Abstract
Objectives
This study focused on the isolation, structural identification, and anti-neuroinflammatory effects of benzyl benzoates from the medicinal plant Solidago decurrens Lour.
Methods
A range of chromatographic techniques were employed to isolate compounds from the ethanol extract of S. decurrens. Their chemical structures were subsequently elucidated via comprehensive spectroscopic analyses, encompassing 1D/2D NMR and HR-ESI-MS data. The isolated compounds were then assessed for anti-neuroinflammatory potential, using lipopolysaccharide (LPS)-stimulated murine BV-2 microglial cell model.
Results
A benzyl benzoate, namely benzyl 2-hydroxy-3-methoxybenzoate (
Conclusions
A benzyl benzoate was identified from S. decurrens as a natural product for the first time, and a new hydrolysate was prepared by acid hydrolysis of leiocarposide (
Introduction
Solidago decurrens Lour. (family Asteraceae) is a herbaceous plant mainly distributed in South China. In traditional Chinese medicine, the whole plant of S. decurrens (known as “Yizhihuanghua”) is used for the treatments of sore throat and pyogenic infections by its effects of clearing heat, detoxifying, and dispelling wind-heat. 1 Rare phytochemical studies of S. decurrens have reported several benzyl benzoate aglycones and glycosides, such as benzyl 2,6-dimethoxybenzoate, 2 2'-methoxybenzyl 23,6-trimethoxybenzoate, 3 and leiocarposide. 4 Some natural benzyl benzoates have shown to have pharmacological potentials including anti-scabies5,6 and α-glucosidase inhibition activities, 7 and contractility effects on human prostate smooth muscle. 8 In addition, some natural benzyl benzoate glycosides showed neuroprotective effects against glutamate-induced oxidative stress damage. 9 However, the anti-neuroinflammatory properties of these benzyl benzoates are still unknown.
Many studies have shown that imbalanced neuroinflammatory responses play a critical role in neurodegenerative diseases
10
and neural damage caused by ischemic stroke.
11
A large number of natural products have been reported for their significant neuroinflammation-regulating effects, making natural compounds a promising source of neuroprotective drug candidates.12–16 Our research group has been consistently engaged in the discovery of neuroprotective compounds from natural organisms.17–23 Given the lipophilicity and favorable blood-brain barrier permeability of benzyl benzoate scaffold, it is hypothesized to have promising potential in neuroprotection. Therefore, we conducted a phytochemical study on the benzyl benzoate components of the medicinal plant S. decurrens. During this study, eight benzyl benzoate derivatives were isolated and identified, including a new compound benzyl 2-hydroxy-3-methoxybenzoate (

Chemical structures of the benzyl benzoates
Materials and Methods
General
Column chromatography (CC) were performed with silica gel (200-300 mesh, Qingdao Marine Chemistry Co. Ltd), Sephadex LH-20 (GE Healthcare Bio-sciences AB), and ODS (50 μm, YMC). Thin-layer chromatography (TLC) analysis was carried out on Merck silica gel 60 (40-63 μm) pre-coated plates. High-performance liquid chromatography (HPLC) was conducted on an Agilent 1100 series apparatus with a YMC-Pack C18 column (10 μm, 250 × 10 mm) and recorded by a UV detector (at 210 and 254 nm). Optical rotations were determined with a PoLAAR 3005 digital spectropolarimeter (Na filter, λ = 589 nm). UV spectra were obtained on a TU 1901 spectrophotometer. IR spectra were measured with an FTIR-850 spectrometer with KBr disks. NMR spectra were obtained with a Quantum-I Plus NMR spectrometer and performed at 400 MHz for 1H and 100 MHz for 13C, using CDCl3 or methanol-d4 as the solvent and tetramethylsilane (TMS) as an internal reference. Standard pulse sequences were employed for the 2D NMR experiments, including the 1H–1H COrrelated SpectroscopY (COSY), Heteronuclear Single Quantum Coherence (HSQC), and Heteronuclear Multiple Bond Correlation (HMBC) experiments. HR-ESI-MS spectra were acquired on a Waters ACQUITY UPLCⅠ-Class/XEVO G2-XS Qtof-MS mass spectrometer in positive-ion mode.
Plant Material
The whole plant S. decurrens was collected at Shenxianju, Taizhou (Zhejiang province, China) in October 2023, and identified by one of the authors (Prof. Jinzhang Cai). A voucher specimen of the plant (ZY202302) was deposited at the Laboratory of Natural Product Chemistry, Wenzhou Medical University, China.
Extraction and Isolation
The whole plant S. decurrens was air dried (dry weight: 1.0 kg) and chopped into small pieces, and then extracted with a mixture of organic solvent (CH2Cl2/MeOH, 1:1) for two times (2 × 4 L) at room temperature. Evaporation of solvent in vacuo afforded a dark residue of 41.0 g. A portion of the organic extract (40.0 g) was dispersed in distilled water (800 mL) and extracted with EtOAc (3 × 400 mL) and n-butanol (2 × 400 mL), respectively. Evaporation of EtOAc and n-butanol under vacuum yielded the EtOAc- and n-butanol-soluble extracts of 15.1 and 4.2 g, respectively. The EtOAc extract (15.1 g) was separated by silica gel vacuum CC (150 × 50 mm), eluting with a stepwise gradient of petroleum ether (PE)/EtOAc (20:1, 10:1, 5:1, 3:1, 2:1, and 1:1, v/v), and MeOH, to obtain seven fractions (A–G), respectively. Fraction B [900 mg, eluted by PE/EtOAc (10:1, v/v)] was fractionated on a Sephadex LH-20 column (1200 × 30 mm), eluting with PE/CH2Cl2/MeOH (5:5:1, v/v) to yield four fractions (B1–B4). Fraction B3 (245.2 mg) was purified by HPLC (MeOH/H2O, 73:27, v/v) to yield
Acid Hydrolysis of 8
Compound
Benzyl 2-hydroxy-3-methoxybenzoate (
1H and 13C NMR Data for
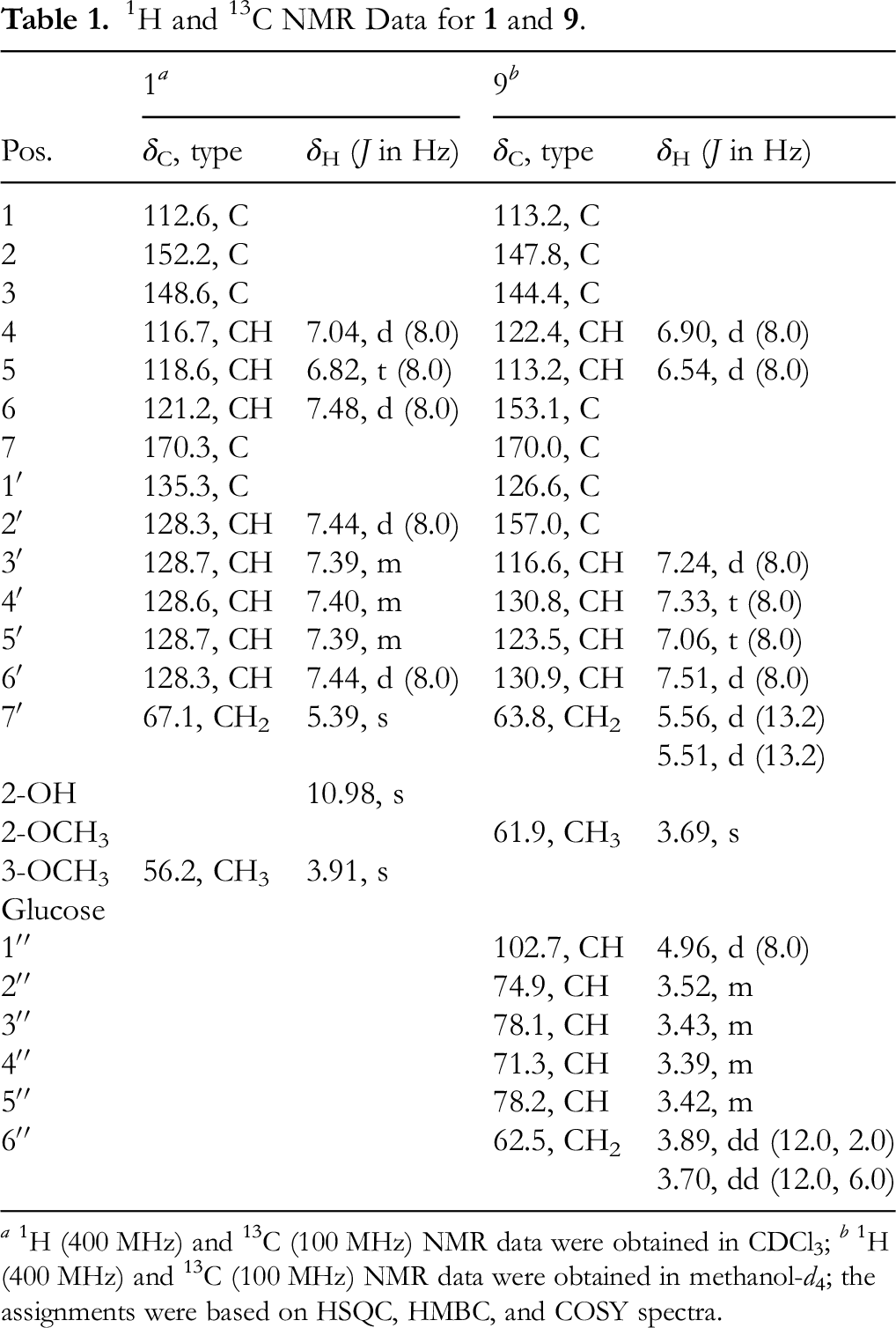
a 1H (400 MHz) and 13C (100 MHz) NMR data were obtained in CDCl3; b 1H (400 MHz) and 13C (100 MHz) NMR data were obtained in methanol-d4; the assignments were based on HSQC, HMBC, and COSY spectra.
3-Deglucopyranosyloxy-3-hydroxyleiocarposide (
Cell Culture
Murine BV-2 microglia cells, obtained from the Cell Culture Center of the Institute of Basic Medical Sciences (Chinese Academy of Medical Sciences), were incubated in Dulbecco's modified Eagle's medium (DMEM, Gibco) supplemented with 10% (v/v) fetal bovine serum and 1% penicillin (both sourced from Gibco), and maintained at 37 °C in a humidified 5% CO2 incubator.
MTT Assay
Cell viability was assessed by the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay, using a 0.5 mg/mL MTT solution (Sigma). First, BV-2 cells in the logarithmic growth phase were seeded into 96-well plates and treated with various compounds for 24 h. The original culture medium was then replaced with the MTT solution, followed by incubation at 37 °C for 4 h. After removing the MTT solution, dimethyl sulfoxide (DMSO; Sigma) was added to solubilize the formazan crystals formed. Absorbance at 490 nm was detected using a microplate reader (Thermo Fisher Scientific), and cell viability was calculated with the formula: cell viability (%) = [OD490 (experimental group)/OD490 (control group)] × 100%.
Assay for Inhibition of LPS-Induced Nitric Oxide Production
A previously established protocol 20 was employed. BV-2 cells were seeded in 96-well plates at a density of 1 × 104cells per well. The cells were then treated with varying doses of the test compounds (or left untreated as a control) for 4 h, after which 1 μg/mL lipopolysaccharide (LPS) was added to induce stimulation for an additional 24 h. The nitric oxide (NO) concentration in the cell supernatant was measured via the Griess reaction, following the protocol provided by the kit manufacturer (Nanjing Jiancheng Bioengineering Institute). Absorbance at 540 nm was measured with a microplate reader (Thermo Fisher Scientific).
Statistical Analysis
All bioassays were performed in triplicate to ensure reproducibility. Statistical analyses were conducted using SPSS 20.0 software, with significance determined via Student's t-test for pairwise comparisons or one-way analysis of variance (ANOVA) for multiple group comparisons. A P value < 0.05 was defined as statistically significant.
Results
Compound

HMBC and COSY correlations for compounds
Compound
In addition, seven known compounds were co-isolated from the ethanol extract of S. decurrens and determined to be benzyl 2-hydroxy-3,6-dimethoxybenzoate (
Extensive studies have shown that neuroinflammation participates in the progression of various neurological diseases, such as ischemic stroke-induced nerve injury,
11
Alzheimer's disease, and Parkinson's disease.
10
Regulating the level of neuroinflammation may contribute to the improvement and treatment of neurological disorders. In this study, the anti-neuroinflammatory effects of compounds
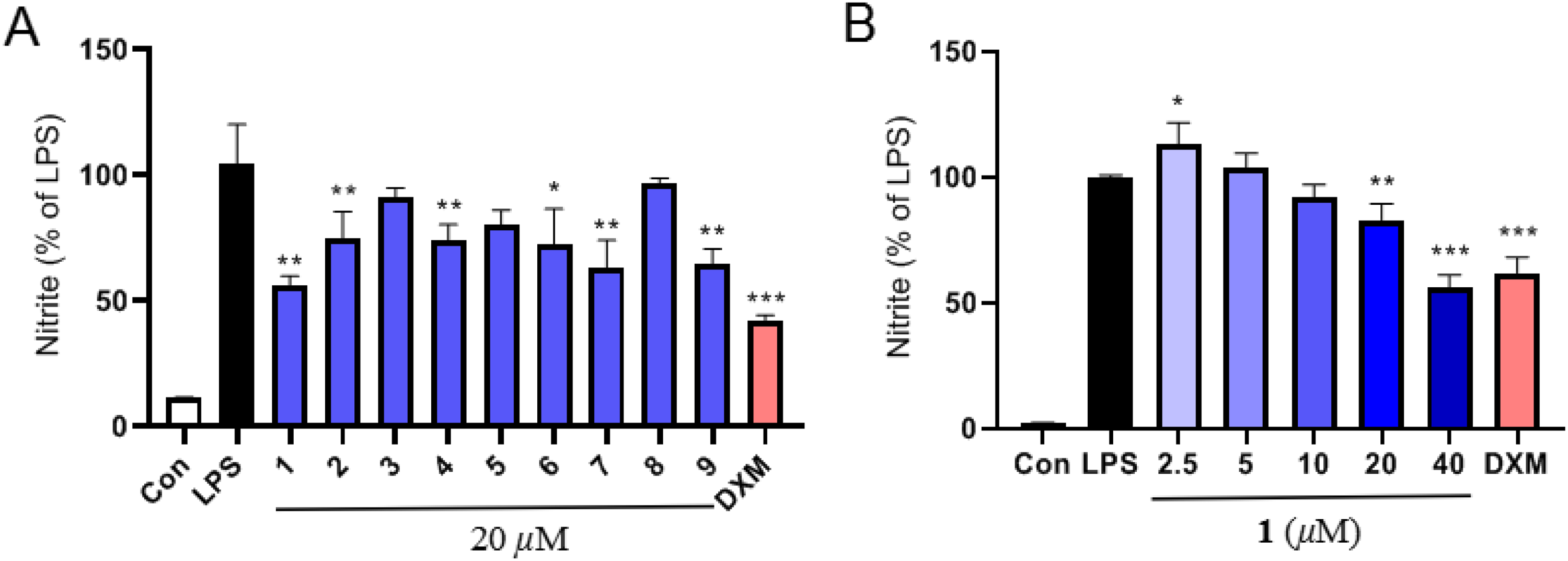
The inhibitory effects of
Discussion
Previous phytochemical studies of the medicinal plant S. decurrens have reported a limited number of benzyl benzoates including aglycones and glycosides.2–4 The present study afforded benzyl 2-hydroxy-3-methoxybenzoate (
A previous study had shown that a natural benzyl benzoate glycoside, 2′-β-D-glucopyranosyloxybenzyl 6-α-L-(4′-O-acetyl)-rhamnopyranosyloxy-3-hydroxy-2-methoxybenzoate from Disporum viridescens roots had neuroprotective property against glutamate-induced oxidative damage in HT22 hippocampal neuronal cells.
9
Another study showed that two benzyl benzoate aglycones, 2'-methoxybenzyl 2,6-dimethoxybenzoate (
There are two major limitations in this study. First, the anti-neuroinflammatory activities of the active benzyl benzoates need to be evaluated by testing their effects on other inflammatory factors, such as interleukin (IL)-6, IL-1β, and tumor necrosis factor (TNF)-α, and further confirmed by in vivo experiments. Second, the potential mechanisms of action remain to be clarified by investigating their regulatory effects on signaling pathways (eg, NF-κB and MAPK). However, in-depth study on the therapeutic effects of benzyl benzoates-containing extract (EtOAc-soluble fraction) from S. decurrens against sepsis-associated encephalopathy is currently conducted both in vitro and in vivo.
Conclusion
In summary, chemical investigation of the medicinal plant S. decurrens afforded benzyl 2-hydroxy-3-methoxybenzoate (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251411175 - Supplemental material for Anti-Neuroinflammatory Benzyl Benzoates from the Medicinal Plant Solidago decurrens
Supplemental material, sj-docx-1-npx-10.1177_1934578X251411175 for Anti-Neuroinflammatory Benzyl Benzoates from the Medicinal Plant Solidago decurrens by Junzhi Pan, Ziqiang Zou, Huayuan Liu, Junpeng Xu, Yali Lv, Jingyang Du, Xiaohong Ma, Yu Qi, Jinzhang Cai, Pengcheng Yan and Yao Ding in Natural Product Communications
Footnotes
Acknowledgments
The authors wish to acknowledge the valuable support received from the Wenzhou Key Laboratory of Research and Transformation of Chinese Medicine during the course of this study.
Author Contribution Statement
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jinhua City Science and Technology Project, Zhejiang Provincial Science and Technology Plan of Traditional Chinese Medicine, (grant number 2023-3-061, 2026ZL0991).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
