Abstract
Background
Rhizome Angelica sinensis and Ligusticum sinense ‘chuanxiong’ are the main medicinal herbs used in traditional Chinese or Asian medicine to treat alopecia likely associated with their expertise in invigorating blood circulation. However, the effects of the volatile essential oils present in their aboveground parts are unclear.
Objective
To evaluate the general safety of essential oils from A. sinensis and L. sinense ‘chuanxiong’ leaves and evaluate their hair grow promoting effect on model mice and cells of dehydrotestosterone-induced androgenetic alopecia.
Methods
Essential oil safety was evaluated based on acute oral/dermal/eye irritation toxicity tests on laboratory animals. Essential oils (5%, 3% and 1%) were topically applied to model mice with dehydrotestosterone-induced androgenetic alopecia for 28 days. They (50-200 mg/L) were appended to the monoculture of model cells of dehydrotestosterone-induced androgenetic alopecia for 48 h.
Results
Both essential oils showed no acute toxicity. Mice dorsal skin indicated faster hair regrowth initiated by essential oils than by 3% minoxidil. Histological analysis shows that both essential oils increased the follicle number and dermal thickness in mice treated with dehydrotestosterone, with 1% A. sinensis and 3% L. sinense ‘chuanxiong’ essential oils performing the best. The 1% A. sinensis and 3% L. sinense ‘chuanxiong’ essential oils exhibited an average number of hair follicles (pcs/field of view) of 69.73 and 70.13, respectively, which were significantly larger than that of the model (14.80) and minoxidil (36.53) treated mice. Both essential oils significantly reversed the dehydrotestosterone-induced inhibition of secretion of hair growth promotors, the vascular endothelial growth factor and keratinocyte growth factor, in cultured cells.
Conclusion
A. sinensis and L. sinense ‘chuanxiong’ essential oils may act as safe and effective adjuvant agents in aromatherapy and cosmetics to treat human hair loss.
Keywords
Introduction
Alopecia is a common autoimmune disease featuring hair loss triggered by genetic and environmental factors. 1 Androgenetic alopecia (AGA), also known as male pattern baldness, is the most common type of alopecia affecting up to 50% men and women characterized by gradual hair shedding and thinning and receding hairline any time after puberty. 2 The disease not only impacts the appearance but also causes psychological/psychiatric problems such as anxiety, depression and lower self-confidence. 3
AGA is marked by progressive miniaturization of scalp follicles, shortened anagen phase and prolonged telogen phase. 4 AGA is majorly instigated by the androgenic substance, dihydrotestosterone (DHT), which is transformed from testosterone by the 5α-reductase enzyme. 5 DHT mainly binds to the androgen receptor on dermal papilla cells (DPCs) located at the bottom of the hair follicle, initiating a complex series of enzymatic reactions and adversely regulating the production of growth-stimulating factors, such as the vascular endothelial growth factor (VEGF), basic fibroblast growth factor and keratinocyte growth factor (KGF), and growth-inhibiting factors, such as transforming growth factor β1 and interleukin 1α. 6 Consequently, hair becomes shorter and finer, and finally baldness appears.
Currently, only two drugs are approved by U.S. Food and Drug Administration (FDA) to treat AGA, which are oral finasteride and topical minoxidil (MXD). But they are costly, require perpetual treatment to sustain results and have various side effects. For example, finasteride induces anaphrodisis, gynecomastia and sexual dysfunction, while minoxidil causes pruritus and irritation. 2 So far, non-FDA-approved AGA adjunctive therapies such as phytomedicine are gaining attention and popularity. Notably, topical application of plant-derived essential oils shows great potential in AGA treatment due to their percutaneous property, multicomponent trait, easy availability and fewer side effects. Traditionally, essential oils from cedarwood (Cedrus atlantica), lavender (Lavandula agustifolia Mill.), thyme (Thymus vulgaris) and rosemary (Rosmarinus officinalis) have been used anecdotally to treat hair loss for over 100 years; however, few associated scientific work has been published. In 1998, a seven-month randomized, double-blind study demonstrated that aromatherapy and massage using these four essential oils significantly improved the hair growth in 44% patients with alopecia areata, while only 15% patients showed improvement in the control. 7 More recently, in a randomized comparative trial with 100 AGA patients, topical application of rosemary essential oil-containing lotion (3.7 mg/mL) showed positive effect on hair loss, which was noninferior to 2% MXD. 8 In another study, topical application of 3% peppermint (Mentha piperita) essential oils was proved via animal experiment to significantly increase dermal thickness and follicle number in the dorsal area of mice, which was more prominent than 3% MXD. 9 These studies provide initial clinical or experimental trials for the therapeutic efficiency of a few essential oils; however, the number of investigated essential oils is a far cry from what is needed to form diversified compound essential oil formulae used in aromatherapy and cosmetics. In addition, these studies fail to include the safety application concentration which is a prerequisite for a safe application. In view of this, more trials with robust experimental design on different essential oils are needed to further illustrate their therapeutic efficiency on hair growth.
Angelica sinensis, known as Dang Gui (in Chinese), and Ligusticum sinense ‘Chuanxiong’ known as Chuanxiong (in Chinese), are two of the most common edible Chinese herbs. Traditionally, their roots are the parts used in alopecia prescriptions in Asia.10,11 The hair growth promoting effects of 70% ethanol extracts of A. sinensis and L. sinense ‘Chuanxiong’ roots have been witnessed on experimental mice.12,13 Nevertheless, the medical value of their aboveground parts is hardly investigated. A. sinensis and L. sinense ‘Chuanxiong’ are widely cultivated in many places of China. Each year after root harvesting, their leaves are discarded as waste for incineration. It is known that the aboveground part of these two herbs also contains aroma and the essential oils extracted from leaves may resemble that from roots in chemical composition. Also, the essential oils penetrate skin more readily than the aqueous extracts. The potential of leaf essential oils from two herbs to treat AGA deserves investigation.
In the current study, we uniquely focus on leaf-derived essential oils of two herbs. Acute toxicity of two essential oils extracted from leaves was firstly evaluated. Subsequently, their effects on hair growth were investigated in model mice with AGA induced by DHT. Finally, two practical cell models of DHT-AGA were established. It is known human hair follicle is mainly composed of hypodermal cells and epidermis cells, with the former represented by DPCs and the later keratinocytes. We used mice DPCs and human immortalized keratinocytes (HaCaTs) as substitutes in DHT-induced AGA modeling to examine secretion of the hair growth-stimulating factors in model cells. This study will provide more scientific support for aromatherapy-based treatment of AGA and help expand the essential oil recipe pool.
Materials and Methods
Animals
Institute of Cancer Research (ICR) mice, aged 5-6 weeks, weighing 26-30 g, and Sprague Dawley (SD) rats, weighing 180-200 g, were purchased from Shanghai Lingchang Biotechnology Co., Ltd, China. Mice were kept at Shanghai Jiao Tong University Laboratory Animal Center (Shanghai, China). The temperature was 22 ± 2 °C and the relative humidity of the feeding environment was 50 ± 5%. The light/dark cycle was 12 h/12 h. The animal experiment started after 7-day adaptive feeding.
The experiments were approved by the Institutional Animal Ethics Committee of Shanghai Jiao Tong University with approval number A2019001. The animals were manipulated in accordance to ARRIVE Guidelines for animal ethics. All efforts were made to minimize both animal suffering and the number of animals used to produce credible data.
Materials and Reagents
The A. sinensis essential oil (AEO) and L. sinense ‘chuanxiong’ essential oil (SEO) were purchased from Sichuan Yingu Aromatic Technology Co., Ltd (Sichuan, China; AEO's batch number:211101; SEO's batch number: 210701). Essential oils were extracted by steam distillation. The species of two plant materials was checked with the planting base.
DHT and ethoxylated hydrogenated castor oil (PEG-40) were provided by Shanghai Meryer Biochemical Technology Co., Ltd (Shanghai, China). MXD was purchased from Beijing Innochem Science & Technology Co., Ltd (Beijing, China). PBS buffer (0.01 M, pH 7.2-7.4) was purchased from Shanghai Beyotime Biotech Co., Ltd (Shanghai, China). Polysorbate 80 (T-80) was purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). DPCs were extracted from mice whisker follicles. HaCaTs were from Shanghai Hongshun Biological Technology Co., Ltd. Collagenase I was from Shanghai Univ-Bio Co., Ltd. Trypsin, Dulbecco's Modified Eagle Medium (High Glucose) (DMEM), 75% ethanol, hexane, cell counting kit-8 (CCK-8), hematoxylin-eosin (HE), streptomycin and penicillin were bought from Shanghai Titan Scientific Co., Ltd. The KGF, VEGF and enzyme-linked immunosorbent assay (ELISA) kits were purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd (Shanghai, China). Wheatgerm oil was provided by Ecoarts Enterprise (Shanghai) Co., Ltd.
Complete culture medium for HaCaTs was made by combing 4 mL fetal bovine serum, 0.4 mL antibiotics (100 μg/mL streptomycin and 100 IU/ mL penicillin) and 35.6 mL DMEM. Complete culture medium for DPCs was made by combing 6 mL fetal bovine serum, 0.4 mL antibiotics and 33.6 mL DMEM. Collagenase I (0.25%) was prepared by dissolving 100 mg collagenase I in 40 mL PBS.
Gas Chromatography-mass Spectrometry Analysis of Essential Oils
Each essential oil was diluted 100 folds with ethanol:hexane (1:1, v/v) and analyzed by gas chromatography-mass spectrometry (Agilent 7890GC-5973MSD) equipped with an EI source and a DB-WAX column (30 μm × 250 μm, 0.25 μm film thickness) referring to our previous method. 14 Ultrapure helium was used as carrier gas and the column flow rate was 1 mL/min. One microliter of the diluted essential oil was injected into inlet with a split ratio of 10:1. The temperature of column was kept at 50 °C for 3 min initially, and then raised to 220 °C at 5 °C/min. A full scanning mode ranged 35-450 m/z was used to acquire the mass spectra. The ion source and the interface temperature of MS detector was set at 230 °C and 280 °C, respectively. The identity of each peak in the chromatogram was judged by searching its mass spectrum against the NIST 14 library. The peak area normalization method was applied to calculate the relative percentage content of each component (Table S1). The dominant components for AEO were γ-terpinene and (Z)-ligustilide which accounted for 29.78% and 24.74% of total compounds identified, respectively. While SEO was dominated by β-selinene (17.09%), (Z)-ligustilide (16.59%) and β-caryophyllene (14.01%).
Essential oil Safety Evaluation Via Acute Toxicity Tests
Considering the essential oils used in aromatherapy for treatment of hair loss are administrated to humans via oral ingestion and external application on head which is close to eyes, we performed oral, dermal and eye toxicity tests to evaluate the general safety of essential oils.
Acute Oral Toxicity Test
The Up-and-Down Procedure (TG 425) regulated by the Organization for Economic Co-operation and Development was performed in the current study to calculate LD50 (Lethal Dose, 50′) for acute oral toxicity evaluation. 15 This procedure allows minimum use of experimental animals and observation of their signs of poisoning. A computer based statistical program, AOT425pgm, was used to implement TG 425, with regard to the sequential dosing, the termination of tests, and the calculation of LD50. According to the program, doses would be selected from the sequence of 1.75, 5.5, 17.5, 55, 175, 550, 2000/5000, using a dose progression factor of 3.2. The first dosing can be initiated at 175 mg/kg if the substance's lethality is unestimated. In the current study, ∼10 ICR mice, half male and half female, were used. The first mouse was administrated intragastrically with each essential oil solution (2 mL essential oil in 4 mL wheatgerm oil) at an initial dose of 175 mg/kg. Mice were observed for signs of toxicity within 24 h and daily, thereafter, for up to 14 days. If the mouse survives, the second one is given a higher dose. If the first mouse dies or becomes moribund, the second animal is given a lower dose. The test was terminated when one of the default criteria set up by the program was met. After termination, individual animal data on survival status was attained and tabulated. LD50 was calculated by the formula: LD50 = XF + Kd, where XF is the last dose administrated, k is the interval between doses, and d is the tabular value. 16
Acute Dermal Toxicity Test
The single-dose toxicity test was employed to assess the acute dermal toxicity of essential oils according to Chinese standard: Procedures and Methods of Safety Evaluation for Cosmetics (GB7919-87). Ten SD rats, half female and half male, were used in the tests. Twenty-four hours prior to essential oil application, the dorsal area (6 × 6 cm) of all rats was shaved. Essential oil solution (2 mL essential oil in 8 mL wheatgerm oil) was smeared onto the test area at a dose of 2000 mg/kg; if no deaths occur, the test can be terminated. To prevent essential oil from animal licking, the test area was sealed with non-irritating tape for 24 h. Meanwhile, appearance and disappearance of poisoning signs as well as deaths were recorded. Twenty-four hours later, sterile water was used to clean the skin of remaining rats which were kept for up to 14 d. Signs of toxicity were recorded at different time nodes and the severity of irritation was assigned.
Acute Eye Irritation Test
The acute eye irritation test was also conducted by referring to Chinese standard: Procedures and Methods of Safety Evaluation for Cosmetics (GB7919-87). Two groups of female SD rats, 3 in each group, were examined 24 h prior to formal tests to ensure no eye irritation, corneal defect and conjunctival injury. To start the tests, 0.1 mL essential oil solution (2 mL essential oil in 4 mL wheatgerm oil) was dipped into the conjunctival sac of one eye of each rat in the first group and the eye was kept closed for 1 s to avoid drug loss. The other eye free of drug was used as control. Afterwards, no eye flushing was allowed within 24 h. Meanwhile, if eye irritation was observed, 3 rats in the second group were repeated the essential oil application and 30 s later, the tested eyes were flushed with mild water stream to test effect of flushing on eye irritation mitigation. If no eye irritation was observed, eyes of rats in the first group were examined 1 h, 24 h, 48 h, 78 h, 4 d and 7 d after essential oil application. If no irritation was found during the time slot, the tests terminated. For observation, a magnifier was used to visualize the cornea, iris, conjunctiva and any other impairment if possible. The scoring criterion of eye impairment was described in Table S2.
Effects of Essential Oils on a Mice Model of DHT-Induced AGA
Establishment of the Mice Model
After one-week adaptive feeding, ICR male mice were administrated i.p. with 1% pentobarbital sodium at dosage of 45 mg/kg. We used DHT to establish mouse model of AGA since DHT is a key hormone involved in AGA induction and has been applied frequently to induce AGA in animal modeling. 17 The scheme of mice modelling is displayed in Figure S1(a). On the first day, the dorsal skin (3 × 3 cm) of >54 mice was shaved using a hair clipper to 1 mm in length. The tested area was cleaned using wipes and skin in this area was pink. On the second day, 54 depilated mice without visible skin damage were selected and randomly divided into a CK group, a model group, a positive group (3% MXD group) and 6 treatment groups, 6 mice in each group. Treatment groups included high dosage group of AEO (5% AEO group), medium dosage group of AEO (3% AEO group), low dosage group of AEO (1% AEO group), high dosage group of SEO (5% SEO group), medium dosage group of SEO (3% SEO group), and low dosage group of SEO (1% SEO group). We chose 1%-5% dilution ratio because this is the typical range used in aromatherapy. 18 The essential oils were dissolved in 1.5% T-80/PEG-40 (1:1, v/v) and MXD was prepared in 75% ethanol/PBS (1:1, v/v).
DHT-induced mouse model was established based on the modified method by Kim et al 19 With the exception of the CK treated with 50% ethanol, all groups were treated with DHT (in 50% ethanol) dosed at 60 mg/kg/d for 21 consecutive days. Each time before DHT application, the tested area was swabbed with saline. After 21-d DHT application, the hair at the dorsal area of the CK mice regrew to normal length while other groups kept bald. Then mice in the CK group were depilated again. Essential oils and MXD were applied to the bald skin at a dosage of 200 μL/d, while sterile water was applied for CK and model groups, once per day for 28 consecutive days. Each time after drug administration, mice were moved back to the cage for normal keeping.
Histological Inspection of Skin Tissue
After the last administration at 28 d, all mice were euthanized via CO2 and sacrificed by cervical dislocation. Skin in the tested area was sampled immediately. A part of the samples was fixed in 4% paraformaldehyde solution for histological analysis. 9
After 48 h, skin was taken out of the fix solution for dehydration and embed in paraffin for sectioning into square blocks (1cm × 1 cm, 2 μm thickness). Before staining, the paraffin has to be removed since it is immiscible with the aqueous staining solution. Deparaffinating was performed by passing the sections sequentially through xylene, decreasing strengths of alcohol (100% to 0%) and water. After staining with HE, the sections were dehydrated again and placed in xylene for histological inspection which was performed on OLYMPUS BX51. Number and depth of hair follicles and dermal thickness were measured. Three tissue sections were analyzed per mouse to confirm reproducibility.
Effects of Essential Oils on the Monocultured Cell Model of DHT-Induced AGA
Isolation of DPCs from Whisker Hair Follicles and Their Propagation
Dermal papillary cells (DPCs) were isolated from ICR mice whisker hair follicles under microscope based on modified method of Gledhill et al 20 Briefly, 5-week mice were euthanized by cervical dislocation. Skin of the whisker region was immersed into 75% ethanol for 30 s for sterilization. Then whisker pad was incised off and washed with PBS (containing 1% double antibiotics) for thrice and then stored in PBS to prevent cell death. The whisker pad was transferred to a petri dish for digestion with 0.25% collagenase I for 2 h at a 37 °C incubator and then flushed with PBS (containing 1% double antibiotics) for thrice (Figure S2a-b). Fascial tissue under the corium layer of whisker pad was removed to expose the follicles bulbs (Figure S2c-d). The dermal papillae were isolated from these follicles. About 10 DPCs were pooled in its complete culture medium and 15 mL of the suspension was inoculated in a 75T culture vessel and incubated at 37 °C and a 5% CO2 level.
The DPCs were incubated until cells migrated from the hair papilla, grew adhering to the vessel wall, and finally extravasated from where they adhered onto. Meanwhile, 4 mL PBS was pipetted into the vessel and discarded, followed by addition of 3 mL 0.25% trypsin solution. The vessel was shaken gently and placed in a 37 °C incubator for 5 min for digestion. When DPCs changed from spindle to oval shapes, 4 mL complete culture medium was added to stop the digestion. The vessel was centrifuged at 1000 r/min for 5 min and the supernatant was discarded. The cell pellet was resuspended in the culture medium for propagation. DPCs within three generations were used for the following experiment. The framework of the experiment was displayed in Figure S1(b).
Effect of Two Essential Oils on Vitality of HaCaTs and DPCs by CCK-8 Assay
DPCs after propagation were suspended in the culture medium and 25 μL of the suspension was pipetted onto a cell counting plate. Cells were counted under a microscope and a 10-fold gradient dilution was conducted with sterile water until the density of cells in the final diluent reached 1.2 × 105/mL, out of which 10 μL was pipetted into each well of a 96-well plate. After inoculation, wells were sealed with sterile water and the plate was incubated at 37 °C and 5% CO2 for 24 h until 80% cells stuck to the vessel wall. Then supernatant was removed from each well, waiting for drug spiking.
Cell viability was evaluated by CCK-8 method. 21 The essential oils were diluted using PEG-40/DMEM (1:1, v/v) to make a concentration gradient, corresponding to different treatment groups. According to the preliminary experiment, essential oil concentrations were set at 5, 25, 50, 100, 200, 250, 300, 400 and 500 mg/L for HaCaTs, and 1, 10, 100, 200, 400, 600, 800 and 1000 mg/L for DPCs. MXD was prepared in 75% ethanol/PBS (1:1, v/v) to make a concentration series of 5, 25, 50, 100 and 200 mg/kg for HaCaTs and 1, 10, 100, 200, 400, 600, 800 and 1000 mg/L for DPCs. The positive control group was set by replacing essential oils with MXD. DMEM added to the wells was set as blank control. CK with cell suspension only was also set. Each group had 6 wells as replicates but only three with stable results were used for final statistics. The DPC-containing wells were each spiked with 100 μL MXD or essential oil solutions and placed into the incubator for 48 h. Subsequently, 10 μL of CCK-8 solution was appended to wells. Then the plate was incubated for 90 min and the optical density (OD) of samples was read at 450 nm by a microplate reader. The vitality of cells was calculated based on the formula: Vitality (%) = (ODtreatment-ODblank)/(ODCK-ODblank) × 100.
According to United States Pharmacopeia evaluation criteria for cytotoxicity and the vitality statistics, safe concentrations of essential oils on cells were decided. Table S3 shows that the positive relationship of cell vitality and the safety level of experimental materials, which can be deemed non-toxic when the safety levels are 0 and 1. Additionally, data between CK and the treatments was statistically compared. Consequently, safe threshold concentrations of the experimental materials were determined for further experiment.
Establishment of the Cell Model of DHT-Induced AGA
The cell model of DHT-induced AGA was set based on the method described elsewhere. 21 Stable HaCaT and DPC suspensions at the density of 1.2 × 105/mL were prepared for AGA modelling. The setting of experimental groups, transferring of cells to 96-well plate, incubating and OD recording were carried out in a way similar to what is described in previous cell culture section except substituting essential oils with DHT which had a concentration gradient of 10−4, 10−5, 5 × 10−5, 10−6, 5 × 10−6, 10−7, 10−8, 10−9 and 10−10 mol/L. An appropriate DHT concentration level corresponding to a successful modeling was selected for the following experiment.
Effects of Essential Oils on the Production of Cell Growth-Promoting Factors by ELISA. 22
After modeling, HaCaT and DPC cell lines at the density of 1.2 × 105/mL were used further to evaluate the effect of essential oils on the secretion of cell growth-promoting factors by the cultured cells. An aliquot of 10 μL of the prepared cell line was pipetted into wells followed by addition of DHT to reach a given concentration. Thirty-six hours later, liquid in the wells was decanted. In the treatment and positive control groups, AEO, SEO and MXD were individually added to the cell to bring their final concentration to preset levels as finalized in previous section. In the model group, an equal amount of DMEM was added. Wells with merely cell lines and no other substance addition were used as CK. Each concentration group had 6 wells as replicates. The plate was incubated for 48 h. Thereafter, liquid in each pair of wells of each group was collected into a 2 mL centrifuge tube and centrifuged within 1 h (3000 rpm for 10 min). After centrifugation, 800 μL of the supernatant in each tube was dispensed to four 0.5 μL–tubes with 200 μL each. Samples were subject to VEGF and KGF analysis using ELISA kits following the manufacture's instruction. Briefly, standard wells, sample wells and blank wells were set on the ELISA plate. Firstly, 50 μL standards at 0, 20, 40, 80, 160 and 320 pg/mL were added to the standard wells, 10 μL test sample together with 40 μL diluent to the sample wells and 50 μL diluent to the blank wells (n = 6). Then 100 μL of horseradish peroxidase (HRP)-conjugated antibodies was added to the wells. Wells were sealed with parafilm and incubated at 37 °C for 60 min. After that, the liquid in each well was discarded and wells were flushed with wash buffer for five times and blotted against paper towels. Thereafter, wells were spiked with 50 μL Substrate A and 50 μL Substrate B and then incubated at 37 °C in dark for 15 min. After incubation, 50 μL stop solution was added to wells to stop the reaction and the plate was read at 450 nm by a microplate reader to obtain OD values of each well. Concentrations of the growth factors were derived from the calibration curve.
Statistics
The experiment results were expressed as Mean ± SD and analyzed using the IBM SPSS 26.0 software. One-way ANOVA followed by a Dunnett test was used for statistical analysis. A threshold probability value of ‘P < .05’ was considered to be statistically significant.
Results
Evaluation of Essential Oil Safety
Acute Oral Toxicity Test
As shown in Table 1, mice administrated with AEO and SEO showed an overall similar response. At the dose of 5000 mg/kg, mice exhibited low spirit, drowsiness and waddle or stayed still and then they were all dead within 24 h. At the dose of 2000 mg/kg, mice showed drowsiness only within 2 h of drug administration and the symptoms disappeared after 2 h and no abnormity was observed till 14 d. In all other dosing groups, mice displayed transient excitement which vanished within 1 h and then turned to be normal till 14 d. Based on AOT425pgm, under a limit dose of 5000 mg/kg and an estimate of sigma of 0.5, 5 reversals occurred in any 6 consecutive mice tested for both essential oils, meeting one of the stopping criteria. Based on the statistic outcome of 14 d, LD50 of AEO and SEO was calculated to be 4721 and 4104 mg/kg, respectively, with a 95% confidential interval of 2000-5000 mg/kg. Therefore, AEO and SEO were judged to be actually nontoxic.
LD50 of AEO and SEO in TG 425-guided Tests.

AEO, Angelica sinensis essential oil; SEO, Ligusticum sinense ‘chuanxiong’ essential oil.
“X” indicates mice dead; “O” signifies mice alive.
Acute Dermal Toxicity Test
Mice administrated with 2000 mg/kg of essential oils appeared anxious and attempted frequently to lick the tested area in the skin. Symptom of anxiety vanished after 1 h while the frequency of licking declined obviously after 24 h and no abnormities were observed till 14 d. According to the regulations of China's Safety and Technical Standards of Cosmetics (2015), if no animal deaths occur at the dose of 2000 mg/kg in the acute dermal toxicity test, the test can be terminated and the LD50 of essential oils is determined to be larger than 2000 mg/kg, indicating the essential oils are mildly toxic.
In the current test, no skin irritation was found at the dose of 2000 mg/kg as shown in Table S4. Therefore, no acute dermal irritation/corrosion tests were needed and both essential oils were considered safe.
Acute Eye Irritation Test
After application of both essential oils, eyes of rats appeared red and swollen in 5 s and squinted with substantial vision decline. Comparatively, the irritation of SEO to rats was heavier than that of AEO, manifested by the severer eye congestion in the swollen eyes of the former than the later. After 60 s, eyes opened and signs of redness and swelling gradually dissipated. Six minutes later, only slight swelling was observed with no other adverse symptoms. Twenty-four hours later, rats restored to normal state. Based on the regulations of China's Safety and Technical Standards of Cosmetics (2015) on the acute eye irritation toxicity test, both essential oils exerted no irritation on cornea, iris and conjunctiva, as displayed by the scoring results in Table S5. Therefore, they were deemed free of acute eye irritation.
Effects of Essential Oils on the Mice Model of DHT-Induced AGA
Figure 1a visualizes the hair growth promotion in mice. Obviously, compared with control, mice in the model group showed thinner and shorter hair, indicating the validness of the model. Compared with model, essential oil and MXD treatments accelerated the hair growth and after 28 d, hair density and length in the treatment groups were similar to that of control. Essential oils at lower concentration levels tended to restore hair growth faster than MXD. Histological analysis (Figure 1b) clearly shows that, compared with control, topical application of essential oils increased the number of follicles and promoted the elongation of hair follicles from cuticular layer to dermis. Further quantitative statistics shows that 1% AEO and 3% SEO exhibited an average number of hair follicles of 69.73 and 70.13 (pcs/field of view), respectively, which were significantly higher than that of the model group (14.80 pcs/field of view) and even performed better than MXD (36.53 pcs/field of view) (P < .01) (Figure 1c). Similarly, low to moderate concentration of essential oils tended to restore thickness of DHT-affected dermis to normal (Figure 1d).
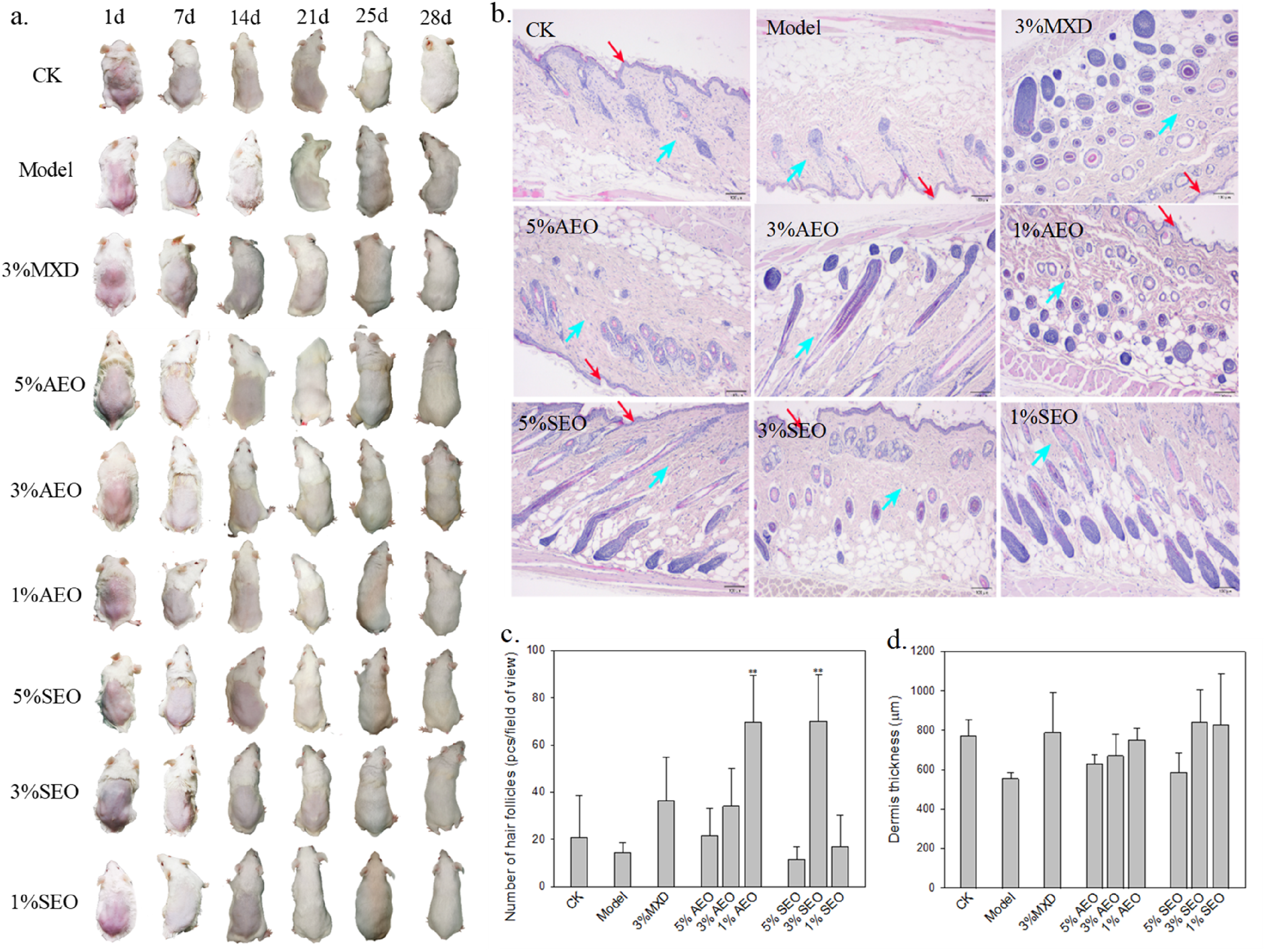
Effects of Essential Oils on Mice Model of Testosterone-induced AGA. (a) Gross Observation of Back Skin in Mice. (b) Histological Observation of Hair Follicles In Mice after 28 d of Drug Application (HE Stain, ×4; Purple Cricles, Follicles; Red Arrows, Cuticular Layer; Blue Arrows, Dermis). (c) Number of Hair Follicles of Mice and (d) Dermis Thickness of Mice Based on The Histological Analysis. Means Were Compared Based on Dunnett Test Relative to Model (n = 3); *P < .05, **P < .01.
Effects of Essential Oils on the Cell Model of DHT-Induced AGA
Cytotoxicity of Essential Oils on HaCaT and DPC Vitality
To figure out a safe experimental threshold concentration for treatment of AGA, the cytotoxicity of essential oils on HaCaTs and DPCs was evaluated. Figure 2 shows the viability of monoculture of cells after exposure to toxins for 48 h. It is shown that, with the increase of concentration, both essential oils exhibited an overall significantly positive effect and a following adverse effect on HaCaT and DPC viability (P < .05), directing a safe threshold concentration of 200 mg/L for both oils, comprehensively considering the criterion of Table S3 and the statistical analysis. Likewise, the thresholds of MDX were determined to be 25 mg/L and 10 mg/L, respectively, in the case of HaCaTs and DPCs.

Effect of Essential Oils on Vitality of (a) HaCaTs and (b) DPCs after 48 h. Comparisons were Made Relative to CK Using Dunnett Test (n = 3); **P < .01, *P < .05.
Effect of DHT on HaCaT and DPC Modeling
To ensure successful modeling, effects of a DHT concentration series on cell viability were evaluated (Figure 3). Overall, HaCaT and DPC viability exhibited an overall decreasing trend with increasing DHT concentration. At the respective threshold concentration of 1 × 10−9 mol/L and 1 × 10−5 mol/L, the DPC and HaCaT viability was significantly lower than that of CK (P < .05). Considering the visual viability drop was not large at lower concentrations for both cells, finally, the concentration for modelling was set at 1 × 10−4 mol/L, corresponding to a cell viability of 58.9% and 50.8% for HaCaT and DPCs, respectively.

DHT-induced HacaT and DPC Viability After 48 h. Comparisons were Made Relative to CK Using Dunnett test (n = 3); **P < .01, *P < .05.
Effect of Essential Oils on Growth-Promoting Factors of AGA Cell Model
Table 2 shows the effect of AEO and SEO on concentrations of VEGF and KGF, two important cell growth-promoting factors secreted by HaCaTs and DPCs. Levels of KGF ranged 849.72-974.76 pg/mL, higher than that of VEGF in CK (229.27-272.04 pg/mL). Compared with CK, secretion of VEGF in HaCaT and KGF in DPCs in the model group was significantly inhibited (P < .01), and secretion of VEGF in DPCs and KGF in HaCaTs was also inhibited though not significantly. Overall, secretion of hair growth promotors was significantly different between the essential oil treatment groups and the model group (P < .05), indicating that application of 50-200 mg/L essential oils reversed the inhibition to different degrees and the concentration dependence of reversing effects was not consistent. Moderate to high concentration of AEO tended to enhance the secretion of hair growth promotors more than low concentration of AEO, while the promoting effect of SEO at all investigated concentrations on the production of VEGF and KGF by HaCaTs was more pronounced than that by DPCs. Notably, the moderate concentration of AEO showed VEGF and KGF levels of 232.52, 304.42, 994.05 and 731.02 pg/mL in two cells, respectively, while low concentration of AEO showed corresponding values of 223.89, 268.78, 1112.06 and 836.48 pg/mL, which were mostly significantly higher than the model and prone to be closer to normal (in CK).
Effect of Two Essential Oils on Secretion of Hair Growth Promotors by HaCaTs and DPCs (pg/mL).
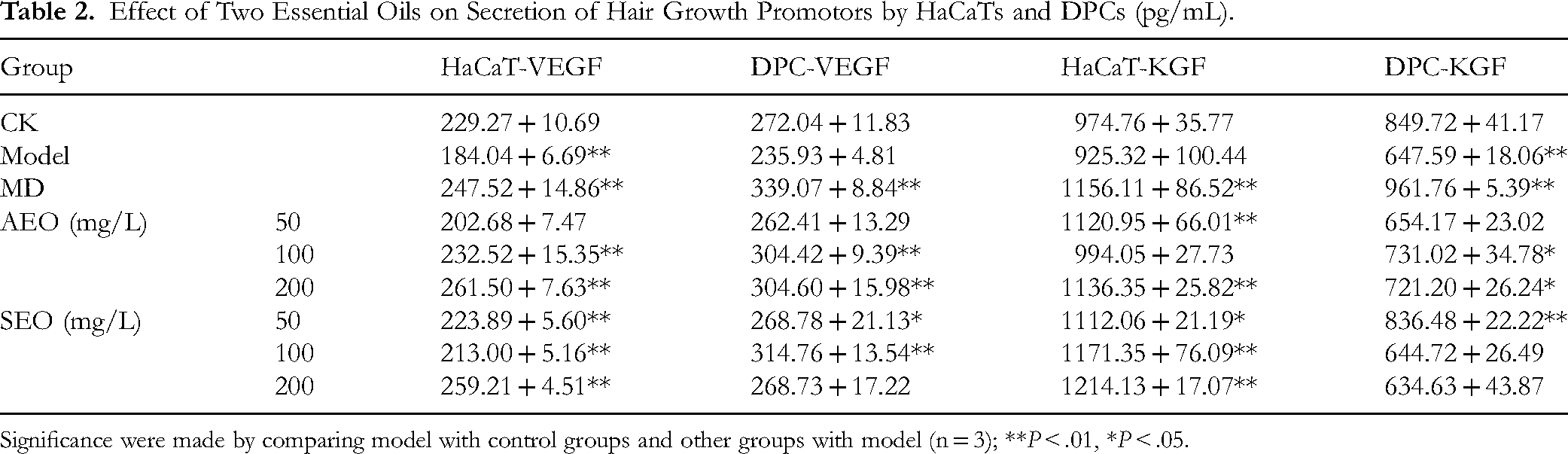
Significance were made by comparing model with control groups and other groups with model (n = 3); **P < .01, *P < .05.
Discussion
In the present study, we demonstrated, using standard methods, that AEO and SEO were actually non-toxic, which guaranteed its possible safe application in future. In a safety evaluation study by Zhang et al, the oral LD50 and intra-peritoneal LD50 of essential oils from rhizomes of L. sinense ‘chuanxiong’ in mice was calculated to be 7.23 g/kg and 2.52 g/kg, with a 95% confidential interval of 6.66-7.79 g/kg and 2.07-2.97 g/kg, respectively. 23 Additionally, the essential oil exhibited slight skin irritation in rabbits. Similarly, we reported an oral LD50 value of 4721 mg/kg for AEO and 4104 mg/kg for SEO, with a 95% confidential interval of 2000-5000 mg/kg, and they caused indiscernible irritation on eyes and skin. The safety results agree well with the chemical composition of the essential oils which were mostly composed of safe components, ie, terpenes. 24 Two essential oils shared one common major component, ligustilide, which was demonstrated by the acute oral toxicity test to be non-toxic to mice at the dosage of 5000 mg/kg, 25 supporting our toxicity results. One thing worth noting is that caution should be made when extrapolating the animal toxicity data to humans due to interspecies differences in xenobiotic metabolism. Humans are more susceptible to toxicants since they metabolize the xenobiotic less extensively than animals and thus prolong the exposure to toxicants. 26 Furthermore, although long-term safety tests were not considered in the current study, it is expected that long-term topical application of essential oils below the thresholds as displayed by the acute toxicity tests would be safe because essential oils have a life-time within one hour in living bodies and eliminated from human body quickly. 27
MXD, one mainstay drug for AGA treatment, mainly promotes hair growth via stimulating follicular proliferation and differentiation, the microcirculation of the bald scalp or VEGF synthesis of hair-related cells.28,29 Specifically, MXD opens the K+-channel which is essential for progression of the early stage of cell proliferation, increases the cellular DNA synthesis in anagen bulbs, and induces β-catenin activity in the anagen phase in dermal papillae. 30 Using mice model of testosterone-induced AGA, we demonstrated the hair growth-promoting effects of AEO and SEO, especially at low to moderate concentration, were even better than that of 3% MXD. Notably, visual observation of back skin of mice indicated that topical application of essential oils stimulated the hair regrowth faster than MXD did (Figure 1a). One possible explanation is that the plethora of components in the essential oil exhibited synergy. Additionally, the terpenes act as skin penetration enhancers associated with their lipophilic nature which render them to break the H-bond between ceramides in the lipid bilayer of stratum corneum.31,32 Consequently, essential oil penetrates the skin and enters into the blood circulation beneath the dermis very fast. Furthermore, essential oils may exhibit hair-growth promoting effects via multifaceted mechanisms of action including the anti-oxidative and anti-inflammatory abilities which MXD is not versed at. 33 Our results are similar to what was reported by Oh et al that 3% peppermint essential oils significantly promoted dermal thickness, follicle number and follicle depth in the bald dorsal area of mice and the effects were not interior to 3% MXD. 9 Essential oils from rosemary, peppermint and so on, were all reported to exhibit hair-growth enhancing effects on experimental mice or humans.34,35
Apart from the animal model for AGA which visualized the hair growth process and facilitated histological examination, in vitro monoculture cell models using mice DPCs and HaCaTs were employed to evaluate the hair growth-promoting effect or mechanism at the cellular level. DPCs comprise the dermal papilla which are essential for hair growth while keratinocytes play a role in the proliferation of hair and are also one of the main targets of toxins. During the hair developmental phase, DPCs and keratinocytes produce various hair growth promoters, among which VEGF and KGF play great roles in the development, differentiation and regeneration of hair follicles. VEGF substantially facilitates follicle vascularization and angiogenesis, thus promoting hair growth. 36 KGF is an essential endogenous mediator of hair follicle growth and differentiation which is expressed in large quantity in skin to exhibit alopecia cytoprotection. 37 In the current study, DHT induced VEGF and KGF secretion decline in cultured DPCs and HaCaTs, which was reversed by essential oils, indicating essential oils may play a positive role in treating AGA via stimulating VEGF and KGF production in related cells. 38 The essential oils may enhance the expression and secretion of VEGF and KGF via the Wnt/β-catenin, ERK, and JNK signaling pathways in hair follicle cells. 22 Similarly, the essential oil of Chamaecyparis obtusa was also reported to promote hair growth through upregulating VEGF expression in HaCaTs and mouse skin tissues.21,39 The authors indicated that VEGF may extend the anagen phase in AGA and these essential oils may thus be used as a prophylactic medicine for human AGA. Besides, the multifaceted molecular mechanism of essential oils in stimulating hair growth was summarized elsewhere, 33 including inhibiting DHT synthesis related enzymes, invigorating blood circulation to hair follicle, increasing the expression of reactive oxygen scavengers and constraining the expression of pro-inflammatory cytokines in follicle cells, etc. In future, the molecular mechanisms of A. sinensis and L. sinense ‘Chuanxiong’ essential oils will be verified experimentally and clinically.
So far, there are no studies on the hair growth-promoting effects of essential oils of A. sinensis and L. sinense ‘Chuanxiong’ for comparison. Nevertheless, the ethanol extract of Angelica sinensis has been reported to induce hair regrowth in mice via inhibiting apoptosis of hair follicle cells. 13 In another study, the Liao Tuo Fang Chinese yao, composed of five Chinese herbs including A. sinensis and L. sinense ‘Chuanxiong’, was proved to significantly restore hair growth in dorsal skin of model mice of DHT-induced AGA via upregulating VEGF expression. 40 The extracts of the two plants featured a major bioactive ingredient, ligustilide, while whether their therapeutic potential for AGA was linked to ligustilide is uncertain. Nevertheless, it has been reported that ligustilide exhibited protective effects on cultured vascular endothelial cells originated from human umbilical vein, dilated blood vessels and promoted blood circulation in human head.41,42 Comparing with other hair-growth promoting studies using common essential oils such as lavender, rosemary and peppermint which feature the chemical constituents of monoterpenes or sesquiterpenes, the uniqueness of the current study lies at the bioactive ingredient, ligustilide, a dihydrophthalide that may have multifaceted effects on hair loss. However, since we did not isolate the monomer from the oil blend, its potency cannot be clarified. Future study will try to figure out the hair growth promoting effect of the major monomer combing animal experiment and clinical trials.
Conclusion
Summing up, essential oils from aerial part of two Chinese traditional herbs, A. sinensis and L. sinense ‘Chuanxiong’, were demonstrated to exhibit hair-promoting effect without acute toxicity. Two essential oils distinguish themselves from traditional terpene-rich essential oils (eg, lavender, rosemary, etc) in its main active component, (Z)-Ligustilide, which is a dihydrophthalide that may have multifaceted effects on hair loss. The volatile essential oils may make the therapeutic effect more pronounced because they penetrate the skin easier than the aqueous extracts. The results suggest that AEO and SEO can be used solely or in combination with other agents in anti-AGA therapies as VEGF and KGF secretion promotors. For example, these essential oils formulated with lavender, rosemary and peppermint essential oils can be applied to scalp in terms of shampoo or cream via massage. The study will add oriental element to the essential oil recipe pool for hair loss treatment. In future, the hair-growth promoting effects will be evaluated clinically before the commercial products come out.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251333902 - Supplemental material for Angelica sinensis and Ligusticum sinense ‘Chuanxiong’ Leaf Essential Oils Promote Hair Growth without Acute Toxicity
Supplemental material, sj-docx-1-npx-10.1177_1934578X251333902 for Angelica sinensis and Ligusticum sinense ‘Chuanxiong’ Leaf Essential Oils Promote Hair Growth without Acute Toxicity by Li Ma, Miyuan Pi, Zhuqing Dai, Yuexing Zhu, Wencui Wang, Haicheng Yang and Lei Yao in Natural Product Communications
Footnotes
Acknowledgements
The authors acknowledge the technical assistance of the Histopathology Department and Biochemistry Laboratory staff at Suzhou Cancercell Biotechnology Co. Ltd, Jiangsu, China.
Ethical Considerations
The experiments were approved by the Institutional Animal Ethics Committee of Shanghai Jiao Tong University with approval number A2019001. The animals were manipulated in accordance to ARRIVE Guidelines for animal ethics.
Informed Consent
Not applicable.
Author Contributions/CRediT
Li Ma: Writing-original draft, Visualization, Writing-review & editing, Conceptualization, Supervision; Miyuan Pi: Resources, Data curation, Visualization; Zhuqing Dai: Resources, Visualization, Validation; Yuexing Zhu: Resources, Validation, Writing-review & editing; Wencui Wang: Methodology, Visualization, Writing-review & editing; Haicheng Yang: Methodology, Visualization; Lei Yao: Conceptualization, Supervision, Visualization, Funding acquisition.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Yingu Aromatic Technology Co., Ltd (grant number [22H020100432]). Author Lei Yao has received research support from Yingu Aromatic Technology Co., Ltd.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
