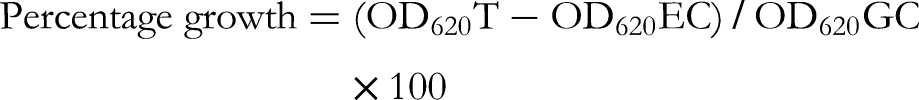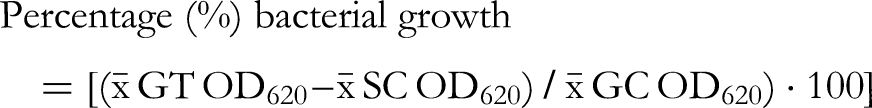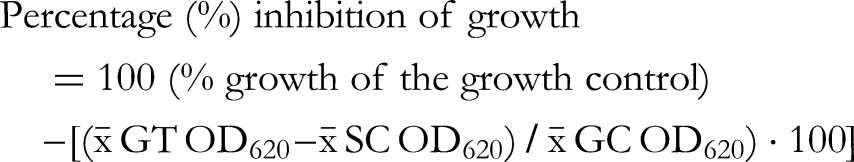Abstract
Bacillus cereus is a gram-positive foodborne enterotoxigenic bacterium that can form endospores and biofilms to resist heating and the effects of antibiotics. The emergence of multidrug-resistant B. cereus strains which are typically resistant to β-lactam antibiotics have made it difficult to treat foodborne bacterial infections. In this study, we investigated the antibiotic potentiating effects of methanol extracts of Salix aurita and Salix myrsinifolia twigs on tetracycline and ciprofloxacin hydrochloride against B. cereus. A two-dimensional checkerboard method was used, and the fractional inhibitory concentration (FIC) index was used to ascertain the synergy. The FIC index values ranged between 0.133–2.0 in all the tested concentration ratios of the antibiotic combinations. Combinations containing low concentrations of both plant species and tetracycline as well as S. aurita and ciprofloxacin gave strong synergistic interactions when compared with combinations containing a high concentration of both the antibiotic and the plant extract. The most significant synergistic effects for tetracycline in combination with the Salix extracts were obtained with the combinations containing 0.061 µg/ml of tetracycline with 19.56 µg/ml of the S. aurita extract and 0.061 µg/ml of tetracycline with 9.77 µg/ml of the S. myrsinifolia extract, both showing a FIC index of 0.133 and resulting in an 8-fold reduction of the MIC of tetracycline alone (MIC 0.488 µg/ml). Besides, a combination containing tetracycline at 0.015 µg/ml and the S. aurita extract at 625 µg/ml gave a synergistic effect resulting in a 32-fold reduction of the MIC of tetracycline. The best combination obtained with ciprofloxacin was observed for a combination containing 15 ng/ml of ciprofloxacin with 313 µg/ml of the S. aurita extract (FIC index 0.250). S. aurita and S. myrsinifolia extracts greatly enhanced the antibacterial efficacy of tetracycline and could be promising antibiotic adjuvants.
Introduction
Antibiotic resistance resulting from improper use and misuse of antibiotics is a public health problem that requires urgent intervention worldwide.1–3 Globally, the mortality rate as a result of treatment failures associated with the emergence of multidrug-resistant Bacillus species and other antibiotic-resistant pathogens, including the ESKAPE bacteria are increasing rapidly.4,5 Bacteria develop resistance and completely alter the efficacy of antibiotic therapies and treatments by resisting the drugs thereby making it difficult to find an antibiotic with long lasting efficacy.6–8 Till date, antibiotics resistant bacteria still pose the greatest risk to humans and animal health.5,9,10 According to the World Health Organization (WHO), the new challenges posed by antibiotic resistant bacteria in the health and medical sector is alarming and this has made infectious diseases more difficult to treat thereby making a common infection to develop into a life-threatening disease.11,12 In recent times, due to this increasing rate of antibiotic resistance and the need for novel and highly effective antibacterial agents from natural products, including traditional medicinal plants, to combat antibiotic resistant pathogens and to minimize adverse drug effects, alternative therapies such as combination therapies, including plant-based extracts and compounds, are constantly of high research interest.9,13
B. cereus is a Gram-positive opportunistic foodborne pathogen which is common in raw and processed foods including fish, meat, milk, rice and noodles and is typically resistant to β-lactam antibiotics and capable of easily developing resistance to other conventional antibiotics such as ciprofloxacin and tetracycline,14–16 of which ciprofloxacin is used as a standard treatment of B. cereus infections in immunocompetent patients. 17 B. cereus is the primary cause of foodborne diseases due to its production of enterotoxins which is often characterized by abdominal pain and watery diarrhea.15,18,19 B. cereus produces haemolysin (Hbl), and non-haemolysin enterotoxins (Nhe) which target cells and tissues thereby causing septicemia, emetic syndromes and diarrhea.20–22 Furthermore, B. cereus can form heat resistant spores making it resistant to the heating of food and biofilms increasing its resistance to antibiotics, thereby leading to an ineffective treatment.19,23,24 Recent pharmacological studies have shown that B. cereus is responsible for 1%–22% of foodborne disease outbreaks globally.22,25
Combinational antibacterial chemotherapies that allow synergistic interaction between plant extracts/bioactive compounds and conventional antibiotics is one of the most effective methods to minimize resistance.9,26 Plant-derived compounds and plant extracts have been known to be potentiators of antibiotics in the fight against antibiotic resistant pathogens and in a bid to minimize the drug/dose-related side effects associated with the constant use of conventional antibiotics.27,28 They could re-purpose conventional antibiotics and effectively enhance the treatment of infectious diseases.29,30 These plants-derived compounds/extracts which are biologically active compounds are being explored as potential therapeutic agents to develop new antibiotic therapies to address the problem of persistent infectious diseases caused by antibiotic resistant pathogens.9,31,32 Furthermore, research has shown that combination of bioactive compounds/extracts from medicinal plants with antibiotics could improve the effectiveness of the antibiotics with their target in the pathogen thereby reducing the emergence of resistance.27,33 Synergistic interactions of the components in combination therapies, consisting of plant extracts/compounds and antibiotics could be used to restore or improve the effects of conventional antibiotics against resistant bacteria by increasing the efficiency of the antibiotics, for example with the aid of efflux pump inhibitors present in plant extracts, and reducing the adverse effects of the antibiotics as they can be used in smaller concentrations in the combinations.9,34–36 Therefore, to modulate B. cereus drug resistance, extracts from Salix aurita and S. myrsinifolia could be valuable targets.
The genus Salix comprising of S. aurita and S. myrsinifolia among around 500 other species, is a large genus of perennial woody plants that belongs to the Salicaceae family and are native to North America, Europe, Africa, and Asia.37,38 Salix extracts and most especially the barks, especially from S. alba L. and S. purpurea L., are known phytopharmaceutical products used globally in pharmaceutical industries and several Salix species are also used traditionally as herbal remedy in the treatment of inflammation, fever, rheumatic disorders and headaches.38–40 Salix species have a high content of polyphenolic compounds such as flavonoids, proanthocyanidins, salicylates and phenolic acid derivatives. 38 In our previous studies, we have found that S. aurita and S. myrsinifolia twig methanol extracts gave good antimicrobial effects against B. cereus and S. aureus.41,42 Besides, some other Salix species and extracts have been reported to show antibacterial activity alone, including hot water extracts of S. myrsinifolia and S. phylicifolia.43,44 However, studies on combination effects of Salix extracts with antibiotics are rare. 45
S. myrsinifolia Salisb., also known as dark-leaved willow is a small tree that grows in damp mixed forest habitats, and it is native to Europe and western Siberia. 46 As all other willow species, S. myrsinifolia is dioecious, meaning that male and female inflorescences (catkins) occur on separate individuals, and besides males and females were found to differ somewhat from each other in their secondary metabolite quantities. 46 S. aurita L., commonly called eared willow, is a small shrub that could grow between 1 to 3 meters in height, occurring in wetlands, mire patches in forests or on rock outcrops, on field margins and roadsides. 47 S. aurita and S. myrsinifolia are rich natural sources of phenolic secondary metabolites38,41,42,48,49 and therefore could be good alternatives for antibacterial therapies either alone or when compared to synthetic therapeutic therapies in a bid to prevent antibiotic resistance.
Previous research has shown that extracts of the leaves of the African species, Salix ledermannii in combination with ciprofloxacin showed significant and remarkable synergistic effects against multidrug-resistant bacteria, including Escherichia coli, Enterobacter aerogenes, Klebsiella pneumoniae, Providencia stuartii, Pseudomonas aeruginosa, and Staphylococcus aureus. 45 However, there are no reported information on the antibiotic potentiating effects of S. aurita and S. myrsinifolia. As part of our continuous search for new antimicrobial extracts, compounds and adjuvants from Salix species growing naturally in Finland, we evaluated the in vitro antibiotic potentiating effects of the methanol extracts of the winter-dormant twigs of S. aurita and S. myrsinifolia against B. cereus. The aim was to investigate the synergistic effect of the methanol extracts of S. aurita and S. myrsinifolia in combination with tetracycline and ciprofloxacin against B. cereus. Testing the synergistic effects of these extracts in combination with conventional antimicrobials could be a good strategy to repurpose these antibiotics that have lost their efficacy due to resistance. Also, it could be a way to minimize the drug/dose-related side effects associated with the use of these conventional antibiotics since lower concentrations of both the antibiotic and the extract could be used (Figure 1).

(A) Salix aurita with characteristic obovate leaves with a bent apex. This specimen was collected from Seurasaari, Helsinki. (B) Salix myrsinifolia male catkins on twigs collected in May 2022 from the Lammi region, Finland. Photos: Pia Fyhrqvist.
Materials and Methods
Plant Collection and Authentication
S. aurita and S. myrsinifolia winter-dormant twigs were collected in April 2022 from Helsinki and Lammi regions, respectively, in Finland (Figure 1). The collected twig samples were spread in the laboratory table for three weeks to dry in room temperature and then cut into smaller pieces for easy milling. The dried twig samples were milled using a grinding machine (AURUM-PHARMAKON Oy Apta Ab, PELITEOS Oy No 7018, Helsinki, Finland) and the milled powdered plant samples were stored in paper bags, labeled properly, and ready for extraction. A voucher specimen was collected from each of the plants in May–June 2022 when the leaves were fully developed for proper authentication. The collected voucher samples were taken to the Finnish Museum of Natural History, University of Helsinki, Finland where the expert in botany and taxonomy, Dr Henry Väre, Curator of the Botanical Museum identified and authenticated the samples. The identified and authenticated voucher specimens were deposited at the herbarium of the Botanical Museum, University of Helsinki, Finland with specimen voucher numbers of H856575 for S. myrsinifolia and H856582 for S. aurita.
Methanol Extraction
Methanol extraction of the S. aurita and S. myrsinifolia twig samples were done using analytical grade methanol (Fisher Scientific). 20 grams of each of the powdered twig samples were extracted with a 500 mL volume of 100% methanol in a big Erlenmeyer flask (Darmstadt, Germany). Extraction was performed overnight using a magnetic stirrer (RCT, digital, Staufen, Germany). After 24 h, the mixture was filtered using the filtration Büchner funnel kit vacuum suction glass flask apparatus and Whatman filter paper (Schleicher & Schuell, ø 150 mm, Dassel, Germany). The filtrates were collected and transferred into a round bottomed flask of known weight and then the methanol was evaporated using a rotary evaporator apparatus (Rotavapor, Heidolph VV2000, Schwabach, Germany) combined with a water bath set at a temperature of 40 °C. The filtrates were covered with perforated parafilm, and stored at −20 °C. Afterwards, the frozen filtrates were lyophilized for 3 days to completely freeze dry using a SCANVAC Coolsafe 110-4 Pro lyophilizer (Labogene, Denmark).
Bacterial Strain and Antibiotics
B. cereus ATCC 10987 collected from the Division of Pharmaceutical Biosciences, Faculty of Pharmacy, University of Helsinki, Finland was used for the in vitro synergistic evaluation of the twig extracts of S. aurita and S, myrsinifolia. Tetracycline hydrochloride (Sigma-Aldrich, St. Louis MO, USA) and ciprofloxacin hydrochloride (Sigma-Aldrich, St. Louis MO, USA) were used as the conventional antibiotics for the synergistic antibacterial investigation.
Antibacterial and Antibiotic Potentiating Testing
Turbidimetric Microplate Screening Method
For the primary screening, a turbidimetric microplate method involving screening at one starting concentration was first conducted using sterile, transparent and flat-bottomed 96-well plates (Thermo Fisher Scientific) to evaluate if the Salix extracts were active at a starting concentration of 2500 µg/mL. Prior to the test, a few colonies of B. cereus were transferred to 20 mL of nutrient broth and grown for 24 h at 37 °C with shaking at 200 RPM in an orbital incubator (Stuart® SI500289, London, UK). To prepare the inoculum, 2 mL of the overnight grown bacterial culture was taken, and divided between two test tubes, with 1 mL in each. The absorbance or optical density (OD) of 1 mL of the overnight bacterial culture was measured at 625 nm, using a UV–visible spectrophotometer (Pharmacia LKB-Biochrom 4060; Pfizer Inc., New York, NY, USA). According to the OD625 result, the suspension in the other test tube was diluted with Mueller–Hinton broth to reach an absorbance of 0.1 at 625 nm (containing approximately 1.0 × 108 CFU/mL). Furthermore, a 100-fold dilution was prepared by taking 100 µL from the diluted A625 = 0.1 suspension and further diluting it in 9.9 mL of Mueller–Hinton broth to obtain the inoculum containing approximately 1 × 106 CFU/mL, as recommended by the Clinical Laboratory Standards Institute. 50 A total of 100 µL of this inoculum, combined with 100 µL of the plant extracts (5 mg/mL) or 100 µL antibiotics (1 mg/mL) were introduced into the wells of the 96 microwell plates to make a total volume of 200 µL per well. Methanol which was used as the solvent control was not toxic at a 5% (v/v) volume in the wells, which was the maximum volume of this solvent used. The growth control (GC wells) contained the bacterial suspension + broth, and the test wells (T wells) contained Salix plant extracts + bacterial suspension, while the extract control wells (EC wells) contained plant extracts and broth. These extract control wells were prepared for each plant extract to be tested to subtract the eventual light absorbance of extracts from the wells containing the corresponding extracts with bacteria. The microwell plates were incubated for 24 h in a BioSan incubator (Thermo-Shaker PST-60HL-4) at +37 °C, 350 RPM. The turbidity of the wells at 620 nm was recorded using a Multiskan Sky Microplate Spectrophotometer (Thermo Fisher Scientific) and the antibacterial activity was subsequently calculated as expressed as the mean percentage growth and growth inhibition of duplicate wells.
Also, the turbidimetric microplate method was used to evaluate the minimum inhibitory concentrations (MICs) using two-fold dilutions of the plant extracts. For the MIC evaluation, the Salix twig extract stock solutions (50 mg/mL in methanol) were first diluted 10-fold with sterile Mueller–Hinton broth to obtain a 5 mg/mL concentration. Two-fold serial dilutions were subsequently prepared in Eppendorf tubes (2 mL volume) starting from 2500 to 19 µg/mL using sterile Mueller–Hinton broth. For the antibiotics, a 1 mg/mL concentration was serially two-fold diluted in Eppendorf tubes (2 mL volume) starting from 500 to 0.007 µg/mL. After the completed test, performed as described above, the minimum inhibitory concentration was estimated from the spectrophotometric data as the smallest concentration that led to 90% or more growth inhibition, resulting in no visible growth. The absorbance of the broth was automatically subtracted from all test samples using the “zero” application in the Multiskan Sky (Thermo Fischer Scientific) spectrophotometer. The percentage growth was calculated according to the formula:
Two-Dimensional Broth Microdilution Checkerboard Method
The antibacterial effects of the extracts in combinations with conventional antibiotics were determined using the two-dimensional checkerboard method modified from Mordmuang et al. 51 Before the test, the plant extracts (methanol twig extracts of S. aurita and S. myrsinifolia) were prepared in Eppendorf tubes (2 ml volume) in concentrations relating to their MICs as follows: MIC, 1/2 × MIC, 1/4 × MIC, 1/8 × MIC, 1/16 × MIC, and 1/32 × MIC. The antibiotics, tetracycline hydrochloride (Sigma-Aldrich) and ciprofloxacin hydrochloride (Sigma-Aldrich) were prepared at 1/8 × MIC, 1/4 × MIC, 1/2 × MIC, and MIC concentrations. The concentration of the plant extracts and the antibiotics were prepared in such a way that their actual concentrations in the wells was one-fourth of that of the originally prepared concentrations in the Eppendorf tubes since in the checkerboard test, 50 µl of antibiotic, 50 µl of the plant extract and 100 µl of the bacterial suspension were pipetted into the microplate wells (200 µl, total volume in well/50 µl = 4). Prior to the test, a colony of the B. cereus was transferred from a Mueller-Hinton agar slant into 10 ml of Mueller-Hinton broth and the bacterial suspension was incubated overnight at 37 °C in an orbital incubator (Stuart® SI500289, London, UK) with shaking at 200 RPM. After 24 h, 2 mL of the overnight grown bacterial suspension was pipetted, and divided between two test tubes, with 1 mL in each. The absorbance or optical density (OD) of 1 mL of the overnight bacterial culture was measured at 625 nm, using a UV–visible spectrophotometer (Pharmacia LKB-Biochrom 4060; Pfizer Inc., New York, NY, USA). According to the OD625 result, the suspension in the other test tube was diluted with Mueller–Hinton broth to reach an absorbance of 0.1 at 625 nm (containing approximately 1.0 × 108 CFU/mL). Furthermore, a 100-fold dilution was prepared by taking 100 µL from the diluted A625 = 0.1 suspension and further diluting it in 9.9 mL of Mueller–Hinton broth to obtain the inoculum containing 1 × 106 CFU/mL, as recommended by the Clinical Laboratory Standards Institute. 50
To begin the test, 10 ml of the bacterial suspension (OD620 = 0.001) was poured into a multichannel pipette reagent reservoir. Also 10 ml of the Mueller–Hinton broth was poured into another multichannel pipette reagent reservoir. The checkerboard test was performed using two replicate microplates for each pair of plant extract and antibiotic; the other plate contained bacterium and the other was without the bacterium. Microplate A (containing bacteria and extract-antibiotic combinations as well as the growth control) was used as the test plate while microplate B contained broth without bacteria and the extract-antibiotic combinations corresponding to plate A. Plate B was used as a control plate to get absorbance measures of the extracts-antibiotics combinations that might absorb light themselves at 620 nm. For microplate A, 100 µl of the diluted bacterial suspension (containing approximately 1 × 106 CFU/ml) was pipetted from the reservoir containing bacterial suspension into all the wells except the rim wells of a 96-well microdilution plate using a multichannel pipette. 100 µl of broth was then pipetted from the other reservoir containing Mueller–Hinton broth into the growth control (GC) wells in column 2. For microplate B (control plate without bacteria), 100 µl of Mueller-Hinton broth was pipetted into all the wells of a 96-well microdilution plate using a multichannel pipette. A specific pipetting scheme was followed for the pair of plant extracts and antibiotics and pipetting was done accordingly for both microplate A (with bacteria) and B (control plate without bacteria). The antibiotics were added on the y-axis so that 50 µl of the two-fold diluted concentrations, with the highest concentration starting from the top left-hand corner of the 96-well microplate (calculated in relation to the MIC) were pipetted into the wells. Therefore, the concentrations in the microplate wells were ¼ of the concentration in the Eppendorf tubes, as each of the tested compounds were pipetted in a volume of 50 µl added to the 100 µl of bacterial suspension (50/200 = 0.25 = ¼). Also, 50 µl of various concentrations calculated according to the MIC of the plant extracts were pipetted into the rows of the wells of microplates A and B accordingly, so that the two-fold dilution pattern followed the x-axis of the plate, starting with the highest concentration from the left uppermost corner of the microplate. The microplates A and B were incubated in a plate shaker set at 37 °C and 350 RPM for 24 h. After 24 h, the optical density (OD) or turbidity of the wells at 620 nm was measured for each of the plates using a Multiskan Sky Microplate Spectrophotometer (Thermo Fisher Scientific). The optical density values for the combination wells in plate A were subtracted from each of their corresponding wells (containing the same extract-antibiotic combinations) in the control plate B. The percentage growth was calculated by dividing the final OD620 of the plant extracts and antibiotics combinations with the OD620 of the growth control × 100. Then the percentage growth inhibition was calculated by subtracting the percentage growth results from 100. The following formulas were used for the percentage growth and growth inhibition calculations:
Fractional Inhibitory Concentration index Calculations
From the results obtained from the two-dimensional broth microdilution checkerboard analysis, the FIC index was calculated. The FIC index is used to describe the nature of the interaction of the agents in a combination and is especially used to find combinations in which the agents enhance the effects of each other, but also to rule out combinations in which the agents antagonize the effects of each other. The FIC values relate the MIC of a combination to the MIC of the respective agents alone and thus the FIC value indicates how much more or less active a combination is compared to the MIC of the single agents in the combination. The FIC index was calculated for all combinations resulting in ≥90% growth inhibition. For the FIC index calculation, the MIC values of the antibiotics in the combination and the MIC values of the Salix extract in the combination were used to calculate FIC (A) and FIC (B), using the equation below:
The FIC values were calculated as follows:
The modulation factor indicates the gain of activity of the antibiotics when combined with the Salix extract compared to MIC of the antibiotic alone and was calculated according to Fankam et al.
6
The Modulation Factor (MF) is calculated as follows:
Statistical Analysis
The checkerboard experiments were performed in duplicate experiments with two pairs of identical microplates (two test plates with bacteria and agent combinations and two control plates containing the agent combinations in broth without the bacteria) under the same experimental conditions. The percentage growth inhibition was obtained from the data of the duplicate experiments (n = 2) using a Microsoft Excel program to calculate the mean OD620 and the standard error of mean (SEM) of the duplicates.
Results
The antibiotic potentiating effects of S. aurita and S. myrsinifolia methanol extracts of winter-dormant twigs were evaluated in combinations with tetracycline hydrochloride or ciprofloxacin hydrochloride against B. cereus. In our previous research, we found that both Salix species are antibacterial against B. cereus, with MIC values of 1250 and 2500 µg/ml, for S. myrsinifolia and S aurita, respectively and the growth inhibition was dose-dependent,41,42 (Figure 2A and B). The MIC value of tetracycline was found to be 0.488 µg/ml while that of ciprofloxacin was 120 ng/ml against B. cereus. Based on these growth inhibitory effects, S. aurita and S. myrsinifolia were chosen to test their effects in combinations with the conventional antibiotics, tetracycline hydrochloride and ciprofloxacin hydrochloride. The aim was to evaluate at which doses the Salix extracts can potentiate the effects of the antibiotics and to find effective combinations with synergistic or additive effects containing a minimum concentration of antibiotics that could serve as an alternative way to minimize antibiotic resistance and at the same time improve the efficacy of these conventional antibiotics against B. cereus infections. Especially, the possibility to find combinations containing low concentrations of both Salix extracts and antibiotics while still showing synergistic or additive effects was explored.

(A) Dose-response effects of a Salix myrsinifolia methanol twig extract against B. cereus. The results are expressed as the means of duplicates ± SEM. (B) Dose-response effects of Salix aurita methanol extract against B. cereus. The results are expressed as the means of duplicates ± SEM.
Tables 1–4 show the results of the interactions of the Salix extracts with tetracycline hydrochloride and ciprofloxacin hydrochloride against B. cereus. Altogether, from the checkerboard calculations, the FIC index values ranged between 0.133–2.0 for the antibiotic-plant extract combinations, and synergy, additivity, or indifferent effects were observed at varying concentrations of the antibiotic combinations (Tables 1–4). No antagonistic effect was observed in any of the tested combinations resulting in ≥90% growth inhibition, as depicted from the FIC calculations. However, this could be due to that 2 × MIC and 4 × MIC concentrations of the extracts and antibiotics in the combinations were not tested.
Checkerboard Calculation Results Showing the FIC index and Potentiating Effects of a S. aurita Methanol Twig Extract with Tetracycline Hydrochloride Against B. cereus. The Modulation Factor Indicates the Gain of Activity of Tetracycline Hydrochloride When Combined with the S. aurita Extract Compared to the MIC of Tetracycline Alone. The FIC index was Calculated for Combinations Resulting in ≥90% Growth Inhibition.
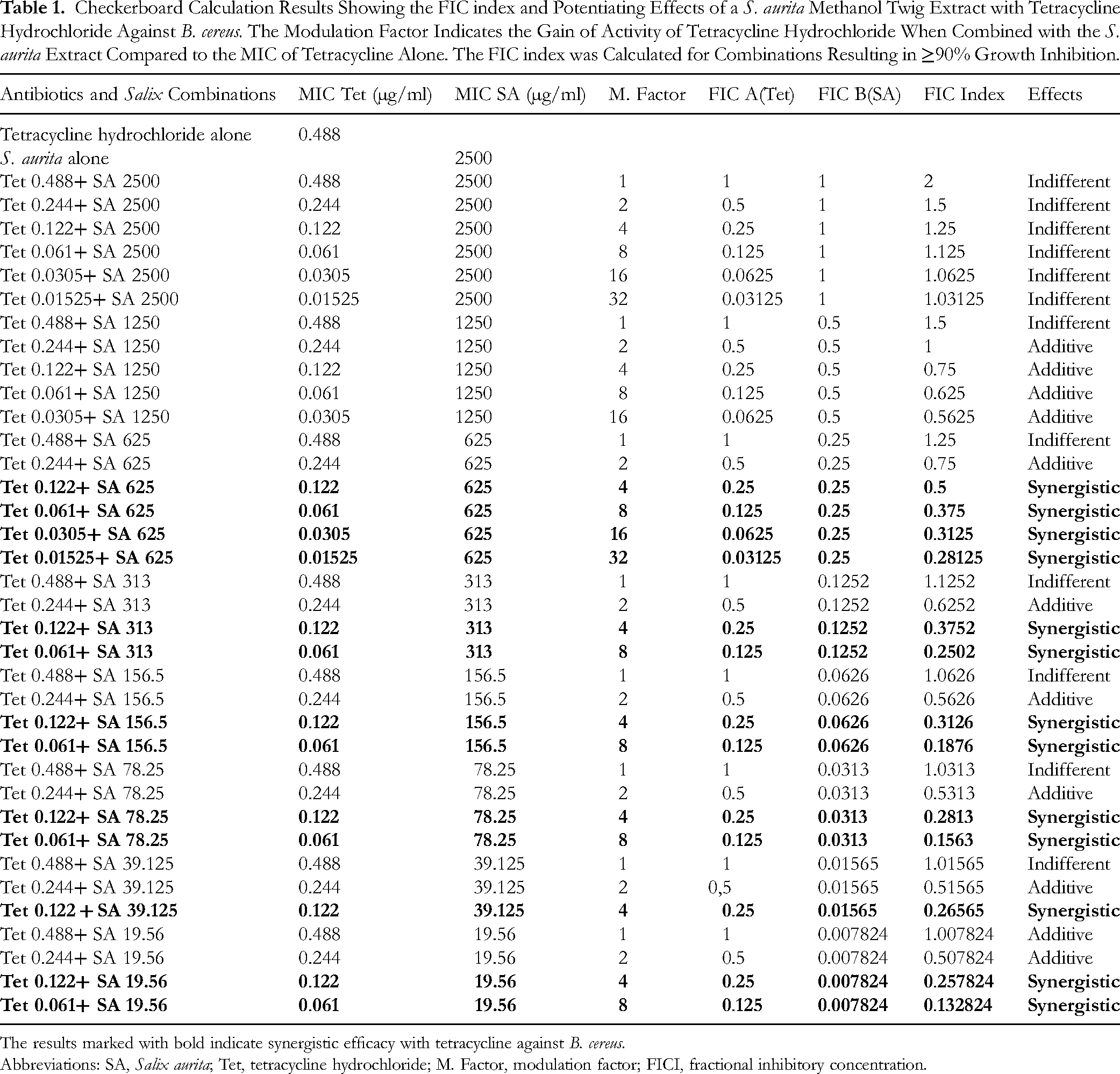
The results marked with bold indicate synergistic efficacy with tetracycline against B. cereus.
Abbreviations: SA, Salix aurita; Tet, tetracycline hydrochloride; M. Factor, modulation factor; FICI, fractional inhibitory concentration.
Checkerboard Calculation Results Showing the FIC index and Potentiating Effects of a S. myrsinifolia Methanol Extract with Tetracycline Hydrochloride Against B. cereus. The Modulation Factor Indicates the Gain of Activity of Tetracycline Hydrochloride When Combined with the S. myrsinifolia Extract Compared to the MIC of Tetracycline Alone.

The results marked with bold indicate synergistic efficacy with tetracycline against B. cereus.
Abbreviations: SM, Salix myrsinifolia; Tet, tetracycline hydrochloride; M. Factor, modulation factor; FICI, fractional inhibitory concentration.
Checkerboard Calculation Results Showing the FIC index and Potentiating Effects of S. aurita Methanol Extract with Ciprofloxacin Hydrochloride Against B. cereus. The Modulation Factor Indicates the Gain of Activity of Ciprofloxacin When Combined with the S. aurita Extract Compared to MIC of Ciprofloxacin Alone.

The results marked with bold indicate synergistic efficacy with ciprofloxacin against B. cereus.
Abbreviations: SA, Salix aurita; Cip, ciprofloxacin hydrochloride; M. Factor, modulation factor; FICI, fractional inhibitory concentration.
Checkerboard Calculation Results Showing the FIC index and Potentiating Effects of a S. myrsinifolia Methanol Extract with Ciprofloxacin Hydrochloride Against B. cereus. The FIC index was Calculated for Combinations Resulting in ≥90% Growth Inhibition. The Modulation Factor Indicates the Gain of Activity of Ciprofloxacin Hydrochloride When Combined with the S. myrsinifolia Extract Compared to MIC of Ciprofloxacin Hydrochloride Alone.

The results marked with bold indicate additive efficacy with ciprofloxacin hydrochloride against B. cereus.
Abbreviations: SM, Salix myrsinifolia extract; Cip, ciprofloxacin hydrochloride; M. Factor, modulation factor; FICI, fractional inhibitory concentration.
Combination Effects of S. aurita and S. myrsinifolia with Tetracycline Hydrochloride
For the combination effects of the methanol extract of S. aurita twigs with tetracycline hydrochloride, the FIC index value showed thirteen synergistic interactions between the S. aurita extract and tetracycline (Table 1, Figure S1). Thirteen combinations containing 1/2× MIC – 1/8 × MIC of tetracycline gave synergistic antibacterial effects when combined with various subinhibitory concentrations of an S. aurita extract, with extract concentrations in the combinations ranging from 625 to 19.56 µg/ml (Table 1). From the results of the FIC index calculation of the various combinations of S. aurita methanol extract with tetracycline hydrochloride, the best synergistic effects in terms of combinations containing low concentrations of both the antibiotic and the extract were obtained with the combinations containing 0.061 µg/ml of tetracycline hydrochloride and 19.56 µg/ml of the S. aurita extract (FIC index 0.133), followed by the combination containing 0.061 µg/ml of tetracycline hydrochloride and 78.25 µg/ml of the S. aurita extract (FIC index 0.156), and finally a combination containing 0.061 µg/ml of tetracycline and 156.5 µg/ml of the S. aurita extract (FIC index 0.188). Besides, 0.015 µg/ml was the lowest concentration of tetracycline hydrochloride resulting in a synergistic effect when combined with the S. aurita extract at 625 µg/ml and thus implying a 32-fold reduction of the MIC of tetracycline alone (0.488 µg/ml) against B. cereus (Table 1). Also, eleven additive effects were recorded between the S. aurita extract and tetracycline hydrochloride in combinations containing both tetracycline and the S. aurita extract in subinhibitory concentrations. The combinations containing the MIC of tetracycline (0.488 µg/ml) and the MIC of S. aurita extract (2500 µg/ml) all gave indifferent effects (Table 1).
For the combination of S. myrsinifolia with tetracycline hydrochloride, eleven synergistic interactions were observed at various combinations containing 1/2 × MIC – 1/8 × MIC of tetracycline hydrochloride with S. myrsinifolia (Table 2, Figure S2). The optimal (meaning the combinations containing the smallest concentrations of extract and antibiotic) synergistic effects for the S. myrsinifolia extract were observed with the combinations containing 0.061 µg/ml of tetracycline and 9.77 µg/ml of the S. myrsinifolia extract (FIC index 0.133), followed by the combination containing 0.061 µg/ml of tetracycline with 19.56 µg/ml of the S. myrsinifolia extract (FIC index 0.141), and a combination containing 0.061 µg/ml of tetracycline with 39.125 µg/ml of the S. myrsinifolia extract (FIC index 0.156). Twelve additive effects were observed for various combinations of S. myrsinifolia with tetracycline hydrochloride (Table 2).
Combination Effects of S. aurita and S. myrsinifolia with Ciprofloxacin Hydrochloride
In this study, the evaluation of the effects of S. aurita on ciprofloxacin hydrochloride gave seven synergistic effects against B. cereus (Table 3, Figure S3). For ciprofloxacin hydrochloride with S. aurita, synergistic effects were observed with combinations containing 15 ng/ml of ciprofloxacin hydrochloride and 313 µg/ml of the S. aurita extract (FIC index 0.250), followed by the combination containing 7.5 ng/ml of ciprofloxacin hydrochloride with 625 µg/ml of S. aurita extract, and then the combination containing 30 ng/ml of ciprofloxacin with 156.5 µg/ml of the S. aurita extract of which the two last mentioned combinations gave low FIC indexes of 0.313 (Table 3).
Notably, S. myrsinifolia in combination with ciprofloxacin hydrochloride in all the tested combinations of various MIC and subinhibitory concentrations did not show synergistic activity against B. cereus (Table 4, Figure S4). The effects were either additive, with ten combinations showing additive effects, or indifferent.
Discussion
B. cereus has mostly received marginal attention in clinical microbiology, although apart from causing foodborne diseases, it can also cause local and systemic infections in both immunocompetent and immunodeficient patients, including soft-tissue and cutaneous infections, respiratory infections, osteomyelitis, central nervous system infections and blood stream infections. 53 B. cereus is becoming increasingly resistant to most conventional antibacterial drugs and most especially the β-lactam antibiotics, and in addition also rifampicin, tetracycline and ciprofloxacin-resistance was reported and this trend of increasing resistance poses a significant risk in the fight against gastrointestinal and foodborne diseases as well as other infections caused by B. cereus.15,16,54,55 More so, the constant use of high doses of conventional antibiotics in the treatment of infections could lead to increased toxicity to normal body cells and could also be characterized with numerous adverse effects, including the high risk for development of antimicrobial resistance (AMR) and adversely affecting the gut microbiome leading to dysbiosis.56,57 Recent interest in natural products, especially plant-derived extracts and compounds, has highlighted their potential in combination therapies to combat resistant bacteria.35,58 The combinational approach enables a faster and cheaper development of novel antibiotic therapies, compared to the development of novel antibiotic molecules which might need decades to be fully developed. 9 The drug/natural product combinational approach has been reported to be one of the most effective strategies to reduce toxicity and resistance thereby increasing the efficacy of an antibacterial drug 59 and several plant extracts and plant-derived compounds, including some flavonoids were found to show antibiotic potentiating and/or restoring effects. 58 While some plant special metabolites have intrinsic antimicrobial activity, others target bacterial virulence, including efflux pumps and quorum-sensing systems among others, and thereby when combined with antibiotics at subinhibitory concentration they could improve antibiotic effectiveness. In the current study, the winter- dormant twigs of two Salix species; S. aurita and S. myrsinifolia, were chosen for our screenings on their antibacterial effects in combination with conventional antibiotics as dormant twigs contain more polyphenols and phenolic glucosides when compared to the summer twigs,60,61 which could significantly affect their antibiotic potentiating activity. In this present study, we found that S. aurita in combination with varying concentrations of tetracycline hydrochloride showed thirteen synergistic interactions and significantly improved the antibacterial activity of tetracycline hydrochloride against B. cereus (Table 1, Figure S1). The most potent synergistic interaction with the lowest FIC index value was found for a combination containing as low concentration as 0.061 µg/ml of tetracycline and 19.56 µg/ml of the S. aurita extract (FIC index 0.133). Thus, in this combination, the S. aurita extract at its subinhibitory concentration of 19.56 µg/ml allowed tetracycline hydrochloride to be active at 61 ng/ml, and thus tetracycline hydrochloride was eight (8) times more active in this combination compared to when used alone (MIC 0.488 µg/ml). Tetracycline is a widely used antibacterial drug which has been reported to cause adverse effects such as hearing impairment, connective tissue diseases, psychiatric disorders, as well as blood and lymphatic system diseases. 62 Our study indicates that S. aurita methanol twig extracts could serve as a natural modulator of antibiotic resistance to address the problem of tetracycline resistant strains of B. cereus. 15 The significant synergistic activity recorded in this study shows that the dosage combination of a low concentration of S. aurita with a low concentration of tetracycline hydrochloride could be a better option with minimum adverse effects for the treatment of infections caused by B. cereus. To the best of our knowledge, this present study is the first report on the antibiotic potentiating activity of a S. aurita twig extract against B. cereus. Also, in this present investigation, we found that a S. myrsinifolia twig methanol extract improved the effects of tetracycline hydrochloride against B. cereus at various concentration combinations. Altogether eleven combinations of S. myrsinifolia and tetracycline hydrochloride at subinhibitory concentrations (1/2 × MIC – 1/8 × MIC) showed synergistic effects against B. cereus. A significant synergistic interaction was observed for a combination containing 0.061 µg/ml of tetracycline and 9.77 µg/ml of the S. myrsinifolia extract (Table 2). From the results of this study, it could be seen that S. aurita and S. myrsinifolia extracts improved the effectiveness of tetracycline hydrochloride against B. cereus at very low concentrations of tetracycline in the combination. However, in combinations containing higher concentrations, especially of the willow extracts, the effects for both S. aurita and S. myrsinifolia with tetracycline hydrochloride were either additive or indifferent. The result of this study shows that S. aurita and S. myrsinifolia enhanced the overall antibacterial activity of tetracycline hydrochloride and enabled the use of very small concentrations of this antibiotic to inhibit all visible growth of B. cereus. Therefore, the result demonstrates that S. aurita and S. myrsinifolia could act as resistance modulating agents to restore or improve the efficacy of tetracycline against B. cereus, as strains of this bacterium were reported to develop high resistance also against tetracycline.15,20,63
In this present investigation, we found that a S. aurita twig methanol extract enhanced the antimicrobial effects of ciprofloxacin hydrochloride, and seven extract-antibiotic combinations gave synergistic effects (Table 3, Figure S3). The optimal synergistic interaction was observed with the combinations containing 15 ng/ml of ciprofloxacin with 313 µg/ml of the S. aurita extract (FIC index 0.250) (Table 3). However, the S. aurita extract was more active with tetracycline hydrochloride than with ciprofloxacin hydrochloride. Ciprofloxacin is an antibiotic used in the treatment of numerous bacterial infections, and it is often characterized by adverse effects such as tendinitis, insomnia, depression and other psychotic symptoms. 64 The synergistic activity recorded in this study agrees with the findings of Demgne et al., 45 who reported that some African medicinal plant extracts are potentiators of ciprofloxacin; among the nine medicinal plants tested in combination with ciprofloxacin, a methanol extract of the leaves of Salix ledermannii gave the highest synergistic effects against multidrug-resistant bacteria. Notwithstanding that a previous research has shown that Bacillus species may have lower sensitivity to ciprofloxacin than to other antibiotics, 4 through our study, it is interesting to note that certain combinations of the S. aurita extract with ciprofloxacin at very low concentrations could enhance the antibacterial activity against B. cereus and this implies that S. aurita may reduce the drug/dose-related side effects associated with the frequent use of this conventional antibiotic. This result further suggests that extracts from S. aurita in combination with ciprofloxacin could be used to produce a new drug for possible alternative antibacterial treatment against infections caused by B. cereus.
In contrast to S. aurita, the S. myrsinifolia extract in combination with ciprofloxacin were devoid of synergy. The FIC index values indicated additive or indifferent effects in all the tested combinations. Thus, our result demonstrates that antibiotic potentiating effects of Salix spp. may vary depending on the species and that the S. myrsinifolia extract, when used in conjunction with ciprofloxacin as a combinational therapeutic alternative approach to conventional antibiotics will not be as effective as the S. aurita-ciprofloxacin combinations in the treatment of B. cereus related infections.
Previous research, including ours, has demonstrated that S. aurita and S. myrsinifolia are rich in polyphenols, including proanthocyanidins, flavonoids and phenolic glucosides41,42,46,48 and these compounds could be responsible for the significant synergistic efficacy that was observed in this present study when combining twig extracts with tetracycline and ciprofloxacin hydrochloride against B. cereus. We found that a S. aurita extract contained mostly proanthocyanidins such as procyanidin B1 and its monomer catechin, procyanidin C1 and other procyanidin derivatives. 41 Similarly, our previous investigation indicated that a S. myrsinifolia methanol extract contained taxifolin, procyanidin B1, salicin-7-sulfate, catechin, p-hydroxycinnamic acid, and salicin. 42 This present study suggests that the polyphenols in S. aurita and S. myrsinifolia potentiate the effects of tetracycline and ciprofloxacin against B. cereus and thus polyphenol combinations or some of the polyphenols present in these Salix species could be valuable targets for novel combinational antibiotic therapies. Furthermore, previous studies have identified synergistic interactions between the bioactive compounds from Salix species and antibiotics, for example, the combination of catechin with ciprofloxacin was found to show synergistic efficacy against chronic bacterial prostatitis in adult male Wistar rats. 65 Extracts from S. aurita and S. myrsinifolia could be good combinational therapeutic options with tetracycline or ciprofloxacin in the treatment of bacterial infections caused by B. cereus since only few combinational antibiotic therapies containing plant-derived components are clinically available.9,34
Furthermore, our previous studies on the time-kill effects of various subinhibitory concentrations of the methanol extracts of S. aurita and S. myrsinifolia twigs against B. cereus, indicated that the extracts are active also when used alone, and showed growth inhibitory actions against B. cereus at all investigated time-points until 24 h of incubation.41,42 The S. aurita extract was bactericidal at the MIC concentration (2500 µg/mL) and at 0.5 × MIC (1250 µg/mL) and totally inhibited the growth of B. cereus at all time-points until 24 h of incubation but was bacteriostatic at a lower concentration of 0.25 × MIC (625 µg/mL). 41 Also, S. myrsinifolia was bactericidal at 2 × MIC (2500 µg/mL) and at MIC (1250 µg/mL) but was bacteriostatic at a lower concentration of 0.5 × MIC. 42 However, in this present study, from the results of the FIC index calculations and modulation effects in Tables 1 and 2, S. aurita and S. myrsinifolia extracts, even at very low concentrations (625-19.56 µg/ml) in combinations with tetracycline hydrochloride (0.122-0.0305 µg/ml) showed mostly potent synergistic effects. For a drug combination to show synergistic effects, the effect of the combination is greater than the sum of the individual single compounds tested. These significant synergistic effects observed in our combination study at very low concentrations of both extracts and antibiotics in some of the combinations indicates that the Salix extracts are more effective when used as a combinational therapy against bacterial infections as compared to the antimicrobial effects of the extracts alone and could be employed to reduce drug adverse effects associated with conventional antibiotics such as tetracycline and ciprofloxacin.
Although this present study resulted in the finding of Salix extract-antibiotic combinations with promising combinational effects against B. cereus there are some limitations of the utilization of combination preparations containing plant extracts and antibiotics as antimicrobial drugs. Plant extracts are complex mixtures of compounds, and the phytochemical composition and quantity of compounds in the extracts vary according to the extraction methods used as well as due to the individual, seasonal and geographic variations between the source plants in terms of their contents of special metabolites. 66 Due to the complexity and variation of the compounds in the extracts, it is difficult to assess the active compounds in the combination mixture as well as their correct dosage. Moreover, for the in vitro checkerboard method, the lack of a standardized interpretation of the results is a drawback and the interpretation of the results remains difficult. 67 For example, there is no explanation on how to calculate the FIC index from microplate wells containing combinations of two agents at their MIC or below and showing antagonistic effects. Thus, the FIC index calculation can miss these obvious antagonistic effects of combinations containing subinhibitory concentrations of the agents in the combination. In addition, in the checkerboard method, the use of two-fold dilutions for the combinations creates an uneven (nonlinear) concentration gradient, that might lead to false positive or negative synergistic or antagonistic interpretations. 68 This could be overcome by using smaller dilution factors, such as 1.5 times steps between the dilutions, and complementing the checkerboard assays with time-kill assays. 69 Moreover, to more precisely mirror the true effects of a combination remedy, in vitro and in vivo studies should be combined.67,68 Novel, alternative in vivo models are promising, such as the use of Caenorhabditis elegans to study the in vivo effects of combinations. 70 Besides, the multiple drugs in the combination can amplify the adverse side effects of each other, and the combination drug might need a complex dosage administration. 71 Thus, a careful evaluation of the toxicity profile of the combination drug is needed. Unfortunately, in this present study, we could not evaluate the cytotoxicity effects of S. aurita and S. myrsinifolia twig extracts and that of the most effective extract-antibiotics combinations due to time constraints. This will be further evaluated in depth both against cell lines and in vivo using Caenorhabditis elegans. Salix extracts could serve as natural modulators of antibiotic resistance to address the problem of tetracycline resistant strains of B. cereus.
Conclusion
This study has given an insight into possible novel plant extract-antibiotic combination strategies that could contribute to the finding of new therapies to fight antibiotic resistance by providing a faster solution than creating novel molecules for antibiotics, since combinations containing low concentrations of both the antibiotics and the S. aurita and S. myrsinifolia extracts showed significant synergistic effects against B. cereus with 8 to 32-fold reductions in the MIC values of the antibiotics in the combinations. Our results demonstrate that especially a S. aurita methanol twig extract in certain combinations with tetracycline or ciprofloxacin hydrochloride could be promising antibiotic potentiating agents for the development of a new and effective antibacterial treatment for foodborne and other infections caused by B. cereus. Our findings indicate that using combination therapies involving S. aurita and S. myrsinifolia extracts will enhance the treatment efficacy of tetracycline and ciprofloxacin and could therefore be a good strategy to repurpose or improve the effects of these conventional antibiotics as they were found to show a diminished or lost effect against resistant strains of B. cereus. 15 Methanol extracts of the twigs of S. aurita and S. myrsinifolia should be further evaluated for their efflux pump inhibitory effects, which are related to the abilities of the extracts to enhance the effects of the antibiotics when used in combinations with them. Foremost, the results of this study give an incentive to evaluate the extracts of S. aurita and S. myrsinifolia in combinations with antibiotics against antibiotic resistant strains of B. cereus. Besides, the toxicity profiles of the Salix extracts alone and of each combination that show promising antibiotic-potentiating effects should be evaluated in vivo and in vitro using cell lines, Caenorhabditis elegans and animal models.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251413012 - Supplemental material for Salix aurita and Salix myrsinifolia Twig Extracts Show Synergistic or Additive Effects with Conventional Antibiotics Against Bacillus cereus
Supplemental material, sj-docx-1-npx-10.1177_1934578X251413012 for Salix aurita and Salix myrsinifolia Twig Extracts Show Synergistic or Additive Effects with Conventional Antibiotics Against Bacillus cereus by Eunice Ego Mgbeahuruike, Enass Salih, Henry Väre, Brigita Novak and Pia Fyhrqvist in Natural Product Communications
Footnotes
Ethical Approval
Ethical approval is not applicable for this article.
Author Contributions
All authors contributed equally to the laboratory experiments and writing of the manuscript. All authors read and approved the final version of the manuscript.
CRediT Authorship Contribution Statement
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Ekhagastiftelsen (Ekhaga Foundation Stockholm, Sweden) for providing the funding for this research (project grant 2021-57 and project grant 2024-39). The article processing charge is funded by the University of Helsinki Library (HuLib), Finland.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human participants in this article and informed consent is not required.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.