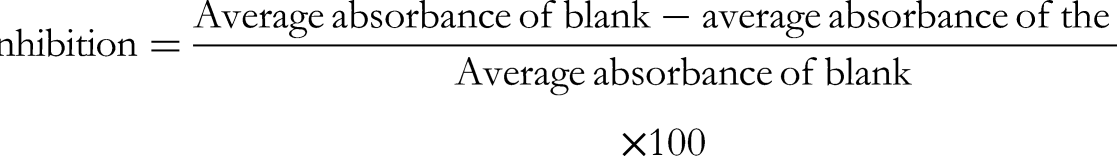Abstract
Objectives
The aim of this study was to investigate the therapeutic properties of Adenium obesum, a succulent plant native to the arid regions of the Arabian Peninsula, with particular reference to specimens from the Reem Valley in Rijal Alma, Asir Province, Saudi Arabia. The study specifically investigated the bioactive compounds in the methanolic cold extract of Adenium obesum flowers (CMEAOF) and evaluated its antioxidant and antibacterial properties.
Methods
The bioactive compounds in CMEAOF were analyzed by various analytical techniques, including gas chromatography-mass spectrometry (GC-MS), liquid chromatography-mass spectrometry (LC-MS), Fourier transform infrared spectroscopy (FT-IR), and nuclear magnetic resonance spectroscopy (NMR). The antioxidant potential of CMEAOF was quantitatively evaluated and its antibacterial activity was tested against both Gram-positive and Gram-negative bacteria.
Results
Several important bioactive compounds were identified in CMEAOF, including hexadecanoic acid methyl ester, ethyl isoallocholate and 9,12-octadecadienoic acid (Z,Z)-methyl ester. The extract showed significant antioxidant activity with a measured potency of 81.29%. In addition, the antibacterial evaluation showed a strong inhibitory effect against both Gram-positive and Gram-negative bacteria, indicating a broad antibacterial potential.
Conclusion
The results suggest that CMEAOF is a promising source of bioactive compounds with remarkable antioxidant and antibacterial properties. Ethyl iso-allocholate was identified as the most abundant compound, exhibiting the highest retention time and largest percent area under the curve, and is considered a potentially bioactive constituent based on both GC-MS and LC-MS analyses. These results support its potential therapeutic applications, especially in the development of natural antioxidant and antibacterial agents.
Introduction
Plants are widely recognized as therapeutic agents and serve as important reservoirs of bioactive substances. The healing properties of medicinal plants have been known since the dawn of human civilization. Studies have shown that in Saudi Arabia, a wide range of plants are used in traditional health care as natural treatments. These plants play a crucial role in local culture and health, providing easily accessible and traditional remedies for a range of health problems.1–3 The World Health Organization (WHO) acknowledges that herbal medicines and phytonutrients are becoming increasingly prevalent worldwide. Around 25% of modern drugs are estimated to be derived from plants. As a result, over the last four decades, research has increasingly focused on scientifically confirming the efficacy of plants and herbs as therapeutic agents for various diseases. 2 Adenium obesum, commonly referred as the desert rose, is a visually striking succulent plant native to the arid regions of Arabian Peninsula. The plant can be recognized by its uniquely tall and often swollen stem. It produces bright pink-red flowers that thrive in large numbers. The distinctive morphology of the stem acts as a water reservoir, enabling the plant to survive prolonged dry periods and demonstrating its ability to combat drought. Adenium obesum is a popular ornamental plant in the garden due to its distinctive appearance and bright flowers. It thrives with minimal watering and plenty of sunlight, reflecting its natural habitat in the desert. 4 Adenium obesum, belonging to the Apocynaceae family, is renowned for its anti-influenza, antioxidant, antibacterial, and cytotoxic characteristics. These effects emphasize the plant's capacity for medicinal uses.5–8 Furthermore, Adenium obesum is highly esteemed for its ethnomedicinal properties, as its diverse bioactive components are utilized in traditional healing methods across numerous cultures. The wide range of therapeutic advantages highlights the significance of this plant in both conventional and contemporary healthcare. 5 Despite the recognized medicinal value of Adenium obesum, most studies to date have focused on the roots, leaves or latex, with minimal focus on its flowers. Moreover, no comprehensive spectral or biological evaluation of the methanolic flower extract has been conducted, especially on plants collected from ecologically diverse regions. The Reem Valley in Rijal Alma, Asir Province, Saudi Arabia, is known for its unique biodiversity and environmental conditions that may affect the phytochemical composition of medicinal plants. Therefore, this study was conducted to investigate for the first time the profile of bioactive compounds, antioxidant and antibacterial properties of Adenium obesum flowers collected specifically from this unique region.
Materials and Methods
Materials
Scharlau, Spain provided the bacteriological media that was utilized in this research. Other materials employed in this study were supplied by Ejadah Medical Supplies Est in Riyadh, Saudi Arabia.
Study Area, Plant Collection, and Identification
Adenium obesum, is a desert plant commonly found Rijal Alma, Asir province, Saudi Arabia (Figure 1). It grows in the Reem Valley area of Rijal Alma, situated approximately 45 km west of Abha city, at coordinates N 18.126896 and E 42.192586. Adenium obesum was harvested in September 2023 and identified by Dr Remesh Moochikkal, herbarium curator identified the plant and deposited a voucher specimen in the herbarium of Jazan University (reference number = JAZUH 1636).

Processing of Flowers
A bunch of red flowers of Adenium obesum (approximately 10-12 flowers), collected randomly from the plant population, was used for each experimental combination. The flowers were carefully plucked and thoroughly rinsed with both tap water and Millipore water to remove surface impurities. They were then air-dried under shade in a well-ventilated area of the laboratory at ambient room temperature (25 ± 2 °C) with relative humidity maintained between 45%–55% for a period of one week. This was done to preserve phytochemical integrity by preventing degradation due to direct sunlight. After complete drying, the flowers were blended and ground into a fine powder using a laboratory grinder, and the powder was stored in a sealed container at room temperature until further use.
Maceration
A 25-gram sample of powdered dried flower material was soaked in 100 mL of methanol (100% v/v) for one week to allow for thorough maceration. After this period, the mixture was spun at a force of 2000 × g for 10 min in a Sigma brand tabletop centrifuge to separate the debris. The clear liquid layer obtained was filtered using Whatman no. 1 filter paper to eliminate any residual particles, and the collected liquid, or filtrate, was left to evaporate at room temperature (25 °C), resulting in a methanolic extract named CMEAOF. From 25 grams of dried powdered flower material, approximately 3.9 grams of methanolic extract (CMEAOF) were obtained after complete evaporation of the solvent. The percentage yield was calculated based on the following formula
The CMEAOF was subsequently evaluated for its antibacterial efficacy in further experiments.
GC-MS Analysis
The CMEAOF was put through an additional purification step by dissolving it in HPLC-grade methanol. The solution was then passed through a Millex-GV Syringe Filter Unit with a pore size of 0.2 µm, composed of polyvinylidene fluoride (PVDF) from Merck KGaA, Darmstadt, Germany. To ascertain phytochemical composition of the resulting filtered CMEAOF, it was subjected to Gas Chromatography-Mass Spectrometry (GC-MS) analysis. The GC-MS analysis utilized Thermo Scientific equipment with an AS 3000 autosampler and an ISQ detector. A 2 µL sample of the extract was introduced into a TR 5MS capillary column for partial component separation. Helium served as the carrier gas, maintaining a flow rate of 1.2 mL/min. Mass spectrometry was conducted, with spectral data analyzed via Xcalibur software. Interpretation of the mass spectra relied on the NIST and MAINLIB software libraries to identify the bioactive compounds present and recorded.
LC-MS Analysis
CMEAOF was subjected to detailed analysis through Liquid Chromatography-Mass Spectrometry (LC-MS) employing a Waters Xevo G2-S system, located in Milford, USA. This comprehensive characterization process was guided by the protocols outlined by Khojali et al (2023), ensuring the accuracy and reliability of the results obtained.
9
The technique facilitated the precise identification and quantification of the molecular constituents within CMEAOF
FT-IR Spectroscopy
FT-IR spectroscopy was employed to analyze the functional groups within CMEAOF (Table 1). The analysis was conducted using a Nicolet iS10 FT-IR spectrophotometer from Thermo Scientific in the USA. Samples were prepared using the KBr pellet technique, and the resulting spectra were recorded with the FT-IR spectrophotometer. These spectra were then compared with those of a reference KBr pellet. The spectral analysis covered a range from 400 to 4000 cm −1, with the resolution set at 4 cm −1.
Nuclear Magnetic Resonance (NMR) Spectroscopy
The air-dried CMEAOF underwent analysis through nuclear magnetic resonance spectroscopy (NMR) after sample preparation in deuterated water (D2O). The NMR spectra, including 1H NMR and 13C NMR, were recorded using a Bruker 400 UltraShield NMR spectrometer. This instrument operated at a frequency of 400 MHz for 1H NMR and 100 MHz for 13C NMR.
Antioxidant activity by 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay
The DPPH stable radical method was employed to measure the antioxidant capacity of CMEAOF. For the preparation of the DPPH solution, 2.4 mg of DPPH was dissolved in 100 mL of methanol. Then 3.995 mL of this methanolic DPPH solution was combined with 5 µL of the CMEAOF solution for the assay. The absorbance at 30 min of the resulting mixture was measured at 517 nm employing a spectrophotometer. In addition, the absorbance of the DPPH radical without the presence of antioxidants, referred to as “blank”,” was measured. Each measurement was performed in triplicate. The percentage of antioxidant capacity was calculated using a specific formula
Determination of IC50 Value Using DPPH Assay
To evaluate the antioxidant potential of the methanolic cold extract of Adenium obesum flowers (CMEAOF), the DPPH radical scavenging test (2,2-diphenyl-1-picrylhydrazyl) was performed and the IC50 value was determined using gallic acid as a reference standard. 2.4 mg DPPH in 100 ml methanol. Serial dilutions of CMEAOF (between 50 and 500 µg/mL) and gallic acid (standard antioxidant) were prepared in methanol. For each concentration, 5 µL of the sample or standard was added to 3.995 mL of DPPH solution in a test tube. The mixture was shaken and incubated for 30 min at room temperature in the dark. The absorbance of each mixture was measured at 517 nm using a UV-Vis spectrophotometer. A control solution (DPPH with methanol, without extract) was also included. Each measurement was performed in triplicate. The percentage of radical scavenging activity was calculated using the following formula:
A graph was plotted with % inhibition against concentration, and the IC50 value (the concentration required to inhibit 50% of the DPPH radicals) was determined by linear regression analysis.
Evaluation of the Antibacterial Potential of CMEAOF
Bacterial Strains Utilization and Culture Standardization
The study employed various bacterial strains, including Bacillus subtilis ATCC 6633, Staphylococcus aureus ATCC 25923, Streptococcus pyogenes ATCC 19615, Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853 and Klebsiella pneumoniae ATCC 700603. A 24-h culture was initiated and adjusted to a standardized concentration by diluting it in a gradient ranging from 10−1 to 10−7 using nutrient broth. The bacterial culture viability was determined by quantifying the colony-forming units per milliliter (CFU/mL).
Assessment of Antibacterial Susceptibility
The antibacterial activity was assessed following the method outlined by Moni et al (2018). 10 The bacterial cultures for antibacterial testing were prepared using Muller-Hinton agar plates and were ready after a 24-h subculture from stock culture. Both the CMEAOF samples and a conventional antibiotic, ciprofloxacin, were tested using the agar well diffusion method. A sterile cotton swab soaked with a culture containing a specific concentration of CFU/mL for each bacterial strain selected in the study was used for inoculation. The cotton swab was swabbed over the Muller-Hinton agar plates and allowed to dry for about 10 min before applying the samples. The agar plates were inoculated and then drilled with a sterile stainless-steel drill to create wells for the agar well diffusion procedure. Antibacterial activity was assessed by measuring the zones of inhibition around the wells following 24 h of incubation at 37 °C.
Structural Features of CMEAOF Extracts by FT-IR Spectrum.

Statistical analysis
The statistical analysis was done using the Prism 9, Graph Pad Instat software system, USA. Statistical analyses were performed using one-way ANOVA, followed by Tukey's test (post hoc test). Values of P < .0001, P < .001, P < .05 was considered statistically significant for all analyses.
Results
In this study, the extraction process yielded 15.6% w/w of CMEAOF from the dried flower material. Numerous bioactive compounds were subsequently identified, as evidenced by the individual peaks in the GC-MS chromatogram presented in Figure 2. The major bioactive compounds identified by GC-MS analysis of CMEAOF are summarized in Table 2 together with their respective retention times. The chemical structures of the b ioactive compounds found in CMEAOF are shown in Figure 3. Hexadecanoic acid, methyl ester was exhibited highest retention time followed by Ethyl iso-allocholate, 9,12-Octadecadienoic acid (Z,Z)-, methyl ester, Tetradecane, 2,6,10-trimethyl-, Benzoic acid, methyl ester, Decane, Undecane, Methyl tetradecanoate, 1,2-Benzenedicarboxylic acid, diisooctyl ester, Tetratetracontane, Octadecane and Heptadecane, 9-hexyl-. The bioactive compounds ethyl iso-allocholate, tetradecane, 2,6,10-trimethyl-, decane, undecane, methyl tetradecanoate and heptadecane, 9-hexyl- were identified in CMEAOF using the positive ionization mode of LC-MS (Figure 4A). In addition, 9,12-octadecadienoic acid (Z,Z)-, methyl ester, benzoic acid, methyl ester and 1,2-benzenedicarboxylic acid, diisooctyl ester were identified in CMEAOF using the negative ionization mode of LC-MS (Figure 4B). The Figure 5 showed FT-IR spectrum analysis of CMEAOF displayed a characteristic bell-shaped peak at 3418 cm−1, indicating the presence of phenolic OH groups. The peak at 2929 cm−1 is associated with both asymmetrical and symmetrical C-H stretching. Additionally, distinct peaks at 1724, 1614, 1399, 1267, 1073, 819, and 609 cm−1 represent specific functional groups and their corresponding biomolecules, as detailed in Table 1.




GC-MS Detection of Possible Bioactive Compounds of CMEAOF.

1H NMR analysis revealed the fingerprint region of CMEAOF, with the most deshielded protons appearing at 3.370 and 4.870 ppm in the proton dimension (Figure 6A). The chemical shifts represent protons that are deshielded, probably due to electronegative atoms such as oxygen or nitrogen or due to their involvement in functional groups such as ether (-O-), alcohol (-OH) or amine (-NH-). The 13C NMR spectrum showed unique peaks at 37.68, 37.74 and 38.06, which are associated with carbon atoms attached to electronegative elements such as nitrogen, oxygen or halogens. It may also indicate carbon atoms in aliphatic chains adjacent to such functional groups. The peaks at 42.18 to 49.80 ppm, indicative of carbon atoms bonded to nitrogen, such as in amines or amides, or to oxygen, such as in ethers. The peaks at 51.37 to 56.11 ppm, are often found in methoxy groups (-OCH3) or RCH2OH in the alpha position to oxygen or nitrogen. Further peaks at 60.55 to 68.11, 70.84, and 75.90 ppm chemical shifts are characteristic of carbons attached to oxygen in alcohols, ethers, or esters. It could also be indicative of carbons adjacent to oxygen in sugars or similar structures. The antioxidant properties of CMEAOF showed considerable potential, which led to a quantitative evaluation of its antioxidant activity. This evaluation revealed that CMEAOF has an impressive antioxidant scavenging capacity of 81.29%. In addition, CMEAOF showed a broad spectrum of antibacterial activity against the tested human pathogenic organisms, demonstrating strong efficacy against both Gram-positive and Gram-negative bacteria (Table 3). Figure 7 shows that Klebsiella pneumoniae exhibited the highest zone of inhibition, which was significantly greater than all other tested bacteria. The difference was extremely high significant compared to Pseudomonas aeruginosa with a P-value of ****P < .0001, and high significant compared to Escherichia coli with ***P < .001. No significant differences (ns) were observed among Bacillus subtilis, Staphylococcus aureus, Streptococcus pyogenes, and Escherichia coli. Pseudomonas aeruginosa showed the lowest zone of inhibition and was significantly lower than Klebsiella pneumoniae.

NMR Study. (A) 1H NMR Analysis of CMEAOF (B) 13C NMR Analysis of CMEAOF.

Antibacterial Activity.

Each value is the mean of 3 batches with standard deviation. The statistical analyses were done using the Prism 9, Graph Pad Instat software system, USA. #CFU- Colony Forming Unit; Cold methanolic extract of Adenium obesum flowers (CMEAOF) All the values of CMEAOF were lesser than Ciprofloxacin significantly at P < .05 level.
Discussion
CMEAOF showed promising medicinal properties in this study, which revealed the presence of numerous bioactive compounds. Hexadecanoic acid, methyl ester is otherwise called methyl palmitate was identified in CMEAOF. Methyl palmitate is an ester formed by the reaction of methanol with palmitic acid. Previous studies have shown that methyl palmitate can inhibit phagocytic activity, which is associated with differential expression of cytokines, nitric oxide and COX-2. Both in vitro and in vivo studies have shown that methyl palmitate can suppress macrophage activity, highlighting its promising anti-inflammatory and antifibrotic properties.11,12
In addition, methyl palmitate is known for its antioxidant and anti-inflammatory effects, as well as its ability to lower blood cholesterol levels.13,14 Ethyl iso-allocholate, a steroidal derivative, was identified as the main component of CMEAOF according to GC-MS and LC-MS analysis, followed by methyl palmitate. A study from 2019 showed that ethyl isoaloxolate has significant cytotoxic effects and effectively reduces tumor growth and liver metastasis. 15 Another study suggested that ethyl isoaloxolate exhibited antioxidant and anti-bacterial activity.16,17 9,12-Octadecadienoic acid (Z,Z)-, methyl ester is otherwise termed as linoleic acid, methyl ester was identified as one of the major active constituent in CMEAOF. It is an organic compound that belongs to the class of fatty acid methyl esters. In our previous research, we identified Octadecanoic acid, methyl ester in the exudate gel of Aloe fleurentiniorum. 18 The present study initially was demonstrated promising antioxidant potential by a quantitative evaluation using the DPPH assay, revealing that the cold methanolic extract of Adenium obesum flowers (CMEAOF) exhibited a free radical scavenging activity of 81.29%, with an IC50 value of 350 µg/mL. The presence of compounds such as methyl palmitate, and 9,12- octadecadienoic acid (Z, Z) in CMEAOF could be responsible for its antioxidant properties. This finding aligns with previous research showing that methyl palmitate exhibited anti-inflammatory, antioxidant, and anti-apoptotic effects. 19 Another study highlighted the potential antioxidant properties of crude extracts from the leaves of Adenium obesum.. 20 Other bioactive compounds such as tetradecane, 2,6,10-trimethyl-, benzoic acid, methyl ester decane, undecane, methyl tetradecanoate, 1,2-Benzenedicarboxylic acid, diisooctyl ester, tetratetracontane, Octadecane, heptadecane, 9-hexyl- were identified in CMEAOF. An earlier study suggested that similar compounds have been reported in the ethanolic leaf extract of Adenium obesum. 5
Analysis of the FT-IR spectrum of CMEAOF provides important information about its chemical composition and the presence of various functional groups. The spectrum shows a characteristic feature associated with phenolic OH groups and indicates the ethyl isoallocholate, which is known for its antioxidant properties. In addition, the spectrum shows features corresponding to asymmetric and symmetric C-H stretching vibrations, indicating aliphatic hydrocarbons. This suggests the presence of compounds such as n-hexadecanoic acid, decane, undecane, heptadecane, octadecane and tetratetracontane. Other notable features in the spectrum are associated with carbonyl stretching vibrations (C = O), which indicate the presence of ester or aldehyde functional groups likely associated with fatty acids or esters in the sample. The presence of unsaturated compounds, such as alkenes or aromatic rings, can be deduced from the features corresponding to the C = C stretching vibrations. Other absorptions in the spectrum can be traced back to C-H bending vibrations, which are typically observed with methyl or methylene groups, and to C-O stretching, which is characteristic of alcohols, esters or ethers.
The ¹H NMR analysis of CMEAOF revealed significant details about its molecular structure, particularly in the fingerprint region. The most deshielded protons were identified at chemical shifts of 3.370 and 4.870 ppm. These shifts suggest that the protons are in environments influenced by electronegative atoms, such as oxygen or nitrogen, or are part of functional groups like ethers (-O-), alcohols (-OH), or amines (-NH-). 21 The ¹³C NMR spectrum provided further insights into the carbon framework of CMEAOF. Peaks observed at 37.68, 37.74, and 38.06 ppm are characteristic of carbon atoms that are either directly bonded to electronegative atoms like nitrogen, oxygen, or halogens or are part of aliphatic chains adjacent to such functional groups. Additional peaks in the range of 42.18 to 49.80 ppm were noted, typically associated with carbon atoms bonded to nitrogen, as seen in amines or amides, or to oxygen, as seen in ethers. Further up field, the chemical shifts from 51.37 to 56.11 ppm are commonly found in methoxy groups (-OCH3) or in RCH2OH groups positioned alpha to oxygen or nitrogen. These shifts suggest that CMEAOF may contain such structural elements, which could influence its solubility and reactivity. Additionally, peaks at 60.55 to 68.11, 70.84, and 75.90 ppm are indicative of carbons attached to oxygen in alcohols, ethers, or esters, or possibly carbons adjacent to oxygen. 22 The ¹H and ¹³C NMR spectra of the crude methanolic extract showed primarily revealed signals corresponding to saturated aliphatic chains and ester functions, indicating the presence of fatty acid esters such as methyl palmitate. The carbon signals were predominantly in the range of 30-75 ppm, with no significant peaks in the lower range above 100 ppm, indicating the absence of aromatic or unsaturated systems. These results are consistent with the GC-MS and LC-MS data and support the conclusion that the extract is rich in lipid-containing compounds. Interpretation was limited to the main peaks due to overlapping signals in the crude extract.22–25
CMEAOF exhibited a broad spectrum of antibacterial activity and showed strong efficacy against both Gram-positive and Gram-negative bacteria. The strongest inhibition observed against Klebsiella pneumoniae, a Gram-negative pathogen often linked to drug resistance. The uniform response among Bacillus subtilis, Staphylococcus aureus, Streptococcus pyogenes, and Escherichia coli suggests moderate antibacterial efficacy across these species. The relatively low inhibition against Pseudomonas aeruginosa may be due to its robust resistance mechanisms. Overall, the CMEAOF demonstrates selective and promising antibacterial potential, especially against Klebsiella pneumoniae. This broad efficacy may be attributed to the presence of compounds such as ethyl isoallocholate, methyl palmitate and methyl benzoate, as previously reported.26,27 This observation is supported by a recent study highlighting the potent antibacterial effect of Adenium obesum flower extracts. 28 In addition, previous research has shown that crude extracts from the leaves of Adenium obesum grown in the National Botanical Garden of Oman also possess significant antibacterial properties. 20 Comprehensive analyses of CMEAOF have revealed a rich profile of bioactive compounds with significant medicinal potential, underscoring its promise as a versatile therapeutic agent. These results improve our understanding of its chemical composition and bioactivity and support its potential use in the development of novel treatments for oxidative stress and bacterial infections. However, there are still some limitations, including the reliance on in vitro assays without in vivo validation for pharmacokinetics and toxicity. The variability of the phytochemical composition, the limited antibacterial activity and the lack of stability studies pose a challenge. Future research should address these gaps through in vivo studies, toxicity assessments, mechanistic insights and clinical trials to fully demonstrate the therapeutic value of CMEAOF.
Footnotes
Acknowledgements
The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through project number: RG24-S0112. The authors express their gratitude to Dr Remesh Moochikkal, curator of the herbarium, for his expertise in identifying Adenium obesum and the specimens are preserved in the herbarium at the College of Jazan (JAZUH).
Ethical Approval
This article does not involve any experiments or studies involving human or animal participants were conducted as part of this research. Therefore, ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not include any research involving human or animal participants and therefore, not applicable.
Statement of Informed Consent
Not applicable as this research did not involve experiments or studies with human or animal subjects.
CRediT Authorship Contribution Statement
SJM & SMS: Principal investigators, conceptualization, experimentation, processing of results, writing and editing; MFA, AMA, AA, AA: Performed experiments; AK, MQ, AI, AMA: Funding sources: SA, KA, ST: Project management.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through project number: RG24-S0112.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.