Abstract
Objective
Lonicera japonica primarily originates from artificial cultivation; however, varying environmental conditions in different production areas lead to inconsistencies in the quality of the medicinal materials. Consequently, it is essential to identify the key factors influencing the quality of Lonicera japonica to enhance its cultivation standards. This study compares the chlorogenic acid content of Lonicera japonica from various production areas, explores the climate and soil factors that influence chlorogenic acid accumulation, and provides a basis for optimizing artificial planting methods and quality control of Lonicera japonica.
Methods
We collected Lonicera japonica samples from three origins: Henan, Hebei, and Shandong, determined the chlorogenic acid content using high-performance liquid chromatography (HPLC), and analyzed the variations in samples from different origins. We tested the soil physicochemical properties according to national standards, detected the soil microbial community composition using 16S amplicon sequencing technology, and analyzed the climatic and soil factors affecting chlorogenic acid accumulation.
Results
Chlorogenic acid content was highest in the Hebei Lonicera japonica samples. The annual mean temperature, precipitation, relative humidity, and wind speed negatively correlate with chlorogenic acid content. In contrast, the annual sunshine duration positively correlates with chlorogenic acid content. Rhizosphere samples from Shandong have higher total nitrogen, sulfur, and carbon content than those from Henan and Hebei. Furthermore, Henan soils exhibit slightly higher nitrate nitrogen content and slightly lower nitrite content, while Hebei soils have higher ammonium nitrogen concentration. The weakly acidic pH value was positively correlated with chlorogenic acid content. The rhizosphere soil of Lonicera japonica in Shandong exhibited the highest bacterial abundance, and the rhizosphere soil of Lonicera japonica from Henan yielded the highest number of ASVs. Strains 27F-1492R, KF-JG30-B3, Pla4_lineage, Bacillus, and bacteriap25 were highly abundant in rhizosphere soil samples from Henan. Ohtaekwangia, Caulobacter, and Larkinella were abundant in the Shandong rhizosphere samples. The abundances of Subgroup_10, Bryobacter, and Uncultured were negatively correlated with the chlorogenic acid content, whereas Terrimonas showed positive correlations.
Conclusion
Lonicera japonica from the Hebei Province had the highest chlorogenic acid content. Environmental factors affect the quality of Lonicera japonica. In artificial cultivation, the quality of Lonicera japonica can be enhanced by controlling the temperature, soil nitrogen content, and the quantity of beneficial bacteria.
Introduction
Lonicera japonica Thunb. is a perennial plant belonging to the Caprifoliaceae family. Dried flower buds or unopened flowers of Lonicera japonica are commonly in medicines. 1 Lonicera japonica flowers initially bloom as white flowers and later turn yellow, which has earned them the name “Golden and Silver Flowers”. 2 Lonicera japonica primarily grows in Russia, China, and Japan but has spread to Europe and North America. 3 The primary regions of Lonicera japonica production in China include Hebei, Henan, and Shandong, there are extensive areas covered by both artificially cultivated and wild varieties. 4 Lonicera japonica boasts high ornamental value, making it suitable for landscaping purposes, such as to cover low walls. They are also renowned for their medicinal properties. Lonicera japonica has been used in local and traditional medicine in clinical practice for the treatment of exopathogenic wind heat, epidemic febrile diseases, sores, carbuncles, furuncles, and infectious diseases.
Modern pharmacological studies have shown that Lonicera japonica possesses a wide range of pharmacological activities, such as antibacterial, anti-inflammatory, antiviral, antiendotoxin, blood fat-reducing, antipyretic activities; however, Due to the high demand for Lonicera japonica, the majority of Lonicera japonica produced currently originates from artificial cultivation. 3 Due to the varying conditions in the planting regions, the quality of Lonicera japonica available in the market is inconsistent. There is an urgent need to identify the factors influencing the quality of Lonicera japonica while optimizing various cultivation conditions to ensure high-quality and stable cultivation and production.
As the use of Lonicera japonica has grown, its chemical composition has been thoroughly investigated. The primary components are essential oils, organic acids, flavonoids, and saponins (among others). Organic acid compounds encompass chlorogenic acid, isochlorogenic acid, and caffeic acid, with chlorogenic acid being a key component. In the current edition of the Chinese Pharmacopoeia, quality control for Lonicera japonica is centered on chlorogenic acid content. 5
The active ingredients in traditional Chinese medicine are primarily secondary metabolites, and their concentrations are crucial indicators for assessing the quality of Chinese medicinal materials. While these metabolites are influenced by genetic factors inherent to the plant, they are also affected by external elements such as climate, soil conditions, and microbial interactions. Previous studies have shown that various plant secondary metabolites are affected by cold stress. The phenolic compounds flavonol, quercetin, kaempferol, and isorhamnetin increased as the temperature decreased in kale (Brassica oleracea L., var. sabellica). 6 Another study showed that monoterpenes increased in thyme in response to drought stress. 7 Researchers generally believe that light intensity may positively affect the accumulation of plant chemicals. 8 Extended photoperiods elevated the concentration of the glycoside ginsenosides in American ginseng (Panax quinquefolius L.). 9 Different soils and their available nutrients contain varying amounts of mineral nutrients and metabolites, which can impact plant quality. Nitrogen, potassium, and phosphate are crucial for plant growth. 10 A study on the Lycium barbarum fruit has found that increasing the application of nitrogen can help improve the content of amino acids and flavonoids in the fruit. 11 Rhizosphere soil microorganisms are the most active component of soil ecosystems and can effectively regulate the synthesis and accumulation of active ingredients.12–13 Microorganisms in ginseng's rhizosphere can enhance ginsenosides’ accumulation, the key active component in ginseng. 14 Similarly, microorganisms in the rhizosphere of Bupleurum chinense DC. Effectively facilitate the accumulation of total sterols and total flavonoids, which are the primary medicinal compounds in Bupleurum chinense DC.15–16
In recent years, numerous studies have explored methods to evaluate the quality of Lonicera japonica, providing reliable evidence. Mass spectrometry has been utilized to quantitatively detect active ingredients in Lonicera japonica from different origins, combined with bioinformatics analysis to establish quality evaluation methods. 17 Another study showed that an ultra-high performance liquid chromatography method was used to establish a quality evaluation method for Lonicera japonica formula granules based on the effective component index. 18 The quality of medicinal materials has always been the focus of research in the development and utilization of traditional Chinese medicines. The properties of traditional Chinese medicinal materials, whether cultivated or grown in the wild, are influenced by climate and soil composition, resulting in variations in quality. In the present study, Lonicera japonica samples were collected from three production areas in Henan, Hebei, and Shandong as research objects. The chlorogenic acid content was determined, and the effects of climatic factors, soil physicochemical properties, and soil microbial communities on chlorogenic acid content were analyzed. Exploring the differences in effective components of Lonicera japonica from different origins, exploring the effects of climate and soil on the quality of Lonicera japonica, providing a basis and indicators for optimizing artificial cultivation methods and quality control of Lonicera japonica.
Materials and Methods
Plant Material
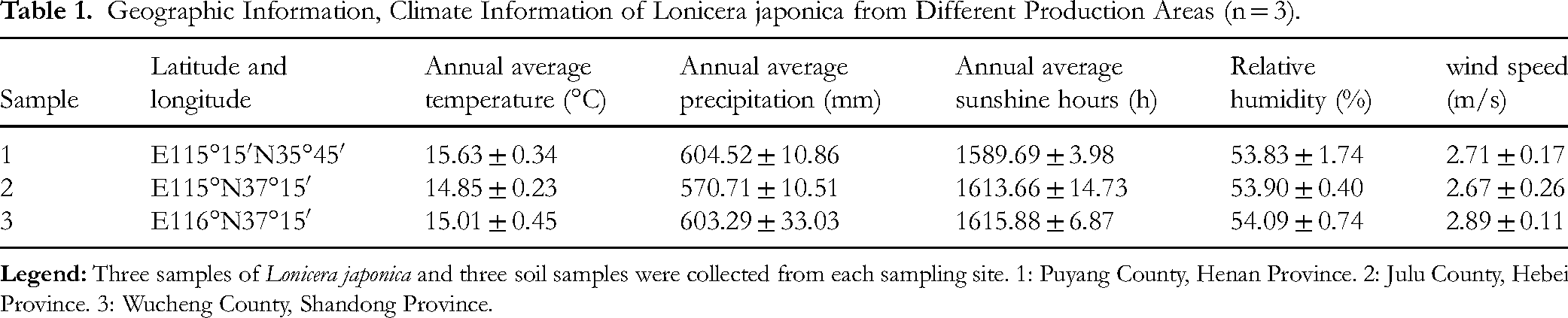
Information for the Lonicera japonica samples is shown in Table 1. 1: Puyang County, Henan Province. 2: Julu County, Hebei Province. 3: Wucheng County, Shandong Province. Professor Zhao Yutang from Chengde Medical College identified dried flower buds or early blooming flowers of Lonicera japonica Thunb., which belongs to the Caprifoliaceae family. The meteorological factor data for the samples used in the research were sourced from Wheat A Wheat Bud - Agricultural Meteorological Big Data (http: //www. wheata. cn/), which includes information such as annual mean temperature (°C), annual mean precipitation (mm), annual mean sunshine duration (h), relative humidity (%), and wind speed (m/s).
Geographic Information, Climate Information of Lonicera japonica from Different Production Areas (n = 3).
Chemicals and Reagents
Chlorogenic acid standard (C23PC489C; Beijing Jinming Biotechnology Company Limited; purity: 98%) were used. Carbinol (WXBB7259 V; Sigma-Aldrich), phosphoric acid (20190520; Froming Tianjin Chemical Reagent Supply and Marketing Company), acetonitrile (WXBB7245 V; Sigma-Aldrich), potassium hydrogen phthalate (P816389; Macklin), potassium dihydrogen phosphate (P815662; Macklin), anhydrous disodium hydrogen phosphate (S818100; Macklin), sodium tetraborate (S70022; Pusitang), sodium hydroxide (DG19852F; Anhui Tiandi High-Purity Solvent Co., Ltd), potassium dichromate, magnesium nitrate, nitric acid (Tianjin Chemical Reagent Factory), MagPure Soil DNA LQ Kit (D6356-02; Magen), Qubit dsDNA Assay Kit (Q32854; Life Technologies), Tks Gflex DNA Polymerase (R060B; Takara) were used in this study.
Instruments
A Shimadzu high-performance liquid chromatograph (LC-20AT; Forming Shimadzu Corporation, Kyoto, Japan) was used. A low-temperature high-speed centrifuge (5804R; Forming Eppendorf China Limited), digestion furnace (SKD-20S2; Shanghai Peiou Analytical Instrument Co., Ltd), electric constant-temperature water bath (HH-2A; Beijing Kewei Yongxing Instrument Co., Ltd), pH Meter (PHS-3C; Shanghai Youke Instrument & Meter Co., Ltd), electric hot plate (LC-DB-2AB; Shanghai Lichen Bangxi Instrument Technology Co., Ltd), UV-visible spectrophotometer (UV45N; Shanghai Youke Instrument & Meter Co., Ltd), centrifuge (Centrifuge 5418; Eppendorf), PCR (580BR10905; Bio-Rad), DNA electrophoresis cell (HE-120; Tanon), gel imaging system (2500; Tanon), bioanalyzer (2100; Agilent), NanoDrop (2000; Thermo Fisher) were also used.
Study Design
The experiment was conducted in the Traditional Chinese Medicine Basic Laboratory at Chengde Medical College in 2023, with the temperature maintained at 22–25 °C and the relative humidity at 65%–75%. Lonicera japonica is produced in three regions: Puyang County in Henan Province, Julu County in Hebei Province, and Wucheng County in Shandong Province. The average annual temperature in each region is approximately 15°C, and the relative humidity is approximately 54%. Plants cultivated for two years are selected for harvesting in June-July 2023. The year's first crop is harvested, and the harvesting period is controlled as the “two white period”. Three samples were collected from each region, resulting in nine samples. The five-point sampling method was applied, and the collected medicinal materials were mixed into one medicinal material sample. Soil samples were collected from 0–20 cm depths beneath the Lonicera japonica medicinal herb samples. The collected Lonicera japonica was dried in the shade for three days. Soil samples from five locations were thoroughly mixed and divided using the quartering method to obtain a final soil sample with a mass of 2 kg. Plant residues, stones, and other impurities were removed from the soil sample; a portion was stored in a −80 °C freezer for subsequent determination of soil microbial diversity, and part of it was processed to determine soil physicochemical properties.
Determination of Chlorogenic Acid Content in Lonicera japonica from Different Habitats
Sample Preparation
The samples were air-dried in the shade for three days. Then, a high-speed pulverizer (Model: FW80, Tianjin Taisite Instrument Co., Ltd) was used to grind the sample into powder, which was passed through a No. 4 sieve (65 mesh). Next, 0.25 g of the powder was measured out and placed into a stoppered conical flask. Then, 25 mL 50% methanol was added, and the flask was weighed and sonicated (250 W, 35 kHz) for 30 min. The flask was allowed to cool, reweighed, and any weight loss was compensated for using 50% methanol. Then, the flask was shaken well, and the solution was filtered and obtained ready for use.
Preparation of Standard Solution
Chlorogenic acid reference substance (10.89 mg) was accurately weighed, transferred to a 10 mL brown volumetric flask, dissolved in methanol, and diluted to the mark. Next, 1 mL of this solution was placed in a 25 mL brown volumetric flask, diluted to the mark with methanol, shaken thoroughly, and a chlorogenic acid standard solution with a concentration of 42.7 µg/mL was prepared (the purity of the chlorogenic acid reference standard was 98%). Analyzed using the external standard one-point method.
Chromatographic Condition
Quantification of chlorogenic acid was performed using a Shimadzu LC-20AT HPLC system (Shimadzu Corporation, Japan) equipped with an SPD-M20A detector and a C18 column (250 mm × 4.6 mm i.d., 5 µm Dikma). Solvent A was acetonitrile, and solvent B was a 0.4% phosphoric acid aqueous solution; the ratio is 13:87, with isocratic elution. The flow rate was 0.8 mL/min, and each run lasted 15 min. The column oven was maintained at a temperature of 35 °C. An injection volume of 10 µL was used for each sample. The detection wavelength was monitored at 327 nm. Data acquisition and quantitative analysis were performed using LabSolutions (v 5.57 SP1).
Physicochemical Analysis of the Rhizosphere Soil of Lonicera japonica from Different Sampling Locations
Determination of Soil pH
A 10.0 g soil sample was weighed and placed in a 50 mL tall beaker. Then, 25 mL of water was added. After sealing the container with a sealing film, it was stirred for 2 min, allowed to stand for 30 min, and the measurement was completed within 1 h. The sample temperature was maintained at (25 ± 1)°C. The electrode was inserted into the sample suspension, ensuring that the electrode probe was submerged to 1/3 to 2/3 of the suspension's vertical height below the liquid surface. The sample was gently agitated. Once the reading stabilizes, the pH value is noted. The measurement was performed three times, and the average value was computed. Using a potentiometric method, the soil pH values were determined according to the National Environmental Protection Standard of the People's Republic of China (HJ962-2018).
Determination of Soil Nitrogen, Sulfur, and Carbon Content
A thin layer of the soil sample, which had been sieved through a 1 mm aperture, was spread onto kraft paper and divided into several small squares. Using a small spoon, we extracted equal amounts of soil samples from each square and ground them in a mortar until they passed through a 0.25 mm sieve. The soil was then mixed thoroughly. The total nitrogen content in the soil was determined using the semi-micro Kjeldahl method. 19
A 1 g soil sample (accurate to 0.0001 g) that has passed through a 0.149 mm aperture sieve was weighed and placed in a 50 mL hard, tall beaker. Use the barium sulfate turbidity method to determine the total sulfur content in the soil. 20
A soil sample (0.5 g, accuracy of 0.0001 g) was weighed and sieved through a 0.25 mm aperture and placed into a hard test tube. Under heating conditions, an excess of potassium dichromate sulfuric acid solution is used to oxidize the soil organic carbon. The excess potassium dichromate is then titrated with a standard ferrous sulfate solution. The organic carbon content is calculated based on the gasification correction factor of the consumed potassium dichromate, and this value is then multiplied by a constant of 1.724 to determine the soil organic matter content. 21
The measurements of soil nitrogen, sulfur, and carbon content are repeated three times, with the relative error between repetitions being controlled within 5%. The final measurement value was the average of the three results.
Detection of Soil Nitrate Nitrogen, Nitrite, and Ammonium Nitrogen Content
A 0.1 g soil sample was weighed, 1 mL of distilled water was added, and the mixture was shaken and extracted for 1 h in a shaker. The mixture was centrifuged at 10,000 rpm and 25 °C for 10 min, and the supernatant was collected. Nitrate nitrogen was measured using a micro method, and ammonium nitrogen was measured using spectrophotometry. A 1 g soil sample was weighed, 2 mL of extraction solution was added, the mixture was shaken at room temperature for 1 h, and then centrifuged at 8000 rpm and 25 °C for 15 min. The mixture was allowed to stand and separate into layers. The supernatant was collected, and the nitrite content was measured using spectrophotometry. Soil nitrate nitrogen, nitrite, and ammonium nitrogen contents were determined using the BC0045, BC1480, and BC1510 reagent kits, respectively.
Microbial Composition Detection of Rhizosphere Soil of Lonicera japonica from Different Sampling Locations
DNA Extraction and PCR Amplification DNA
Total genomic DNA was extracted using the MagPure Soil DNA LQ Kit, following the manufacturer's instructions. DNA concentration and integrity were measured using NanoDrop 2000 and agarose gel electrophoresis. Extracted DNA was stored at −20 °C until further processing. The extracted DNA was used as a template for PCR amplification of bacterial 16S rRNA genes using barcoded primers and Takara Ex Taq (Takara). For bacterial diversity analysis, the V3-V4 variable regions of the 16S rRNA genes were amplified using the universal primers 343F (5′-TACGGRAGGCAGCAG-3′) and 798R (5′-AGGGTATCTAATCCT-3′).
Library Construction and Sequencing
Electrophoresis was performed to detect the PCR products, which were purified using magnetic beads. A second round of PCR amplification was conducted, the above steps were repeated, the PCR products were quantified using Qubit, and the samples were mixed in equal amounts and sequenced. Sequencing was performed by Shanghai Ouyi Biotechnology Company, Ld
Bioinformatics Analysis
Cutadapt software was used to trim the primer sequences from the raw data. Using DADA2, the raw data were quality-filtered, denoised, and merged according to the default parameters of QIIME 2 (2020.11) for quality control analysis, resulting in representative sequences and ASVs. QIIME 2 was used to select representative ASV sequences and perform database alignment and annotation. The Silva (version 138) database was used for 16S alignment, while the q2-feature-classifier was employed for species alignment and annotation. QIIME2 software was used for alpha and beta diversity analysis. The linear discriminant analysis effect size (LEfSe) method was used to compare the abundance spectra of the taxa. This analysis was performed by Shanghai Ouyi Biomedical Technology Company Limited.
Statistical Analysis
Statistical analyses were performed using SPSS version 23.0 software. Normally distributed measurement data were expressed as the mean ± SD. The median M (Q1, Q3) was used to express non-normally distributed measurement data, and intergroup differences were compared using the rank-sum test. Correlation analysis was conducted using Spearman's correlation analysis, with P < 0.05 indicating statistical significance.
Results
Analysis of Chlorogenic Acid Content in Lonicera japonica from Different Regions
In this study, we measured the content of chlorogenic acid content in Lonicera japonica in different regions. The results are presented in Table 2. The active ingredients in Lonicera japonica primarily consist of organic acids, with chlorogenic acid being its representative component. The sample from Hebei Province showed the highest chlorogenic acid content; this may be due to factors such as local climate, moisture, and soil that have influenced the accumulation of chlorogenic acid, which can be used to investigate external factors that may affect the accumulation of chlorogenic acid.
Chlorogenic Acid Content of Lonicera japonica from Different Production Areas (n = 3).

Correlation Between Chlorogenic Acid Content and Climatic Factors
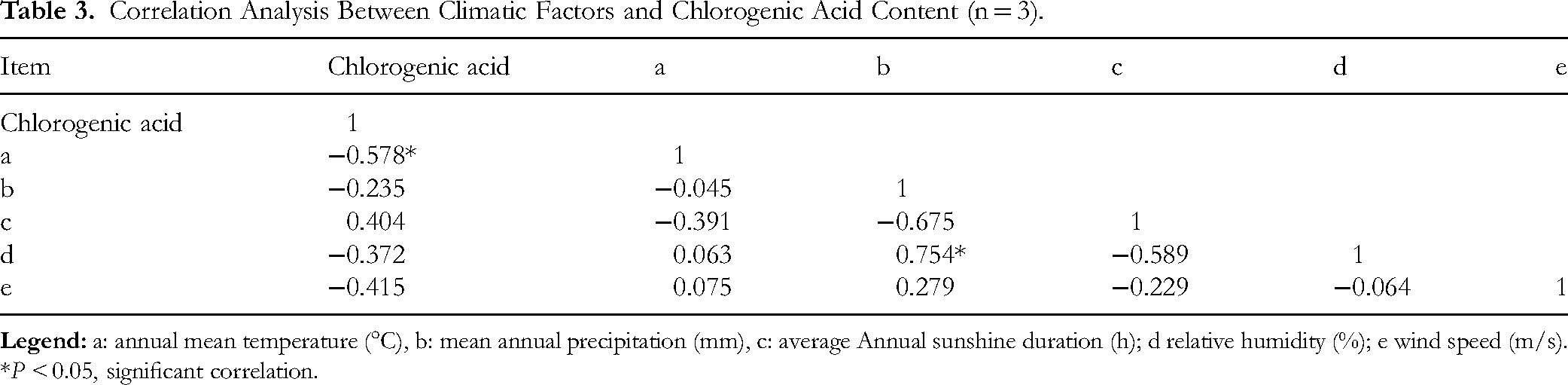
To analyze the impact of differences in the climate factors of different producing areas on chlorogenic acid content, five climatic factors were studied: annual mean temperature (°C) (a), annual mean precipitation (mm) (b), annual mean sunshine duration (h) (c), relative humidity (%) (d), and wind speed (m/s) (e)(Table 1). Correlation analysis between chlorogenic acid and climatic factors showed that the annual average temperature, precipitation, relative humidity, and wind speed were inversely correlated with chlorogenic acid levels. Conversely, the average annual sunshine duration directly correlates with chlorogenic acid. Notably, a significant negative correlation existed between the annual average temperature and chlorogenic acid content, as indicated by a correlation coefficient of −0.578 in Table 3, suggesting that higher temperatures are detrimental to chlorogenic acid accumulation.
Correlation Analysis Between Climatic Factors and Chlorogenic Acid Content (n = 3).
Correlation Between Chlorogenic Acid Content and Rhizosphere Soil Properties
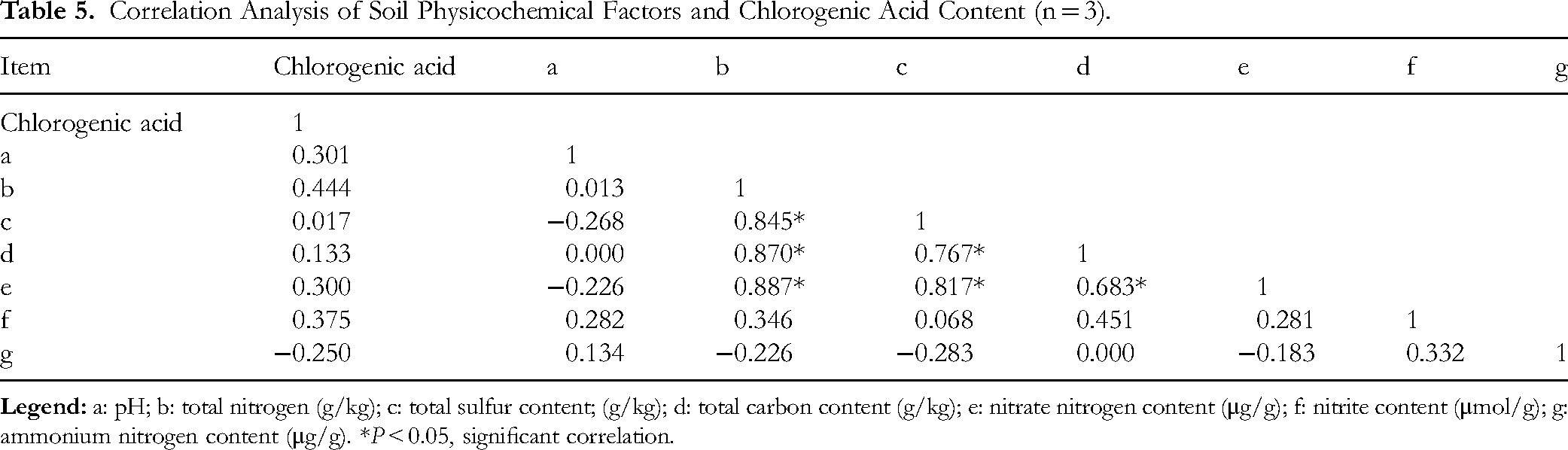
Comparison of the physicochemical properties of the rhizosphere soil from different origins of Lonicera japonica (Table 4) indicated that the soil pH values in Henan, Hebei, and Shandong are all weakly acidic. Rhizosphere samples from Shandong have higher total nitrogen, sulfur, and carbon content than those from Henan and Hebei. Furthermore, Henan soils exhibit slightly higher nitrate nitrogen content and slightly lower nitrite content, while Hebei soils have higher ammonium nitrogen concentration. Chlorogenic acid content appeared to be influenced by the nitrogen (Table 5), as indicated by a correlation coefficient of 0.444 with total nitrogen content. Generally, in addition to ammonium nitrate, within a certain range, the higher the levels of these elements, the greater the chlorogenic acid content.
Physical and Chemical Properties of Rhizosphere Soil of Lonicera japonica Georgi from Different Origins (n = 3).

Correlation Analysis of Soil Physicochemical Factors and Chlorogenic Acid Content (n = 3).
Correlation Between Chlorogenic Acid Content and Rhizosphere Microorganisms
Microbial Community Composition and Diversity of the Rhizosphere of Lonicera japonica from Different Origins
Following quality control and deduplication of high-throughput sequencing data from the rhizosphere soil samples, 19,437 ASVs were obtained. Upon taxonomic classification, these ASVs were found to belong to 35 phyla, 84 classes, 222 orders, 351 families, 655 genera, and 1349 species. There were variations in the composition of the rhizosphere soil microorganisms of Lonicera japonica originating from different sources. The top 15 bacterial species at the genus level, based on species abundance, were selected for display (Figure 1) and included Sphingomonas, MND1, and AKAU4049. At the microbial species level (Figure 2), the rhizosphere soil of Lonicera japonica in Shandong exhibited the highest bacterial abundance, particularly those of Pseudoxanthomonas mexicana g _ Pseudoxanthomonas and Citrobacter amalonaticus g _ Citrobacter, which have significant advantages. Visual analysis of the correlations and differences in the rhizosphere soil microbial communities of Lonicera japonica from different origins was performed using Venn diagrams (Figure 3). Among the samples, the rhizosphere soil of Lonicera japonica from Henan yielded the highest number of ASVs (5135), followed by Hebei (4623) and Shandong (4027). These three regions shared 696 common ASVs, including k__ bacteria, p__Proteobacteria, c__Alphaproteobacteria, and o__Dongiales. The ternary phase diagram in Figure 4 displays the relative abundance of microorganisms in the rhizosphere soil of different origins. In the samples from Hebei, Actinobacteria, Dependentiae, and RCP2-54 had the highest abundances; Planctomycetota, Cyanobacteria, and Firmicutes were the dominant species in the samples from Henan, and Proteobacteria and Actinobacteria had the highest relative abundances. Therefore, the rhizosphere soil of Lonicera japonica from the three production areas showed high microbial diversity and significant correlations.

Histogram of Community Structure of Samples from Different Sampling Locations, the Figure Shows the Top 15 Species in Terms of Microbial Abundance in the Rhizosphere Soil of Lonicera japonica from Different Origins, with Dominant Species Including Sphingomonas, MND1, AKAU4049, etc.
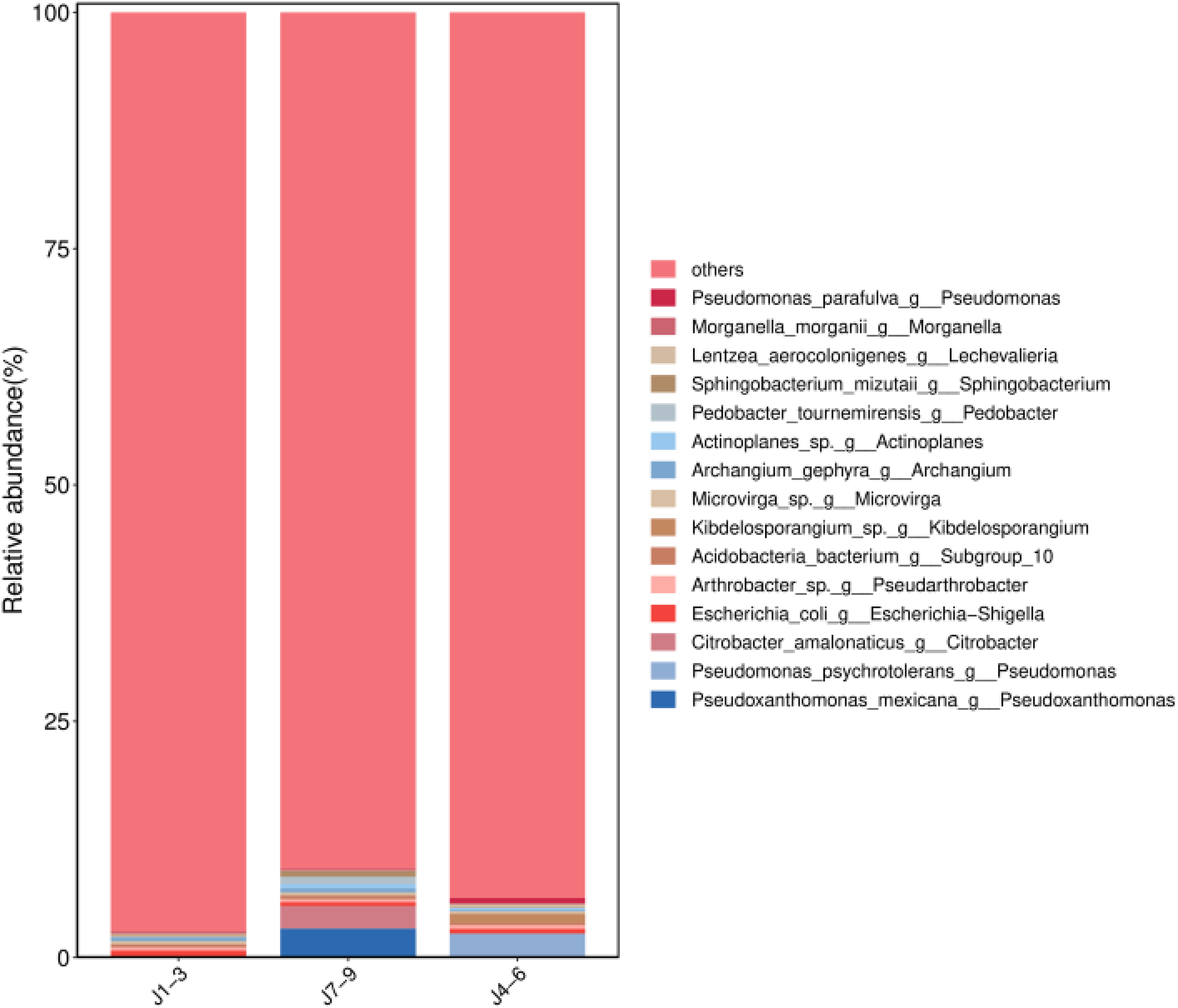
Histogram of Community Structure of Samples from Different Sampling Location. The Figure Shows the Microbial Species Level Identified by Species Selection at the Genus Level in the Rhizosphere Soil of Lonicera japonica from Different Origins.

Using Venn Diagram to Demonstrate and Analyze the Correlation and Differences of Rhizosphere Soil Microbial Communities in Lonicera japonica from Different Origins.

Using a Ternary Phase Diagram to Display the Relative Abundance of Microorganisms in Rhizosphere Soils from Different Origins.
As indicated by the alpha diversity index analysis (Table 6), the goods-coverage index for all samples was 0.999, suggesting that the sequencing depth sufficiently covered nearly all species within the samples, thereby accurately reflecting the species composition. High ACE and Chao1 indices indicate high microbial abundances in the samples. Simpson indices above 0.97 suggest high microbial diversity in the samples. Shannon indices greater than seven reflect high microbial community diversity and even distribution in the samples. In the PCoA (Figure 5), the samples from different origins exhibited significant clustering, with those from Henan showing a higher degree of clustering, and individual samples from Hebei and Shandong were scattered. The contribution rate of PC1 is 59.17%, and that of PC2 is 17.15%, respectively. The combined contribution rate of these two principal components was 76.32%, indicating that they explained 76.32% of the microbial information in the samples and accurately reflected sample conditions.

Utilizing Principal Coordinates Analysis (PCoA), it is Evident that Samples from Different Regions Exhibit Distinct Clustering Patterns. Specifically, Samples from Henan Display a Higher Degree of Clustering, whereas those from Hebei and Shandong are more Dispersed. The First Principal Component (PC1) Contributes 59.17% to the Variance, the Second Principal Component (PC2) Contributes 17.15%, and Together, these Two Components Account for a Total Variance of 76.32%.
Alpha Diversity Index of Rhizosphere Soil microbiota from Various Origins.

Correlation of Rhizosphere Soil Microbial Communities in Lonicera japonica from Different Habitats
At the genus level, the eight most abundant microorganisms across all samples were selected for differential analysis, and their relative abundances in each sample are shown in Figures 6 and 7. Strains 27F-1492R, KF-JG30-B3, Pla4_lineage, Bacillus, and bacteriap25 were highly abundant in rhizosphere soil samples from Henan, but were less abundant in samples from Hebei and Shandong. Conversely, Ohtaekwangia, Caulobacter, and Larkinella were highly abundant in the rhizosphere samples from Shandong but were not significantly increased in the samples from Henan. The correlation of the top 50 species was calculated based on horizontal abundance. The abundance of Ohtaekwangia, which belongs to the genus Bacteroidetes, was significantly correlated with various microorganisms. For example, its abundance was negatively correlated with bacteriap25 and positively correlated with Novosphingobium and Allorhizobium-Neorhizobium-Pararhizobium-Rhizobium. The abundance of Flavobacterium was negatively correlated with MB-A2-108, YC-ZSS-LKJ147, 67-14, and 0319-7L14. The abundance of Sphingobacterium showed a negative correlation with that of Rubrobacter, YC-ZSS-LKJ147, and 67-14 but a positive correlation with that of Pseudoxanthomonas. for Differential Analysis.

Microbial Clustering Heatmaps of Samples from Different Regions, Correlation of Rhizosphere Soil Microbial Communities of Lonicera japonica from Different Origins Classified by Order, and Selection of the Top 16 Microorganisms with Relative Abundance in All Samples.

Differential Microbial Correlation Network of Samples from Different Regions.
Correlations Between Rhizosphere Soil Microorganisms and Chlorogenic Acid Content in Lonicera japonica from Different Habitats
To analyze the effect of rhizosphere soil microorganisms on chlorogenic acid content, the top 50 microorganisms in terms of relative abundance were selected. Utilizing SPSS23.0 software, the correlation between these microorganisms and the chlorogenic acid content was analyzed (Table 7). Following this screening process, the top 10 microorganisms correlated with chlorogenic acid content were identified and displayed. The results indicated that the abundances of Subgroup_10, Terrimonas, Bryobacter, and Uncultured were significantly correlated with the chlorogenic acid content. Specifically, the abundances of Subgroup_10, Bryobacter, and Uncultured were negative correlated, whereas Terrimonas showed positively correlations. Furthermore, AKAU4049, 67-14, Subgroup_7, PLTA13, Lysobacter, and 0319-7L14 were also associated with chlorogenic acid content. These findings suggest that Terrimonas, PLTA13, and Lysobacter can enhance chlorogenic acid accumulation, whereas other microorganisms suppress it. The synthesis of active ingredients is a complex process influenced by various environmental factors, but further research is required to ascertain the precise regulatory mechanisms.
Correlation Analysis Between Rhizosphere Soil Microorganisms of Lonicera japonica from Different Origins and Chlorogenic Acid Content. (n = 3).

Discussion
Environmental factors have a significant impact on the quality of Lonicera japonica. This study analyzed the climatic characteristics of three different production areas, explored the correlation between climatic factors and active ingredient content, and identified potential exogenous factors affecting chlorogenic acid accumulation.
Chlorogenic acid is one of the active ingredients with a high content in Lonicera japonica. By measuring the content of chlorogenic acid, the quality of Lonicera japonica medicinal materials and their preparations can be effectively controlled, ensuring the stability and consistency of their clinical efficacy, 5 and providing a scientific basis for standardized planting, harvesting, processing, and quality evaluation of Lonicera japonica. Therefore, we tested the chlorogenic acid content in Lonicera japonica from Henan, Hebei, and Shandong provinces and found that Lonicera japonica from Julu County, Hebei Province, has significant advantages in several major production areas, and its quality is outstanding. Some researchers have studied the quality differences between Lonicera japonica grown in Hebei, Henan, and Shandong provinces. They found that the content of chlorogenic acid in honeysuckle from Hebei province is relatively higher compared with that in those from the other provinces. 22 Studies have used high-performance liquid chromatography (HPLC) to determine phenolic acid content in Lonicera japonica from multiple production areas and evaluate their quality. The results showed that the content of chlorogenic acid in Lonicera japonica from Hebei Province was generally higher than that in other provinces, 23 which is consistent with the findings of this study. The ecological environment is an extrinsic factor affecting plants’ growth, development, and distribution, and internal compositions. 24 Studies have shown that the chlorogenic acid content in Lonicera japonica significantly increases under low temperature stress. 25 This study also found a negative correlation between temperature and chlorogenic acid content, which may be related to the activation of the chlorogenic acid synthesis pathway under low-temperature stress.
Organic matter and trace elements in the soil contribute to plant growth processes and their variation indicates the dynamics of plant root system development. The organic acids released by plant roots play a role in regulating soil pH. Through the interactions of root microbial communities, soil organic matter is decomposed, leading to an increase in nitrogen and phosphorus content, while trace elements are consumed.26–27 In our study, the total nitrogen levels exhibited an upward trend as the chlorogenic acid content increased. Studies also show that chlorogenic acid's content increases first and then decreases with the increase of nitrogen application rate, 28 suggesting a potential metabolic regulation mechanism that warrants further in-depth investigation.
Microbial activity is crucial for soil fertility. The composition and diversity of microbial communities in the rhizosphere soil differ across various production regions, thereby influencing plant secondary metabolic processes. Terrimonas was the dominant bacterial species associated with the accumulation of chlorogenic acid. A significant amount of land in China is affected by salinization. Lonicera japonica possesses a degree of salt-alkali tolerance. 29 Terrimonas is a member of the Bacteroidetes phylum. Bacteroidetes are primarily involved in decomposing soil organic matter and metabolizing polysaccharides in secretions, which aids in maintaining the balance of microbial communities. 30 They are the dominant microbial community in saline-alkali soils 31 and are salt-alkali-resistant bacteria. They help alleviate the salt-alkali stress experienced during the growth of Lonicera japonica. Studies indicate that Bacteroidetes are the predominant bacterial group in soil samples taken from Lonicera japonica, aligning with the findings of this study. 32 The mechanisms by which microorganisms resist saline-alkali stress are complex. Salt-tolerant bacteria possess a stronger ability to activate Na+/H+ antiporter and K+/Na+ transporter proteins, facilitating the regulation of intracellular ion osmotic pressure to withstand saline-alkali stress. 33 These findings provide a foundation for understanding the regulatory relationship between chlorogenic acid accumulation and microorganisms in Lonicera japonica.
The quality of Lonicera japonica is influenced by several factors. Upon reviewing the entire experimental process, we found that this study had some shortcomings. This study primarily investigated the impact of climate, soil, and other factors on chlorogenic acid. However, it does not delve into research on molecular mechanisms or plant genes, and has a small sample size and incomplete analysis of the influencing factors. In the future, we will conduct in-depth analyses of regulatory patterns from the perspective of climate-metabolite-gene interactions to establish more comprehensive cultivation methods and stably enhance the quality of Lonicera japonica. Consequently, more in-depth research to address these should be conducted in the future.
Conclusion
In this study, Lonicera japonica from Hebei Province had the highest chlorogenic acid content. Low temperatures are conducive to chlorogenic acid accumulation in Lonicera japonica; therefore, the total nitrogen content in the planting soil should be appropriately increased. Terrimonas promote the accumulation of chlorogenic acid. Environmental factors affect the quality of Lonicera japonica, and controlling the influencing factors is conducive to improving the cultivation and quality of this plant.
Footnotes
Ethical Approval
All experimental data in this study did not involve animals or humans; therefore, ethical approval was not required.
Consent for Publication
All of the authors are aware of and agree to the content of the article and their being listed as a co-author of the article.
Statement of Human and Animal Rights
All experimental data in this experiment do not involve animal or human rights issues.
Authors’ Contributions
Yan Liu conceived and designed the experiments, wrote the manuscript, and created the figures. Zhixu Gao, Yintao Zhao, Lingjuan Kong, and Xiaoqing Ji analyzed the data. Jinyang Wu and Zhanhua Gao collected and prepared the related papers. All the authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work was supported by Hebei Natural Science Foundation (H2022406056).
Natural Science Foundation of Hebei Province, (grant number H2022406056).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
