Abstract
Background and objectives
The therapeutic properties of local Cannabis sativa varieties remain underexplored. This study aimed to evaluate the antinociceptive effects of a Moroccan C. sativa ethanolic extract (CEE) in acute pain models.
Methods
Phytochemical analysis of CEE was conducted using HPLC, and antioxidant activity was assessed via DPPH and FRAP assays. Antinociceptive effects were evaluated using writhing and formalin tests after a single oral administration. Mice treated with CEE (30 mg/kg) or cannabidiol (CBD; 30 mg/kg) were compared to a diclofenac-treated group (10 mg/kg). Both treatments were tested for cannabinoid receptor-1 effects known as the cannabinoid tetrad.
Results
Of several polyphenolic compounds identified from CEE, kaempferol formed the main component. In vitro, CEE exhibited antioxidant activity, with IC50 values of 289.01 ± 0.003 μg/mL in the DPPH assay and 57.29 ± 6.7 μg/mL in the FRAP assay. Both CEE and CBD significantly reduced writhing responses, paw licking/biting time, and paw swelling, indicating antinociceptive and anti-inflammatory effects. The histopathological analysis supported these findings. Neither CEE nor CBD induced cannabinoid tetrad-related effects.
Conclusions
CEE exhibited significant antinociceptive and anti-inflammatory activities comparable to CBD and diclofenac, without cannabinoid tetrad-associated symptoms. Moroccan C. sativa extract shows promise for developing novel anti-inflammatory drugs.
Introduction
Acute pain is a universal experience and a normal physiological response to any noxious stimulus. The duration of acute pain is limited (<1 month), but it can develop into chronic pain if left untreated or not treated properly. 1 First-line treatment options for mild to moderate acute pain include acetaminophen and non-steroidal anti-inflammatory drugs (NSAIDs). 2 NSAIDs are widely used to treat various types of pain and many of them are available in pharmacies for self-medication and without any specialist medical advice, raising concerns about public health problems. The essential mechanism of action of NSAIDs is based on the inhibition of the cyclooxygenase enzymes (COX) responsible for the production of prostaglandins and, therefore, inducing their anti-inflammatory, analgesic, and antipyretic effects. 3 Despite their wide use, they are accompanied mainly by gastrointestinal problems that are pronounced in the case of prolonged use. NSAIDs may affect other organs (ie, liver, kidneys, lungs), and can also cause cardiovascular problems, with high risks of heart attack and stroke.3,4 As a result, the choice of analgesics depends on the type of pain, its severity, side effects, and the risk/benefit ratio. The development of new alternatives to NSAIDs is necessary to improve patient health and reduce the burden of adverse effects associated with their consumption. Consequently, the investigation of plant extracts may be regarded as a viable alternative approach in the search for novel analgesic medications.
For many decades, various plant-derived extracts have been used as analgesics, containing a complex list of synergistically acting compounds. One of the most widely used and extensively studied plants in recent years is Cannabis sativa. Many scientific studies have proposed cannabinoids as strong candidates for the treatment of various pathologies, including different types of pain.5‐8 The majority of anterior studies have focused on delta-9-tetrahydrocannabinol (Δ9-THC) as the essential component of this plant, producing interesting analgesic effects through its action on the cannabinoid type 1 receptor (CB-1R). 9 The use of Δ9-THC is limited by its euphoric effects, with a high risk of dependence. Consequently, research has turned towards the second non-psychoactive compound of the cannabis plant, cannabidiol. CBD, in turn, has proved its potential therapeutic effects on a variety of health problems, supported by its safe toxicological profile and its multi-target effects.10‐12 It is interesting to note that Cannabis sativa contains about 113 phytocannabinoids, not only Δ9-THC and CBD, but 120 terpenes, and other compounds that act in a complex process to exert their effects.13,14 Therefore, studies conducted with purified cannabinoids may not accurately reflect the potential effect of the whole plant's extracts.
Moroccan cannabis, traditionally known as “kif”, has been cultivated for centuries in the Rif region of Morocco, which offers a distinctive environment that contributes to the plant's characteristics. The local strains are particularly well adapted to the challenging climate of the Rif mountains, which may influence their phytochemical composition, including cannabinoid and terpene profiles. Recently, a study revealed a remarkable lack of data on Moroccan local varieties of cannabis in terms of phytochemical properties, genetics, and therapeutic applications. 15 Therefore, the present study aims to investigate the analgesic effects of an ethanolic extract of Moroccan cannabis (CEE) compared with an isolated cannabinoid (CBD) in an animal model of acute pain.
Results
Determination of TPC, TFC, TTC, TSC
The quantification of specific chemical compounds in the extract revealed the existence of significant levels of polyphenols (18,31 ± 0.06 mg GAEq/g DM), flavonoids (32.01 ± 1.46 mg CEq/g DM), condensed tannins (42.03 ± 3.93 mg CEq/g DM), and saponins (1.21 ± 0.03 mg OAEq/g DM) (Table 1).
The Total Phenolic, Flavonoids, Condensed Tannins, and Saponins Content in Cannabis Ethanolic Extract.

Values are expressed as mean ± SEM.
CEE Antioxidant Activity
The in vitro antioxidant activity of our extract, assessed via DPPH and FRAP assays, is detailed in Table 2. The CEE exhibited a notable activity in both tests, with IC50 values of 289.01 ± 0.003 and 57.29 ± 6.7 µg/ml, respectively. It is noteworthy that the extract exhibited an antioxidant capacity slightly comparable to that of quercetin in the FRAP assay.
In Vitro Antioxidant Activity of Cannabis Ethanolic Extract Determined by DPPH and FRAP Methods.

Values are expressed as mean ± SEM.
HPLC Profile
The high-performance liquid chromatography analysis of the polyphenol constituents in the CEE identified several compounds, with kaempferol being the predominant component. Other identified polyphenolic compounds included gallic acid, catechin, ferulic acid, naringin, ellagic acid, and naringenin (Figure 1).

HPLC Analysis of CEE Polyphenols. a: Gallic Acid; b: Catechin; c: Ferulic Acid; d: Naringin; e: Ellagic Acid; f: Naringenin; g: Kaempferol.
Cannabinoid Tetrad Assay
Cannabis ethanolic extract and cannabidiol did not induce CB receptor-mediated effects characterized by hypothermia, catalepsy, and hypomotility. Meanwhile, tested compounds exhibited a significant antinociceptive effect in the hot-plate test (Figure 2).

The Effects of Treatment (p.o.) with Cannabis Ethanolic Extract (30 mg/kg, n = 6) and CBD (30 mg/kg, n = 6) in the Cannabinoid Tetrad assay. (A) Temperature; (B) Locomotion Activity; (C) Nociception; (D) Catalepsy. The Data Represent the Mean ± SEM. *p < 0.05 versus Control Group; ns: Not Significant.
Antinociceptive Effect in the Writhing Test
As shown in Figure 3, oral administration of CEE and CBD significantly reduced the number of writhes (p < 0.001) when compared with the vehicle group. The percentage of inhibition in CEE, CBD, and Diclo groups reached 30.23%, 32.41%, and 34.30%, respectively (Table 3). Meanwhile, no significant differences were observed between the three groups.
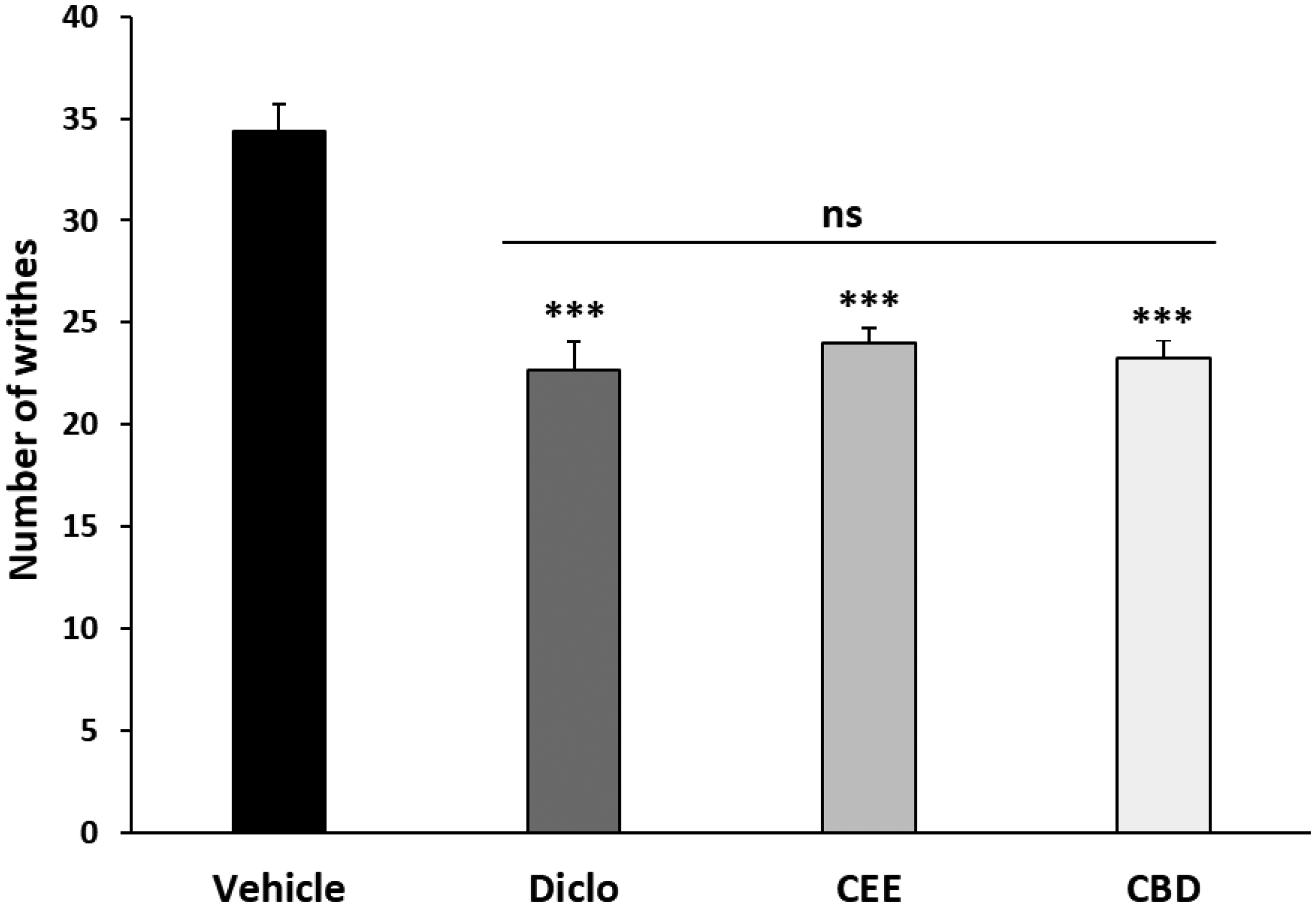
Acetic Acid Induced Writhing Responses in Mice Orally Administered with Cannabis Ethanolic Extract, Cannabidiol, and Diclofenac, and Intraperitoneally Injected with 0.7% Acetic Acid. the Data Represent the Mean ± SEM. ***p < 0.001 Versus the Vehicle Group; ns: Not Significant.
The Inhibition Percentage of CEE, CBD, and Diclofenac Administered by Oral Gavage on Abdominal Contractions in Mice.

***p < 0.001 as compared to vehicle group.
Antinociceptive Effect in the Formalin Test
CEE and CBD had similar effects and significantly reduced the duration of licking/biting the injected paw when compared with the vehicle group in both phases of the test. In contrast, diclofenac treatment failed to exhibit any effect in the first phase but significantly alleviated pain responses in the second phase without being significantly different from other treatments (Figure 4).

The Effect of Cannabis Ethanolic Extract, Cannabidiol, and Diclofenac on Formalin-Induced Paw-Licking Test in Phase One (0-5 min) and Phase Two (20-30 min). The Data Represent the Mean ± SEM. *p < 0.05, **p < 0.01 Versus the Vehicle Group; ≠p < 0.05 Versus the Diclofenac Group; ns: Not Significant.
Formalin-Induced Paw Edema
Paw thickness (Figure 5A) and paw weight (Figure 5B) were significantly decreased after oral administration of CEE, CBD, and Diclo compared with the vehicle group following formalin injection.

Paw Edema Measurements. Delta paw Thickness (A) and Paw Weight (B) Calculated by Ipsilateral and Contralateral Differences. the Data Represent the Mean ± SEM. *p < 0.05, **p < 0.01, *** p < 0.001 Versus the Vehicle Group; ≠p < 0.05, ≠≠p < 0.01 Versus the Diclofenac Group; ns: Not Significant.
Histopathology
The histopathological examination of formalin-injected paws stained with Hematoxylin/Eosin revealed a significant inflammatory reaction in the dermal region and near the basal part of the epidermis in the vehicle group. Treatment of mice with cannabis ethanolic extract, cannabidiol, and diclofenac caused a decrease in the level of inflammatory cell infiltration (Figure 6).

Photomicrographs of Mice Hind Paw Tissues Stained with H&E. (A) Control Group; (B) Vehicle Group; (C) Diclofenac Group; (D) Cannabidiol Group; (E) Cannabis Ethanolic Extract Group; Der: Dermis; Ep: Epidermis; Hy: Hypodermis; KL: Keratinous Layer. Arrows: Inflammatory Cells Infiltration. Magnification: × 200 and ×400.
Discussion
The methodology employed for cannabis extraction in the present study was described by Bowen and colleagues. 16 In their investigation, the authors employed several commonly used extraction protocols to characterize the chemical composition of a cannabis cultivar, and based on their results, a higher abundance of phytocannabinoids is observed in an alcoholic extract with ethanol, justifying our extraction protocol. The use of ethanol as an extraction solvent is well known for its power to extract molecules with antioxidant effects such as polyphenols, flavonoids, and others. 17 In light of the limitation regarding the inability to assay cannabinoids, efforts have nevertheless been made to phytochemically characterize the extract, particularly with regard to other compounds. Indeed, this phytochemical analysis revealed the presence of substantial quantities of polyphenols, flavonoids, tannins, and saponins, which provide an explanation for the antioxidant capacity observed in the DPPH and FRAP assays. The results obtained from the aforementioned tests indicate that CEE is capable of reducing Fe3+ to Fe2+ ions and scavenging free radicals in a manner consistent with the findings of several other studies in the literature.18‐21 Subsequently, an HPLC analysis was conducted to ascertain the composition of CEE in terms of polyphenols, which revealed kaempferol to be the predominant compound. Other identified polyphenolic compounds included gallic acid, catechin, ferulic acid, naringin, ellagic acid, and naringenin. Flavonoids and phenolic compounds, such as kaempferol and naringenin, have been reported to exert analgesic, anti-inflammatory, and antioxidant properties, which may contribute to alleviate pain. Nevertheless, potential genotoxic concerns associated with kaempferol have been noted in some studies and should be taken into consideration. 22 Of note, the cannabinoid tetrad assay demonstrated that neither the cannabis ethanolic extract nor cannabidiol induced CB receptor-mediated effects. Therefore, we have concluded that our extract does not contain a high level of Δ9-THC.
The main objective of this study was to demonstrate the antinociceptive potential of a Moroccan cannabis ethanolic extract in comparison with pure cannabidiol and a reference drug. Consequently, the antinociceptive activity of our extract was assessed by 2 commonly used tests to screen potential therapeutic agents for anti-inflammatory and analgesic properties: the acetic acid-induced writhing test and formalin-induced paw-licking test.
The injection of acetic acid into the peritoneal cavity induces painful responses and has been shown to activate nociceptors innervating the peritoneum, inducing inflammation through the release of cytokines and inflammatory mediators such as TNFα, IL-1β, IL-8, PGE2, and PGF2a.23,24 These endogenous changes result in typical physiological effects of writhing such as abdominal contractions, rotation of the pelvic region, and stretching of hind limbs. 24 In this test, CEE significantly reduced the number of abdominal constrictions compared with the vehicle group, and remarkably with a similar effect as cannabidiol and diclofenac.
The formalin test is characterized by two distinct phases. The first phase (phase one) represents acute pain resulting from C-fiber activation after formalin injection and lasts up to 5 min. The second phase (phase two), starting 20 min after injection, is an inflammatory phase mediated by the secretion of several inflammatory factors such as substance P, bradykinins, histamine, serotonin, and prostaglandins, which activate the TRPV and TRPA receptors and downstream signaling pathways.25‐27 Our results showed that CEE and CBD significantly decreased the paw licking/biting behaviors in both phases. Interestingly, no significant differences were observed between the tested groups and diclofenac in the second phase. Paw edema measurements demonstrated, as expected, that both CBD and CEE significantly reduced the thickness and weight of injected paws. To corroborate the findings observed in animal behavior and paw measurements, a histopathological study was conducted on the injected paws of all groups. The vehicle group exhibited edema associated with a notable rate of inflammatory infiltrates in comparison to the control group. The histological analysis of the mice paws treated with CEE, CBD, or diclofenac revealed a relatively mild inflammatory reaction in comparison to the vehicle group. CEE administration showed a protective effect of paw tissue against inflammatory reactions.
Several studies are consistent with our results. Early investigations showed that in a comparative study, cannabis extract was as effective as Δ9-THC, while CBD showed no analgesic effect in the writhing and hot-plate tests. 28 Also, an ethanolic cannabis extract was able to inhibit phenyl-p-benzoquinone (PBQ)-induced extension movements, and CBD was effective at low doses. 29 A clinical trial in 2003 showed that cannabis extracts rich in Δ9-THC, CBD, or with a 1:1 ratio significantly reduced pain after two weeks of treatment in patients suffering from chronic pain. 30 More recently, an aqueous extract of cannabis roots (p.o.) was shown to significantly reduce the number of abdominal contortions and pain behaviors in both phases of the formalin test. 31 Regarding CBD, studies have demonstrated its antinociceptive effect at a range of doses in the formalin test, 32 in the writhing test, 33 and recently in a cream form. 34 On the other hand, a study carried out in 2022 showed that 10 mg/kg CBD had an antinociceptive effect in phase one of the formalin test in male mice, while it had no effect in female mice in the two phases of the test. 35 Another study showed that CBD at a dose of 30 mg/kg was ineffective in both phases of the orofacial formalin test. It is important to note that although the formalin test induced in the paw and orofacial regions evaluates the antinociceptive effect induced by the same agent, there is a difference in the underlying mechanisms between the two. In the paw, spinal control mechanisms are involved, whereas at the orofacial level, they are not. 36
The combination of two cannabinoids has also been studied. It is widely known that CBD mitigates certain adverse effects of Δ9-THC when combined, including intoxication, sedation, tachycardia, and dependence.37‐39 A recent study in 2022 evaluated the analgesic effect of cannabidiol in combination with a cannabis sesquiterpene, β-caryophyllene (BCP). This study showed that CBD, BCP, or their combination had no effect on phase one but produced a dose-dependent effect in phase two via anti-inflammatory mechanisms. 40
In addition, other contradictory studies need to be addressed. Recently, a study carried out on six chemovars with different cannabinoid profiles showed that their antinociceptive effect, as assessed by the hot-plate, was comparable to that of Δ9-THC alone. 41 Another investigation in 2019 revealed that a THC-rich extract administered intraperitoneally reduced the number of abdominal contractions and increased latency time in hot-plate and tail-flick tests. 42 The main conclusion of these studies is that the extract effect was attributed essentially to Δ9-THC, with the other constituents exerting minimal to no influence on these effects.
When studying a total extract containing several molecules, it is difficult to understand the mechanism by which its components exert their effect. Indeed, as evidenced by HPLC, kaempferol, the primary polyphenol in our extract, renowned for its antioxidant properties, has also been shown to possess antinociceptive and anti-inflammatory effects, as shown by several tests, including the acetic acid writhing test and the formalin test.43,44 It has been demonstrated that intracerebroventricular microinjections of kaempferol induce the aforementioned effects via modulation of TRPV1 receptors and the opioidergic and GABAergic systems.45,46 It is therefore plausible that kaempferol may be responsible, at least in part, for the observed CCE activity.
Several scientific studies have shown that cannabinoids, too, like the non-cannabinoid fraction, possess antioxidant and anti-inflammatory properties.18,47‐50 Cannabinoids are believed to act by mechanisms including those within the spinal, supraspinal, and peripheral regions. 51 Another possible mechanism is targeting the endocannabinoid system, which is involved in many physiological systems, including pain perception, emotional state, learning, memory, and reward behaviors. Modulation of this system by cannabinoids appears to be effective in many disorders and diseases, including pain.52,53 We also believe that the observed effects of the whole extract may result from complex interactions between its various constituents, including both cannabinoid and non-cannabinoid compounds. 54
Limitations
While this study explores the promising potential of the CEE in acute pain models compared to a well-known purified cannabinoid (CBD), some limitations need to be considered. This study was limited by its acute single-dose design, which does not provide insight into the long-term effects of the studied treatments. Additionally, the extract did not exhibit significantly greater effects compared to cannabidiol. This could be explained in part by the short treatment duration and in part by the complex composition of the extract, where interactions among constituents may influence the overall activity. This underscores the importance of thoroughly investigating the cannabinoid content within the extract. Furthermore, no mechanistic studies were performed, limiting understanding of how these compounds exert their effects. Consequently, future work should explore chronic administration, isolate key compounds, and investigate underlying mechanisms.
Conclusions
The administration of a single dose of CEE (extract) at 30 mg/kg had an antinociceptive and anti-inflammatory effects, as evidenced by a reduction in the number of writhes (writhing test), as well as decreased paw licking/biting time, paw edema, and inflammatory cell infiltration (formalin test). These effects were comparable to those of CBD and diclofenac. Of several identified compounds in the extract, kaempferol was the most prominent. The extract exhibited noteworthy antioxidant activity in both the DPPH and FRAP assays. The cannabinoid tetrad assay was performed to determine whether the extract had a THC-like effect. The results showed that the extract did not induce hypothermia, hypolocomotion, or catalepsy, which could suggest the absence of a THC-like effect. However, the extract did have an analgesic effect on the hot plate, which exhibits the potential to alleviate acute pain. These encouraging results require further in vitro studies as well as preclinical and clinical tests for efficacy and safety based on extended duration of treatment that considers other types of pain.
Materials and Methods
Cannabis Ethanolic Extract Preparation
The extract was prepared from dried aerial parts powder of Cannabis sativa and subjected to a decarboxylation step at 135° C for 40 min and then macerated with pure ethanol (1 g of powder in 10 mL EtOH) for 48 h with continuous shaking. 16 After filtration, the solution was submitted to a winterization process (−22° C) for another 48 h. The solution was filtered and then condensed by a rotary evaporator (45° C and 90 rpm). The extract gives a sticky, dark green residue and was placed at 4° C in glass vials covered with aluminum until use. A dose of 30 mg/kg of the CEE was selected for investigation in this work based on our preliminary work and supported by other studies with modifications.55,56
Phytochemical Analysis of CEE
Determination of Total Phenolic, Flavonoids, Saponins, and Condensed Tannins Contents
The total phenolic content (TPC) of CEE was determined according to the Folin–Ciocalteu method. 57 A total of fifty microliters of CEE were mixed with distilled water and Folin–Ciocalteu reagent. After 3 min of incubation, 20% sodium carbonate was added, and the solution was stirred and incubated for 60 min in the dark at room temperature. The measurement was done in triplicate, and the absorbance was set to 725 nm. The results were expressed in milligrams equivalent of gallic acid per gram of dry plant material (mg GAEq/g DM).
The total flavonoids content (TFC) of CEE was determined by using the aluminum trichloride method. 58 A volume of one hundred microliters of the extract was mixed with distilled water and 5% sodium nitrite solution. After 5 min of incubation, 10% aluminum trichloride solution was added and then incubated for another 5 min. Thereafter, sodium carbonate solution (1 M) and distilled water were added and stirred. The measurement was done in triplicate, and the absorbance was set to 510 nm. The results were expressed in milligrams equivalent to catechin per gram of DM (mg CEq/g DM).
The total condensed tannin content (TTC) in CEE was evaluated by the vanillin method with slight modifications. 59 Briefly, vanillin solution (4%) and pure HCl were added to the extract. The solution was stirred and incubated for 15 min in the dark at room temperature. The measurement was done in triplicate, and the absorbance was set to 550 nm. The results were expressed as mg catechin equivalent per gram of DM (mg CEq/g DM).
The determination of total saponin content (TSC) in CEE was performed according to a previous method with minor modifications. 60 Briefly, 0.1 mL of vanillin–glacial acetic acid solution (5% v/v) was added to 0.1 mL of extract. Then, 0.6 mL of perchloric acid was added to the mixture and incubated for 5 min at 50 °C. The mixture was then cooled to room temperature. Finally, 1.7 mL of ethyl acetate was added. The measurement was done in triplicate, and the absorbance was measured at 550 nm. The results were expressed as mg oleanolic acid equivalent/g of DM (mg OAEq/g DM).
In Vitro Antioxidant Activity of CEE
DPPH assay. The CEE antioxidant power was evaluated by using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging test.
61
Various concentrations of CEE in methanol (50 μL) were prepared and mixed with 2 mL of DPPH methanol solution (0.004%). After 20 min of incubation in the dark at room temperature, the absorbance was read at 517 nm. For each concentration, the measurement was carried out in triplicate and the activity was calculated as the percentage of decrease of DPPH absorbance versus control. The following formula was used:
Where Aa is the absorbance of DPPH alone in methanol (control), and Ab is the absorbance of DPPH in the presence of CEE.
The results were expressed as half-inhibitory concentration (IC50 μg/mL), which represents the concentration of the sample required to scavenge 50% of the DPPH free radicals. Quercetin and butylated hydroxyl toluene (BHT) were used as positive controls.
Ferric reducing antioxidant power (FRAP). The ability of CEE to reduce Fe3+ to Fe2+ was evaluated according to Oubella and colleagues. 62 Briefly, the extract was mixed with phosphate buffer and potassium ferricyanide (1%). After 20 min of incubation (50° C), trichloroacetic acid (10%) was added, and the mixture was then centrifuged for 10 min (3000 rpm). Finally, the upper layer of the mixture was combined with ferric chloride (0.1%) and distilled water. The measurement was carried out in triplicate, and the absorbance was read at 700 nm. The results are expressed as IC50 (μg/mL). Quercetin and butylated hydroxyl toluene (BHT) were used as positive controls.
HPLC Analysis
To characterize the composition of CEE in terms of phenolic compounds, a high-performance liquid chromatography (Knauer) equipped with a K-1001 pump and a PDA detector (200-700 nm UV-V) was used. The analysis was conducted using Eurospher II 100–5 column (4.6 × 250 mm) at a temperature of 25° C. The flow rate was set at 1 mL/min, the injected volume of the sample was 10 μL, and the wavelengths of detection were set at 280 nm. The mobile phase consisted of formic acid 0.1% (A) and methanol (B) mixture with a total running time of 30 min. 63 The identification of the phenolic compounds was conducted through a process of comparison involving the retention time and UV spectra with external standards.
Animals
Adult Swiss mice weighing between 30 and 35 g were used in this study. The animals were housed in the Faculty of Sciences Semlalia, Cadi Ayyad University facilities under standard conditions with controlled temperature (23° C), 12 h dark/light cycle, and free access to food and water. Efforts were made to minimize animal suffering, and all procedures were approved by the local Committee of Directors of the Faculty Research Laboratories and carried out in accordance with Directive 86/609/EEC of the European Council on the protection of animals used for scientific purposes and the ARRIVE guidelines. 64
Assessment of Cannabinoid Tetrad Assay
In a separate experiment, to determine whether the molecules tested had a cannabimimetic effect as a result of their action on the CB-1R, we evaluated the tetrad in female mice (locomotion, temperature, catalepsy, and nociception) in that same order as previously described. 65 Animals (six mice/group) were orally administered with saline, CEE, or CBD, and 1 h later, they were subjected to testing. Locomotion was evaluated in the open field apparatus (50 × 50 × 40 cm) and the distance moved over 5 min was calculated. Rectal temperature was measured using a digital thermometer accurate to 0.1 °C at a depth of 2 cm. Catalepsy was measured by calculating the time taken by the animal to descend a horizontal bar, and nociception was measured by the hot-plate test (55 ± 0.2 °C) with a cut-off threshold of 20 s.
Acetic-Acid-Induced Writhing Test
A total of twenty-eight adult male Swiss albino mice were assigned to receive an intraperitoneal injection of 0.7% acetic acid, causing abdominal constrictions as described previously.
66
Animals were divided into four groups (n = 7) and administered orally (10 mL/kg) either with olive oil as a vehicle group, 30 mg/kg CEE, 30 mg/kg CBD (99%, Spectrums Europe),
67
or 10 mg/kg of a non-steroidal anti-inflammatory drug diclofenac sodium (Diclo: Pharma 5). All treatments were administered 1 h before acetic acid injection. The animals’ behavior was observed and recorded for 10 min to calculate the total number of writhes starting 5 min after injection. A “writhe” response was indicated by the contraction of the abdomen, together with twisting and stretching of the hind limbs. One incomplete writhe is considered one-half writhe. Accordingly, two half writhes are considered one full writhe. Analgesic activity was determined by calculating the percentage of inhibition with the following formula:
Formalin-Induced Paw-Licking Test
The formalin test was performed as previously described. 25 In this experiment, mice were given testing drugs by oral gavage (10 mL/kg) 1 h before formalin injection. Thereafter, animals received an injection of 20 μL of either saline or 5% formalin solution into the plantar surface of the right hind paw. Immediately, animals were put into a transparent chamber to start the observation period of the first phase (0-5 min), and then the second phase (20-30 min). Time spent licking/biting the injected paw was used as a pain response. Animals were assigned into five groups (n = 7) as follows: control group received intraplantar injection of 20 μL saline and oral gavage of saline, vehicle group received intraplantar injection of formalin and oral gavage of olive oil, while CEE, CBD, and diclofenac groups received intraplantar injection of formalin and oral gavage of 30 mg/kg cannabis ethanolic extract, 30 mg/kg cannabidiol, and 10 mg/kg diclofenac, respectively.
Paw Edema Measurements
Paw edema induced by formalin was calculated by quantifying differences in hind paws’ weight and thickness. 25 One hour after formalin injection, animals were euthanized and both hind paws were amputated at the ankle joint. They were weighed on a microbalance and the maximal dorsoventral thickness of each hind paw was measured with a digital Vernier caliper. The difference between the injected (right) and non-injected (left) hind paws was used to calculate edema.
Histopathological Analysis
After paw measurements, ipsilateral paws were collected and fixed in 10% buffered formalin. Fixed tissues underwent paraffin embedding, 5 μm thick sections, and then stained with hematoxylin–eosin (H&E). All tissues were observed and photographed using a Zeiss-Axioskop 40 microscope with a Nikon digital camera.
Statistical Analysis
Results are expressed as mean ± standard error of the mean (SEM). Statistical analysis was performed using one-way ANOVA, followed by the Holm–Sidak post hoc test to compare means between different groups. The normality was assessed using the Shapiro–Wilk test. For data exhibiting a non-normal distribution, the Kruskal–Wallis one-way ANOVA for ranks was employed, followed by Dunn's multiple comparisons test. P-values less than 0.05 (p < 0.05) were considered significant. All statistical analyses were conducted using SigmaPlot 12.5 software.
Footnotes
Ethical Considerations
All experimental procedures involving animals were approved by the local Committee of Directors of the Faculty Research Laboratories and were conducted in accordance with Directive 86/609/EEC of the European Council on the protection of animals used for scientific purposes, as certified by the Dean of the Faculty.
Author Contributions
Conceptualization, E.G.H.; methodology, E.G.H. and O.M.; formal analysis, E.G.H.; investigation, E.G.H., O.M., and B.K.; resources, S.R.; writing—original draft preparation, E.G.H.; writing—review and editing, O.M., B.K., S.R., B.L., G.H., and K.N.; supervision, S.R., B.L., G.H., and K.N.; All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this study are available in this article.
Statement of Human and Animal Rights
All animal housing and experiments were conducted in strict accordance with the institutional Guidelines for Care and Use of Laboratory Animals at the Faculty of Sciences Semlalia, Cadi Ayyad University.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
