Abstract
Background
The Sidama nation of southern Ethiopia traditionally uses various medicinal plants, locally referred to as Ameessa, for the management of childhood ailments. This study investigated the antibacterial activities of six most frequently reported Ameessa plants and identified the most active fractions responsible for these effects.
Methods
Powdered plant materials were extracted with methanol, filtered, and concentrated to dryness. Antibacterial activity was initially evaluated using the disc diffusion assay. The minimum inhibitory concentration (MIC) was determined by the broth microdilution method, and the minimum bactericidal concentration (MBC) was established through sub-culturing from non-growth wells. Extracts showing strong antibacterial activity were subjected to column chromatography for fractionation, and the resulting fractions were tested for antibacterial properties.
Results
Among the six medicinal plants screened, Ekebergia capensis and Plectranthus garckeanus exhibited better antibacterial activity, with zones of inhibition ranging from 7 to 16 mm. Staphylococcus aureus was the most sensitive strain, whereas Escherichia coli showed the least susceptibility. Fractions derived from E. capensis showed MIC values between 0.0867 and 0.1734 mg/mL against S. aureus and Enterococcus faecalis, while fractions from P. garckeanus displayed MICs ranging from 0.0867 to 2.775 mg/mL. The most active E. capensis fraction demonstrated a potent bactericidal effect with the MBC of 0.1734 mg/mL.
Conclusion
Methanolic extracts and chromatographic fractions of E. capensis and P. garckeanus exhibited significant antibacterial activity—particularly against S. aureus and E. faecalis—highlighting as potential sources of novel antibacterial agents. Further phytochemical and pharmacological studies are warranted to purify and characterize the bioactive compounds and evaluate their in vivo efficacy and safety for potential therapeutic development.
Keywords
Introduction
Plant-derived medicines have played a large role in human and livestock. 1 African traditional medicine is steeped in the use of medicinal plants, with cultural communities that primarily rely on. In many parts of Africa, particularly remote areas with limited access to modern health facilities, medicinal plants are a vital component of healthcare delivery systems. Traditional herbalists work closely with residents of these communities, utilizing diverse plant species to address a wide range of illnesses and ailments. Increasingly, herbalists are gaining recognition beyond traditional medicine settings as scientific studies validate the value of herbal remedies in managing various conditions. 2
The rapid emergence and global spread of antimicrobial resistance (AMR) is increasingly threatening the ability to successfully treat infectious diseases. According to the Centres for Disease Control and Prevention (CDC) and the World Health Organization (WHO) many first-line antibiotics are ineffective due to the widespread of multidrug-resistant pathogens. Thus, this situation creates a, critical need for novel antimicrobial agents. 3 The antibiotic discovery pipeline has stagnated despite technological advances in these days. Since the late 1980s, no new structural class of antibiotics has been introduced largely due to scientific and economic constraints. 3
Out of the estimated 390,000 vascular plant species on Earth, approximately 9% are documented as being used in traditional medicine, between 70%–95% of people in developing countries still rely on plant-based remedies for primary healthcare, 3 and only a tiny fraction has been studied for their medicinal properties. 4 There is a remarkably high diversity of classes of chemical in plants which offer the complex self-protection mechanisms against microbial attack. 3 Terpenoids and phenolic derivatives are highly abundant chemical structures among natural products possessing antibacterial activity, representing 26.6% and 50.8% of compounds, respectively according to recent evidence. 3 The progress in the field highlights the translational promise of anti-virulence therapy as a complementary strategy to traditional antibiotic development, bridging traditional medicine knowledge and modern pharmacological innovation. 5
The contribution of herbal remedies to modern medicine is profound, with approximately 25% of prescription drugs dispensed in the United States containing at least one active ingredient derived from plant sources. 6 The WHO study 7 identified 119 pharmaceuticals originating from plants, with roughly 74% having direct links to their traditional applications by indigenous societies. For millions worldwide, herbal remedies remain a significant source of traditional medicine and contribute meaningfully to modern healthcare. 8
Many available antimicrobials represent the last line of defence against infections and are threatened by the alarmingly high rate AMR nowadays. According to the report surveyed in 76 countries in 2022 by the Global Antimicrobial Resistance and Use Surveillance System (GLASS), 42% of E. coli isolates exhibit resistance to third-generation cephalosporins, and 35% of S. aureus are methicillin-resistant (MRSA), both posing significant threat. Moreover, approximately about 20% of the urinary tract infections are caused by E. coli showed reduced susceptibility to common antibiotics in 2020, further complicating treatment efforts. 9
Reports indicated that more than 80% of the people used traditional medicine to treat various systems based on indigenous knowledge. 10 Our recent meta-analysis revealed that the prevalence of herbal medicine use in Ethiopia is 46%. 11 Various medicinal plants have therapeutic potential to treat infections, are still given by traditional healers without scientific knowledge and optimization of the right doses.
The search for novel agents with antibacterial activity is intensifying, driven by the desire to reduce hospitalization costs, prevent severe complications and death. 12 Current synthetic drugs often present limitations such as high costs, adverse effects, and the development of drug resistance, prompting scientists to explore alternative therapeutic options. 13 Previous studies have revealed promising combinatory effects 14 and existence of beneficial synergistic effects between the two extracts. 15 Medicinal plants offer a rich source of biologically active ingredients for treating various diseases and disorders, presenting a potential alternative. 16 Ethiopia has about 800 species of plants that are used in such traditional healthcare system characterized by various shape of the ecological diversities of the country. 17
Several medicinal plants, including Lactuca inermis (Haato), Ekebergia capensis (Oloncho), Englerina woodfordioides (Bunu kishee), Plectranthus garckeanus (toontoona), Sida rhombifolia L. (Kirkite), and Viscum tuberculatum (Masinchote kishee), are traditionally used by the Sidama nation and other communities in Ethiopia for treating childhood ailments believed to be associated with bacterial infections. However, most of these traditional practices lack scientific validation regarding their efficacy.
Therefore, this study aimed to scientifically assess the antibacterial potential of six medicinal plants traditionally used in the Sidama community. The research focused on screening their antibacterial activities, determining their minimum inhibitory and bactericidal concentrations, and isolating the most active fractions for further analysis. The findings are expected to validate traditional knowledge, support evidence-based use of herbal medicines, and contribute to the global search for new antibacterial compounds from plant origins.
Materials and Methods
Collection and Identification of the Plant Material
Plant specimens were collected from Sidama National Regional State, Ethiopia. Adequate fresh herbal materials such as (leaves, aerial, root, and bark) were collected from their natural habitat using vernacular names with the assistance of local healers and the direct support and supervision of professional botanists. The scientific names of the collected species were verified and authenticated by taxonomists from Addis Ababa University. Corresponding voucher specimens were prepared and deposited at the National Herbarium, College of Natural and Computational Sciences, Addis Ababa University, for future reference.
GPS coordinates of each collection site were recorded. The collected plant materials were placed in clean, labelled polyethylene bags and transported to the laboratory on the same day. Upon arrival, leaves were rinsed gently with distilled water to remove surface debris and shade-dried at ambient temperature (25 ± 2 °C) in a well-ventilated area, avoiding direct sunlight. Dried samples were pulverised into a fine powder and stored in airtight containers at room temperature until extraction.
The plant collection site is presented in Figure 1. The GPS sites of collection were: E. woodfordiodes (6.9166739, 38.5066969), E. capensis (6.9170121, 38.5061376), L. inermis (6.9597285, 38.3142855), V. tuberculatm (6.9166639, 38.5067054), S. rhombifoli (6.9179787, 38.346937), and P. garckeanus (6.9166649, 38.5066899).

Map of study area depicting plant collection area (map drawn using QGIS).
Extraction of the Plant Materials
Fresh leaf of the plant material of each species was air dried at room temperature and then ground using a mortar and pestle. The powdered samples were weighed using an electrical weighing balance (Sartorius, Ag Gottingen, Germany). The resulting powder was kept in a polyethylene bag to avoid exposure to moisture, air, and dust until used for the study. 18
A portion of the powders of plant material from each species (150 g each) was weighed and soaked in 750 ml of methanol at a solid-to-solvent ratio of 1:5 (w/v) for three days in a conical flask, covered with aluminium foil, and kept for 72 h with intermittent shaking at room temperature. Extracts from each specimen were subsequently filtered with Whatman N
Antibacterial Assay
The antimicrobial activity was evaluated against four bacterial strains. They were obtained from the American Type Culture Collection (ATCC). The bacterial strain was selected as representative of both classes of gram-negative and gram positive, the microbial strains gram positive: S. aureus (ATCC 259223), and E. faecalis (ATCC29212), and gram negative: E. coli (ATCC25922), and P. aeruginosa (ATCC27853). The bacterial strains selected in this study— S. aureus, E. faecalis, E. coli, and P. aeruginosa—are among the major pathogens implicated in paediatric skin, respiratory, and gastrointestinal infections, making them clinically relevant targets for evaluating traditional remedies used for childhood illnesses.
The inocula were prepared following the Clinical and Laboratory Standards Institute (CLSI) recommendation and standardized to a 0.5 McFarland turbidity standard equivalent to 1 × 108 CFU/ml. 19 Then it was further diluted to obtain the desired cell density of 5 × 105 CFU/ml.19,20 Briefly, an equivalent of 0.5 McFarland suspensions (1 × 108 CFU/ml) inoculum was prepared and then diluted 1:20 to yield 5 × 106 CFU/ml using saline media.
Disc-Diffusion Assay
The antibacterial activity of methanol extracts of six different plant extracts was evaluated by using the paper disc diffusion method 21 adapted for plant extracts. 22 Two Gram-positive bacteria (S. aureus (ATCC25923), E. faecalis (ATCC29212)) and two Gram-negative bacteria Escherichia coli (ATCC25922), and P. aeruginosa (ATCC27853) were used. The aliquot was spread evenly onto Muller Hinton agar plates by a sterile cotton swab, and then a filter paper disc (6 mm in diameter, which is prepared from the Whatman filter paper Number 1 and sterilized in an autoclave at 121 °C for 15 min) soaked them in the plant extract solution for 30 min. The discs were allowed to dry for 15 min to avoid excess liquid on the agar. Then, by using sterile forceps, the prepared disc was placed on the inoculated agar surface.
The Petri dishes were placed at room temperature for 30 min to allow diffuse into agar before it is incubated. The negative control disc contained distilled water without plant extract. Antibiotic discs containing ampicillin (for Gram-positive) and ciprofloxacin (Gram-negative) were used as positive controls. Then, the plates were incubated at 37 °C for 24 h. At the end, zone of inhibition (ZOI) was observed around the discs where microbial growth has been inhibited, ZOI was measured in millimetres (mm) using a ruler. The experiment was performed in triplicates, and average values were taken as the ZOI.
Each paper disc was impregnated with mg/mL of 2 µL of plant extract. All assays were conducted in triplicate, and mean ZOI were recorded.
Determination of minimum Inhibitory Concentration
An INT based microdilution assay was used to determine minimum inhibitory concentration (MIC) of the plant extracts using 96-well. The first row of the microtiter plate was filled with 80μL of test materials in 2% dimethyl sulfoxide (DMSO). The solvent DMSO (less than 2%) would not inhibit the growth of the microorganisms 23 was used as the negative control for all the experiments. All the wells of microtiter plates were filled with 80 μL of Muller-Hinton broth.
A two-fold serial dilution was made by mixing the contents in each well of the first row and transferring 80 μL of test material to the second well of the same column, mixed thoroughly before transferring, and the same was done up to the last well of the same column. At the end, after the last dilution 80 μL from the last well were discarded, so each well had 80 μL. Finally, a volume of 20 μL, a final concentration of 5 × 106CFU/mL, bacterial suspension was added to each well.
To avoid dehydration of the bacterial culture, each plate was wrapped loosely with cling film. Each microtiter plate has a set of two controls: (a) a column with all solutions except the test extract, and (b) a column with all solutions except the bacterial solution replaced by 80 μL of Muller-Hinton broth. The results were observed after 24 h of incubation at 37 °C, followed by the addition of 20 μl of INT solutions in each well as an indicator after a further incubation of 2 h at 37 °C. The colour change in the well was then observed. A colour changes from purple to pink or colourless was taken as growth. The lowest concentration of plant leaf extract at which no colour change occurred was recorded as the MIC value. All the experiments were performed in triplicate. The average values were taken for the MIC of the test material. 20
Determination of minimum Bactericidal Concentration
The minimum bactericidal concentration (MBC) of plant extracts was determined by taking a loop full of samples from wells with no visible growth in the MIC assay and sub-cultivating them into Mueller-Hinton agar medium. 23 In this procedure, the loop was sterilized and dipped into each well with a concentration of plant extract greater than or equal to the MIC and inoculated into agar medium. All test bacterial species were then incubated at 37 °C for 24 h. The concentration of the plant extract that inhibited colony formation on solid agar medium was MBC. The entire test was done in triplicate for each bacterial species, and the average values were taken for the MBC of the test material.
Fractionation of Bioactive Compounds from Plant Extracts
Column chromatography separation was conducted using silica gel 60–120 (Loba Chemie PVT.LDUK) with gravity elution. Large 30/40 sized, long, cylindrical glass column stands firm on a column chromatography stand. Silica gel (100 g) was mixed with a hexane solvent system to form a homogenous suspension (slurry) and stirred using a glass-stirring rod to remove bubbles. The silica gel slurry was then poured into the column. A completely dried herbal extract sample was mixed with silica gel to produce a fine form of powder for easy sample distribution in an already sealed silica gel column. A sample powdered mass was placed on top of the pre-packed silica column and covered with a layer of cotton.
The column was eluted with Hex: EAc mixtures of increasing polarity of 5% (100, 0, to 0:100) and then to EAc: MeOH (95:5 to 0:100) at uniform rate under gravity. Each fraction was collected separately in a test tube and numbered consecutively for further evaluation on thin-layer chromatography (TLC) (DS Kieselgel 60 GerF254 Germany).
All fractions obtained from column chromatography were subsequently evaluated for antibacterial activity against sensitive bacteria (S. aureus and E. faecalis) using the disc diffusion method.
Each fraction was applied on activated TLC plates with the aid of a capillary tube 0.5 inch apart from the bottom edge of the TLC plate, and the plate was kept in a developing chamber with a suitable solvent system for a specific time until the developing solvent reaches the top of the upper edge of TLC plate. The plate was removed from the developing chamber; the solvent front was marked by a lead pencil and dried. The compound-containing bands/spots displayed on the TLC chromatoplate was observed by visual detection under UV light detector (4132 Muttenz Switzerland) (254 nm and 366 nm) for the presence of specific compounds.
24
The observed spots of the components in the chromatoplate were marked, and the Rf value of each spot was calculate using the following formula:
Statistical analysis: All experiments were performed in triplicate, and the results are presented as mean ± standard deviation (SD). No additional statistical tests were applied, as the primary objective of the study was descriptive, focusing on comparison of the observed values with established literature references to assess clinical relevance. This approach ensures clarity and reproducibility while highlighting consistency with previously reported standards.
Result
The results of this study presented the extraction, fractionation, and antibacterial activity of selected medicinal plants.
Yield of Extracts
The study evaluated the yield percentages of extracts obtained from various medicinal plants. E. woodfordioides produced a yield of 10.4%, S. rhombifolia yielded 7.27%, P. garckeanus 7.6%, and V. tuberculatum yielded 8.06%. Among the tested species, E. capensis showed the highest yield at 17.2%, whereas L. inermis recorded the lowest yield of 6.33%.
Preliminary Antimicrobial Activity Screening
The antibacterial activity of methanol extracts derived from the bark, leaves, and whole aerial parts of L. inermis, E. capensis, E. woodfordioides, P. garckeanus, S. rhombifolia, and V. tuberculatm was investigated against four bacterial strains: E. coli, P. aeruginosa, S. aureus, and E. faecalis. Ampicillin and ciprofloxacin were used as positive controls, while DMSO served as the negative control. Four of the six medicinal plants exhibited antibacterial activity, with inhibition zones ranging from 7.00 mm to 16.05 mm.
Ekebergia capensis showed the highest antibacterial effect, particularly against S. aureus (16.05 ± 0.06 mm), and demonstrated activity against P. aeruginosa (8.2 ± 0.07 mm) and E. faecalis (8.15 ± 0.07 mm). E. woodfordioides inhibited P. aeruginosa (7.05 ± 0.06 mm), S. aureus (12.01 ± 0.00 mm), and E. faecalis (7.00 ± 0.00 mm). Plectranthus garckeanus was active against P. aeruginosa (11.06 ± 0.05 mm), S. aureus (8.02 ± 0.00 mm), and E. faecalis (8.03 ± 0.00 mm), while V. tuberculatum showed moderate inhibition against S. aureus (8.01 ± 0.00 mm). In contrast, S. rhombifolia and L. inermis did not exhibit any antibacterial activity against any of the tested strains. Notably, none of the plant extracts showed inhibition against E. coli.
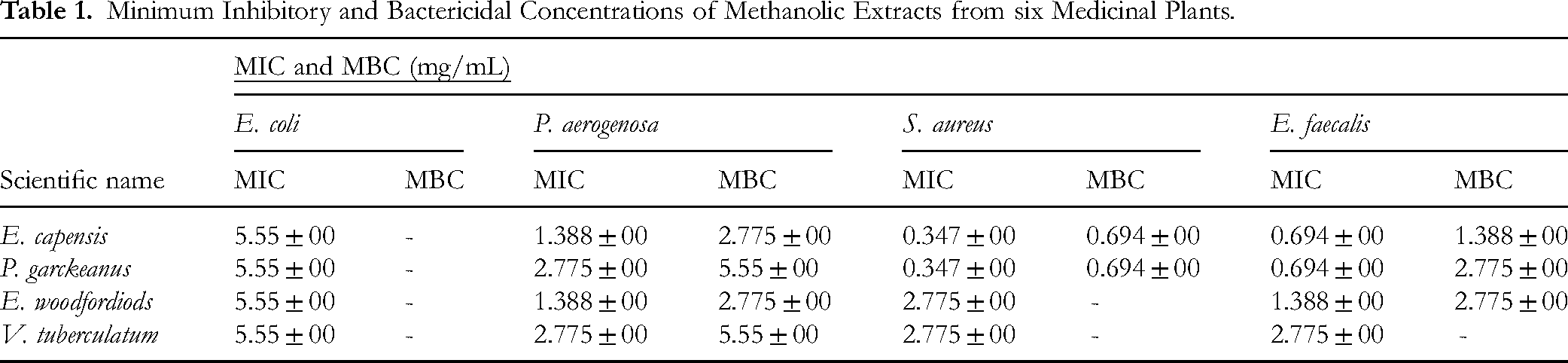
Minimum Inhibitory Concentration and Bactericidal Concentration of the Crude Methanol Extracts of the Plant
The extracts of four plants (E. woodfordiods, P. garckeanus, E. capensis and V. tuberculatum) with better ZOI were selected for the determination of MIC. The extracts of P. garckeanus and E. capensis exhibited the best antibacterial activity; with a minimum inhibitory concentration (MIC) of 0.347 mg/mL against S. aureus. The crude methanol extract of the V. tuberculatum plant was less active against all test microorganisms compared with other plant extracts.
Both E. capensis and P. garckeanus showed the highest bactericidal activity against S. aureus, with a MBC of 0.694 mg/mL, followed by an MBC of 1.388 mg/mL against E. faecalis. V. tuberculatum and P. garckeanus displayed the lowest bactericidal activity against P. aeruginosa. In general, the plant extract was more active against Gram-positive than Gram-negative bacteria at lower concentrations, Table 1.
Minimum Inhibitory and Bactericidal Concentrations of Methanolic Extracts from six Medicinal Plants.
Fractionation of Crude Extracts from Ekebergia capensis
Among all the four plants, E. capensis showed better activity and the extract was subjected to further fractionation. Crude stem bark extract (17 g) of E. capensis was subjected to gradient column chromatography. A total of 55 fractions were collected. Fractions with similar TLC profiles were combined and as a result, eight fractions (Fc1– Fc8) were obtained. Fractions 1–8 were merged as Fc1, fractions 9–14 were merged as Fc2, fractions 15–20 were merged as Fc3, fractions 21–29 were merged as Fc4, fractions 30–35 were merged as Fc5, fractions 36–46 were merged as Fc6, fractions 47–51 were merged as Fc7, and fractions 52–55 were merged as Fc8. The retention factors, colour and numbers of spots of eight different fractions in the hexane: chloroform: ethyl acetate (7:2:1) is shown, Table 2.
Different Fractions of Column Chromatography of Ekebergia capensis and Their Rf Values, Solvent System, №of Spots, Yield and Colour.

Note: Hex = hexane, EAc = ethyl acetate, MeOH = methanol, FC = Ekebergia capensis plant fraction.
Fractionation of Crude Extracts from Plectranthus garckeanus
The crude leaf extract of P. garckeanus (7 g) was subjected to gradient column chromatography, yielding a total of 75 fractions. Fractions exhibiting similar TLC profiles were combined, resulting in eight pooled fractions designated as Fp1–Fp8 (Table 3). Specifically, fractions 1–3 were combined as Fp1, 4–5 as Fp2, 7–10 as Fp3, 11–24 as Fp4, 25–36 as Fp5, 37–48 as Fp6, 49–60 as Fp7, and 61–66 as Fp8. Fractions collected as F67–F70 disappeared following solvent evaporation. Thin-layer chromatographic analysis using a hexane: chloroform: ethyl acetate (7:2:1) solvent system showed distinct retention factors and spot patterns among the eight fractions, indicating effective separation (Table 3).
Different Fractions of Column Chromatography of Plectranthus garckeanus and Their Rf Values, Solvent System, no of Spots, Yield and Colour.

Note: Hex = hexane, EAc = ethyl acetate, MeOH = methanol, Fp = P.garckeanus fraction.
Antimicrobial Activity of Ekebergia capensis and Plectranthus garckeanus Fractions Against S. aureus and E. faecalis
The antibacterial activity of two plants with better activity in MIC determination have undergone column chromatography fractionation. Each fraction was screened for the antibacterial potential by measuring ZOI. The fractions of E. capensis and P. garckeanus showed better activity, results depicted in Table 4.
Antibacterial Activity of E. capensis and P. garckeanus Plant Fractions Against S. aureus and E. faecalis Using Disc Diffusion Method.
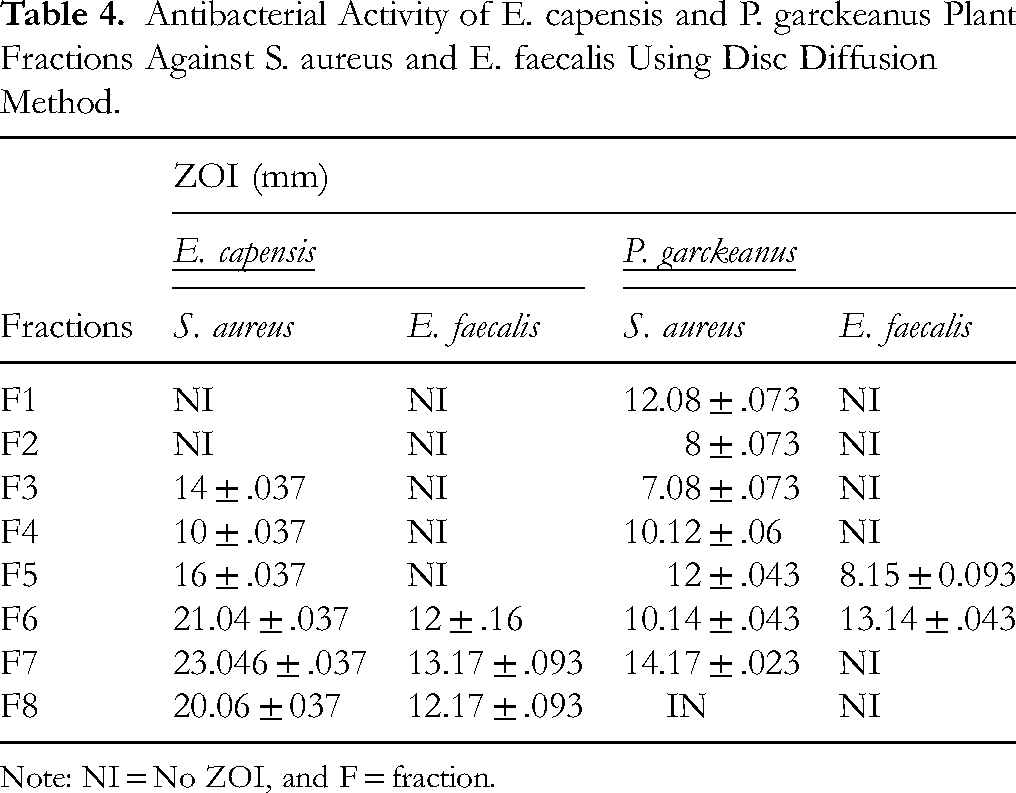
Note: NI = No ZOI, and F = fraction.
Of the two medicinal plant fractions tested for antibacterial activity, thirteen fractions demonstrated inhibitory effects against the test microorganisms, with ZOI ranging from 7 to 23 mm. Overall, the fractions exhibited greater activity against S. aureus than against Enterococcus faecalis. In contrast, three fractions—E. capensis fractions Fc1 and Fc2, and P. garckeanus fraction Fp8—showed no detectable antibacterial activity.
The fractions of both plants with better antimicrobial activity were selected for determination of MIC. The lowest MIC (0.0867 mg/mL) was exhibited by the E. capensis plant fractions (Fc6, and Fc7) against S. aureus and E. faecalis, whereas the highest MIC (2.775 mg/mL) was exhibited by P. garckeanus fraction (Fp7) against S. aureus.
The E. capensis plant fraction (Fc7) exhibited the strongest bactericidal activity against both S. aureus and E. faecalis, with a minimum bactericidal concentration (MBC) of 0.1734 mg/mL. Similarly, the P. garckeanus plant fraction (Fp6) also demonstrated significant bactericidal activity against E. faecalis, achieving an MBC of 0.1734 mg/mL. In contrast, the P. garckeanus plant fraction (Fp7) showed the weakest bactericidal activity against S. aureus, with an MBC of 2.775 mg/mL, Table 5.
The minimum Inhibitory Concentration and minimum Bactericidal Concentration of E. capensis and P. garckeanus Crude Methanol Extract and Their Constituents Against S. aureus, and E. faecalis.

Note: Fc6 - E. capensis plant fraction six, Fc7 - E. capensis plant fraction seven, Fc8 - E. capensis plant fraction eight, Fp6 - P. garckeanus plant fraction six, and Fp7 – P.garckeanus plant fraction seven.
Discussion
In the study, the biologically active phytochemicals and antibacterial activities of six traditionally useful medicinal plants from Sidama were evaluated. Among the medicinal plants, E. capensis and P. garckeanus emerged as the most promising for their antibacterial potential, particularly against E. faecalis and S. aureus. The E. capensis fractions showed a clear pattern of activity, with fractions 6, 7, and 8 exhibiting the strongest inhibition against S. aureus.
Gram-positive bacteria in this study showed a higher sensitivity compared to Gram-negative possibly due to a relatively porous peptidoglycan layer, which allows easier penetration of phytochemicals, whereas the outer membrane of Gram-negative bacteria acts as an additional barrier. 25 The absence of activity against E. coli and P. aeruginosa supports this notion.
Plant extracts exhibit stronger antibacterial activity against Gram-positive bacteria than Gram-negative bacteria.26,27 This difference is largely attributed to structural variations in their cell envelopes; Gram-negative bacteria possess an additional outer membrane rich in lipopolysaccharides, which acts as a permeability barrier that limits the entry of many phytochemicals, while Gram-positive bacteria lack this layer. 28 The observed variation in susceptibility also reflects the influence of bacterial efflux pumps and enzyme-mediated degradation in Gram-negatives. 29 The degree of antibacterial activity may depend on factors such as the extraction method, solvent polarity, and phytochemical composition of the plant material.
Plant-derived antibacterial compounds exhibit a wide range of mechanisms of action, reflecting their structural and chemical diversity. Many phytochemicals target the bacterial cell wall and membrane, leading to disruption of permeability and leakage of cellular contents, as observed with terpenoids and phenolic compounds. Alkaloids can intercalate into DNA or inhibit key enzymes involved in nucleic acid synthesis, impairing bacterial replication. Flavonoids and coumarins often interfere with bacterial energy metabolism and enzyme activity by forming complexes with proteins or metal ions. In addition, several plant metabolites act through antivirulence mechanisms, inhibiting quorum sensing, biofilm formation, and the production of bacterial toxins and pigments, thereby reducing pathogenicity without exerting strong selective pressure for resistance. 5 These multifaceted mechanisms—spanning direct bactericidal effects and indirect modulation of virulence—underscore the potential of plant secondary metabolites as valuable scaffolds for developing novel antibacterial and resistance-modifying agents.
The current study differs from a previous study, 30 which reported that the methanol extract of E. capensis did not show activity against P. aeruginosa. Methodological variation like microbial inoculum density, strain used, and the environment where plant grown could be reasons for the possible variability. Our study used broth dilution different from the previous study which used agar dilution test for MIC determination which could have underestimated antibacterial activity. 30 Agar dilution methods, were deemed unsuitable for plant extracts due to the inability of non-polar compounds to diffuse effectively in the aqueous agar matrix, leading to inconsistent and non-reproducible results across laboratories. The INT-based microplate serial dilution method was found to be sensitive, efficient, and widely applicable for both bacterial and fungal pathogens, making it a preferred approach for studying plant extracts. 31
The activity of E. capensis stem bark extract against S. aureus and P. aeruginosa reported to be 3.13 mg/mL and 6.25 mg/mL respectively 32 which is higher compared to our study (0.347 mg/mL and 1.388 mg/mL). South African study, 33 reported an MIC of 0.78 mg/mL against S. aureus and 3.125 mg/mL against E. coli for ethanol extracts of E. capensis bark. In previous studies it was indicated that extracts from E. capensis possess various bioactive compounds that contribute to its therapeutic and pharmacological properties. Specifically, a phytochemical analysis of the stem bark of E. capensis conducted in Sudan revealed the presence of several secondary metabolites, including alkaloids, flavonoids, glycosides, phenols, tannins, saponins, anthraquinones, and triterpenoids. 34
The crude methanol extract of P. garckeanus exhibited notable antibacterial activity against P. aeruginosa, S. aureus, and E. faecalis, with MIC of 2.775 mg/mL, 0.347 mg/mL, and 0.694 mg/mL, respectively. It was most effective against S. aureus compared to the other bacteria, but less effective against P. aeruginosa than the E. capensis extract. A study 35 found an MIC of 16 µg/mL for the ethanol extract against S. aureus, which is significantly higher than the MIC of 0.347 mg/mL observed in this study.
In this study, the crude methanol extracts of L. inermis and S. rhombifoli did not exhibit any antibacterial effects against all tested bacterial strains. This finding contrasts with a previous study, 36 which reported that the methanol extract of S. rhombifoli was activity against S. aureus and P. aeruginosa, with zone of inhibition 8.7 mm and 7 mm respectively.
The crude methanol extract of the V. tuberculatum plant showed a notable ZOI against S. aureus (8 mm), consistent with previous findings. 37 In our study, however, the crude methanol extract did not demonstrate any inhibitory effect against P. aeruginosa and E. faecalis, which contradicts previous reports. 38
The crude methanol extract of the E. woodfordioides plant demonstrated significant antimicrobial activity, exhibiting zones of inhibition of 12 mm against S. aureus, and 7 mm against both P. aeruginosa and E. faecalis. This extract was more effective against S. aureus compared to other bacterial strains tested. In previous studies conducted in Ethiopia reported no observable effects of E. woodfordioides against E. coli and P. aeruginosa. 37
However, our findings indicate better activity of the crude methanol extract against P. aeruginosa, with a ZOI of 7 mm and a minimum inhibitory concentration (MIC) of 1.388 mg/mL supporting these findings, previous research suggests that extracts of E. woodfordioides contain diverse bioactive compounds that contribute to its therapeutic and pharmacological effects. A phytochemical analysis of E. woodfordioides leaves conducted in Ethiopia identified the presence of several secondary metabolites, including alkaloids, quinones, terpenoids, coumarins, phenols, saponins, steroids, and tannins. 37 Notably, plants rich in alkaloids, flavonoids, terpenoids, steroids, xanthones, phenolic compounds, and polyphenols are known to exhibit significant antimicrobial activity. 39
Overall, the results indicated that the crude methanol extracts of E. capensis and P. garckeanus produced notable antibacterial activity. However, certain fractions derived from these extracts exhibited markedly higher antibacterial activity than the crude extracts, while others showed little or no activity. These variations could be attributed to the increased concentration of bioactive constituents resulting from the removal of inactive components during fractionation.
Although this study provides valuable preliminary data, it is limited by its in vitro design. The active constituents were not fully characterised chemically, and only a limited number of bacterial strains of wild type were tested. Future research should include in vivo evaluation, detailed phytochemical characterisation, toxicity profiling, and mechanism-of-action studies to validate these promising findings.
Conclusion
This study supports the traditional use of E. capensis, E. woodfordioides, P. garckeanus, and V. tuberculatum for their notable antibacterial properties. While S. rhombifolia and L. inermis showed no antibacterial activity, E. capensis and P. garckeanus stood out as the most effective antibacterial agents in both crude resulting to fractions with promising antibacterial potential making them prime candidates for further drug development.
There is a need to conduct a focused, stepwise chemical and pharmacological follow-up on the most promising fractions F6 and F7 from E. capensis and P. garckeanus to detailed phytochemical characterisation to purify and identify the compounds responsible for the observed antibacterial activity. Once candidate constituents are isolated, performing mechanism-of-action studies alongside comprehensive in vitro cytotoxicity screening on relevant mammalian cell lines to establish therapeutic index and safety margins is warranted. Expand the antimicrobial panel to include a wider range of Gram-positive and Gram-negative species — importantly, contemporary multidrug-resistant clinical isolates and determine MICs, MBCs and synergy with standard antibiotics where appropriate. Finally, if potency and safety data are favourable, explore early formulation work for topical and oral dosage forms, together with preliminary pharmacokinetic and local tolerability considerations, so the most active fractions or isolated molecules can be advanced toward translational evaluation in traditional or modern therapeutic contexts.
Footnotes
Abbreviations
Acknowledgements
The author(s) gratefully acknowledge the technical support provided by Hawassa University Research and Collaboration vice president. We appreciate the technical assistance of Solomon Sintayehu Lecturer of Geography and Environmental Studies at Hawassa College of Teacher Education in the creation of the maps presented in this manuscript.
Ethical Approval
An ethical clearance to conduct this study was obtained from the Hawassa University institutional review board after the aim and benefits of the study were adequately explained (Ref. №IRB/280/16).
Statement of Informed Consent
This study does not involve human subjects, and informed consent is not applicable.
Authors contributions
BG, SD, MD, SF, NT, HE and TG: conceptualized the study. BG and SD experimented and collected data. SD, SF and NT: performed the analysis and interpretation of the data. BG, NT, SD: wrote the original manuscript. All authors have made an intellectual contribution to the work and have approved the final version of the manuscript for submission.
Funding
This research did receive a fund from Hawassa University Research and collaboration vice president.
Declaration of Conflicting Interests
Data Availability
All data obtained in this study are included in the manuscript.

