Abstract
This is a visual representation of the abstract.
Introduction
Secondary metabolites produced in plants may play diverse functions in their ecological interactions and are widely used in various fields, such as pharmaceutical, cosmetic and agricultural. 1 One of the most important promising aspects is their use as potential drug and their combination with drugs.2-4 In this sense, plants showed a key role in both conventional and folk medicines and still remain crucial for health promotion or disease prevention and even in chronic disease management.5,6 In the past two decades, there has been a huge shift from synthetic to herbal derived medicines, which is mainly due to the adverse effects of the synthetic compounds on health and the environment, as well as the growing problem of the appearance of multidrug-resistant strains. 7 There is a need to discover new natural compounds that could replace or potentiate synthetic ones; this is particularly challenging in the case of those with antimicrobial activity and antioxidant properties. Therefore, many plants have been researched and different extraction and analytical methods and approaches have been developed in order to obtain potent compounds. 8
As volatile and lipophilic complex mixtures of various secondary biomolecules, mainly of terpene and phenylpropanoid nature, essential oils (EOs) are among the most investigated and used plant products. Species belonging to the Apiaceae (Umbelliferae) family (umbellifers or carrot/parsley family) are well-known to contain essential oil (EO) in different plant organs, especially fruits and underground parts.9,10 Besides being edible plants or used as common spices and condiments, there are a number of studies reporting different bioactivities of umbellifers and their EOs. 11 With around 120 species, Peucedanum L. is a heterogeneous and polyphyletic genus of umbellifers distributed in Europe, Asia, Africa and North America. 12 However, based on recent phylogenetic studies, many species (mainly American and African) have been excluded to establish new or transfer into other genera.13,14 Consequently, the genus in its broader sense (Peucedanum sensu lato) has been reduced to 74 predominantly Eurasian species, 15 some of which are used in folk medicine to treat various pathologies, such as gastrointestinal disorders, cough or asthma, cardiovascular diseases, epilepsy and different kinds of pain. However, pharmacological studies are not extensive.16,17 Coumarins, flavonoids and terpenoids are the most reported phytochemicals in these plants and are considered to form the basis of their traditional uses. Some species are also used in Balkan countries for the treatment of cardiovascular, respiratory, urogenital and nervous system. 18 Such is, for example, P. longifolium Waldst. & Kit. (PL) which is sometimes considered as a subspecies of P. officinale L. This is a glabrous perennial (Figure 1) up to 150 cm, with a stock about 5 cm in diameter densely covered with fibres. Angled stems are stout and branched above with 2- to 6-ternate leaves 30-80 cm long and linear leaf-lobes, narrowed at both ends, often keeled and rarely more than 1 mm wide. 15-30-rayed compound umbels are nod in bud and with small yellow flowers; ripe fruits are 5-7 mm long, narrowly elliptic to oblong-obovate, as long as pedicels.19,20 This plant inhabits dry grasslands and rocky places, usually limestone, at medium altitudes throughout the entire Balkan Peninsula, Georgia, the Asian portion of Turkey and the North Caucasus in Russia.12,21 Different studies were performed on both areal and underground parts of PL,9,18,22-25 but the EOs were mostly extracted by a standard 3-4-h distillation procedure. However, previous studies on some Apiaceae species26-28 highlighted the importance of extending the extraction duration, therefore, a fractionated 24-h hydrodistillation procedure 29 was applied to the PL flower and ripe fruit materials collected on two Dinaric mountains in Montenegro (Lovćen and Rumija). The chemical composition of the obtained EOs and the associated antioxidant efficacy are reported herein.

Peucedanum Longifolium Waldst. & Kit. at the Vrsuta Peak (Rumija) Above the Town of Bar (Left), Flowering (Middle) and Fruiting Umbels (Right) (Photo: Mijat Božović, 2022).
Materials and Methods
Plant Harvesting
Flowers and ripe fruits of PL were collected from two localities in Montenegro: Kuk (42°23′49.4″N 18°47′48.1″E) and Vrsuta (42°09′13.1″N 19°05′07.3″E). Kuk is a plateau (1310 m) on Lovćen, a mountain in southwestern Montenegro that rises from the borders of the Adriatic basin, closing the Bay of Kotor and forming the hinterland to the coastal town of Kotor. Vrsuta is the highest peak (1183 m) of the Sutorman mountain belonging to the Rumija massif. Rumija, in its wider sense, is the southernmost mountain of Montenegro that rises above the town of Bar, separating the Adriatic from the Skadar basin. The both mountains are parts of an uninterrupted coastal Dinaric Mountain chain that runs along the Montenegrin coast. PL material was collected in July and August of 2022 for flowers and fruits, respectively. Air-drying of the collected material was performed in a shady place for approximately 25 days. Voucher specimens have been deposited in the Department of Biology at the University of Montenegro (numbers BDPMF-PLFL/K, BDPMF-PLFL/V, BDPMF-PLFR/K and BDPMF-PLFR/V). Taxonomic identification of the species was conducted according to the official European flora. 19
EOs Extraction
EOs were isolated by the process of hydrodistillation using a Clevenger-type apparatus with the extraction method previously reported. 29 Dried PL material in toto (50 g) was subjected to fractionated distillation collecting EOs at interval times of 1, 2, 3, 6, 12 and 24 h. At each interval, the accumulated oil/water double phase was extracted 3 times with 20 mL of diethyl ether, then the organic layers were dried over anhydrous sodium sulphate (Na2SO4), filtered, and then deprived of the solvent in vacuo to furnish EOs. Thus, 6 EO fractions from each PL sample (two organs per locality) were obtained, and the prepared EOs were stored in tightly closed dark vials until further analysis.
EOs Chemical Composition Analysis
Gas Chromatography/Mass Spectrometry (GC/MS) analysis was performed on a Perkin-Elmer-Clarus 500 model of a gas chromatograph (GC) directly coupled to a mass spectrometer (MS). The analyses were carried out twice, showing reproducible results. The GC was equipped with two columns, one of which was a Restek Stabil wax (fused-silica) polar capillary column, and the other was a Varian (VF-1 ms) apolar column. Helium was used as the carrier gas (1.0 mL/min). The column temperature was programmed as follows: from 60 °C to 220 °C at a rate of 5 °C/min, and held for 10 min. The MS parameters were ionization voltage taken at 70 eV, the mass range was from 40 to 500 m/z, the ion source temperature of 200 °C, and a scan time of 0.2 s. The identification of the components separated by GC/MS was performed first by comparing the mass spectra for each compound with that reported in the MS libraries database (Wiley and Nist 02) and then by comparison of Linear Retention Indices of each compound calculated using a mixture of n-alkanes (C8-C30 aliphatic hydrocarbons, Ultrasci, injected into both polar and apolar columns under the same operating conditions), with available retention indices in the literature. GC-FID (flame-ionization detector) analysis was performed under the same experimental conditions using the polar column as described for the GC/MS measurements. Relative percentages for quantification of the components were calculated by electronic integration of the GC-FID peak areas using the normalization method without using corrections factors (RRFs).
Determination of Antioxidant Activity
A number of different assays have been used to assess the antioxidant activity of plant extracts. In this study, 2,2-diphenyl-1-picrylhydrazyl radical scavenging activity (DPPH) and ferric-reducing antioxidant power (FRAP) assays were carried out on the obtained EOs. Prior to analyses, stock solutions (1 mg/mL) were prepared by dissolving the EOs in methanol.
DPPH Assay
Free radical scavenging activity of the extracted EOs was measured by DPPH test.
30
In brief, 0.1 mM solution of DPPH in methanol was prepared. 3 mL of the EO of the appropriate dilution and 1 mL of DPPH solution were mixed in test tube. The mixture was shaken vigorously and left in the dark place at room temperature for 30 min. After that, the absorbance was measured at λmax = 517 nm. Ascorbic acid was used as a reference standard compound. The experiment was performed in triplicate. The percent DPPH scavenging effect (RSA) or inhibition percentage of antioxidant activity (%AA) was calculated by the equation:
Accordingly, the antioxidant strength is considered weak when the AAI is less than 0.5, moderate when it is between 0.5 and 1, high when ranges from 1 to 2, and very strong when the AAI is more than 2.31,32
FRAP Assay
The FRAP test was used to determine the reducing capacity of the EOs. 33 First, the FRAP reagent was prepared by mixing 300 mM sodium acetate buffer solution at pH 3.6, 10 mM 24,6-tripyridyl-s-triazine (TPZT) diluted in 40 mM hydrochloride acid and 20 mM ferric chloride in a ratio of 10:1:1, respectively. Then, the reaction mixture was prepared with 3 mL of FRAP reagent, 1 mL of distilled water and 100 µL of each EO stock solution. The tubes were incubated for 15 min at 37 °C. After that, the tubes were allowed to stand at room temperature for additional 5 min, and then the absorbance was read at λmax = 595 nm. From the calibration curve of ferrous sulphate heptahydrate, used as a standard (0-1000 µmol/L), antioxidant capacity values were read and expressed as mmol Fe2+ equivalents per g of EO dry weight. The higher the Fe2+ concentration, the higher the ferric-reducing antioxidant power.
Results
EOs Extraction
An extended extraction process to a total of 24 h was applied in accordance with previous studies26-29 suggesting that the Apiaceae species yield a large amount of EO after a standard 3-h distillation process. A total of 24 EOs were obtained including 3 one-hour, 1 three-hour, 1 six-hour and 1 twelve-hour fractions per each extraction (Table 1) for either locality and including both reproductive organs. Relative yield percentages calculated per weight of dried plant material for each EO sample and cumulative yields over the entire extraction time are shown in Table 2.
A Total of 24 Essential Oils (EOs) Obtained with Different Extraction Duration.

L: Lovćen, R: Rumija, FL: flower, FR: fruit, 1-24: the hour in which EO was collected.
Yield % of Flower and Fruit EOs of PL from Montenegro.

Yield % calculated on the dried PL plant material.
Cumulative yield % of EOs over time.
Shared % of the total yield.
EOs Chemical Analysis
The GC/MS analysis of 24 EOs revealed the presence of 66 different chemical constituents (Tables 3-6). Generally, myristicin (MYR) showed as the most prominent EOs’ component. This phenylpropanoid was particularly abundant in the material originating from Lovćen, reaching even 72.9%, both in flower (LFL24h) and fruit EOs (LFR3h). Lesser amount was reported in the sample from Rumija: up to 64.1% and up to 50.1% in the flower and fruit EOs, respectively. MYR was accompanied by significant amounts of its structural analogues, elemicin (ELE) and isoelemicin (ILE). EOs from flowers originating from Rumija were generally the richest in ELE (7.9-13.4%), although its large amount was exceptionally found in some EO fractions from Lovćen (eg, 18.8% in the fraction LFR2h). Contrary of that, ILE was abundantly present in the Lovćen-originating plant material, particularly in the flower EOs (up to 47.0%).
Chemical Composition (Percentage Mean Values ± SD) of the Flower EOs from Rumija.

Linear Retention indices from literature.
Linear Retention indices measured on apolar column; tr – traces < 0.1%.
Chemical Composition (Percentage Mean Values ± SD) of the Fruit EOs from Rumija.

Linear Retention indices from literature.
Linear Retention indices measured on apolar column; tr – traces < 0.1%.
Chemical Composition (Percentage Mean Values ± SD) of the Flower EOs from Lovćen.

Linear Retention indices from literature.
Linear Retention indices measured on apolar column; tr – traces < 0.1%.
Chemical Composition (Percentage Mean Values ± SD) of the Fruit EOs from Lovćen.
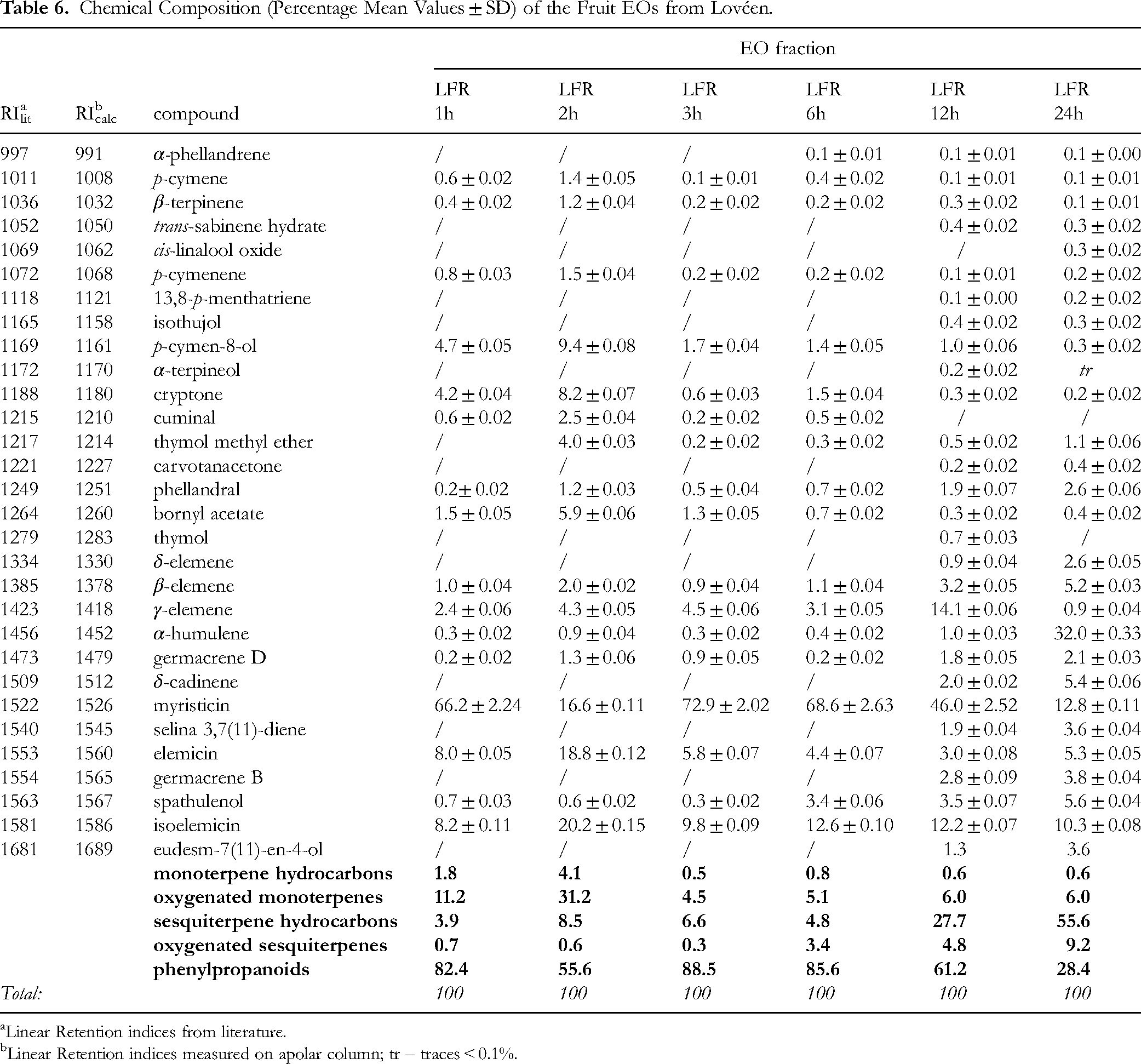
Linear Retention indices from literature.
Linear Retention indices measured on apolar column; tr – traces < 0.1%.
These alkenylbenzenes together constituted up to 93.6% of EO (LFL6h) and were mainly distilled during the first 6 h of the extraction process, although in the case of the sample from Lovćen, considerably high amount was extracted even after that period. The last two EO fractions (after 6 h) were rich in sesquiterpene hydrocarbons, especially in the case of the Rumija sample, where this group of constituents accounted for more than 50% of EO (RFL12h, RFL24h, RFR12h and RFR24h). However, different sesquiterpenes were present depending on the sample locality: the EOs from Lovćen contained considerable amounts of β-elemene (up to 7.1% and 5.2% in the flower and fruit EOs, respectively) and γ-elemene (up to 4.6% and 14.1% in the flower and fruit EOs, respectively), with the exception of the last fruit EO fraction (LFR24h) containing even 32% of α-humulene. In the case of the sample originating from Rumija, in addition to γ-elemene (up to 25.7%) and α-humulene (up to 25.9%), significant amounts of germacrene D (up to 14.3%) and trans-α-bergamotene (up to 8.9%) were found in the flower EOs; the fruit EOs were characterized not only by γ-elemene (up to 9.5%) and trans-α-bergamotene (up to 7.9%), but also by a significant addition of β-elemene (up to 36.2%) and β-copaene (up to 23.9%). Oxygenated sesquiterpenes were poorly present; spathulenol was the most frequently reported (up to 3.6 and 5.6% in the flower and fruit EOs, respectively). All EO fractions were almost deprived of monoterpene hydrocarbons. However, the oxygenated forms were considerably present (up to 13.7 and 31.2% in the flower and fruit EOs, respectively); these compounds were mainly extracted in the first hours of the extraction process, which was particularly evident in the case of fruit plant material. p-cymen-8-ol was found in all EOs with the percentage ranging 0.4-4.9 and 0.3-9.4 in the flower and fruit EOs, respectively. In addition, some of the fruit EOs were characterized by a significant content of certain monoterpenoids. For instance, the RFR1h fraction contained 5.1% of bornyl acetate and 4.7% of cis-sabinol, while 5.9% of bornyl acetate and 8.2% of cryptone were found in the LFR2h fraction.
Antioxidant Activity Analysis
Antioxidant potential can be measured by various assays with specific mechanisms of action; thus, it is recommended that more than one type of antioxidant measurement be perform to account for the different modes of action. In that context, antioxidant activity of the EOs of PL was assessed by two in vitro tests: the DPPH assay, which measures proton-radical scavenging activity, and the FRAP method that estimates the ability of an antioxidant to reduce the ferric (Fe3+) to the ferrous (Fe2+) ions. The antioxidant effect determined by the DPPH assay was expressed as IC50 value (mg/mL), and for comparison purposes, RSA (%) value related to the starting, stock solution (1 mg/mL) was also added (Table 7). In most cases, the IC50 was lower than 1 mg/mL with no significant differences between the localities and organs. The highest activity was observed in the LFR24h fraction (RSA 72.96%, IC50 0.61 mg/mL) whereas the LFR1h fraction showed the weakest activity (RSA 29.21%, IC50 2.54g/mL) (Figures S1 and S2, Supplemental Material). FRAP values were expressed in mmol of Fe2+ per g of EO; the reducing power was ranging from 3.48 to 11.56 mmol Fe2+/g.
Antioxidant Capacities of the EOs of PL from Montenegro; Values Represent Mean ± SD of Three Measurements (n = 3).

IC50 value of the standard ascorbic acid was 0.053 ± 0.01 mg/mL.
Radical scavenging activity of the EOs stock solutions.
Limitations of the Study
The extraction process is time-consuming which can be considered as a main limitation. Thermal degradation and/or structural modifications of EO compounds (with prolongation of the distillation process) might also be pointed out. Assessment of antioxidant activity is always preferable to be carried out with several tests.
Discussion
EOs Yields and Chemical Composition
Total yields for both flower and fruit EOs were quite different among the localities: 3.417% and 1.675% for the flower and 4.819% and 1.701% for the fruit EO samples from Lovćen and Rumija, respectively. A great difference in EO yield is observed since the extraction from Lovćen generally furnished a higher amount of EO, even up to 3 times – as in the case of fruit EOs, than from Rumija. Comparing the plant organs, the fruits yielded more EO than the flowers, although this difference was not so noticeable for the samples from Rumija. Inspecting the separate fractions (Figure 2), the highest EO amount from the flowers was obtained in the first hour in PL sample from Rumija (37.43% of the total EO yield), whereas in the case of Lovćen, the highest amount (24.93%) was obtained between the third and sixth hours of the fractionation process. This distinction clearly makes the overall yield dynamics quite different for each locality, highlighting a significant yield even after the first 3 h in the sample from Lovćen. Differently, the fruiting material from Lovćen gave the highest EO amount in the first fraction (25.56% of the total EO yield), while in the case of Rumija, the fourth fraction was the most abundant (30.22%). However, the overall dynamic by which the fruit PL material gives EO is quite similar for both Lovćen and Rumija, with the peak in the fourth fraction representing a notable addition to the total EO content. Taking into consideration the last fractions’ yield, it is evident that extending the extraction time beyond the first 2 or 3 h (as usual duration) significantly increased the yield, particularly in the fruiting material (up to 69% as in the case of the sample from Rumija). Nevertheless, the amount of EOs extracted in 3 h was higher (with the exception of the fruits from Rumija which yielded 0.52%) than those found in the literature reporting 0.38% in the flowering 18 and 0.91% in the fruiting material. 23

Graphical Representation of Relative Yield % of Each Fraction.
Previous investigations of the EOs from PL are very limited; two studies were performed on material collected in Serbia and represent the only data on EO from PL reproductive organs. Flower EO was reported to be very rich in monoterpenes (92.5%) with myrcene (23.1%), α-phellandrene (22.5%) and β-phellandrene (16.4%). 18 Similarly, EO from fruit contained 79.7% of monoterpenes mainly represented by α-phellandrene (26.2%), β-phellandrene + limonene (21.0%) and myrcene (9.5%). 23 Besides, other studies reported chemical composition of EOs from PL aerial parts and the results clearly showed the terpenes as the main constituents. α-pinene (36.3%) was reported as the most characterizing monoterpene in the material collected from Montenegro. 9 Monoterpene-rich EO was also extracted from the Serbian PL material and myrcene (15.9%) and α-phellandrene (11.3%) were reported as the main ingredients. 22 Another study on the Serbia-originating material, however, revealed sesquiterpene β-elemene (24.7%) along with monoterpene (E)-β-ocimene (11.7%). 24 PL from Turkey gave EO consisting mostly of sesquiterpenes (68.18%); 8-cedren-13-ol (33.74%), germacrene-D (7.73%) and Δ-3-carene (6.38%) were reported as the most dominant ones. 34 EO rich in monoterpenes (96.6%) was also obtained from the PL rootstock collected in Serbia; α-pinene (60.3%) and sabinene (20.9%) were the predominantly present. 25
The results presented herein differ significantly from those found in the literature, in particular due to the high amount of phenylpropanoids MYR, ELE and ILE. As reported by several authors,35,36 both MYR and ELE (with its isomeric form) are biosynthesized from the common precursor methyl eugenol, which is directly derived from eugenol, a product originated from phenylalanine via the shikimate pathway. 37 A genetically controlled biosynthetic route leads to the formation of either ELE or MYR, and the latter can be further converted to dillapiole. 38 The parent compound methyl eugenol was also identified in this study (at least in traces) in flower EOs (Tables 3 and 5). Although phenylpropanoids are not uncommon and occur in a range of umbellifers, these constituents are preferably found in EOs from roots and/or rhizomes. 10 Moreover, some Apiaceae species are well-known source of phenylpropanoids. For instance, Foeniculum vulgare L. and Pimpinella anisum L. are rich in anethole and estragole,26,28,39 while Crithmum maritimum L. 40 and Ridolfia segetum L. 27 often contain dillapiole. Although it was first discovered in nutmeg seed (Myristica fragrans Houtt., Myristicaceae), 41 high MYR content was found in some Apiaceae species as well: eg, Portenschlagiella ramosissima (Port.) Tutin,42,43 Anthriscus sylvestris (L.) Hoffm. 44 and Silaum silaus (L.) Schinz & Thell.45,46 Structurally, MYR is similar to ELE, differing only in the methyl group that joins the two oxygen atoms; these two compounds are often present together in a biological source. However, some Apiaceae species belonging to the genera Thapsia, 47 Daucus48,49 and Heracleum 50 are reported to contain significant amounts of ELE. Besides, its isomer has been found in several umbellifers from the genera Diplotaenia, 51 Pycnocycla 52 and Heracleum. 53
Peucedanum species are not generally recognized as a source of these aromatic compounds and according to the available literature, they are commonly characterized by the predominance of monoterpenes10,54-56 or sesquiterpenes.57-59 Moreover, this is confirmed by a recent multivariate statistical analysis of the literature data for the EOs of numerous Peucedanum species and related taxa (Peucedanum sensu lato). 23 Phenylpropanoid MYR was found only in P. zenkeri Engl. EO 60 and there is also one report on PL containing this compound 34 ; however, its amount in all those reports was much lower (less than 10%). In addition, a related species Demavendia pastinacifolia (Boiss. & Hausskn.) Pimenov (syn. Peucedanum p. Boiss. & Hausskn) from Asia was reported to contain significant amount of ELE (31.1%) along with MYR (8.2%). 61 Thus, the prevalence of MYR and ELE (in both forms) in the EOs of PL gathered from two Montenegrin localities, suggests the existence of another EO chemotype previously unreported. This is further supported by the fact that these EOs were deprived of large quantities of monoterpenes (in many cases less than 10%), which are often reported as the main compound fraction not only for this but also other Peucedanum species.
Antioxidant Activity
Confronting the values obtained in this study with those found in the literature seems rather difficult, since various methods were used as well as different standard compounds. In addition, there is a lack of standardization of the results, therefore, the interpretation the antioxidant power of the results often seems controversial. 32 According to the AAI values calculated (0.01-0.06), the tested EOs generally showed weak antioxidant capacity. In spite of that, a difference in the efficacy assessment was observed between the methods: while the IC50 values estimated by DPPH test were quite similar, there were significantly elevated FRAP values in some EOs that could be characterized as having moderate reducing power.
A survey of the literature revealed a lack of data on PL, but also about other species from the Peucedanum genus. Only one study was performed on EO from PL (from aerial parts) collected from Turkey 34 ; the activity reported (RSA 41.87%) is consistent with the herein presented results. However, some other Peucedanum species showed much weaker antioxidant activity. The endemic P. akaliniae Akpulat from Southern Turkey showed RSA of 68.79% but at 14 times higher concentration, 62 whereas an IC50 value of 9.13 mg/mL was reported for P. dhana Buch.-Ham. ex C.B.Clark from Northern Thailand. 17 EOs of some closely related species from the genus Dichoropetalum (Peucedanum sensu lato) were analysed as well: D. palimbioides (Boiss.) Pimenov & Kljuykov (RSA 47.26%) 34 and D. knappii (Bornm.) Kljuykov (IC50 0.12 mg/mL). 63 The FRAP values obtained in this study cannot be adequately compared because no literature data were found related to the Peucedanum EOs. However, there are few reports for the Peucedanum species extracts, but with much lower activity assessed; for example, a FRAP value of 0.04 mmol Fe2+/g was reported for the root extract of P. japonicum Thunb. 64
Antioxidant activity of EO is directly correlated with its chemical composition and it is known that some EO constituents possess stronger efficacy than others. For instance, the phenolic compounds thymol, carvacrol and eugenol are characterized by very strong RSA whereas many monoterpenes, such as menthol or camphor, have no or limited capacity.65,66 On the other side, the processes of synergism and/or antagonism largely define the overall activity of EO, making it often impossible to explain and fully understand the mentioned correlation. Thus, it seems to be poorly explained when an activity is associated with a specific EO constituent. However, the influence of phenylpropanoids on the antioxidant potential determined in the present study is quite unquestionable – not only because these are quantitatively the most defining fraction but also show significant antioxidant potential individually as reported in several studies.67,68 In addition, some phenylpropanoid-rich EOs are characterized by strong antioxidant properties. For example, the EOs of two umbellifers from the genus Heracleum (with more than 95% of MYR) exhibited high DPPH scavenging capacity with the IC50 values of 0.05 and 0.08 mg/mL 69 ; the IC50 value of 0.04 mg/mL is reported for the EO rich in ELE (77.2%) of Asarum cordifolium C.E.C.Fisch. 70 Moreover, it appears that these compounds could be more effective in combination: eg, Ferula heuffelii Griseb. ex Heuff. EO rich in both ELE (35.4%) and MYR (20.6%) showed substantial activity with IC50 of 0.02 mg/mL. 71 Similarly, in the herein presented study, some of the fractions with the highest activity contained a significant amount of ELE and/or ILE, in addition to MYR (LFL6h, RFL3h, RFL6h). Further, some authors indicated the meta position of the functional group on the phenyl ring may be more potent in the expression of bioactivity 72 ; ELE and its isomeric form bear two meta-methoxy groups relative to the allyl side chain, thus possibly enhancing their activity comparing to MYR. Some studies suggest presumably antagonistic interactions between MYR and other EO constituents. For example, the EO of another Apiaceae species Petroselinum crispum (Mill.) Fuss, rich in MYR (36.15%), exhibited very poor activity assessed by DPPH test with the IC50 of 12.91 mg/mL 73 ; it also contained significant amounts of apiol (20.9%), which is a structural analogue of MYR, and monoterpene hydrocarbons (more than 30%). Several studies showed strong antioxidant activity of aromatic monoterpenes.65,74 Differently, the hydrocarbon forms of monoterpenes often lack activity whereas the oxygenated forms, especially alcohols, can manifest appreciable effect. 75 Accordingly, some EOs from the herein presented study with the weakest potency contained monoterpenes in large amounts (LFR1h and LFR2h). Notwithstanding, the RFR1h fraction with the second highest FRAP value (10.84 mmol Fe2+/g) contained 21.2% of monoterpenes; however, that fraction included a reasonable amount (8%) of p-cymen-8-ol, an aromatic monoterpene that may have enhanced the reducing power activity. In addition, that EO contained 19.8% of sesquiterpenes. Although exhibiting the least efficacy in comparison with other EO components, 75 it is possible that sesquiterpenes act synergistically, to exert a higher antioxidant potency. In fact, a similar pattern was observed in some other fractions as well; namely, more active EOs were those containing a greater amount of sesquiterpenes along with phenylpropanoids (usually last fractions of each extraction; Table S1, Supplemental Material). Based on that, β- and γ-elemene were the most frequently and abundantly found and some authors reported appreciable activity of EOs rich in these sesquiterpenes. 76 In addition, the LFR24h fraction associated to the lowest IC50 value contained 64.8% of sesquiterpenes and it was exceptionally rich in α-humulene (32%), the influence of which can be assumed and supported by some studies. 77
Conclusions
Flower and fruit EOs from PL were characterized for their chemical composition and associated antioxidant potency. A 24-h fractionated hydrodistillation method was applied leading to 6 different fractions per each extraction. Phenylpropanoid MYR was the main EOs’ chemical constituent accompanied by its structural analogues ELE and ILE. Majority of EOs showed weak antioxidant effect as showed by the DPPH test with the IC50 comprised from 0.61 to 2.54 mg/mL (assessed by DPPH), while the FRAP values ranged from 3.48 to 11.56 mmol Fe2+/g indicating weak to moderate reducing power. Nevertheless, this is the first report on the antioxidant potential of flower and fruit EOs of PL. There is no doubt that biological activities of EOs or other plant extracts are related to their chemical profile. Moreover, it is known that the intercomponents’ synergistic or antagonistic effects could play a crucial role in the expression of those bioactivities as highlighted by artificial intelligence applications.78-81 In the study presented herein, these effects are evident; the impact of terpene fraction in defining the overall antioxidant power has been elaborated. A search of the literature available has revealed little data regarding this species. To the best of the authors’ knowledge, there is only one previous report on EO distilled from Montenegrin PL; there, the PL aerial parts were collected from the northern region of its native range and was characterized by monoterpenes-rich EO. 9 Differently, the PL investigated in this study were gathered from the central and southern parts of its range in Montenegro and the results indicate a phenylpropanoids rich chemotype. Peucedanum genus is well known for its great morphological heterogeneity resulting in numerous phylogenetic studies in recent years that have led to significant taxonomic rearrangements and the exclusion of a plenty of species.12,13,82 In this regard, the distinction of this new chemotype was not only based on the limited literature data on PL, but also due to other Peucedanum species that were taken into consideration. From a literature survey, a predominance of terpenes in the EOs of these species and related taxa (Peucedanum sensu lato) revealed a good agreement with a recent chemometric statistical analysis. 23 Moreover, the most closely related P. officinale L. contains EO rich in monoterpenes.54,83,84 From the chemotaxonomic point of view, the obtained results may serve as a good basis for further investigations.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251332394 - Supplemental material for Phenylpropanoid-rich Essential Oils of Peucedanum longifolium Waldst. & Kit. from Montenegro and Their Antioxidant Properties
Supplemental material, sj-docx-1-npx-10.1177_1934578X251332394 for Phenylpropanoid-rich Essential Oils of Peucedanum longifolium Waldst. & Kit. from Montenegro and Their Antioxidant Properties by Mijat Božović, Rino Ragno, Dragana Petrović, Danka Caković and Stefania Garzoli in Natural Product Communications
Footnotes
Acknowledgments
The authors are thankful to Mrs. Ana Uskoković and Mr. Vukoica Despotović for their help with field work.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Ethical Considerations
Ethical Approval is not applicable for this article.
Author Contributions/CRediT
Conceptualization: MB; Methodology: MB, DP, DC, SG, RR; Writing – original draft preparation: MB; Writing – review and editing: MB, SG, RR; All authors approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The authors confirm that the data supporting of this study are available within the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


