Abstract
Objective
The objective of the study was to evaluate the antibacterial and antibiofilm efficacy of three essential oils (EOs), two derived from Tetraclinis articulata and one from Cedrus atlantica, against selected endodontic pathogens including Enterococcus faecalis, Streptococcus mitis, Streptococcus sanguinis, and Streptococcus gordonii.
Methods
The three EOs were extracted by means of hydrodistillation. The antibacterial activities expressed through Minimum Inhibitory Concentrations (MICs) and Minimum Bactericidal Concentration (MBCs), were evaluated in the microdilution assay. The biofilm inhibition and the biofilm disruption were tested for T. articulata2 EO in crystal violet assay.
Results
The total yield of C. atlantica, T. articulata1 and T. articulata2 obtained was 0.1%, 0.2% and 0.45%, respectively. The MIC values of C. atlantica and T. articulata1 EOs were above 20 mg/mL, while the MIC values of T. articulata2 ranged from 4.78 ± 1.48 to 19.43 ± 5.48 mg/mL. Among the isolates, S. gordonii exhibited the highest sensitivity, while E. faecalis was the least sensitive. All tested concentrations (MIC/8–MIC) of T. articulata2 EO successfully inhibited biofilm formation of the endodontopathogenic biofilms, with inhibition ranging from 34.91% to 78.90%. T. articulata2 EO also proved disruptive potential on pre-formed biofilm of S. sanguinis and S. mitis at all tested concentrations.
Conclusion
T. articulata2 proved promising antibacterial and antibiofilm potential which may be considered in dentistry.
Introduction
Treating endodontic infections remains a significant challenge for dental practitioners. 1 These infections result from bacterial invasion within the root canal. They are characterized by intense pain, and if left untreated, they can lead to inflammation of surrounding tissues, causing bone resorption, and potential tooth loss. 2 Standard treatment involves removal of infected pulp, thorough canal cleaning and disinfection, followed by sealing to prevent bacterial reinvasion. 3 In some cases, the complexity of the root canal system, with its tubules and accessory canals, presents a significant obstacle in eradicating all bacteria,4,5 making the commonly used methods insufficient.6,7 Therefore, addressing these infections necessitates innovative strategies to eliminate bacteria within the root canals effectively. 8
Increased interest in scientific communities has been observed in exploring natural antibacterial agents, particularly essential oils (EOs). This transition towards natural options is driven by several factors, including the increasing prevalence of antibiotic-resistant bacteria, concerns about the side effects of synthetic drugs, and in general trend towards more holistic and environmentally friendly approaches in healthcare.9–11 Due to their complex chemical compositions and diverse biological activities, EOs offer a promising approach for developing new antibacterial strategies in endodontics. 12 Herbal medicine has a long history in Morocco, with its population among the first to incorporate plant-based remedies into health practices. 13 Over centuries, this population has utilized the diverse native flora for medicinal purposes, particularly to treat infections. Two EOs that have gained particular attention for their antibacterial and antibiofilm potential are derived from Cedrus atlantica (Atlas cedar) and Tetraclinis articulata (Arar tree). These EOs have been traditionally used in Moroccan medicine for their antiseptic and healing properties.14,15 C. atlantica is an endemic pine tree species, belonging to the family of Pinaceae and it has been traditionally used to treat bronchitis, and certain skin diseases. 16 T. articulata, or Barbary thuja, is native to the western Mediterranean countries and dendrologically belongs to the Cupressaceae family. Historically, its resin gum was commonly used to seal decayed or perforated teeth. 17 As evidenced in the literature, these EOs have demonstrated promising antibacterial properties against a range of gram-positive and gram-negative bacterial strains, as well as fungi, making them potential candidates for use in endodontic treatments.16,18
Among the bacterial strains commonly involved in endodontic infections, Enterococcus faecalis and various Streptococcus species, including Streptococcus mitis, Streptococcus sanguinis, Streptococcus gordonii are recognized as highly important. S. mitis, S. sanguinis, and S. gordonii are primarily responsible for initiating biofilm formation, while E. faecalis is broadly considered the most involved in endodontic treatment failures.3,8 Interestingly, currently there is no available data regarding the efficiency of C. atlantica nor T. articulata EOs against these Streptococcus species, while data on their efficacy against E. faecalis is also limited.19,20
Therefore, this article's aim was to explore both the antibacterial and the antibiofilm activity of C. atlantica and T. articulata EOs against key bacterial strains in endodontics, ie S. mitis, S. sanguinis, S. gordonii, and E. faecalis.
Material and Methods
Plant Material
The aerial parts of C. atlantica were collected from the city of Ben-smim located in Ifrane province (Morocco: 33°25′28.3″N 5°10′35.6″W), in October 2023. The aerial parts of T. articulata were harvested from two different regions in Morocco: (i) T. articulata1, was collected from a town called Ouad Ifrane located in Ifrane province (northern Morocco; latitude 33°17′11.7″N; longitude 5°29′45.5″W) and (ii) T. articulata2, from Imintissakht village in the province of Marrakesh-Tensift-El Haouz (central Morocco; latitude 31°27′29.7″N; longitude 17°19′26.2″W). The herbariums have been confirmed at the plant ecology laboratory in the Scientific Institute of Rabat and identified by Professor Ouafae BENKHNIGUE. The voucher numbers were RAB114767 (C. atlantica), RAB114769 (T. articulata1) and RAB114768 (T. articulata2). Figure 1 shows the regions of collection of the three plants. The aerial parts of the plants were dried in the shade at room temperature.
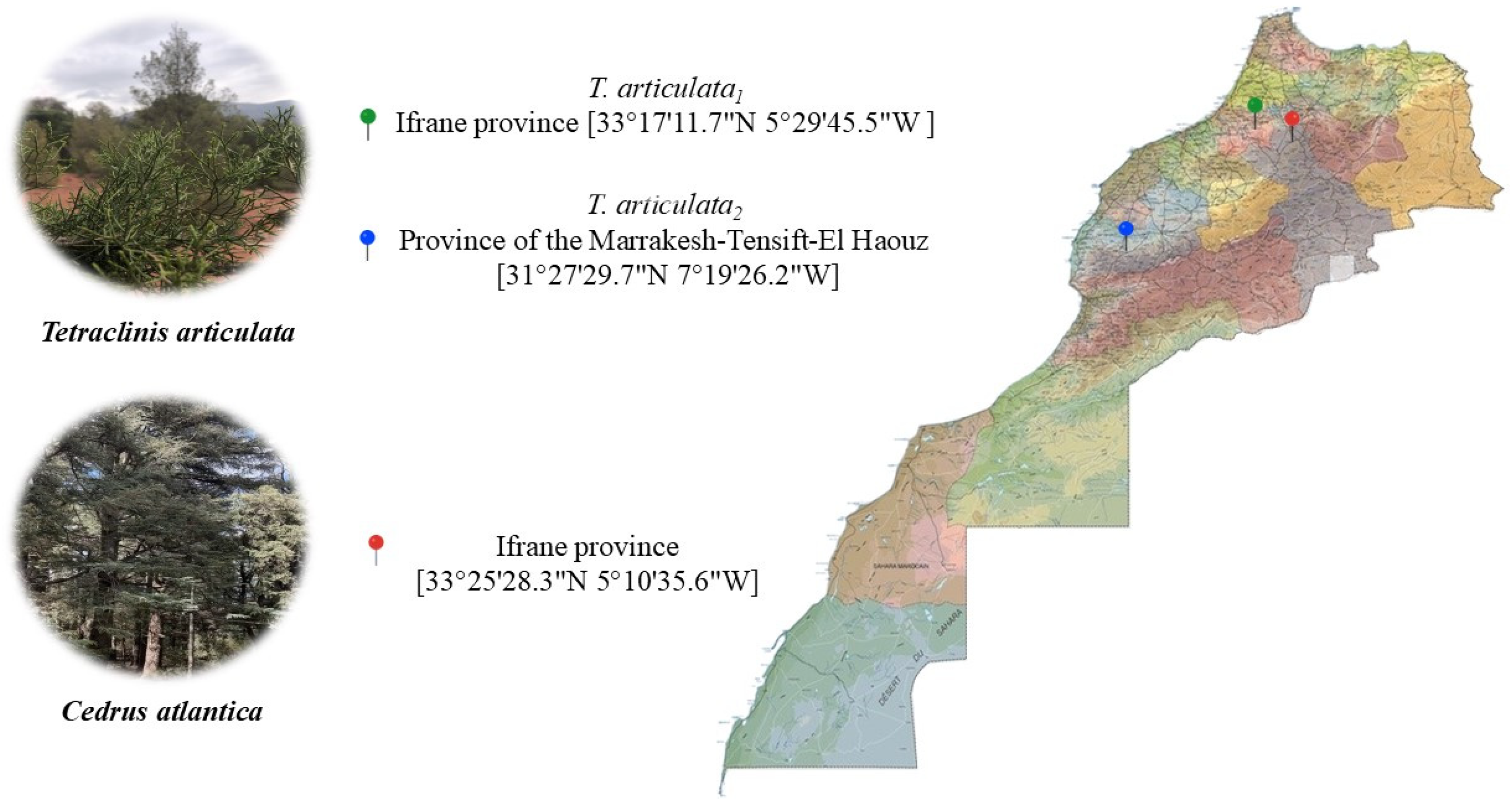
Regions of collection of Cedrus atlantica and Tetraclinis articulata in Morocco. T. articulata1 : Tetraclinis articulata harvested from northern Morocco; T. articulata2 : Tetraclinis articulata harvested from center of Morocco.
Hydrodistillation Apparatus and Procedure
The dried plants were subjected to extraction by hydro distillation by the Clevenger system associated with an Alambic. Figure 2 illustrates the hydrodistillation apparatus used for EO extraction. In the Alambic, water was introduced followed by the plant material (the whole aerial part for T. articulata1 and T. articulata2, and only leaves for C. atlantica, separately). Once the system was heated, the water vapor loaded with oils slowly entered the condenser. These condensed vapors were collected and formed two phases of which the upper low-density phase contained the EOs, and the lower high-density phase contained the hydrolates. The duration of each distillation was about 3 h. The resulting EOs were mixed with sodium sulfate (anhydrous) and stored in 10 mL opaque glass vials at 4 °C. The yield was determined by calculating the ratio between the volume of the EO obtained and the mass of the plant material used.

Hydrodistillation Apparatus and Procedure. Created with BioRender.com.
Bacterial Strains
S. mitis, S. sanguinis, S. gordonii, and E. faecalis were obtained from infected root canals of patients consulting at the School of Dental Medicine, University of Belgrade (Serbia). 21 Sampling was approved by the University of Belgrade Ethics Committee (Approval No 36/7). The strains were stored at – 80 °C at the Microbiology Department within Laboratory of Physical Chemistry 050, VINČA Institute of Nuclear Sciences at the National Institute of The Republic of Serbia. 6
Bacterial Cultivation
Frozen stock cultures of S. mitis, S. sanguinis, S. gordonii, and E. faecalis were streaked onto Mueller-Hinton agar (MHA) plates and incubated for 24 h at 37 °C. Individual colonies were then selected from the MHA plates, transferred to Triptone Soy Broth (TSB), and incubated overnight at 37 °C. To standardize bacterial inocula, suspensions were adjusted spectrophotometrically to an OD600 of 0.2, corresponding to approximately to 1 × 108 CFU/mL.
Assessment of Antibacterial Activity
The microdilution assay, based two-fold dilution in 96-well microtiter plates with sterile TSB as growth medium, was implemented to determine the Minimal Inhibitory Concentrations (MICs) and the Minimal Bactericidal Concentrations (MBCs) of the EOs. 22 Each EO was dissolved in Tween 80 (1:1 ratio) and tested in concentrations ranging from 0.16 mg/mL to 21.2 mg/mL, 0.19 mg/mL to 24.91 mg/mL, and 0.17 mg/mL to 21.5 mg/mL, for T. articulata1, T. articulata2 and C. atlantica, respectively. After the serial dilution, each well was introduced with 20 µL of the standardized bacterial suspension at the concentration of 1 × 106 CFU/mL. After 24 h of incubation at 37 °C, resazurin dye was added to all wells, and microtiter plates were incubated in the dark at the same temperature for an additional 3 h. The lowest concentration of EOs with no visible color change was determined as MIC. To determine the MBC, 15μL from the wells corresponding to concentrations equal to or higher than the MIC were seeded onto blood agar plates (BAP). The plates were incubated for 24 h at 37 °C. Two independent experiments performed in triplicate were provided. Tween 80 was tested solely as a negative control, had no activity at the highest tested concentration which was used to dissolve the oils. Tri-antibiotic paste (TAP), being a reliable reference in root canal disinfection, was used as a positive control. 23 It was composed of ciprofloxacin (Ciprofloxacin®; Remedica LTD, Limassol, Cyprus; 200 mg), metronidazole (Orvagyl®; Galenika, Belgrade, Serbia; 400 mg) and minocycline (Minocin®; Pfizer, New York, NY, USA; 100 mg).22,24
Antibiofilm Activity Assessment
After the antibacterial screening, the EO obtained from T. articulata2 was selected for the antibiofilm activity assessment as it demonstrated superior efficacy compared to the other EOs. The antibiofilm assessment was provided using crystal violet assay 25 as previously explained in Nemoda et al, 26 to investigate their potential in the prevention of biofilm formation, as well as their reductive potential against already formed biofilms.
The Evaluation of the Potential of T. articulata2 EO to Prevent the Biofilm Formation of Selected Endodontic Isolates
In order to estimate the potential of T. articulata2 EO in the prevention of biofilm formation, bacterial suspensions (105 CFU/well) of each isolate were co-incubated with diluted EO at various concentrations (MIC/8, MIC/4, MIC/2, and MIC) in sterile TSB supplemented with 1% glucose at 37 °C for 48 h in 96 well microtiter plates with flat bottom (Sarstedt, Germany). Ater incubation, the medium was aspirated, and non-adherent (planktonic) cells were removed by washing with sterile saline. The remaining biofilms were air-dried, stained with crystal violet (0.1% Bio-Merieux, France) washed again after 15 min, and suspended in 96% ethanol. Absorbance was measured at 570 nm and the inhibition of biofilm inhibition (I) was calculated using the formula:
The Evaluation of the Potential of T. articulata2 EO to Disrupt Already Formed Biofilm of Selected Endodontic Isolates
In the case of the evaluation of T. articulata2 EO's potential to disrupt the biofilms, bacterial suspensions (2 × 105 CFU/well) of each strain was added into polystyrene microtiter plates (200 μL/well) and incubated for 48 h at 37 °C. Following biofilm formation, the medium was replaced, and the biofilm was treated with T. articulata2 EO at MIC/2, MIC, 2MIC, and 4MIC concentrations. After 24 h incubation period at 37 °C temperature, the disruption of biofilms was spectrophotometrically quantified at 570 nm, using the same staining method. 27 In both assays, Tween 80 was used as negative control, and its influence on biofilm formation and disruption was considered when interpreting EO's activity. Figure 3 illustrates the difference in EO's concentrations used in biofilm disruption and biofilm prevention assays.
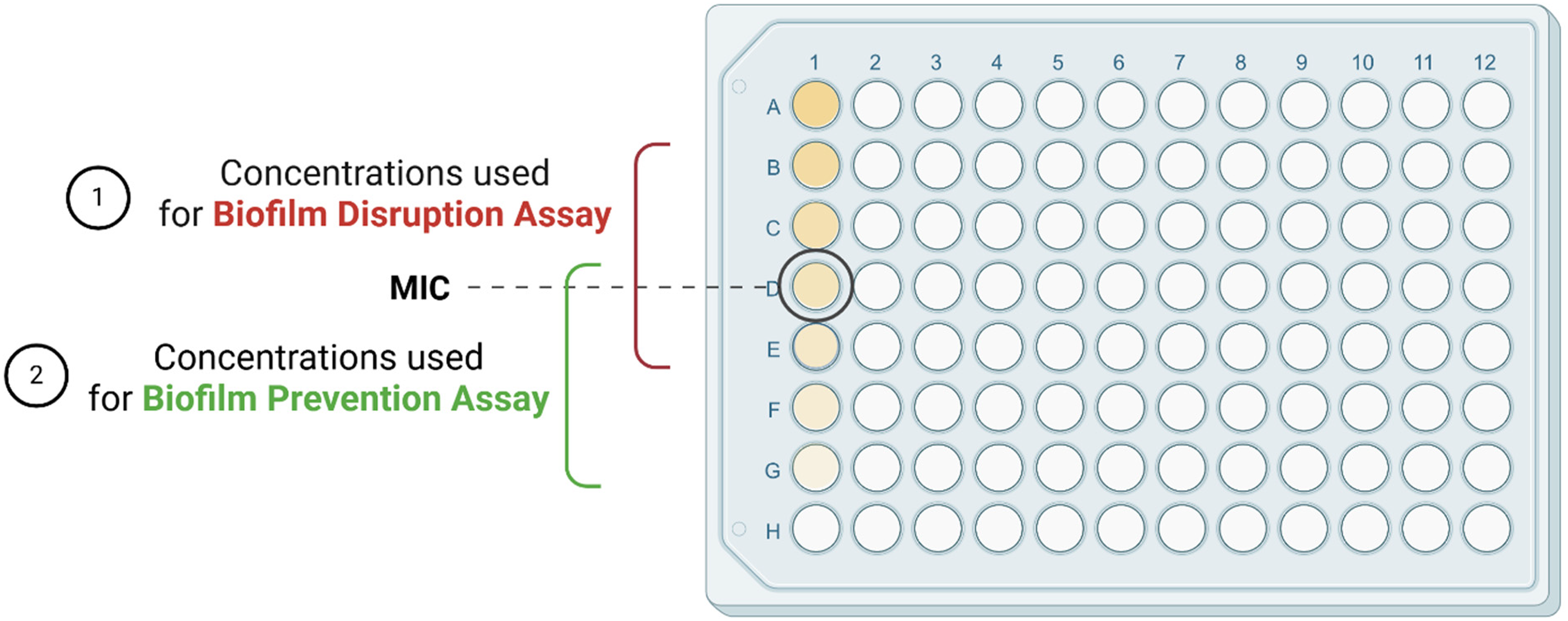
the Concentrations of the Essential Oils and Hydrolates Used for Biofilm Disruption Assay and Biofilm Formation Assay. Created with BioRender.com. MIC : Minimal Inhibitory Concentration.
Statistical Analysis
All experiments were performed in triplicate and results are expressed as mean ± standard deviation (SD). The statistical analyses were performed using One-way analysis of variance (ANOVA) statistical test from SPSS software, version 20.0 (SPSS 10 statistical package for Windows, IBM, Armonk, NY, USA). P values < .05 were accepted as the level of significance.
Results
Characteristics of Essential Oils
The EO originating from the aerial parts of T. articulata2 had a yellow color, agreeable smell, and yield of 0.45%. The EO from the aerial parts of T. articulata1, had a pale-yellow color with a less-accentuated odor and a yield of 0.2%. Leaves of C. atlantica provided a yield of 0.1% and the EO was colorless.
Antibacterial Potential of Essential Oils
The MIC and MBC values for all essential oils against the tested bacterial strains are summarized in Table 1. The inhibitory potential of T. articulata1 and C. atlantica EOs was low with MIC values of above 20 mg/mL, while the MIC values of T. articulata2 ranged from 4.78 ± 1.48 to 19.43 ± 5.48 mg/mL. Notable bactericidal potential was observed only against S. sanguinis and S. gordonii. Among the isolates, S. gordonii and S. sanguinis were the most sensitive, less than S. mitis, and the least sensitive was E. faecalis.
Antibacterial activity of the essential oils against E. faecalis and the Streptococcus isolates (mg/mL).

MIC : minimal inhibitory concentration ; MBC: minimal bactericidal concentration; T. articulata1 : Tetraclinis articulata harvested from northern Morocco ; T. articulata2 : Tetraclinis articulata harvested from center of Morocco. TAP: Tri-antibiotic paste composed of metronidazole, minocycline, and ciprofloxacin.
MIC values are given as mean ± SD of triplicate experiments.
Antibiofilm Activity of Essential Oils
Concerning the antibiofilm potential of T. articulata2, all tested concentrations (MIC/8–MIC) successfully inhibited endodontopathogenic biofilm formation, with inhibition rates ranging from 27.91% to 84.39%. Figure 4 shows the biofilm inhibition percentages of T. articulata2 EO at different concentrations against S. mitis, S. sanguinis, S. gordonii and E. faecalis. All concentrations demonstrated more than 60% inhibition of S. sanguinis and S. mitis biofilms, with the strongest effect observed at MIC, where T. articulata2 prevented S. mitis biofilm formation most effectively by 84.39% (D, Figure 4). Similarly, S. gordonii and E. faecalis biofilms were sensitive to T. articulata2, with the highest concentration being the most effective concentration, achieving 72.08% inhibition for S. gordonii (A, Figure 4) and 83.39% for E. faecalis (B, Figure 4).

Prevention of S. mitis, S. sanguinis, S. gordonii and E. faecalis biofilm formation by T. articulata2 essential oil. MIC : Minimal Inhibitory Concentration; C: Control. The results are expressed as the Mean Values ± Standard Deviation of two individual experiments. Each performed in five replicas. Statistical significance was tested using the One-way ANOVA. *Statistical significance (P < .05) in the biofilm biomass compared to the untreated Control.
Figure 5 shows the biofilm disruption percentages of T. articulata2 EO at different concentrations against S. mitis, S. sanguinis, S. gordonii and E. faecalis. T. articulata2 EO exhibited a disruptive effect on pre-formed biofilm of S. sanguinis and S. mitis across all tested concentrations (C and D, Figure 5). S. sanguinis biofilm was most sensitive at the lowest concentration (MIC/2), with a disruption rate of 74.93%. The effect on S. mitis biofilm was relatively uniform, with the highest disruption observed (69.76%) at MIC (D, Figure 5). Contrary to that, EO was not efficient against the biofilms of S. gordonii and E. faecalis and even estimated the production of their biofilms (Figure 5A and B).
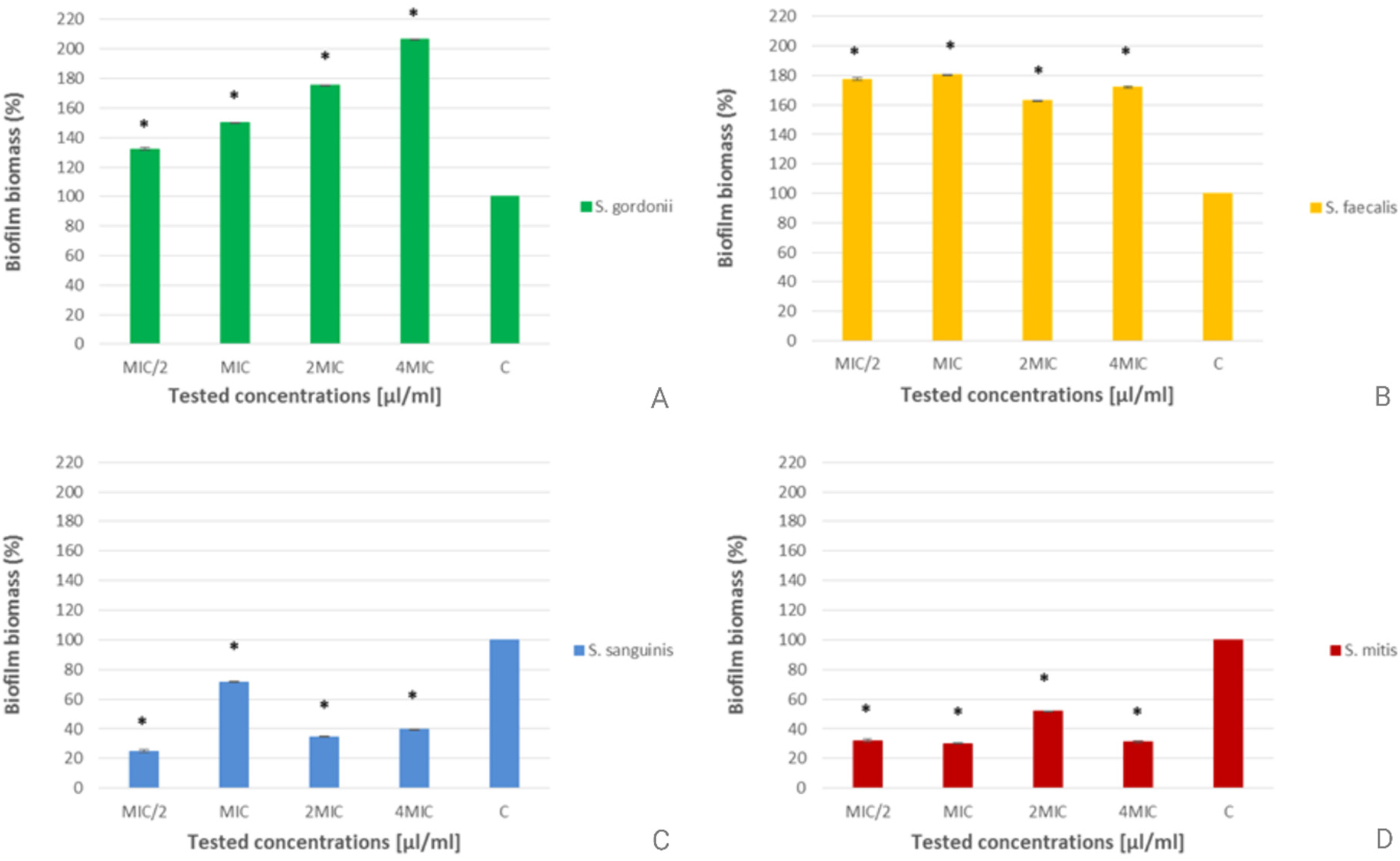
Disruption of S. mitis, S. sanguinis, S. gordonii and E. faecalis biofilms by T. articulata2 Essential oil. MIC : Minimal Inhibitory Concentration; C: Control. The results are expressed as the Mean Values ± Standard Deviation of two Individual experiments, each eerformed in five replicas. Statistical significance was tested using the one-way ANOVA. *Statistical significance (P < .05) in the biofilm biomass compared to the untreated control.
Discussion
In this study, two EOs from T. articulata and one from C. atlantica were extracted and screened for their antibacterial and antibiofilm potential against endodontic bacteria. Among the bacterial strains commonly involved in endodontic infections, several Streptococcus isolates, ie S. mitis, S. sanguinis and S. gordonii, and E. faecalis have been selected as model bacteria due to their prevalence and to their role in formation of the biofilm responsible for root canal infections.3,8 Up to our knowledge this study is the first to evaluate the antibacterial potential of the aforementioned oils against the tested Streptococcus isolates.
The EOs of T. articulata and C. atlantica have been extensively studied for their chemical composition in the literature. T. articulata EO, extracted from various plant parts, exhibits distinct chemical profiles. The EOs from the leaves are dominantly rich in bornyl acetate (16.5-35.05%), camphor (11.17-24.21%), and α-pinene (7.16-23.77%), 15 while the EOs derived from the wood primarily contain cedrol (28.2%), 1,7-di-epi-cedrol (17.9%), and cedrene (23%). 28 On the other hand, C. atlantica EO, characterized by its high sesquiterpene content, are commonly composed of α-himachalene (9.40-54.21%), β-himachalene (9.89-31.24%), γ-himachalene (7.62-15.54%), cedrol (up to 10.32%), and α-pinene (up to 14.85%) as a main constituents.29,30 Concerning their yield and color, it was previously suggested in the literature that the yield of EOs from T. articulata in Algeria ranged from 0.03% to 0.86%. 31 In Morocco, the yield was reported at 0.84%, 32 which is in line to the presented in this work. On the other hand, the yield of EOs from C. atlantica was reported to be approximately 0.9 to 1.1% 29 which is higher than the results of this study.
In the first section of the study, antibacterial evaluation showed that inhibitory potential of both T. articulata1 (originating from northern Morocco) and C. atlantica EOs were low. On the other hand, T. articulata2 EO (originating from central Morocco) showed antibacterial activity particularly against S. gordonii and S. sanguinis. According to the literature data, C. atlantica EO was previously screened for its antibacterial potential against E. faecalis reference strains ATCC 1034 33 and two EOs of T. articulata, one sourced from Khemisset and another from Oulmes, were tested against E. faecalis clinical isolates.20,34 In these studies, a higher antibacterial potential was observed than that presented in our study. The dissimilarity in oil activity could probably be attributed not only to the variability in the origin of the bacterial strains tested across the studies, but also to the difference in the oils composition. As the EO composition and yield from the same plant species can vary, notable differences in their antibacterial efficacy among samples collected from two different regions could be observed. For instance, EO of T. articulata2 demonstrated superior yield and a superior antibacterial property compared to T. articulata1, despite being derived from the same plant species. Similar was also previously suggested by Raal et al 35 and explained with the differences in climate, soil composition, altitude, and precipitation patterns.
The second part of the study was dedicated to exploring the antibiofilm potential of T. articulata2 EO, which emerged as the most effective EO against the tested planktonic bacterial species. This assessment is particularly important given that bacteria in the endodontic canal exist within biofilms, which provide protection against antimicrobial agents. 36 Endodontic biofilms are highly complex, and they exhibit increased resistance that goes up to 1000 times greater than in planktonic counterparts, due to their ability to adhere to unreachable parts of the intracanal system such as apical and lateral ramifications, and isthmuses. 37 Therefore, evaluating antibiofilm activity offers a more clinically relevant insight into the potential effectiveness of T. articulata2 EO.
In this study, both biofilm disruption and inhibition of biofilm formation were assessed. This distinction is essential, as an effective antimicrobial agent in endodontics should not only disrupt established biofilms in infected root canals but also prevent biofilm reformation, thereby reducing the risk of reinfection and treatment failure. 38 The results of the screening of the antibiofilm activity of T. articulata2 were quite promising especially when the prevention of the biofilm formation was screened. The prevention of the biofilm formation was observed for isolates responsible for the initiation of the formation process such as S.mitis, S. gordonii and S. sanguinis but also for the Enterococcus sp. Being known that E. faecalis poses biofilm that is commonly resistant to standard therapy 39 this is more than satisfactory result especially when the inhibition is observed in low concentrations such as MIC/8. Another encouraging result is that T. articulate EO have a potential against pre-formed biofilm of S. mitis and S. sanguinis, as well. This is encouraging in the light that it was suggested that that the mechanisms involved in biofilm inhibition and disruption may be distinct and that EOs may target different stages of biofilm development with varying efficacy. 40
The present study provides novel findings regarding the antibacterial and antibiofilm potential of T. articulata and C. atlantica EOs against endodontic clinical isolates. Yet, the following methodological constraints should be acknowledged. First, the absence of chemical characterization of oils, which makes the association of the observed activities with specific compounds challenging. Second, the experiments used clinical isolates rather than reference strains, and although it mimics clinical settings, it still may limit comparison with the existing literature. Additionally, the potential of the tested EO was screened against biofilm biomass using crystal violet staining, which could be additionally supported with viability testing to provide more profound information.
Conclusion
This study highlights the impact of regional variations when evaluating the potential of EOs for antimicrobial applications is required. EO of T. articulata from the Central Morocco emerged as the most effective EO against the tested bacterial species, demonstrating promising antibacterial properties. Among the four bacterial species examined, S. mitis and S. sanguinis showed the highest sensitivity to the EOs, S. mitis was less susceptible and E. faecalis was the least sensitive. However, the antibiofilm potential of the T. articulata2 EO was significant against all biofilms in their formation and against already formed biofilms of S. mitis and S. sanguinis. These results show the potential of T. articulata2 EO to be used as a part of novel strategies to combat endodontic bacterial infections.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Reporting Guidelines Statement
Reporting guidelines such as CONSORT or ARRIVE do not directly apply to this in vitro study; however, we have followed best practices for transparent and reproductible reporting.

