Abstract
Objective
Isatin (2,3-indolinedione, ISA), as an indole oxide, is widely distributed in mammalian tissues and fluids, with concentrations varying from less than 0.1 to greater than 10 μM. Indirubin (2-(2-oxo-1h-indol-3-ylidene)-1h-indol-3-one, IND), as a bisindole alkaloid, is a dimer of indigo. Recent research indicated these two medications could regulate neural activity, however the precise effects of these two drugs on neural cells remained undefined This study aims to investigate the effects of ISA and IND on neural cells development and the underlying mechanisms.
Methods
Zebrafish embryos at 24 hpf were treated with different concentrations of ISA and IND (0.001, 0.01, 0.1, 1, and 10 μM) for 24 h. The number of neural cells and the length of motor neuronal axons, as well as the length of intersegmental vessels at 72 hpf, were observed by confocal microscopy. The body length of zebrafish larvae was measured through bright-field photography using a Nikon stereomicroscope. Movement distance and trajectory of zebrafish were detected by the Zebralab video tracking system.
Results
The results demonstrate that during the early embryonic development of zebrafish, ISA can inhibit the number of zebrafish neurons, the length of motor neuronal axons, the movement distance of zebrafish, and the development of intersegmental vessels (ISVs) in a dose-dependent manner. Conversely, IND not only promotes the number of zebrafish neurons but also enhances the length of motor neuronal axons, and the movement distance of zebrafish, without the growth of ISVs in zebrafish. Besides, both drugs have no significant impact on the early morphological development of zebrafish.
Conclusion
ISA played an inhibitory effect on neural development, possible by inhibiting ISVs development in zebrafish. While IND had a promotional effect on neural development of zebrafish, and was expected to be a potential neuroprotective agent. Furthermore, present study suggested that two drugs with a dimeric relationship could exert different effects.
Introduction
Isatin (2, 3-indolinedione, ISA, Figure 1) is predominantly present in indigo plants (such as Polygonum tinctorium and Isatis indigotica), and is a secondary metabolite of plants. 1 Minute quantities of isatin can also be detected in the central nervous system, blood and urine of mammals, and it might exist as an endogenous metabolite. 2 Meanwhile, ISA, as an antibiotic produced by a strain parasitic in the Marine organism lobster, is a biologically active substance necessary for the survival of lobster. 3 It has been identified in traditional Chinese medicinal herbs such as ISA naturalis and folium isatidis. The synthesis, structure, and pharmacological properties of ISA have garnered significant interest. For example, research indicates that ISA exhibits various biological effects including anti-Parkinson's disease, anticonvulsant activity, anticancer potential, as well as antibacterial and antiviral capabilities.4–6 Due to its core structure, ISA has become a focal point for the development of numerous compounds used as apoptosis inhibitors, anticonvulsants, anxiolytics, antiviral agents, and possible antitumor drugs6. Additionally, ISA is found abundantly in the brain along with peripheral tissues and bodily fluids of mammals; notably higher concentrations are present in reproductive organs compared to other tissues, and its endogenous production has been experimentally validated. 7 When administered externally, ISA had low toxicity levels along with minimal mutagenic or genotoxic effects on the body while easily crossing the blood-brain barrier.8,9 Although current understanding regarding its impact on nerve cell growth and survival remains limited, there is a considerable promise for using ISA in treating Parkinson's disease.4,5

Molecular structure of Isatin (ISA) and Indirubin (IND).
Indirubin (2-(2-oxo-1h-indol-3-ylidene)-1h-indol-3-one, IND, Figure 1) is fabricated through the fermentation and processing of plants like Polygonum tinctorium and Isatis indigotica, and it constitutes one of the principal active components of the traditional Chinese medicine “Qingdai” and “Danggui Longhui Pill”, which is mainly utilized in the treatment of chronic myeloid leukemia.10,11 IND and its derivatives exhibit extensive biological activities including inhibiting CDK, regulatory effects on glycogen synthase kinase-3β, signal transducer and activator of transcription-3, Jak-STAT signaling pathway, etc. 12 Recent research has discovered that IND and its derivatives possess anti-tumor activities against solid tumors such as breast cancer, liver cancer, lung cancer, and hematological malignancies. 13 What's more, several studies have demonstrated that IND could protect cerebellar granule neurons from apoptosis. 14 Hence, IND holds considerable prospects in the development of anti-cancer, anti-inflammatory, and neurological drugs.
Zebrafish exhibits strong conservation with humans in numerous aspects, including genetics, physiology, and the developmental process.15–18 Besides, the embryos of zebrafish are visually transparent, enabling convenient in vitro observation of their morphological alterations. 19 Zebrafish serves as an ideal in vivo model platform, facilitating the identification of natural active drugs with therapeutic potential and has been extensively applied in studies on toxicity and early development.20–27 Hence, in this research, we selected the zebrafish model to explore the effects of ISA and IND on neural development.
In this work, we directly demonstrated the impacts of ISA and IND on nerve cells, as well as their roles in the early development, morphology, and behavior of zebrafish. Prior research has shown that bioactive compounds with dimeric configurations possess improved multifunctional properties. 28 In particular, dimeric medications exhibit greater efficacy in comparison to their monomeric counterparts. While we observed that drugs with a dimer relationship of ISA and IND exhibited markedly different effects on neural developmen of zebrafish.
Materials and Methods
Materials
ISA (with a purity >97%) was purchased from Sigma-Aldrich (Shanghai), China Trading Co., Ltd, and IND (with a purity >98%) was purchased from Shanghai Aladdin Biochemical Technology Co., Ltd, China.
Zebrafish Drug Treatements
The drug administration experiment was carried out as previously described. 21 Firstly, (huc:mcherry) zebrafish embryos were obtained through natural mating and maintained in E3 solution (5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2, 0.33 mM MgSO4) at 28.5°C according to previous reports. 29 We used a dissecting microscope to screen and eliminate morphologically abnormal embryos at 8 h post-fertilization (hpf). Ten healthy embryos were loaded in each well of a 12-well plate, and then the E3 solution was replaced with treatment solutions of ISA and IND at different concentrations (0.001, 0.01, 0.1, 1, and 10 μM). Zebrafish culture medium (E3 or PTU) containing no more than 0.5% DMSO was used as the solvent for the drug ISA and IND. The control group was treated with the same solvent. At 24 hpf after fertilization, the medium was changed to a drug solution formulated with PTU to prevent the generation of melanin in zebrafish embryos, facilitating confocal imaging. At 72 hpf, zebrafish embryos were collected, anesthetized and embedded in low-melting-point agarose gel for confocal imaging using OLYMPUS FV3000. Analysis was performed using Image J software (version v1.54k). Fluorescence signal observation were performed using a confocal microscope with a × 20 objective for the zebrafish neurons. Split the fluorescent signal using Image-Color-Split Channels tool of Image J software. Adjust the threshold, select the signal, and use the measure tool to get the quantitative Integrated Density (IntDen) for comparative analysis as previously report. 30 This study was conducted between September 2024 and June 2025. All zebrafish related experiments were raised and completed at the zebrafish core facility of Nantong University. The reporting of this study conforms to ARRIVE 2.0 guidelines. 31
The Detection of the Overall Development of Zebrafish larvae
The zebrafish embryos were incubated in a constant-temperature incubator. At 48 h and 72 h, the zebrafish embryos were retrieved to count the heart rate of the embryos (the number of heartbeats within 60 s); meanwhile, at 72 h, bright-field photography was performed using a Nikon stereomicroscope and the body length of the zebrafish larvae was measured.
Locomotion Analysis of Zebrafish
As previously stated,26,29 the swimming distance of zebrafish was determined under different drug administrations. At 24 hpf, the E3 solution was replaced with treatment solutions of ISA and IND at various concentrations (0.001, 0.01, 0.1, 1, 10 μM). At 96 hpf, one zebrafish larva was placed in each well of a 96-well plate filled with 1 mL of E3 culture medium. The Zebralab video tracking system was employed to detect data such as movement distance and trajectory. Each zebrafish was individually placed in a well of the 96-well culture dish, and 300 μL of E3 culture medium was added to each well. The instrument was activated in advance, and the 96-well plate containing the larvae was placed in the observation chamber equipped with a camera for 30 min of adaptation before the formal test, during which the swimming behavior was recorded for 5 min. The light/dark periods, each lasting 30 s, were alternated and recorded continuously for 5 times. The ESOvision behavior analysis software was utilized for analysis as our previously described. 22
Statistical Analysis
GraphPad Prism (version 9.0, Graphpad Software, La Jolla, California) was employed for the statistical analysis and graphical presentation of the data. All data are expressed as the mean ± SD. For multiple comparisons, one-way analysis of variance (ANOVA) was adopted, and Dunnett's multiple comparisons test was used for the specific post hoc test. A P value of less than 0.05 was considered statistically significant (*), a P value of less than 0.01 was considered highly statistically significant (**), and a P value of less than 0.001 was considered extremely statistically significant (***).
Results
The Effects of ISA and IND Treatments on the Morphological Development and Body Length of Zebrafish Embryos
Zebrafish embryos develop rapidly, and the early formation of the nervous system and other systems is essentially completed by 72 hpf. Microscopic imaging was carried out on the hatched zebrafish to observe the morphology of the larvae, which revealed ISA had no significant influence on the morphological development and the body length of zebrafish embryos (Figure 2A and B). Meanwhile, IND also had no significant influence on the morphological development and the body length of zebrafish embryos (Figure 2C and D). In addition, within the concentration range investigated, neither ISA nor IND was found to exert any significant effect on the survival rate of zebrafish embryos. Above results suggested that the treatments of ISA and IND had no significant effect on the overall development of zebrafish larvae.

Effects of zebrafish embryo body length by ISA and IND treatment. A, C): The bright field images of zebrafish at 72 hpf. B, D): Statistics of body length, n ≧ 6, biological replicates per group. Scale bar 100 μm.
The Impact of ISA and IND Treatments on the Number of Zebrafish Neurons
To further explore the effects of ISA and IND on neural development of zebrafish, the red fluorescence-labeled Tg (huc:mcherry) zebrafish neurons were employed to investigate the influence of ISA and IND treatments on the quantity of neurons. The results demonstrated that, in comparison with the control group, ISA inhibited the neuronal development and the inhibitory effect on the quantity of neurons was in a concentration-dependent manner at concentrations of 0.001, 0.01, 0.1, 1, and 10 μM at 72 hpf (P < 0.05) (Figure 3A and B). In contrast, IND treatment promoted the neuronal development with an increased quantity of neurons (P < 0.05) (Figure 3C and D).
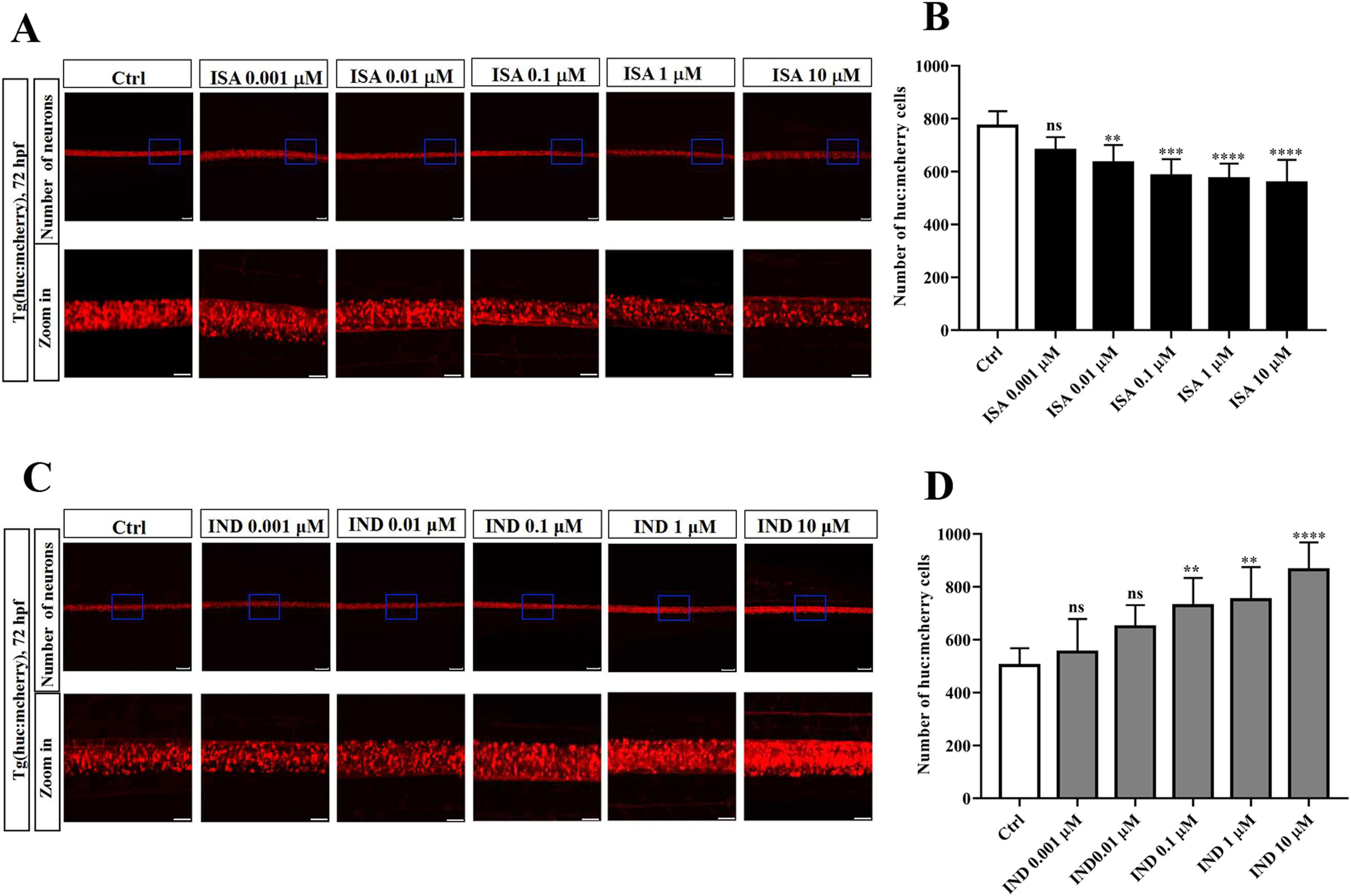
Effects of ISA and IND treatments on Tg(huc:mcherry) embryonic zebrafish neurons. A, C): Phenotype of zebrafish by ISA and IND treatment at 72 hpf. B, D): Number of huc:mcherry cells, n = 6, biological replicates per group. Scale bar 100 μm.
The Impact of ISA and IND Treatment on the Length of Motor Neuronal Axons in Zebrafish
The green fluorescence-labeled Tg (hb9:EGFP) zebrafish neurons were employed to study the impact of isatin and indirubin treatment on the length of motor neuronal axons in zebrafish. The results indicated that, compared to the control group, ISA exerted an inhibitory effect on the length of motor neuronal axons in zebrafish and the inhibitory effect was in a concentration-dependent manner at concentrations of 0.001, 0.01, 0.1, 1, and 10 μM at 72 hpf (P < 0.05) (Figure 4A and B). Nevertheless, compared with the control group, IND promoted the length of motor neuronal axons in zebrafish (P < 0.05) (Figure 4C and D).

Effects of ISA and IND treatments on Tg (hb9:EGFP) embryonic zebrafish motor neuronal axons. A, C): Phenotype of motor neuron by ISA and IND treatment at 72 hpf. B, D): Statistics of the neuronal axon lengths, n = 6, biological replicates per group. Scale bar 100 μm.
The Influence of ISA and IND Treatments on the Neurobehavior of Zebrafish Larvae
To assess whether the early ISA and IND treatments would result in abnormal neurobehavior in zebrafish, behavioral assays were performed on zebrafish larvae on the 96 hpf after incubation. Under a brief light/dark cycle, the movement distance of zebrafish larvae treated with ISA was significantly shortened only to 70 mm (Figure 5). Conversely, the movement distance of the zebrafish larvae treated with IND were significantly lengthened up to 700 mm (Figure 5). These data further substantiated that ISA had an inhibitory effect on neuronal development, while IND played a promoting role in neuronal development of zebrafish.

The swimming distance of zebrafish at 96 hpf (swimming behavior assay (5 min)). A): ESOvision behavioral analysis software for swimming distance by ISA and IND treatments. B): Statistics of the swimming distance, n = 6, biological replicates per group.
The Impact of ISA and IND Treatments on the Development of Intersegmental Vessel in Zebrafish
To explore the influence of ISA and IND on the development of intersegmental vessel (ISVs) in zebrafish, we utilized double-fluorescent-labeled zebrafish Tg (hb9:EGFP::kdrl:mcherry), where endothelial cells were marked as mcherry and motor neurons were labeled as EGFP. At 48 hpf, ISA had an inhibitory effect on the growth of ISVs in zebrafish, while IND had no significant effect on the growth of ISVs (Figure 6A–C and G–J). What's more, ISA inhibited the length of motor neuronal axons, while IND enhanced the length of motor neuronal axons in zebrafish (Figure 6D–F and K).

Effects of ISA and IND treatments on Tg (hb9:EGFP::kdrl:mcherry) embryonic zebrafish. A-C): ISV phenotypes of control group, and 10 μM ISA or IND treated groups at 48 hpf. D-F): Phenotype of motor neuron of control group, and 10 μM ISA or IND treatment at 48 hpf. G-I): Merged of images. J): Statistical analyses of ISV length, n = 6, biological replicates per group. K): Statistical analyses of neuronal axon length, n = 6, biological replicates per group. Scale bar 100 μm.
Discussion
Here, we set out to explore for the first time the effects of ISA and IND on neuronal development of zebrafish. We found that both ISA and IND had no significant effect on the overall development of zebrafish larvae (Figure 2). While, ISA inhibited the quantity of neurons, the length of motor neuronal axons, the movement distance of zebrafish, and the growth of ISVs in zebrafish (Figures 3–5). On the contrary, IND treatment promoted the quantity of neurons, the length of motor neuronal axons, and the movement distance of zebrafish, without the growth of ISVs in zebrafish (Figures 3–5). By comparing above experimental results, we can clearly see that despite having a dimeric relationship, ISA and IND have completely different effects on neuronal development of zebrafish.
As an indole derivative, ISA is an endogenous, multifunctional compound widely present in plants, marine organisms, fungi, and mammals. 32 Further research shows that ISA, as a precursor of pharmacologically active compounds, has a variety of pharmacological characteristics, including anticancer, antibacterial, and antidiabetes. 32 However, the impact of ISA on the growth and survival of nerve cells is not yet clear, and it has only been demonstrated to inhibit the proliferation of human neuroblastoma (SH-SY5Y) cells and induce their apoptosis. 33 Our research showed that ISA could suppress the number of zebrafish neurons and the length of motor neuronal axons in a dose-dependent manner (Figure 4). By monitoring the movement distance and trajectory in zebrafish behavior, we discovered that ISA conspicuously shortened the movement distance of zebrafish (Figure 5). Furthermore, by contrasting the confocal microscopic imaging results of double fluorescently labeled zebrafish Tg (hb9:EGFP::kdrl:mcherry), we inferred that the inhibitory effect of ISA on nerve cells of zebrafish was predominantly achieved by impeding the development of intersomitic vessels (Figure 6). Some studies have demonstrated that pharmacological agents can promote angiogenesis, thereby supplying nutrients, immune mediators, and other essential substances to support neural growth and development. Conversely, inhibition of angiogenesis may produce opposing effects. However, whether the suppressive effects of ISA on vascular and neural growth are directly causal remains to be established and requires further experimental validation. The phenotypic outcomes induced by ISA and IND observed in this study may be linked to complex neuro-vascular interactions, which are fundamental to proper brain development and function. In zebrafish, neuronal activity regulates angiogenesis through mechanisms such as NMDAR-mediated upregulation of vascular endothelial growth factor (VEGF), ensuring the coordinated development of neural and vascular systems. 34 This tight physiological coupling suggests that the primary neuroactive effects of the investigated compounds might secondarily influence vascular patterning. Glycogen Synthase Kinase-3β (GSK-3β) serves as a critical signaling hub capable of integrating both neurogenic and angiogenic processes. Its inhibition promotes neurogenesis via stabilization of β-catenin, while also modulating the expression of key angiogenic factors such as VEGF through downstream pathways including Wnt/β-catenin and PI3K/AKT. Moreover, cyclin-dependent kinases (CDKs), including CDK2—whose activity is regulated by factors such as the RNA demethylase FTO—contribute to cell cycle regulation during vascular development. Therefore, we propose that the pleiotropic phenotypes induced by ISA and IND may originate from their interaction with a GSK-3β-centered regulatory network, potentially involving crosstalk with CDK-related pathways, which concurrently coordinates neurogenic and angiogenic outcomes within the neurovascular unit. Further studies are warranted to elucidate this putative mechanism. Some studies have shown that drugs may promote angiogenesis, providing nutrients, immune and other substances for nerve growth and development, thereby promoting nerve growth. 22 Inhibition of angiogenesis may lead to the opposite result. However, whether the inhibition of ISA on vascular and neural growth is directly causally related still requires further experimental evidence.
IND, a bisindole alkaloid, exists in traditional Chinese medicine Isatis root, indigo naturalis, and a variety of bacteria and mollusks, and can exert various biological activities, such as anti-tumor, anti-inflammatory, and neuroprotective effects. 35 Our research disclosed that IND not only promotes the increase in the number of zebrafish neurons but also facilitates the development of the length of zebrafish motor neuronal axons. Through the monitoring of zebrafish behavior, we observed that IND significantly lengthened their movement distance of zebrafish. Studies show that IND has inhibitory effects on CDKs and GSK-3β signaling pathways, which are important therapeutic targets for neurodegenerative diseases such as AD and PD, 36 further highlighting the medicinal value of IND. Previous research also demonstrated that the neuroprotective effect and anti-angiogenic effect of IND were accomplished through distinct molecular mechanisms and were not correlated directly.13,37–39 Here, our data showed that ISA had an inhibitory effect on the growth of ISVs in zebrafish, while IND had no significant inhibitory effect on the growth of ISVs (Figure 7). Therefore, the neuroprotective mechanism of IND may not directly associated with the supply of nutrients and growth factors related to vascular development in zebrafish. It is still unclear whether the potential indirect or compensatory effects of ISA and IND on other signaling pathways, neurotransmitters or glial cells. The neuroprotective mechanism of IND requires further investigation.

The structure-activity relationships of ISA, and IND in transgenic zebrafish.
Previous research has shown that dimeric compounds in nature generally exhibited more favorable biological activities compared to their monomeric counterparts, and dimeric natural products have become increasingly appealing to researchers in areas such as medicinal chemistry and pharmacology. 40 Dimerization can expand the spectrum of biological activity, overcome drug resistance, enhance drug activity, and improve physicochemical, pharmacokinetic and pharmacological properties. These may be the reasons for the functional divergence caused by dimerization. Therefore, dimerization has become a promising strategy for the development of new drugs. 41 Considering the close structural relationship between ISA and IND, namely, IND is the dimer of ISA, and previous research found the combined effects of IND and ISA on the antitumour properties and anti-inflammatory activities. 41 While our present study showed that despite having a dimeric relationship, ISA and IND have completely different effects on neuronal development of zebrafish (Figure 7). So we discerned that despite having similar molecular structures, two drugs with a dimeric relationship could exert different effects, and hypothesized that this discrepancy might be related to subtle differences in target binding, target selectivity or multi-target effects, as well as differential regulation of signaling pathways. So far, the side effects and structure-activity relationships of ISA and IND have not been reported. Here, our research offered novel insights into the toxicity, functions, and structure-activity relationships of ISA and IND, and promoted the study of the role of drug structures in pharmacological effects.
The current study has several limitations such as the absence of rescue experiments, effects of drugs on physiology of zebrafish, and the study of prescise molecular mechanism of the ISA and IND on zebrafish neurons. Furthermore, the concentration-response window was narrow. In addition, the relevant research has not been carried out in other models of mammal animals or human beings. Therefore, further studies are needed to investigate these issues.
Conclusion
In general, our study showed that during the early embryonic development of zebrafish, ISA could inhibit the number of zebrafish neurons in a dose-dependent manner and suppress the length of motor neuronal axons, the movement distance of zebrafish, and the growth of ISVs in zebrafish. Conversely, IND treatment promoted the quantity of neurons, the length of motor neuronal axons, and the movement distance of zebrafish, without the growth of ISVs in zebrafish. By comparing above experimental results, we can clearly see that despite having a dimeric relationship, ISA and IND have completely different effects on neuronal development of zebrafish. Furthermore, our data indicated that IND, as an ISA dimer, exhibited enhanced pharmaceutical versatility, and could be a potential neuroprotective agent.
Footnotes
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Project of Nantong Health Commission (MSZ2023018 to Y. L.), the Jiangsu Province College Students'innovation and entrepreneurship training program (S202510304167 to K. Z. and S202510304162 to C. X.), Nantong Project of Science and Technology (JC2025071 to Y. C. and MS2024066 to X. T.), and Large Instruments Open Foundation of Nantong University (KFJN2544 to C. W.).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures involving animals were conducted in accordance with the Animal Protection Ethics Committee of Nantong University's (permission no. S20240715-008) approved protocols.
