Abstract
Sickle cell disease (SCD) remains a major public health burden, especially in sub-Saharan Africa, where access to conventional therapies is limited. Consequently, herbal medicines/medicinal plants are increasingly utilised for SCD management. However, standardisation and quality control of these medicinal plants are hindered by variability in phytochemical composition, underscoring the relevance of marker compounds. This systematic review applies the Herbal Marker Ranking System (Herb MaRS) to identify and prioritise appropriate marker compounds for quality control of the most frequently reported anti-sickling medicinal plants. Selected plants include Carica papaya, Terminalia catappa, Annona muricata, Ceiba pentandra, Ocimum gratissimum, Garcinia kola, and Rauvolfia vomitoria. Data on phytochemical composition, anti-sickling bioactivity, toxicity, compound concentration, and availability of analytical methods and standards were obtained through a systematic literature review. Marker compounds were ranked based on adapted Herb MaRS criteria prioritising anti-sickling activity, safety, and analytical feasibility. Overall, 35 compounds were identified, with rutin and ellagic acid achieving the highest scores (8/8), indicating their suitability as marker compounds. Quercetin (7/8), carpaine (6/8), gallic acid (6/8), campesterol (6/8), and stigmasterol (6/8) also ranked highly. Several compounds demonstrated toxicity concerns requiring monitoring, while others lacked sufficient data for full evaluation. This study demonstrates the utility of the Herb MaRS framework for systematic marker selection to support the quality control of herbal formulations for SCD management. It highlights promising chemical markers that can facilitate standardisation, advancing the safe and effective use of plant-based therapies for SCD.
Keywords
Introduction
Sickle cell disease (SCD) is a monogenic condition characterised by mutation in the beta-globin gene, leading to the formation of uncharacteristic haemoglobin S (HbS). 1 The abnormality results in deoxygenated HbS, which polymerises, causing erythrocytes of victims to assume a sickle shape. 2 The malformed cells contribute to vaso-occlusion, chronic haemolytic anaemia, organ damage, and a range of lethal complications. 3 SCD is the most prevalent monogenic disorder globally, with an estimated 300 000 infants born annually with the condition; a figure projected to exceed 400 000 by 2050. 4 This burden is unduly high in sub-Saharan Africa, which accounts for approximately 75% of global SCD births. Alarmingly, due to limited access to early diagnosis and adequate healthcare services, the majority of affected children in these regions do not survive beyond the age of five.4–7 The management of SCD incurs substantial healthcare costs for individuals affected by the condition. In Sub-Saharan Africa, most individuals live below the poverty line and cannot afford conventional treatments such as hydroxyurea or blood transfusions. 7 Consequently, indigenes in this region have increasingly resorted to medicinal plants to manage SCD and other diseases, leveraging traditional knowledge to access affordable and culturally accepted remedies.8,9
Medicinal plants have been extensively used in Africa and other regions to manage SCD and its symptoms, offering affordable alternatives to conventional treatments. Ethnobotanical studies have reported these uses in countries including Nigeria8,10,11, Cameroon 9 and the Ivory Coast. 12 Some of these medicinal plants have been validated for SCD management, with Amponsah et al 7 reporting a total of 117 plant species with anti-sickling activities. Out of the 117 reported species, seven (7), Carica papaya L. (Caricaceae), Terminalia catappa L. (Combretaceae), Annona muricata L. (Annonaceae), Ceiba pentandra (L.) Gaertn. (Malvaceae), Ocimum gratissimum L. (Lamiaceae), Garcinia kola Heckel (Clusiaceae), and Rauvolfia vomitoria Afzel. (Apocynaceae) were the most frequently reported. These plants contain various bioactive compounds which show promise as lead molecules for alternative product development for SCD management.
The efficacy and safety of herbal medicines depend on consistent quality, which is often affected by variability in phytochemical content due to environmental factors, harvesting practices, and processing methods. 13 This underscores the importance of regular quality control measures in standardising the medicinal plants and their formulations. The usage of marker compounds has become a popular and dependable strategy among suggested quality control techniques.14,15 Particularly in complex multi-herb formulations, marker compounds provide a dependable means of assessing batch-to-batch consistency, detecting product variability, and monitoring the concentrations of active constituents. 16 Nevertheless, selecting appropriate markers for standardisation remains challenging due to the complex and diverse chemical composition of medicinal plants. This highlights the need for a structured approach, such as the Herbal Marker Ranking System (Herb MaRS).
The Herbal Marker Ranking System (Herb MaRS), created by Bensoussan et al 17 and adapted by Addotey et al 16 , provides a systematic framework for selecting marker compounds. This selection focuses on biological activity, concentration of potential marker compounds in plant material, reports of analytical assays and standards, and safety profiles of potential markers. However, its application has not been explored in SCD management plants, leaving a critical gap in the standardisation of formulations incorporating such medicinal plants. Therefore, this study aims to identify and rank potential marker compounds for quality control of herbal medicinal products formulated from the most reported anti-sickling plant species reported by Amponsah et al 7 , using the Herb MaRS framework.
Method
Medicinal Plants Selection
This study builds upon the findings of a systematic review by Amponsah et al 7 , which identified medicinal plants most frequently reported for SCD management. The plants included Carica papaya, Terminalia catappa, Annona muricata, Ceiba pentandra, Ocimum gratissimum, Garcinia kola, and Rauvolfia vomitoria. The plants were chosen for Herb MaRS evaluation to rank and choose potential marker compounds for standardising medicinal plants and/or herbal medicines that incorporate them in formulations.
Data Collection Methods
A systematic, literature-based approach was employed to compile detailed information on the selected plants. A comprehensive desktop review was conducted to extract data on their anti-sickling properties, isolated compounds, safety profiles, and potential chemical markers. Data were retrieved from multiple scientific databases, such as Google Scholar, PubMed, ScienceDirect, Semantic Scholar and Web of Science. The terms included in the search were the botanical names of the plants in combination with keywords such as “anti-sickling activity,” “sickle cell disease management,” “phytochemicals,” and “bioactive compounds.” The date of publication of obtained articles was not constrained to any specific year, and only manuscripts that were published in the English language were considered.16,18
Reports of Bioactivity and Toxicity of Medicinal Plants Selected
A database was compiled cataloguing the selected medicinal plants, with each entry detailing the plant's scientific name, family, and documented anti-sickling properties. An extensive review was conducted to gather evidence of biological activity related to SCD management, focusing on mechanisms such as inhibition of haemoglobin S polymerisation and erythrocyte membrane stabilisation. Safety profiles were assessed by reviewing toxicity data from sources such as the Pharmacopoeias (West African Herbal 19 and the Ghana Herbal 20 ), peer-reviewed articles, and pharmacological databases. The compiled data comprised information on documented studies and, where available, clinical evidence on the activity and toxicity profile of selected plants.16,18
Potential Chemical Markers Identification
Potential chemical markers for the selected plants were identified using a modified version of Herb MaRS 17 , adapted to prioritise compounds with anti-sickling activity.16,18 The criteria for selection entailed reported biological (anti-sickling) activity, reported bioactivity pertinent to SCD management, reported concentration of compounds in respective plant materials, commercial reference standards’ availability, existence of validated analytical methods, and documented toxicity profiles. 18
Bioactivity and Toxicity Profiles of Potential Markers
A thorough literature review was conducted in order to identify compounds that were obtained from the plants being studied and to compile a list of their documented anti-sickling properties.
The review focused on compounds with reported effects on haemoglobin polymerisation, oxidative stress, or erythrocyte stability, as these are critical mechanisms in SCD management. Toxicity profiles of the compounds were similarly systematically documented. Searches were conducted across Web of Science, PubMed, Google Scholar, and Semantic Scholar, using criteria combining plant names, compound names, and terms like “anti-sickling activity” and “sickle cell disease.” Only full-length peer-reviewed manuscripts written in the English language were considered. The date of publication was not a barrier to manuscript consideration. 16
Review of Marker Compounds Concentration in Selected Plants and Analytical Methods Availability
Literature was reviewed to identify reports/studies quantifying marker compounds in the respective plant materials. Each marker compound's concentration in the respective plant was assessed, which is intended to facilitate reliable detection and standardisation. Additionally, reports on validated assay methods for assaying chemical markers were assessed to ensure their applicability in quality control. The analytical assay procedures were prioritised based on their sensitivity, reproducibility, and accessibility, particularly in resource-limited settings.16,18
Review of Analytical Reference Standards Availability
The commercial accessibility of reference standards of potential chemical markers was reviewed and documented to support their use in quality control. This was done following the method by Amponsah et al, 18 where the data was sourced from reputable suppliers. The unit prices and availability of standards were recorded to assess feasibility for routine analysis. Compounds lacking commercially available standards were noted as less suitable for immediate standardisation purposes 18 .
Prioritisation of Appropriate Markers for Standardisation
The catalogued compounds were prioritised using a modified scoring system adapted from Amponsah et al, 18 with compounds scoring from 0 to 8. 0 represented the least ideal chemical marker, while 8 represented the most ideal chemical marker. Scoring was conducted as follows: evidence of anti-sickling activity (0 or 2 points); evidence of bioactivity related to SCD management (0 or 1 point); concentration of potential chemical marker in raw material (<5 µg/g: 1 point; 5-50 µg/g: 2 points; >50 µg/g: 3 points); availability of a commercial reference standard (1 point), and existence of a validated analytical method (1 point). Compounds with documented toxicity were flagged for cautious use and adherence to safety protocols. Consequently, they were assigned a score of 8 points. The highest-scoring compounds were prioritised as potential marker compounds for standardising herbal products formulated from these plants.
Results
Evidence of Bioactivity and Toxicity of Medicinal Plants Selected
There have been reports of the selected medicinal plants demonstrating anti-sickling activity, indicating their potential as therapeutic agents for managing SCD (Table 1). In addition to the anti-sickling activity, Garcinia kola also exhibits erythrocyte membrane stabilisation, a unique bioactivity that may enhance its efficacy in preventing sickling. Carica papaya, Terminalia catappa, Annona muricata, and Ceiba pentandra showed low acute toxicity with LD50 values exceeding 2000-5000 mg/kg. Garcinia kola is markedly more toxic, with an LD50 of 358 mg/kg, while doses above 300 mg/kg of Rauvolfia are associated with liver damage.
Evidence of Activity and Toxicity of Catalogued Plants.

Potential Chemical Markers, and Their Bioactivity, Toxicity and Analytical Assay Methods
Several catalogued compounds demonstrate various bioactivities implicated in the management of SCD. Compounds including rutin, quercetin, ellagic acid, and naringenin, primarily from Carica papaya, Terminalia catappa, and Annona muricata, demonstrate anti-sickling properties, positioning these plants as promising candidates for SCD management. Other compounds have been reported to exhibit bioactivities, including antihaemolytic, antiplatelet, antithrombotic, and erythrocyte membrane stabilisation effects, which play a supportive yet significant role in symptomatic management of SCD (Table 2).
Reported Bioactivities, Toxicity Profiles, and Analytical Methods of Compounds Isolated from Selected Plants.

Compounds, such as carpaine, kaempferol, rutin, ellagic acid and β-sitosterol, are reported as non-toxic or safe at specific doses, with some showing no adverse effects in acute, chronic, or human studies. However, compounds like punicalagin and punicalin, eugenol, thymol, reserpine, and yohimbine pose significant toxicity risks, including liver, neurological, reproductive, and carcinogenic effects at higher doses (Table 2).
Analytical methods for identifying and quantifying the compounds are well-developed for most, with high-performance liquid chromatography (HPLC) and its variants (eg, HPLC-UV, HPLC-DAD, RP-HPLC) being the most prevalent due to their precision and versatility (Table 2).
Concentration of Potential Marker Compounds in Plant Materials and the Availability, Cost, and Purity of Their Analytical Standards
The concentrations of the potential markers in plant materials vary widely, with some compounds present in trace amounts and others in higher quantities. For example, the concentration of campesterol is 99 200 µg/g in seed oil, and loliolide is 55 800 µg/g in leaves, indicating high natural abundance that could facilitate extraction. Conversely, kaempferol and apigenin occur at low concentrations, posing challenges for cost-effective isolation. Compounds including quercetin, isorhamnetin, myricetin, and luteolin, which are present across multiple plants, lack reported concentration data, suggesting limited quantitative studies on their natural occurrence (Table 3).
Summary of the Availability, Cost, Purity, and Concentration of Analytical Standards for Selected Marker Compounds.
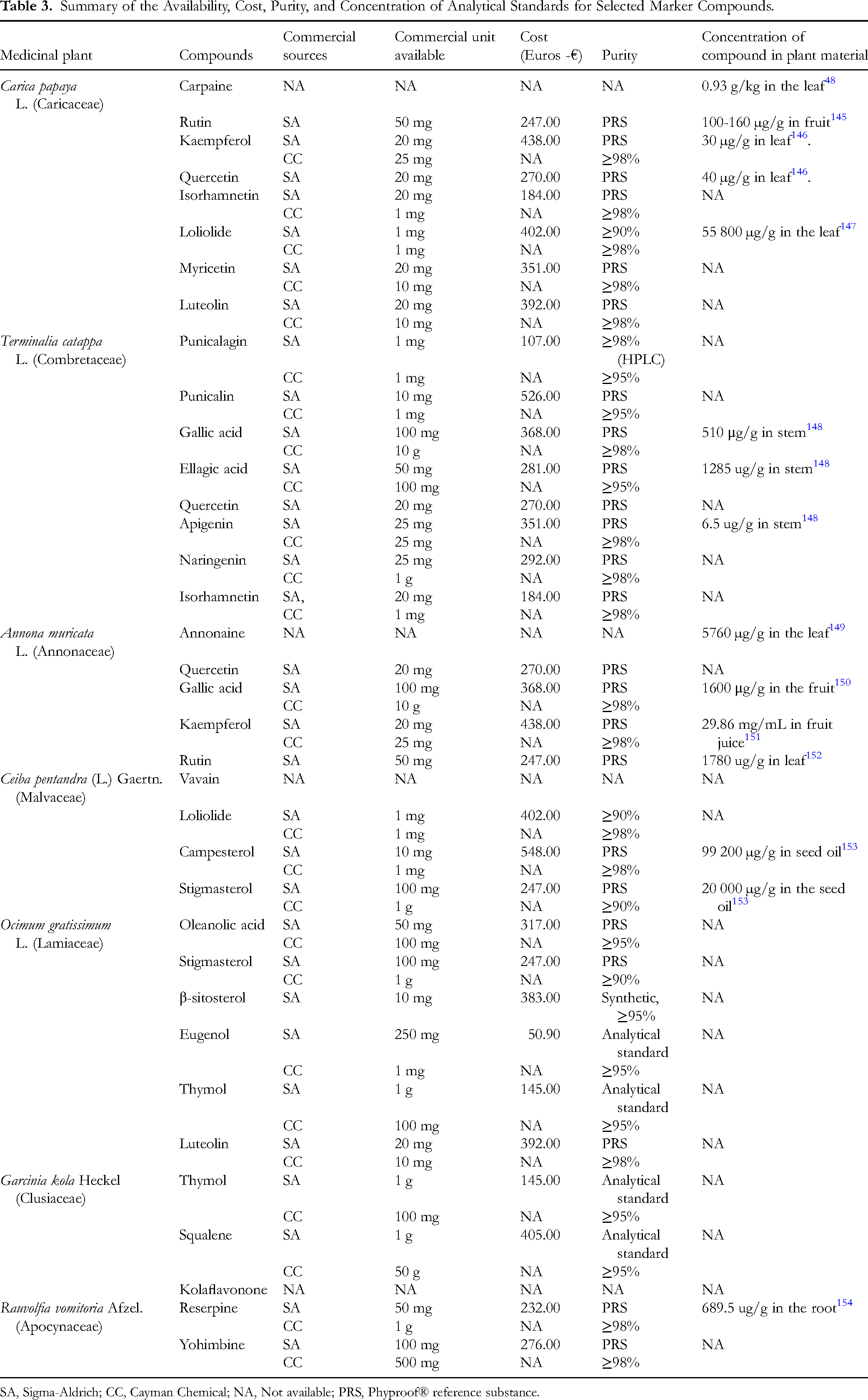
SA, Sigma-Aldrich; CC, Cayman Chemical; NA, Not available; PRS, Phyproof® reference substance.
Several compounds are commercially available, primarily through reputable suppliers like Sigma-Aldrich and Cayman Chemical, with unit sizes ranging from 1 mg to 50 g and costs spanning €50.90 to €548.00. Quercetin, kaempferol, rutin, and luteolin, which are present in multiple plants, are commercialised with high purity (≥95% or phyproof® Reference Standard) but at higher prices (ranging between €247.00 and €438.00). Gallic acid and ellagic acid are similarly available at high purity (≥95%) but moderate costs (€281.00-€368.00). Carpaine, annonaine, vavain and kolaflavonone lack commercial sources, highlighting their limited accessibility for research or industrial use (Table 3).
Prioritisation of Appropriate Markers Using the Herb MaRS Framework
Most of the potential markers scored 1/1 for exhibiting bioactivity relevant to SCD management (A = 1). Carpaine, rutin, quercetin, ellagic acid, and naringenin scored 2/2 due to reported anti-sickling activities (B = 2). The variations in the concentrations of compounds in their respective plant materials led to different scores (C = 1, 2 or 3). About 19 compounds lack reported data on concentration in plant materials, and thus, scored 0/3 (C = 0). Availability of reference standards was recorded for 33 compounds, scoring 1/1 (D = 1), while carpaine, annonaine, and vavain scored 0/1 (D = 0). Additionally, analytical methods have been reported for 34 compounds (E = 1), except for punicalin and vavain (E = 0).
Toxicity (F = 8) is reported for 16 compounds, including rutin, quercetin, punicalagin, gallic acid, eugenol, thymol, and reserpine, while 21 compounds, such as carpaine, kaempferol, ellagic acid, and naringenin, show no evidence of toxicity (F = 0).
The Herb MaRS scores range from 1 to 8, with rutin and ellagic acid achieving the maximum score of 8/8. Quercetin scores 7/8, while carpaine, gallic acid, kaempferol, loliolide, campesterol, stigmasterol, and reserpine score 6/8. Punicalin and vavain had low scores of 2/8 and 1/8, respectively (Table 4). Overall, the chemical markers prioritised for standardising the selected medicinal plants include carpaine, rutin, kaempferol, quercetin, loliolide, gallic acid, ellagic acid, apigenin, naringenin, campesterol, stigmasterol, thymol, reserpine, yohimbine and annonaine (Figure 1).
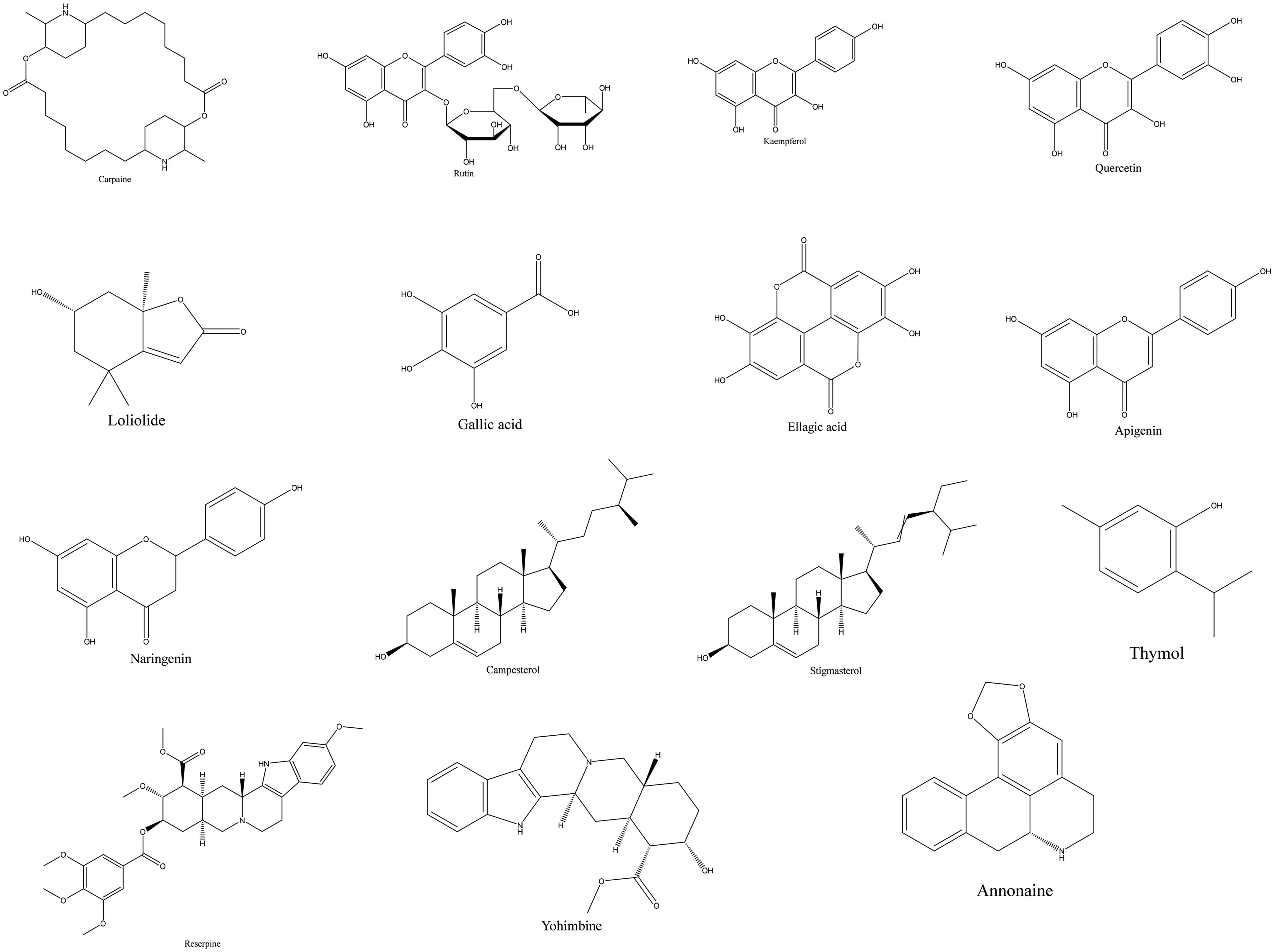
Chemical Structures of Selected Marker Compounds Isolated from Medicinal Plants.
Scoring and Ranking of Potential Markers for Medicinal Plants Selected for the Study.

Discussion
Carica papaya L. (Caricaceae) has been widely studied for its potential in managing sickle cell disease (SCD), particularly its leaves, seeds, and fruit. Ethnobotanical studies, including Amponsah et al 7 document Carica papaya as a key component in polyherbal formulations for SCD in Ghana and Nigeria, often used as leaf decoctions or seed extracts. Studies have confirmed the anti-sickling, membrane-stabilising and sickling reversal effects of papaya leaf extracts.21,22 Additionally, Singh et al 155 reported that papaya leaf extract significantly decreased sickle cell crises by enhancing antioxidant defences, attributed to its high phenolic content. Carica papaya is generally considered safe at therapeutic doses, with minimal toxicity reported.23,24 However, excessive consumption of unripe papaya or high doses of seed extracts may cause gastrointestinal irritation due to benzyl isothiocyanate, a compound requiring monitoring in formulations. 156 Carica papaya contains bioactive compounds such as flavonoids (quercetin, kaempferol, myricetin, luteolin), terpenoids (loliolide), and alkaloids (carpaine), which contribute to its therapeutic potential. Muhammad et al 61 has demonstrated that quercetin inhibits sickle cell haemoglobin polymerisation by stabilising the oxygenated state of haemoglobin, thus reducing sickling. Similarly, rutin has shown anti-sickling effects in vitro by modulating red blood cell membrane stability. 49 These compounds align with Herb MaRS criteria, scoring high (quercetin at 7/8 and rutin at 8/8) due to their anti-sickling activity, adequate plant concentrations (>50 µg/g), and availability of analytical standards for quality control (Table 4). It should be noted that both compounds scored 8/8 for toxicity, warranting caution in their use. Carpaine, a dimeric piperidine alkaloid, showed the highest inhibitory activity in a molecular docking study against the 2HBS protein, which drives sickling and associated complications. 45 It scored 6/8 in the analysis owing to limited commercial standards and insufficient concentration data. Similarly, loliolide, a monoterpenoid lactone, scored 6/8 but lacks reported data on anti-sickling activities. It, however, exhibits antioxidant and anti-inflammatory properties, which may indirectly benefit SCD management by reducing oxidative stress and inflammation, key drivers of sickle cell crises. 69 Likewise, kaempferol, lacking reported anti-sickling data, scored 5/8, while isorhamnetin, myricetin and luteolin each scored 3/8 (Table 4). The application of Herb MaRS to Carica papaya highlights quercetin and rutin as ideal chemical markers.
Terminalia catappa L. (Combretaceae), commonly known as Indian almond or tropical almond, is a medicinal plant widely reported in ethnobotanical studies for its use in SCD management.7,25,26 It exhibits significant anti-sickling activity, primarily attributed to its ability to inhibit haemoglobin S (HbS) polymerisation. Studies have demonstrated its efficacy in both in vitro and in vivo models, where extracts from its leaves and seeds reduce red blood cell sickling and improve erythrocyte stability.25–27 The plant has a favourable safety profile, with an LD50 exceeding 5000 mg/kg in animal models28,29, indicating low acute toxicity. Key bioactive compounds include quercetin, apigenin, ellagic acid, naringenin, isorhamnetin, gallic acid, punicalagin, and punicalin, which contribute to its anti-sickling and antioxidant properties (Table 2). Punicalagin and punicalin, respectively, scored 3/8 and 2/8, owing to limited data on anti-sickling activity and concentrations in plant materials (Table 4). Gallic acid, although lacking direct anti-sickling activity, protects against oxidative stress-induced osmotic fragility in erythrocytes, supporting symptomatic relief in SCD. 82 It scored 6/8 in the analysis, signifying a valuable marker for standardisation. Moreover, gallic acid has a favourable safety profile, with an LD50 exceeding 900 mg/kg in animal models, indicating low acute toxicity. This was further confirmed in sub-acute toxicity studies in albino mice. 83 Nevertheless, caution should be applied in its use, especially at high doses. Ellagic acid, a polyphenolic compound, exhibits significant anti-sickling activity by directly inhibiting haemoglobin S (HbS) polymerisation. 86 Studies, including those using Berkeley mice, show that ellagic acid reduces pathophysiological manifestations of SCD, such as vaso-occlusion, by stabilising erythrocyte membranes and mitigating oxidative stress.86,87 Its antioxidant properties further protect erythrocytes from oxidative damage, complementing its anti-sickling effects. 87 In the absence of reported toxicity88,89, ellagic acid serves as the ideal chemical marker, scoring 8/8 in the Herb MaRS analysis (Table 4). Apigenin and naringenin, both flavonoids, each received a score of 5/8 in the analysis (Table 4). While there have not been any reports on the anti-sickling activity of apigenin, naringenin exhibits significant anti-sickling activity in vitro on human sickle red blood cells, where it inhibits haemoglobin S (HbS) polymerisation. 95 Additionally, contrary to apigenin, which has studies indicating potential hepatotoxicity at doses exceeding 200 mg/kg 93 , limited toxicity data are available for naringenin, with no comprehensive studies reporting acute or chronic toxicity in animal or human models (Table 3). From our study, gallic acid, ellagic acid, apigenin, naringenin and quercetin emerge as ideal markers for quality control purposes. However, owing to toxicity concerns, ellagic acid and naringenin are recommended for standardisation.
Annona muricata L. (Annonaceae), commonly known as soursop, is a medicinal plant widely used in sub-Saharan Africa for SCD management, particularly in countries like Nigeria and Cameroon.7,9,30 It exhibits significant anti-sickling activity, as demonstrated in vitro and in vivo, with its extracts inhibiting haemoglobin S (HbS) polymerisation.30,31 The plant has a relatively safe profile, with an LD50 exceeding 5000 mg/kg in animal models. 32 However, some studies report genotoxic potential, particularly with prolonged use or high doses, necessitating careful monitoring. 33 Key bioactive compounds, including quercetin, rutin, gallic acid, and kaempferol, contribute to its anti-sickling and antioxidant properties (Table 2). Annonaine, an isoquinoline alkaloid, exhibits antioxidant and vasorelaxant properties, which are relevant to SCD management by reducing oxidative stress and improving vascular function.101,102 Limited data exist on annonaine's toxicity profile, with no comprehensive studies reporting acute or chronic toxicity in animal or human models, resulting in 0/8 in the toxicity scoring. It, however, scored 5/8, indicating potential as a chemical marker. Quercetin, likewise, scored 5/8, kaempferol and gallic acid each scored 6/8, and rutin scored 8/8 (Table 4), signifying their ideal selection as markers for Annona muricata in SCD management formulations.
Ceiba pentandra L. (Malvaceae), commonly known as kapok or silk-cotton tree, is a tropical tree widely used in traditional medicine in sub-Saharan Africa, particularly in the Democratic Republic of Congo, for SCD management. 7 It exhibits significant anti-sickling activity, with aqueous extracts from its trunk bark and branches demonstrating the ability to inhibit HbS polymerisation. 34 The plant also shows antithrombin effects by activating heparin cofactor II and modulating fibrinolytic activity. This reduces the secretion of tissue-type plasminogen activator (t-PA), urokinase-type plasminogen activator (u-PA), and plasminogen activator inhibitor type-1 (PAI-1) without affecting their synthesis. 35 These properties help mitigate thrombotic crises, a major complication in SCD. The LD50 exceeds 5000 mg/kg in animal models, indicating low acute toxicity. 36 Bioactive compounds such as campesterol, stigmasterol, vavain, and loliolide contribute to its anti-sickling, antioxidant, and membrane-stabilising effects (Table 2). Vavain exhibits antioxidant properties, which are relevant for SCD management by reducing oxidative stress, a major contributor to erythrocyte damage and vaso-occlusive crises. 106 Additionally, its ability to inhibit cyclooxygenase-1 and -2 (COX-1 and COX-2) catalysed prostaglandin biosynthesis suggests anti-inflammatory effects that may support symptomatic relief in SCD. 105 That notwithstanding, the limited data on vavain led to the scoring of 1/8 in the analysis (Table 4). Campesterol and stigmasterol each scored 6/8 (Table 4), indicating their suitability as chemical markers for C. pendandra and its formulations. Although direct evidence of anti-sickling activity is limited, they can stabilise erythrocyte membranes, supporting the prevention of haemolysis, a common complication in SCD. 107 Additionally, the suitability is enhanced by the absence of reported toxicity data.
Ocimum gratissimum L. (Lamiaceae), commonly known as African basil, exhibits significant anti-sickling activity, with methanolic leaf extracts inhibiting HbS polymerisation in vitro.37,38 Furthermore, the antioxidant properties of Ocimum are attributed to compounds such as stigmasterol, β-sitosterol, eugenol, thymol, and luteolin (Table 2), which mitigate oxidative stress, reducing erythrocyte damage and vaso-occlusive events.77,107,118,124 Eugenol and thymol also demonstrate membrane-stabilising effects, further supporting SCD management by preventing haemolysis.118,124 With an LD50 exceeding 5000 mg/kg in animal models, Ocimum has a generally safe toxicity profile. 39 However, specific compounds present risks: oleanolic acid may cause cholestatic liver injury at doses above 1350 mg/kg and has antifertility effects111,112, while eugenol 118 and thymol 124 exhibit liver and neurotoxicity at high doses. β-sitosterol shows no clear toxicity in chronic studies, but careful dose monitoring is needed for safe therapeutic use. 116 All the compounds scored 3/8 in the Herb MaRS analysis, indicating low scores for potential chemical markers. However, consideration was given to their reported toxicity profiles, which are crucial in selecting safe doses for therapy. Thus, luteolin, stigmasterol and β-sitosterol were prioritised to serve as potential markers owing to their safety profiles.
Garcinia kola Heckel (Clusiaceae), commonly known as bitter kola, is a tree indigenous to West and Central Africa, widely used in traditional medicine for managing SCD.7,10 Kola extracts exhibit anti-sickling activity, with methanolic leaf extracts and aqueous fractions demonstrating inhibition of HbS polymerisation in vitro at concentrations of 0.1-10 mg/mL, comparable to p-hydroxybenzoic acid. 40 The erythrocyte membrane stabilisation properties of the aqueous extracts of the seeds have also been reported. 41 The methanol extract of the stem bark has been reported as very toxic, with an LD50 of 358 mg/kg 42 , highlighting the need for cautious dosing in therapeutic applications. Bioactive compounds isolated from G. kola, including thymol, squalene, and kolaflavonone, all demonstrate antioxidant activity (Table 2). However, thymol exhibits significant toxicity, with LD50 values of 2462.23 mg/kg in rats 125 and 1350.9 mg/kg in mice 126 , and doses of 20-40 mg/kg may induce neurotoxicity, memory impairment, and blood-brain barrier breakdown in mice. 127 This toxicity necessitates careful monitoring in the selection of doses for SCD management. Per the Herb MaRS criteria, thymol scored 3/8, which is below the acceptable score for a marker for standardisation. However, its 8/8 score for toxicity could be used to select it as a toxicological marker for the plant. Thus, formulations incorporating bitter kola could be assayed for toxicity by monitoring the levels of thymol. Squalene and kolaflavonone lack comprehensive toxicity data, and kolaflavonone is not commercially available as an analytical standard, limiting its immediate use for standardisation (Table 3).
Rauvolfia vomitoria exhibits anti-sickling activity, with root and leaf extracts inhibiting HbS polymerisation in vitro, reducing sickling of erythrocytes under hypoxic conditions. 43 The plant has an LC50 of 1.205 mg/mL, and doses >300 mg/kg may cause liver damage. 44 Its biological activities have been attributed to indole alkaloids, including reserpine and yohimbine, which mitigate oxidative stress (Table 2). Reserpine, in particular, has erythrocyte membrane stabilisation effects 136 , which indirectly contribute to the symptomatic management of SCD. Its use should, however, be monitored owing to reports of hypothermia-related testicular toxicity 137 and CNS toxicity. 138 Reserpine scored 6/8 (Table 4) in the analysis, indicating a suitable marker for standardising R. vomitoria and formulations that incorporate it. Yohimbine, on the other hand, scored 3/8, mainly due to a lack of reports on its concentration in the plant material. It, however, scored 8/8 for reported toxicity (Table 2). Both reserpine and yohimbine are therefore prioritised as markers, serving as a toxicological marker for the plant.
The application of Herb MaRS for the identification and prioritisation of chemical markers for the quality control of medicinal plants used in the management of SCD represents a significant advancement in the standardisation of such herbal formulations. A major contribution of this study is the systematic adaptation of the Herb MaRS framework to prioritise markers specifically associated with anti-sickling activity, thereby addressing a critical gap in existing literature. This targeted approach improves the precision and relevance of marker selection for complex herbal products intended for SCD management. However, certain limitations were identified, including the dependence on secondary, literature-derived data, which may overlook unpublished findings or region-specific studies. Additionally, the absence of concentration data for 19 compounds restricted their full evaluation within the Herb MaRS framework. Future studies should incorporate primary phytochemical investigations to address these limitations and undertake clinical validation of the proposed markers.
This study has certain limitations that warrant acknowledgement. As a systematic review and analytical application of the Herb MaRS framework, the findings depend largely on published literature, and as such, minor but pharmacologically potent compounds may be underrepresented where their activities have not yet been reported. Furthermore, the analysis may not fully capture geographical and environmental influences, including soil type, climate, harvest season, and genetic variation, which can significantly affect phytochemical composition. The use of the Herb MaRS framework also introduces a reductionist dimension, as it focuses on individual chemical markers for standardisation and quality control. While this approach does not encompass the holistic principles and synergistic interactions fundamental to traditional herbal medicine, it serves as a complementary scientific tool to support the consistency, safety, and reliability of herbal formulations. Future studies should therefore incorporate region-specific phytochemical analyses, experimental validation, and integrative approaches that bridge traditional therapeutic concepts with modern quality control systems.
Conclusion
The application of the Herbal Marker Ranking System (Herb MaRS) has enabled the systematic identification and prioritisation of chemical markers for quality control of medicinal plants used in SCD management. The study highlights several compounds as ideal markers due to their demonstrated anti-sickling activity, high concentrations in respective plant materials and analytical accessibility. Compounds including thymol, reserpine and yohimbine are also prioritised as toxicological markers for their respective plant materials. Despite promising outcomes, data gaps persist regarding the concentration and toxicity of several compounds, underscoring the need for further phytochemical and clinical investigations. Overall, this approach enhances standardisation efforts, ensuring the quality, safety, and efficacy of herbal medicines used for managing SCD.
Footnotes
Acknowledgements
The authors wish to acknowledge the Departments of Pharmacognosy and Herbal Medicine, Faculty of Pharmacy and Pharmaceutical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana.
ORCID iDs
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Desmond Nkrumah: Conceptualisation, Methodology, Data curation, Writing – original draft. Reinhard Isaac Nketia: Writing – review & editing, Formal analysis. Michael Kwesi Baah: Writing – review & editing, Methodology. Linda Mensah Sarpong: Writing – review & editing, Data curation. George Adjei-Hinneh: Data curation, Formal analysis. Bernard Kofi Turkson: Supervision, Data curation, Formal analysis. Isaac Kingsley Amponsah: Conceptualisation, Writing – review & editing, Supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All datasets used and/or analysed during the current study are available in the manuscript and could also be requested from the corresponding author.
