Abstract
Background
Migraine is a common primary headache characterized by paroxysmal features and associated symptoms that significantly disrupt patients’ daily lives, work, and social interactions, rendering it the second leading disease cause of disability worldwide. Toutongping granule (TTP) is a well-established compound prescription in Traditional Chinese Medicine (TCM) for the treatment of migraine. However, the underlying mechanisms remain unclear.
Objective
This study aims to investigate the multi-pathway mechanisms through which TTP exerts its effects on migraine.
Methods
The therapeutic effects of TTP on migraine were assessed through behavioral observations, Nissl staining, Iba-1 staining and immunofluorescence (IF) for reactive oxygen species (ROS). Additionally, enzyme-linked immunosorbent assay (ELISA) was employed to measure serum cytokines (TNF-α, IL-6 and CGRP). The mechanism of TTP was examined in a nitroglycerin-induced migraine rat model using IF (CGRP, RAMP1, AKT, mTOR, p38 MAPK, p-STAT1, NRF2 and GPX4), Western blotting (PKA, cAMP, and p-CREB), and qRT-PCR (miRNA382-5p, circNR3C1, TACR1, HSP60, and ATF4 mRNA) to evaluate neuroprotective, anti-inflammatory, and antioxidant effects.
Results
TTP reduces headache-like behaviors, diminishes mitigates neuronal damage, inhibits aberrant activation of microglia and ROS production, decreases pro-inflammatory cytokines (TNF-α, IL-6), and modulates key signaling proteins (CGRP, RAMP1, AKT, mTOR, p38 MAPK, p-STAT1, NRF2, GPX4, cAMP, PKA, p-CREB) along with genes (miRNA382-5p, circNR3C1, TACR1, HSP60, and ATF4 mRNA) associated with migraine.
Conclusion
TTP demonstrates multi-pathway and targets, exhibiting neuroprotective, anti-inflammatory, and antioxidant effects, thereby supporting its application in migraine treatment.
Introduction
Migraine is a common, recurrent neurovascular disorder, typically manifested as moderate to severe pulsatile headaches, frequently accompanied by nausea, vomiting, photophobia, phonophobia, and various autonomic nervous system dysfunction. The global prevalence of migraine is approximately 14%-15%. 1 The Global Burden of Disease Study (GBD) ranks migraine as the second leading disease causing disabilities worldwide, 2 especially among the working population aged 15-49. Furthermore, migraine increases the risk of other neurological disorders, including depression and anxiety. 3 Overall, the current global epidemiology of migraine underscores its prevalence and complexity, emphasizing its significant impact on physical and mental health, work productivity, and social interactions, as well as its extensive public health and economic ramifications, which require urgent attention.
Nonsteroidal anti-inflammatory drugs are the most commonly used medicine for migraine management. However, the use may cause gastrointestinal adverse reactions and drug resistance.4,5 Ergotamine has been withdrawn from use due to its cardiac and hepatic toxicity. Additionally, the long-term administration of preventive drugs may cause central or peripheral adverse effects. More importantly, these traditional preventive drugs are nonspecific medications for migraine. Calcitonin gene-related peptide (CGRP) is recognized as a primary target for migraine therapy. 6 The first class of migraine-targeted drugs was introduced in 2018, with Rimegepant being the first orally disintegrating tablet formulation of a CGRP receptor antagonist, approved for both the treatment and prevention of migraine. Numerous randomized controlled trials and real-world studies have confirmed that CGRP monoclonal antibodies can significantly decrease the number of migraine days per month in patients with both chronic and episodic migraine.7,8 Despite this revolutionary progress in migraine prevention and treatment, challenges remain, including unsatisfactory therapy success rates, economic burdens, long-term safety concerns, and the need for individualized treatment strategies.
The pathogenesis of migraine is highly intricate. The interplay among oxidative stress, neuronal damage, and inflammatory responses forms a complex network that facilitates both the onset and persistence of migraine. This complexity suggests that a single targeted medication is insufficient to address the multifaceted nature of the disease. MiRNAs, as a novel class of biological markers offer several advantages, including high sensitivity, stability, specificity, efficient delivery, and accessibility. Reports indicate that miR-382-5p levels are elevated in the blood of migraine sufferers during attacks, remaining higher than those in healthy individuals even during periods of pain relief.9,10 This finding positions miR-382-5p as a potential new biomarker and therapeutic target for migraine. CircNR3C1, as a miRNA sponge, sequestering significant amounts of miR-382-5p and preventing it from interacting with its targets. Up-regulation of miR-382-5p and down-regulation of CircNR3C1 have been observed in the hippocampus of stressed rats. 11 Most migraine studies have concentrated on direct symptoms relief and the use of CGRP inhibitors. However, these approaches carry side effects and a risk of recurrence. In contrast, existing studies on non-coding RNAs have elucidated the underlying regulatory mechanisms of gene expression, particularly focusing on epigenetic processes. These mechanisms may intervene in migraine pathology at its roots by modulating pathways associated with inflammation, oxidative stress and neuronal damage, etc. This exploration at the molecular level provides a novel perspective for personalized therapy, characterized by reduced potential side effects and the capacity to design precise treatments that target specific non-coding elements. In recent years, the strategy of combining CGRP targeted drugs with non coding RNA regulation has emerged as a potent approach to enhance the therapeutic efficacy or migraine. 12 MiRNA modulates the synthesis and release of the CGRP protein by binding to CALCA, the gene encoding CGRP, 13 thereby influencing the pain transmission process.14,15 Non-coding RNA is anticipated to synergistically reduce neuronal damage and improve treatment tolerance when used in conjunction with CGRP targeted drugs. 16
TTP is a well-established compound prescription of TCM, in China. It comprises Paeoniae Radix Alba, licorice, Viticis Fructus, Cicadae Periostracum, Concha Haliotidis, Bombyx Batryticatus, Cyathulae Radix, A. Dahurica (Fisch.) Benth. Et Hook, Radix Puerariae, Asari Radix Et Rhizoma, and Chuanxiong Rhizoma. Clinically utilized for over 20 years, TTP has garnered positive reception among patients. Randomized controlled trials (RCTs) indicate that the cure rate and total effective rate of TTP for migraine are 76.7% and 93.4%, respectively. 17 Early animal studies have demonstrated that TTP exhibits significant effects of analgesic, vascular permeability regulating, neuroprotective, reducing serum CGRP and inflammatory factors,18,19 highlighting its multi-level therapeutic effects. Although TTP has a solid clinical application and significant therapeutical effect, the specific mechanism of action remains unclear.
Multi-targeting, linkage, and dynamic regulation are the defining characteristics of TCM compound prescription. Accordingly, our study focused on CGRP, a core target in migraine, and comprehensively investigated the mechanism of TTP from multiple perspectives, including CircNR3C1, miR-382-5p, neuroprotective, oxidative stress, and inflammation in both blood and brain tissue. We anticipate that this study will provide substantial evidence and candidate drugs for new therapies aimed at migraine, offering novel insights and strategies in this area. Moreover, this research underscores the importance of systematic evaluation to guide both clinical practice and future research directions.20,21
Methodology
Drugs and Reagents
Nitroglycerin injection (Shandong Shenglu Pharmaceutical Co., Ltd, batch number: 2303181); TTP (The Second Hospital Affiliated to Liaoning University of Traditional Chinese Medicine, Lot No.: 230726); Rimegepant (Nurtec@, Lot No.: 5231395PA); ELISA Kits for Tumor Necrosis Factor (TNF)-α, Interleukin (IL)-6, and CGRP (Shanghai Yuanju Biotechnology Center, Lot No.: 202405); SGExcel FastSYBR Master Premix (Sangon Biotech@, batch number: JC19KA2350); miRNA fluorescent quantitative PCR kit (staining method) (Sangon Biotech@, lot number: K506KA 4049); microRNA Extraction Kit (Sangon Biotech@, lot number: J719KA 0200); CGRP antibody (Proteintech@, Lot No. 00011200); RAMP1 Antibody (Proteintech@, 00116158); AKT Antibody (Abways, AY0420); mTOR Antibody (Abways, CY5306); p38 MAPK Antibody (PTG, 14064-1-AP); p-STAT1 Antibody (Abways, CY6498); NRF2 Antibody (PTG, 16396-1-AP); GPX4 Antibody (PTG, 67763-1-IG); PKA antibody (Cell Signaling Technology, 47825); cAMP antibody (ABclonal, A1640); p-CREB antibody (Cell Signaling Technology, 9198 T).
Experimental Design
Eighty SPF healthy male Sprague-Dawley (SD) rats, weighing 180-220 g and aged 3 to 4 months, were obtained from Liaoning Changsheng Biotechnology Co., Ltd (license number: SCXK (Liao) 2020-0001). The rats were housed at a temperature of 20 °C to 26 °C and a relative humidity of 40% to 70%. They had unrestricted access to food and drinking water, and were maintained under a 12-h natural light/dark cycle.
Following a 3-day acclimatization period, the rats were randomly assigned to four groups based on body weight: natural control group (CG), model group (MG), positive control group (XY) and TTP treated group (PG), with 20 rats in each group. The interventions were as follows:
Rats received no additional treatment.
(untreated migraine rats) rats received subcutaneous injections of nitroglycerin (10 mg/kg) into the back of the head and neck once daily for 10 consecutive days. Frequent head-shaking, double forelimb scratching, or unilateral hind limb scratching were considered indicative of headache-like behaviors and headache attacks.
(Rimegepant treated) Rats were undergone the same procedures as MG, and administrated with Rimegepant at a dose of 6.75 mg/(kg•d).
(TTP treated) Rats were followed the same protocols as MG, and administrated with TTP at a dose of 5.4 g/(kg•d).
Animal dose = Human dose × Human-Animal equivalence coefficient. 22
The reporting of this study conforms to ARRIVE 2.0 guidelines. 23
Sampling
Following the final NTG injection, headache-like behaviors were recorded during two intervals: 0 to 30 min and 30 to 60 min. Sixty minutes post-administration, blood samples were collected from the abdominal aorta for serum separation. The serum levels of TNF-α, IL-6, and CGRP were quantified using ELISA. The entire brain was collected, and frozen sections were prepared for ROS detection. Neuronal morphology was evaluated through Nissl and Iba-1 staining after fixation in a 10% paraformaldehyde solution. The protein expression of CGRP, RAMP1, AKT, mTOR, p38 MAPK, p-STAT1, NRF2, and GPX4 was detected by IF staining. Brain tissue from trigeminal nucleus caudalis (TNC) region was collected and stored at −80°C. miRNA382-5p was quantified using qRT-PCR (tail method), with U6 serving as an internal reference. The mRNA levels of circNR3C1, TACR1, HSP60, and ATF4 were measured using β-Actin as an internal reference (Table 1). Protein expressions of PKA, cAMP and p-CREB were analyzed by Western blot (WB). Per group, 8 rats were used for Nissl and Iba-1 staining (n = 8), among which three underwent IF staining (n = 3); 6 rats were used for frozen sections of ROS (n = 6); 3 rats were used for WB (n = 3); 3 rats were used for qRT-PCR (n = 3); serum of 20 rats were used for ELISA (n = 20); 20 rats were tested for frequency of headache-like behaviors (n = 20).
qRT-PCR Primer Sequences.

Three sections per rats and six non overlapping fields, fixed in TNC region, were randomly selected for analysis, with single blind for operator, adhering to consistent randomization criteria. ImageJ was used for quantifying Mean Fluorescence Intensity (MFI). Images were acquired with identical exposure settings, dark background and systematic threshold. The quantification used to statistic analysis is the average across multiple fields/sections of per rats.
Statistical Analysis
Statistical analyses were conducted using SPSS 26.0 software. Data are presented as mean ± standard deviation. The Shapiro-Wilk test was used to verify the normality of the data, and Levene's Test was applied to evaluated the homogeneity of variance. The frequency of headache-like behaviors was analyzed using two-way ANOVA, whereas other comparisons were performed using One-way ANOVA. When the data met the criteria for homogeneity of variance, LSD post-hoc tests conducted; otherwise the Games-Howell method was applied. P value of less than 0.05 was deemed statistically significant.
Results
Effect of TTP on Headache-Like Behaviors in Rats
Scratching head with double forelimbs or unilateral hind limbs is recorded as a single occurrence. TTP delays and decreases the frequency of headache-like behaviors (Figure 1, Table 2).

Effect of TTP on the Frequency of Headache-like Behaviors. The natural control group (CG) exhibited only a few instances of headache-like behaviors. In contrast, the model group (MG) demonstrated a significant increase in these behaviors following NTG injection. Compared to the MG, the frequency of headache-like behaviors significantly devreased in both the positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) after NTG injection. Data are presented as means ± SD (n = 20). **P < 0.01 versus MG.
Frequency of Headache Like Behaviors (Mean ± SD).

Each group consisted of 20 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using two-way ANOVA. **P < 0.01 versus MG.
Effect of TTP on the State of Neurons in TNC Region of Rats
As illustrated in Figure 2, TTP has been shown to protect neurons and inhibit the aberrant activation of microglial.

Effect of TTP on Neuron Cells and Microglia in TNC Region of Rats. (A) Structure and morphology of neuron cells (representative images from Nissl staining). Neuron cells in natural control group (CG) exhibit a granular and plump appearance with clear identification; in contrast, neuron cells (blue-stained) in the model group (MG) displayed loss of nuclei, light cytoplasmic staining, significantly reduced Nissl bodies, unclear edges, and signs of atrophy (red arrow). Neuron cells in positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) showed clearer morphology, a gradual recovery, an increase in normal cells, and a decrease in damage. (B) Structure and morphology of microglia in TNC region (representative images from Iba-1 staining, with microglia brown staining). Compared to Natural control group (CG), the model group (MG) rats demonstrate a significant increase in positive expression of microglia in TNC region, along with cell body deformation, protrusions, and branching (red arrow). The positive expression of microglia and the number of abnormal cell bodies in positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) decreased significantly. Scale: 50 µm. 8 biological replicates per group, each consisting of 3 technical replicates.
The MFI of ROS in PG was reduced compared to MG. TTP also inhibited oxidative reactions in TNC region of migraine rats (Figure 3, Table 3).

Effect of TTP on the state of ROS in TNC Region of Rats. ROS is displayed in red, while the nucleus is shown in blue. Increased brightness indicates stronger fluorescence intensity and higher expression of ROS. Compared to natural control group (CG), the fluorescence intensity of ROS in the model group (MG) was significantly elevated. In contrast to the MG, ROS fluorescence intensity was markedly reduced in both positive control group (Rimegepant treated group) (XY) and TTP treated group (PG). Scale: 100 µm. 6 biological replicates per group, each consisting of 3 technical replicates.
MFI of ROS (Mean ± SD).

Each group consisted of 6 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.

Effect of TTP on CGRP and RAMP1 in TNC Region of Rats. Representative images of IF, show that CGRP was displayed in green, RAMP1 was displayed in red, and nucleus in blue. Increased brightness indicates stronger fluorescence intensity and higher protein expression. Compared to natural control group (CG), the fluorescence intensity of CGRP and RAMP1 in the model group (MG) was significantly enhanced. In contrast, the fluorescence intensity in both positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) was significantly reduced compared to the MG. Scale: 100 µm. 3 biological replicates per group, each consisting of 3 technical replicates.
Effect of TTP on Serum Levels of TNF-α, IL-6 and CGRP in Rats
TNF-α, IL-6 and CGRP are widely recognized blood biomarkers for migraine. As shown in Table 4, serum levels of TNF-α, IL-6 and CGRP were significantly elevated (P < 0.01) in MG, compared to CG. TTP treatment reduced the inflammatory factors TNF-α, IL-6 and the common migraine biomarker CGRP in serum. The therapeutic effect of TTP is associated with its anti-inflammatory properties and its impact on CGRP.
Serum Content of TNF-α, IL-6 and CGRP (Mean ± SD).

Each group consisted of 20 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.
MFI of CGRP and RAMP1 (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.
MFI of AKT and mTOR (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.
MFI of p38 MAPK and p-STAT1 (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed by One-way ANOVA. **P < 0.01 versus MG.
Effect of TTP on CGRP, RAMP1, AKT, mTOR, p38 MAPK, p-STAT1, NRF2 and GPX4 Protein Expression in TNC Region of Rats
In the TNC region of MG, levels of CGRP and its receptor RAMP1 were significantly elevated (P < 0.01). Additionally, the expression of AKT, mTOR, p38 MAPK and p-STAT1, which are associated with repair and inflammation, was markedly increased. TTP treatment adjusted those expressions, resulting in a reduction of CGRP release and its biological effects, as well as inflammatory responses (P < 0.01) (Figures 4–6, Tables 5–7). Due to oxidative stress, NRF2 was activated and shifted to the nucleus to perform its antioxidant functions, while GPX4 was consumed in large quantities in the brains of migraine rats (P < 0.01) (Table 8, Figure 7). TTP effectively mitigated the oxidative stress chain reaction (P < 0.01).

Effect of TTP on AKT and mTOR in TNC Region of Rats. Representative images of IF, show AKT in green, mTOR in red, and the nucleus in blue. Increased brightness indicates higher fluorescence intensity and greater proteins expression. Compared to natural control group (CG), the fluorescence intensity of AKT and mTOR in the model group (MG) was significantly enhanced. In contrast, the fluorescence intensity in both positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) is significantly reduced compared to the MG. Scale: 100 µm. 3 biological replicates per group, each consisting of 3 technical replicates.

Effect of TTP on p38 MAPK and p-STAT1 in TNC Region of Rats. Representative images of IF show p38 MAPK in green, p-STAT1 in red, and nucleus in blue. Increased brightness indicates higher fluorescence intensity and greater proteins expression. Compared to natural control group (CG), the fluorescence intensity of p38 MAPK and p-STAT1 in the model group (MG) was significantly enhanced. In contrast, the fluorescence intensity in both positive control group (Rimegepant treated group) (XY) and TTP treated group (PG) is significantly reduced compared to the MG. Scale: 100 µm. 3 biological replicates per group, each consisting of 3 technical replicates.

Effect of TTP on NRF2 and GPX4 in TNC region of rats. Representative images of IF show NRF2 in green, GPX4 in red, and nucleus in blue. Increased brightness indicates higher fluorescence intensity and greater proteins expression. Compared to natural control group (CG), the fluorescence intensity of NRF2 in the model group (MG) was significantly enhanced, while that of GPX4 was decreased. In both the positive control group (Rimegepant treated group) (XY) and TTP treated group (PG), fluorescence intensity was significantly reduced compared to MG, with GPX4 exhibiting the opposite trend. Scale: 100 µm. 3 biological replicates per group, each consisting of 3 technical replicates.
MFI of NRF2 and GPX4 (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.
Effects of TTP on the Expression of PKA, cAMP, and p-CREB Proteins in the TNC Region of Rats
Our study displayed that, in migraine rats, the expressions of PKA, cAMP, and p-CREB proteins in the TNC region of brain tissue were significantly elevated (P < 0.01) (Figure 8). Administration of TTP resulted in down-regulation of cAMP, p-CREB protein expressions (P < 0.05) as well as PKA protein expression (P < 0.01), thereby providing protection to neuronal cells (Table 9).

Effect of TTP on PKA, cAMP and p-CREB in TNC region of rats. Representative WB bands show protein expression in black with GAPDH serving as the loading control. The relative expression is defined as the ratio of the optical density (OD) of the target protein band to the OD of the corresponding GAPDH band. Compared to natural control group (CG), the expressions of PKA, cAMP, and p-CREB in the model group (MG) were significantly enhanced. In contrast, the expressions of these protein were markedly reduced in both positive control group (Rimegepant treated group) (XY) and TTP treated group (PG). 3 biological replicates per group, each consisting of 3 technical replicates.
Relative Expression of PKA, cAMP, and p-CREB (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG.
Relative Expression of circNR3C1, miRNA382-5p (Mean ± SD).

Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. n = 3 biological replicates per group, each consisting of 3 technical replicates. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG, * P < 0.05 versus MG.
Relative Expression of TACR1, HSP60 and ATF4 mRNA (Mean ± SD).
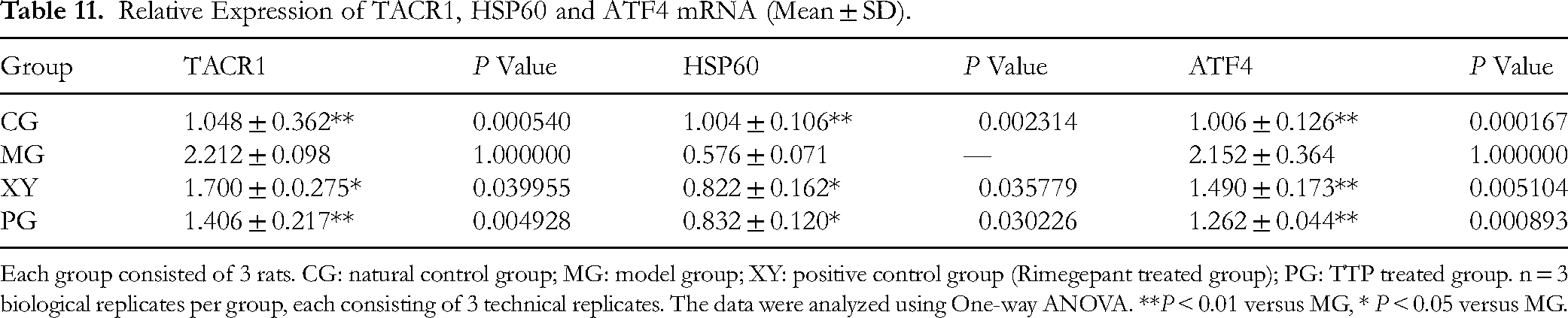
Each group consisted of 3 rats. CG: natural control group; MG: model group; XY: positive control group (Rimegepant treated group); PG: TTP treated group. n = 3 biological replicates per group, each consisting of 3 technical replicates. The data were analyzed using One-way ANOVA. **P < 0.01 versus MG, * P < 0.05 versus MG.
Effect of TTP on circNR3C1, miRNA382-5p, TACR1, HSP60, ATF4 mRNA Expression in TNC Region of Rats
CT values of target genes were identified the in each group, with β-actin serving as an internal reference. The relative expression levels of target genes across different groups were calculated using the 2−ΔΔCT method. In migraine rats, circNR3C1 exhibited a significant decrease (P < 0.01), indicating a diminished capacity to sponge miRNA382-5p. miRNA382-5p, TACR1 and ATF4 mRNA increased, while HSP60 decreased significantly (P < 0.01). These findings suggest that TTP treatment adjusted the mRNA levels associated with the CGRP pathway, as well as those linked to inflammation and oxidative stress (Tables 10 and 11).
Discussion
Currently, the trigeminal neurovascular reflex hypothesis serves as the predominant theory elucidating the pathogenesis of migraine. Activation of the trigeminal nervous system triggers the release of neurotransmitters, including CGRP, which increases vascular permeability and induces cerebral vasodilation. Simultaneously, microglia in TNC region become aberrantly activated, leading to local inflammatory responses and oxidative stress. This cascade enhances the transmission and processing of pain signals, resulting in persistent pain. These interconnected mechanisms and feedback loops establish into a network that is crucial for the onset and progression of migraine.
Different from exiting studies, we conducted a comprehensive investigation into inflammation, oxidative stress and neuronal damage, revealing that migraine arises from multiple pathways rather than a singular one. This finding partially elucidates the complexity of migraine and highlights the limited efficacy of single-target drugs. Previous research has primarily emphasized the role of CGRP, while our study not only addresses the upstream factors that initiate complex reactions but also identifies the involvement of circNR3C1/miR-382-5p in TNC region in the context of migraine. Furthermore, we demonstrated a previously unrecognized role of TTP in migraine management. Consequently, our research may offer novel therapeutic strategies with significant clinical translation potential.
We employed continuous subcutaneous injections of nitroglycerin to establish a migraine model to investigate the multi-pathway intervention mechanisms of TTP.
We assessed the frequency of headache-like behaviors at intervals of 0-30 min and 30-60 min following injection. Administration of TTP significantly reduced the frequency of these behaviors, indicating its therapeutic efficacy in treating migraine. TNF-α and IL-6, known inducers of visceral pain, 24 are common inflammatory markers associated with migraine in serum. TTP administration resulted in decreased serum levels of TNF-α and IL-6 in migraine rats, thereby alleviating the inflammatory environment. In TNC region of the brain, TTP inhibited the aberrant activation of microglia, and suppressed the release of inflammatory mediators. Additionally, TTP reduced the transcriptional activity of Tachykinin receptor 1 (TACR1) and activating transcription factor 4 (ATF4) genes in the TNC, which further inhibited the activation and synthesis of inflammatory factors. The expression of p38 MAPK protein was also diminished, causing a decrease in the phosphorylation of the signal transducer and activator of transcription 1 (STAT1), and decreased the activity of inflammatory mediators. Ultimately, TTP diminished both peripheral and central inflammatory substances, exerted anti-inflammatory and analgesic effects in treatment. Concurrently, inflammatory factors stimulated the expression and release of CGRP. 25
CGRP is positively correlated with miR-382-5p expression, 26 indicating that miR-382-5p plays a role in regulating the CGRP pathway. In animal models, CGRP receptor antagonists have been shown to decrease the expression of miR-382-5p, further supporting the relationship between miR-382-5p and the CGRP pathway. 27 Over-expression of miR-382-5p induces spontaneous production of central pro-inflammatory cytokines, including TNF-α and IL-6, in chronic migraine rats, 28 and reduces SOD2 activity leading to ROS accumulation. 29 Inflammation and hypoxia are primary factors that stimulate CGRP release from neurons, contributing to neuronal damage30,31 and serving as independent factors in migraine pathogenesis. In our study, we observed a decrease in CGRP levels in both serum and TNC region in PG. Our results demonstrated that TTP can up-regulate the expression of circNR3C1 in the TNC of migraine rats, enhance the adsorption of circNR3C1 to miRNA382-5p, and subsequently down-regulate the expression of CGRP. TTP's modulation of the circNR3C1/miR-382-5p axis represents a novel connection between non-coding RNA regulation and CGRP signaling in migraine.
CGRP exerts its biological effects primarily by binding to and activating the receptor RAMP1, which is widely recognized in migraine studies as the classical CGRP receptor. CGRP as a pain mediator by activating receptors stimulates intracellular signaling pathways associated with pain, resulting in increased intracellular cAMP levels. The elevation of cAMP is crucial for the activation of protein kinase A (PKA). PKA promotes the release of pain-related neurotransmitters including glutamate and Substance P. By modulating neurotransmitter release, PKA enhances the efficacy of synaptic transmission, thereby increasing pain sensitivity and responsiveness to pain stimuli. This sensitization can result in “spontaneous pain” or “hypersensitivity”, enabling otherwise painless stimuli to elicit significant pain. Additionally, PKA activation influences the contraction and relaxation of vascular smooth muscle. PKA also affects brain sensitivity to light by phosphorylating cAMP response element-binding protein (CREB), 32 which promotes the expression of pain-related genes such as neurotransmitter receptors and inflammatory mediators, thereby influencing long-term pain responses. Following the administration of TTP, the expression levels of RAMP1, cAMP, PKA, and p-CREB in the TNC region were down-regulated, leading to the inhibition of CGRP - mediated biological effects.
Oxidative stress plays a significant role in the pathogenesis of various diseases.33,34 Numerous migraine triggers, including stress and sleep deprivation, are associated with oxidative stress. 35 Furthermore, markers of oxidative stress have been positively correlated with both the frequency and severity of migraine. 36 This condition can alter neuronal excitability by producing ROS, activating TRPA1 ion channels, and promoting the release of CGRP, thereby enhancing the transmission of pain signals in neurons. This mechanism is regarded as a crucial link in the pathogenesis of migraine. 35 Additionally, oxidative stress can activate microglia, which leads to the release of pro-inflammatory factors, and subsequently triggering local neuroinflammatory responses. 37 A substantial accumulation of ROS has been observed in the TNC region of migraine rats, resulting in neuronal damage, a decrease in the number of Nissl bodies, and heightened oxidative stress. Activated NRF2 shifts to the cell nucleus to exert antioxidant effects; however, due to the antioxidant reaction, GPX4 and HSP60 are consumed more rapidly than they are synthesized, leading to a significant reduction in their expression. Concurrently, the degree of cell damage has reached the threshold that actives AKT and mTOR to initiate tissue repair. TTP can restore the antioxidant functions of GPX4 and HSP60, reduce tissue oxidative stress, ROS levels, consequent more cytoplasmic localization of NRF2. Those actions have protected neurons and Nissl bodies. Thereby, the expression of AKT and mTOR diminished, due to lower degree of cell damage.
Migraine is a disease characterized by highly complex mechanisms, never resulting from a single target acting in isolation. The circNR3C1/miR-382-5p and the over-activation of microglia in migraine initiate an inflammatory response in both central and peripheral environments, which in turn leads to an increased release of CGRP and various biological effects. This amplification of pain signals results in headache-like behaviors, while oxidative stress exacerbates these processes. Ultimately, neuronal damage occurs, prompting the body to activate autophagy and antioxidant programs for self-repair.
TTP not only targets the core pathways of migraine therapies involved in CGRP, but also addresses critical disease-modifying factors such as inflammation, oxidative stress, and neural damage. A novel aspect of this approach is the modulation of the circNR3C1/miR-382-5p axis. The multi-pathway strategy employed by TTP complements single-target drugs to a certain extent, thereby accommodating personalized treatment needs and broadening applicability to a wider patient population, which ultimately enhances efficacy. We anticipate that this study will introduce a new paradigm in drug development by transcending the limitations of directly targeting pain pathways and incorporating non-coding RNAs that regulate key gene expression through epigenetic mechanisms.
Conclusions
Based on the multidimensional data analysis and thorough consideration, TTP not only influences the expression of neurotransmitters and pain-related genes but also inhibits the transmission of pain signals and protects neuron cells. Additionally, it reduces excessive synaptic transmission and pain perception by anti-infammatory and antioxidant effects, thereby diminishing sensitivity to painful stimuli. Such hypersensitivity can render otherwise innocuous stimuli capable of eliciting significant pain and also affects the contraction and relaxation of vascular smooth muscle, either directly or as a secondary consequence. This phenomenon is a key feature of migraine attacks. In summary, TTP exhibits multi-target effects in a rat model of migraine, indicating its potential therapeutic value.
TTP, as a TCM compound prescription, possesses the notable advantage of multi-target activity; however, this characteristic also brings the risk of off-target effects, which may result in adverse reactions, or therapeutic deviations. Our previous acute toxicity studies in mice presented that the maximum tolerated dose of TTP was 212.8 times the daily oral dose for adults. Additionally, long-term toxicity study involving continuous administration for 90 days revealed no abnormal changes in general behavioral status, body weight, blood parameters, or histopathological examinations in rats. 38 The field of toxicity research has completed a long time ago, but earlier limitations in technology precluded the use of high-throughput screening and systems biology to comprehensively analyze potential off-target effects. Moving forward, we will maintain our focus on this area of research, systematically evaluating potential off-target effects to improve the selectivity and safety of TTP.
This study has several limitations, including its narrow focus and the necessity for further research to address these issues. The mechanisms underlying migraine are intricate, involving multiple components such as the trigeminal nerve, cerebral cortex, and cerebral vasculature. This study concentrated solely on TNC region, which is currently recognized by the research community, thereby limiting the comprehensiveness of the findings. The study analyzed key factors involved in the inflammatory response, oxidative stress response, and central nervous system sensitization within the TNC region, as well as the regulatory effects of non-coding RNA. However, the mechanisms by which interactions among these components ultimately influence the disease remain inadequately understood. Although correlations among circNR3C1, miR-382-5p, and CGRP have been observed, causality has not been functionally verified. Future research, including in vitro miRNA inhibition or over-expression, should confirm the regulatory hierarchy. Additionally, validation across different migraine models, human studies, and exploration of circRNA/miRNA interactions will be pursued. Many mechanistic studies rely on minimal feasible sample size, which is the scientific and statistical limitations, especially for claims spanning many pathway. Mechanistic investigations require the validation of regulatory relationships at the cellular level, which is critical component of future research agendas. This study was conducted using a rat model, which introduces limitations concerning its clinical translation applicability. Obtaining brain tissue from migraine patients poses both ethical and practical challenges; consequently, relevant data may be derived from biomarkers in peripheral blood samples, meriting further exploration.
Footnotes
Acknowledgments
The authors acknowledge the assistance of the Pharmacology Department and Animal Experimental Center, Liaoning Academy of Traditional Chinese Medicine (The Second Hospital Affiliated to Liaoning University of Traditional Chinese Medicine).
Ethical Approval
This study was approved by the Animal Care and Welfare Committee of Liaoning Academy of Traditional Chinese Medicine (The Second Hospital Affiliated to Liaoning University of Traditional Chinese Medicine), Shenyang, China. The approval number is LZYY240302. This study does not involve individuals.
Authors’ Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shenyang Bureau of Science and Technology, Liaoning Province, China (grant number RC230094); Department of Science and Technology of Liaoning Province, China (grant number 2025-MSLH-400).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
