 299 preterm infants (gestational age (GA) ≤32 weeks, birth weight <1500 grams) underwent percutaneous PDA closure. Characteristics and periprocedural outcomes were compared between early (≤30 days) (n = 120) and late PDA closure (>30 days) (n = 179).
Results
Mean age (±standard deviation) for early PDA closure was 18.6 days ± 7 and 67.3 days ± 36.5 in late PDA closure. Comorbidities including respiratory distress syndrome/bronchopulmonary dysplasia, heart failure, and pulmonary hypertension were comparable between the two groups. There was no difference in mortality or referral for surgical ligation between early and late closure. Postprocedural vascular complications were low, but higher in patients with early PDA closure (3.3% vs 0%; p = 0.025). While the total length of hospital stay was longer in late PDA closure group (121.1 days ± 51.7 vs 81.5 days ± 48.8; p = <0.01), postprocedural length of stay was paradoxically longer in the early closure group (69 days ± 43.6 vs 57.5 days ± 43, p = 0.03).
Conclusion
There was no statistical difference in in-hospital mortality; however, there is a trend toward higher mortality in the early closure group. Major periprocedural complications are comparable between early and late PDA closure, except vascular complications that were higher with early PDA closure.
Background
Patent ductus arteriosus (PDA) is a common congenital lesion in preterm infants, with an estimated incidence as high as 60%. 1 It is associated with significant morbidity including necrotizing enterocolitis (NEC), intraventricular hemorrhage (IVH), prolonged respiratory support, and even death.2,3 There is a strong association between prolonged PDA exposure and development of bronchopulmonary dysplasia (BPD) and subsequent pulmonary hypertension.4,5 Medical management or surgical ligation of a hemodynamically significant PDA (hsPDA) failed to demonstrate a decrease in these adverse events.6,7 The efficacy of pharmacological agents such as indomethacin, ibuprofen, and acetaminophen used for PDA constriction is controversial in premature infants.8,9 Surgical ligation is rarely performed in the contemporary practice due to potential periprocedural adverse outcomes, particularly respiratory complications.10–12 The emergence of transcatheter PDA device closure (TCPC) has become the standard management for larger children and adults, 13 and is increasingly performed in preterm neonates.12,14 The Amplatzer Piccolo™ Occluder (Abbott Structural Heart, Plymouth, MN) was approved by the Food and Drug Administration (FDA) in 2009 for closure of PDA in newborn infants ≥700 grams. Evidence demonstrated acceptable short- and mid-term outcomes of this device.12,15
In the premature infant population, the optimal timing of TCPC is controversial and data are lacking. Previously published reports advocate for earlier closure, as it is associated with better outcomes including shorter time on the ventilator and improved somatic growth.3,16–18 The Kids’ Inpatient Database (KID) provides an opportunity to examine characteristics and clinical outcomes of early (≤30 days ) and late (>30 days of age) TCPC in premature infants across United States centers.
Methods
Study design
A retrospective review of hospitalizations from KID was performed to identify preterm infants who were born ≤32 weeks’ gestation and their birth weight was less than 1500 grams. Of them, 299 patients underwent TCPC in 2009, 2012, 2016, and 2019. The data was obtained from these specified years. KID is a component of the Healthcare Cost and Utilization Project (HCUP) (https://hcup-us.ahrq.gov/kidoverview.jsp). It is the largest publicly all-payer inpatient care in pediatrics in the United States. The KID gathers data from approximately 1000 hospitals on the basis of size (small, medium, and large), region (Northeast, Midwest, South, and West), teaching status (nonteaching rural or urban vs nonteaching urban), which reflects the real world patient population in the US. It is structured to be released every 3 years and contains approximately three million pediatric discharges each year across US hospitals. The study was waived by the Institutional Review Board because the data are obtained from a deidentified national database. The International Classification of Diseases Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes were utilized to identify comorbidities, index procedure and periprocedural complications. The ICD-9 and ICD-10 are the discharge codes for diagnoses related to the entire hospitalization.
We used 30-day a threshold to identify early versus late transcatheter PDA for a number of reasons. First, the first month of life represents the transitional period in neonatal cardiopulmonary physiology as pulmonary vascular resistance drops in 4–6 weeks of life. Second, the first month of life is an important period in decision making regarding the strategies to close the PDA which include observation, pharmacological treatment and referral to cardiology for consideration of PDA closure. Last, using 30-day as a cutoff aligns with existing literature facilitating comparisons. 3
Statistical analysis
Statistical analyses were performed using IBM SPSS statistics version 28 (IBM Corporation, Armonk, NY). The patient sample was divided into two groups based on postnatal age (early TCPC ≤30 days and late TCPC >30 days). Continuous variables are reported as mean with standard deviation (SD), and t-test was used to compare continuous variables. Categorical parameters are presented as counts and percentages. Comparisons between categorical parameters were done using χ2 test, and Fisher’s exact test was performed when cell counts were ≤5. p-value less than 0.05 was deemed statistically significant.
Results
Demographics and baseline characteristics
Demographics and baseline characteristics of premature infants undergoing transcatheter patent ductus arteriosus closure.
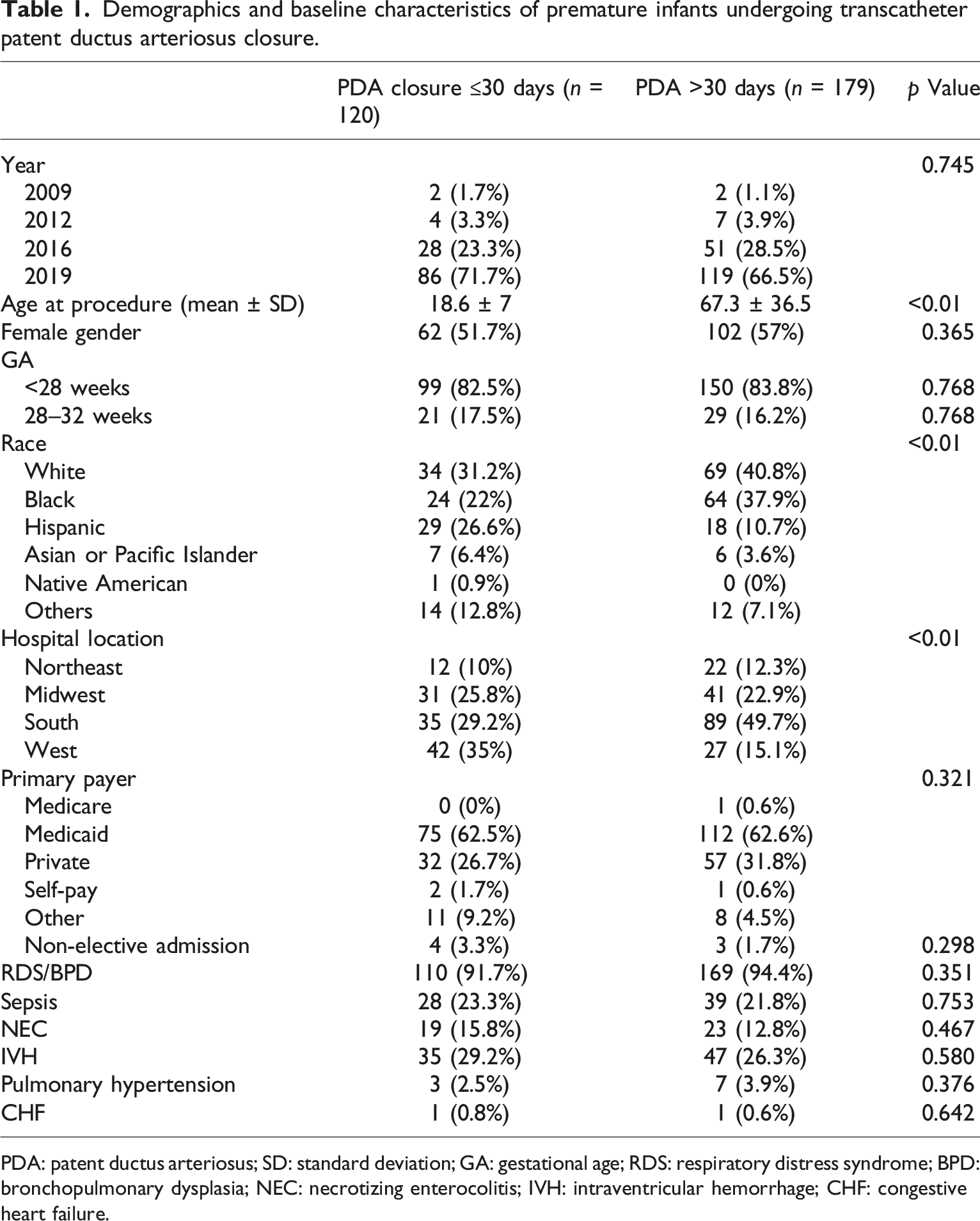
PDA: patent ductus arteriosus; SD: standard deviation; GA: gestational age; RDS: respiratory distress syndrome; BPD: bronchopulmonary dysplasia; NEC: necrotizing enterocolitis; IVH: intraventricular hemorrhage; CHF: congestive heart failure.
In-hospital complications and outcomes
In-hospital periprocedural outcomes of premature infants undergoing transcatheter patent ductus arteriosus closure.
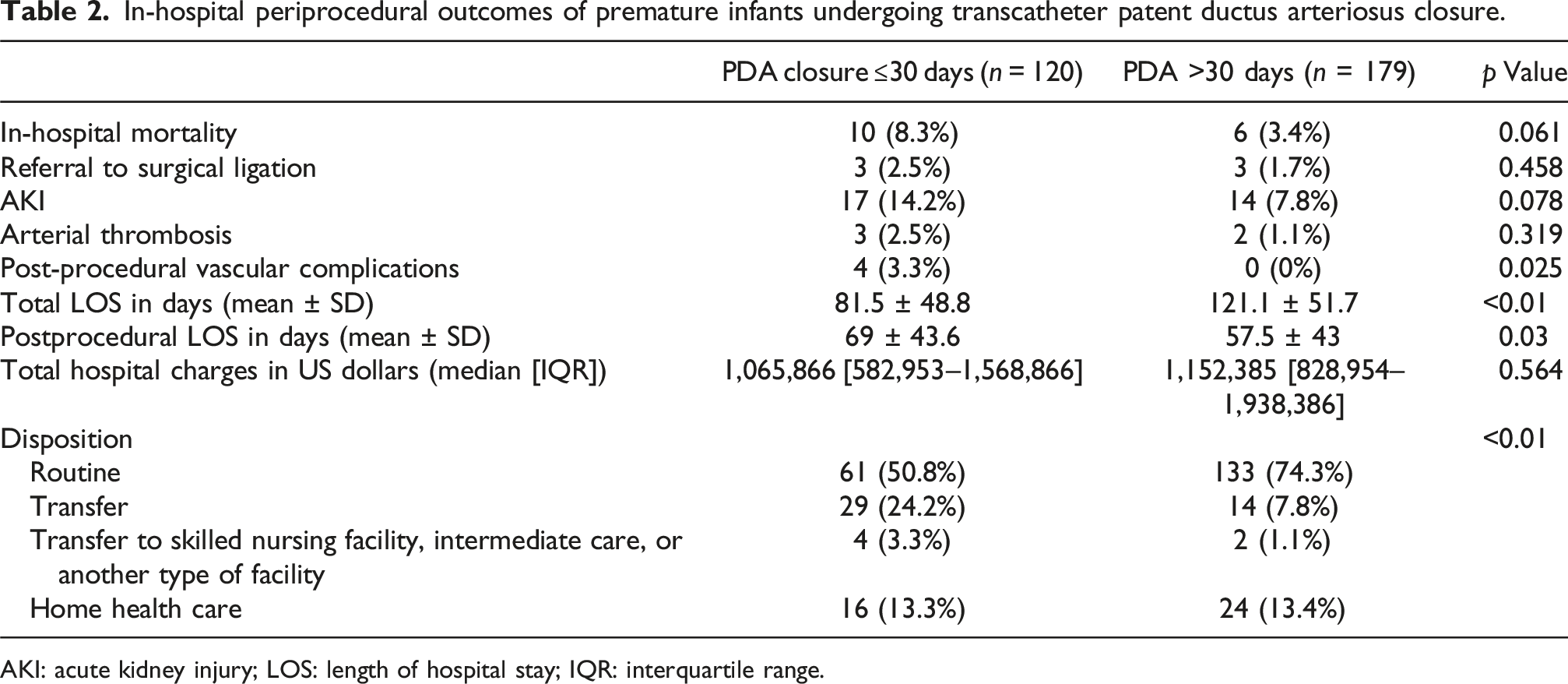
AKI: acute kidney injury; LOS: length of hospital stay; IQR: interquartile range.
Discussion
Our main findings
The main findings of our investigation are (1) TCPC in premature infants was increasingly performed in the most recent era with very high success rate. (2) In-hospital mortality and morbidity were acceptable following TCPC in premature infants. (3) Periprocedural complications were comparable between early and late TCPC.
Uncertainty exists regarding the optimal timing of TCPC, and the number of courses of pharmacological therapy before proceeding with TCPC. The decision regarding referral to TCPC among premature infants has been influenced by many factors. Consideration of early referral to TCPC has been affected by concerns that early referral may cause missed chances for effective NSAID (non-steroidal anti-inflammatory drugs) treatment or early spontaneous closure which is not uncommon this group. 19 Data from randomized clinical trials demonstrated no relationship between efficacy of NSAID and postnatal age. In the PDA-TOLERATE trial, the incidence of PDA closure following NSAID treatment was similar in early treatment group (mean age 8 ± 2 days) compared to late treatment group (mean age 21 ± 8 days). Consequently, there is no postnatal age cutoff at which medical treatment is considered ineffective. On the other hand, this clinical trial also demonstrated no significant reduction in PDA ligation or presence of a PDA at time of discharge in early routine medical treatment compared to those who received conservative treatment. Hence there is no postnatal cut off where medical management is deemed effective. 20 Nevertheless, among the high-risk subset of premature infants, such as infants born <26 weeks gestation with evidence of a hemodynamically significant PDA and resultant significant comorbidities, adoption of a conservative management approach will be questionable, and clinical instability may necessitate sooner definitive PDA closure.
When the decision is made to proceed with TCPC, it remains unclear if adverse events following TCPC is related to postnatal age.21,22 In the absence of direct comparisons, the choice of optimal timing for TCPC among premature infants remains unclear. Our study reveals there is no statistical difference in the majority of periprocedural complications and requirement of surgical ligation between early and late TCPC. Although the difference in in-hospital mortality between early versus late TCPC groups is not statistically significant, there is observed trend toward higher mortality in the early closure group. The observed trend may have clinical relevance and warrants cautious interpretation. Our study demonstrated shorter total hospitalization in the early closure group; however, postprocedural length of hospital stay was paradoxically longer in the early closure group. This finding can be potentially explained by higher patient acuity in the early closure group prompting early intervention but requiring longer recovery. Additionally, infants in the late closure group might have had more time to stabilize and become more mature, allowing for quicker postprocedural recovery. Unfortunately the administrative database we utilized did not include granular data regarding important outcomes such as weight at procedure, weaning of ventilatory support reaching full feeding or other illness severity indicators that may attenuate the difference or explain it better. Philip et al. 3 demonstrated that early closure of a hemodynamically significant PDA (first 4 weeks of life) may be beneficial before the onset of increased pulmonary vascular resistance in extremely low birth weight (ELBW) infants. They also found that early closure may allow for shorter ventilatory support and better weight gain. A recent meta-analysis performed by Yan and colleagues 23 investigated outcomes of premature infants who underwent early surgical PDA ligation compared to late surgical ligation. This meta-analysis concurred with our results and revealed no difference in mortality or postoperative adverse events; however, early ligation affords faster weaning ventilator support and oxygen support and better nutritional status. Our conclusions were consistent with previously reported studies.17,24 Our investigation demonstrates low rate of vascular complications (1.3%) which is similar to the incidence of non-serious cardiovascular injury in the premarket trial of Amplatzer Piccolo Occluder. 25 Literature reported that incidence of serious cardiovascular injury ranges from 0%–8%.26–31 The low rates of vascular complications in our cohort may be explained by the clinical practice adoption of antegrade transvenous approach only in this patient population and sparing arterial access solely for emergencies. In our cohort, all vascular injury occurred in the early TCPC group which may be due the relatively smaller vessel size or technical challenges in younger and lower weight infants. As expected in this fragile population, the overall hospital charges were significantly high across both early and late TCPC groups. These high charges likely reflect the complexity of care in this population, including prolonged NICU stays, respiratory support, specialized monitoring, several investigations, and requirement multiple interventions.
Surgical PDA ligation is associated with significant postoperative complications, especially in preterm infants.32,33 These complications include chylous effusion, bleeding, iatrogenic occlusion of left pulmonary artery, aorta, or left main stem bronchus.32,33 Additionally, left vocal cord paresis has been frequently reported in premature neonates.33–35
Given the variability that exists across centers in the early management of PDA in premature infants, prospective randomized clinical trials are required to compare the outcomes of the management modalities to help guide decision making regarding the optimal timing of TCPC in preterm infants.
Limitations
Our study has some limitations. First, KID is derived from hospital claims data without access to the medical records of the patients, hence there was no way to obtain important clinical information such as data regarding weight and definitive corrected gestational age at time of procedure, echocardiographic measurements, devices, and procedural details. Additionally, follow-up data beyond hospital discharge were not available. Second, the codes used for procedures and diagnoses may be subject to reporting errors. However, major procedures codes are unlikely to be inaccurate because they are the principal methods for the hospital re-imbursement. Due to the limitations of the administrative dataset and the way diagnoses are coded, we were unable to reliably distinguish between these conditions in all cases. BPD and RDS were combined in one variable as several patients had the two codes for these entities among their discharges’ codes and there is potential for diagnostic overlap. Furthermore, the published error rates of diagnoses and procedures codes in the 2003 nationwide inpatient sample database (a part of HCUP database) are as extremely low as 0.08% and 0.01% Despite the limitations, this study represents a comprehensive and contemporary outcomes analysis for neonates undergoing percutaneous PDA closure. It also reflects “real world” data from all type of centers in the US.
Conclusion
Percutaneous PDA device closure is safe and feasible with low periprocedural complications. There is no statistical difference in in-hospital mortality and morbidity between early and late TCPC, but there is observed trend toward higher mortality in the early closure group. Randomized clinical trial and post-discharge follow-up data are required to validate our results.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
