Abstract
Objectives
Dentin, a highly mineralized tissue, is primarily composed of collagen fibers that play essential roles in mineralization and act as a protective barrier against disease. However, the exposure of collagen to dentin decay or acid erosion activates Matrix Metalloproteinases (MMPs), which lead to collagen degradation. This study aims to explore the potential of exogenous polyphenols, specifically purified proanthocyanins (PAs), to enhance collagen stability and mitigate degradation, with a focus on their effectiveness compared to mixed polyphenol extracts.
Methods
Epigallocatechin gallate (EGCG), methylated EGCG, and dimeric PAs (EGCG-EGCG and EC-EC) were used to assess their impact on collagen. Attenuated Total Reflection Fourier Transform Infrared Spectroscopy (ATR-FTIR) was employed to analyze the chemical structure of collagen after treatment with these polyphenolic compounds. Additionally, thermal stability and resistance to enzymatic degradation were evaluated to determine the efficacy of these polyphenols in stabilizing collagen.
Results
Our findings revealed that dimeric PAs significantly improved collagen stability, exhibiting superior results compared to monomeric catechins (EGCG and methylated EGCG). The dimeric PAs were more effective in enhancing thermal stability and resistance to enzymatic degradation. Furthermore, the content of phenolic hydroxyl groups was identified as a critical factor contributing to the stabilization of collagen fibers.
Conclusion
This study highlights the potential of natural polyphenols, particularly dimeric proanthocyanins, in enhancing collagen stability. The findings suggest that these polyphenols could be valuable for dental applications, contributing to improved dentin health by preventing collagen degradation and enhancing its resilience to external stresses.
Background
Dentin is a highly complex, mineralized three-dimensional structure, where type I collagen fibers constitute approximately 90% of the organic matrix. 1 The type I collagen fiber serves as the structural foundation of dentin matrix, playing a crucial role in both the mineralization and remineralization of dentin. 2 So, collagen fibers are crucial to the structural integrity of the dentin, contributing to its strength and resilience. The mineral component of dentin, which is largely hydroxyapatite, provides hardness and rigidity, while the collagen component provides flexibility and toughness. 3 This intricate network of collagen fibers functions not merely as a passive structural element but as an active framework that facilitates essential physiological processes. Moreover, collagen network is pivotal in combating dental diseases, particularly in its role of inhibiting disease progression. When the integrity of the collagen network is maintained, it presents a barrier to bacterial penetration and enzymatic degradation. 4 However, if this barrier is compromised—whether dentin decay or acid erosion, exposed collagen fibers are vulnerable to various environmental factors in the mouth, such as saliva, acids, and enzymes. 5 Then the fragile collagen fibers activate Matrix Metalloproteinases (MMPs) that degrade collagen, causing irreversible histopathological damage. Therefore, enhancing the stability of collagen fibers is crucial for maintaining the structural integrity of dentin. 6
MMPs and collagenase are a part of the MMP family. They play a crucial role in the breakdown of the extracellular matrix (ECM) during physiological processes such as embryogenesis, tissue remodeling, and wound healing.
7
However, when dentin was exposed, the deregulated MMP will cleave fibrillar collagens like type I, II, and III collagen which result in tissue destruction.
8
Exogenous cross-linking is a technique that has been increasingly recognized for its effectiveness in enhancing the molecular stability of collagen, which will result in more robust and more resistant collagen to enzymatic degradation and tear.
9
One hot direction is the use of plant extracts, polyphenol
Proanthocyanidins (PAs) are a class of polyphenols that widely distributed in grape seeds, cocoa seeds, pine bark and tea. The basic building blocks of PAs are flavan-3-ol units, which include catechin and epicatechin.
13
PAs have gained attention in dental research due to their ability to interact with collagen, enhancing its strength and resistance to enzymatic degradation which present potential value in preventing dental caries and promoting dentin remineralization.
14
However, most studies are about the extract of PAs which are mixtures of different degrees of polymerization, and there is a lack of research on the use of pure polymers. The structural composition and biological activity of natural polyphenols on dentin collagen still require further experimental validation.
15
Herein, we focused on the effect of pur
Materials and Methods
Dentin Sample Preparation
All procedures performed in this study were in accordance with the ethical standards of Anhui Medical University. Thirty intact human molars were collected at the Affiliated Stemmatological Hospital of Anhui Medical University and stored at −20°C prior to use. After removing the enamel crown using a slow-speed cutter under running water, dentin slices of 5 mm × 5 mm × 0.5 mm were extracted perpendicular to the long axis of each tooth's mid-crown region. The dental plates we selected were parallel to the occlusal surface, perpendicular to the major axis of the tooth. The dentin slices were then randomly divided into five groups: EGCG, EGCG-3"Me, EGCG-EGCG, EC-EC, and a blank group, each group with 10 slices. All dentin samples underwent demineralization with 10% phosphoric acid for 5 h followed by repeated washing with distilled water. Subsequently, they were immersed in their respective pretreatment solutions from the five groups for one hour before relevant assays. Appropriate amounts of EGCG, EGCG-3"Me, EGCG-EGCG, and EC-EC were weighed and completely dissolved in 20% DMSO. They were then diluted in HEPES buffer (20 mM, pH 7.2) to obtain a final concentration of 0.65% (w/v) as pretreatment solution. For the blank group, an equal volume of HEPES buffer was used along with an addition of less than 5% DMSO.
Reagents
EGCG, EGCG-3Me, EGCG-EGCG, and EC-EC were provided by the State Key Laboratory of Tea Plant Biology and Resource Utilization of Anhui Agricultural University. Bacterial collagenase I and BioVisions Collagenase were purchased from Sigma-Aldrich (St. Louis, MO, USA). A human hydroxyproline enzyme-linked immunosorbent assay kit was obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
ATR
One slice from each of the five groups of pre-prepared dentin slices was utilized for ATR spectroscopy analysis. The ATR spectra were collected using a Thermo Nicolet iS50 infrared spectrometer equipped with a diamond internal reflection accessory. The scanning range was set at 400-4000 cm−1 with a resolution of 4 cm−1. Subsequently, the obtained infrared spectra were analyzed using OMNIC software.
Swelling Rate
Five dentin slices were selected from each group to determine the swelling rate. Initially, the weight of cross-linked dentin matrix collagen was measured under air drying conditions (W0), followed by inflation in distilled water for 30 min. Excess liquid was removed using filter paper and the wet weight was recorded (W1).
The amount of dentin collagen adsorption can be calculated as follows: swelling rate (%) = (W1 - W0) / W0 × 100%. Each sample underwent three duplicate weighing to obtain an average value. There were five samples in each group (n = 5).
Ultimate Tensile Strength
The demineralized dentin strips were subjected to the ultimate tensile test using a digital display electronic universal testing machine (WDS-20). Each dentin strip was securely fixed onto a specialized test fixture, and the loading rate was set at 0.5 mm.min
Dentin Collagen Thermal Stability Test
The denaturation temperature of dentin collagen in each group was determined using differential scanning calorimetry (DSC). Experimental tests were conducted on a DSC analyzer (DSCQ2000, TA, USA). One treated dentin slice from each group was dried and placed in an aluminum crucible specifically designed for DSC analysis. After capping and sealing the crucible, it was heated from 10°C to 150°C at a heating rate of 5°C/min, while maintaining a nitrogen flow rate of 20 mL/min in the sample chamber. An empty crucible served as the reference.
Bacterial Collagenase-Mediated Collagen Degradation
Five groups (consisting of 5 slices per group) of pretreated dentin slices were immersed in 1 mL bacterial collagenase I solution (100 U/mL) and incubated at 37°C with constant shaking for 7 days. The concentration of hydroxyproline in the solution was quantified using the Human hydroxyproline ELISA Kit. Following kit instructions, after one week of enzymatic reaction, supernatant samples (400 μL) were collected from each reaction solution group. These samples were transferred to polypropylene tubes and mixed with 6 M HCl (400 μL). The resulting mixture underwent hydrolysis at 98°C for 24 h before completion. After lyophilization, the amount of hydroxyproline was measured upon dissolution with distilled water (600 μL), with three replicates performed for each group.
Ninhydrin Test
5 g of solid indophenol blue powder was dissolved in 125 mL of boiled deionized water. Subsequently, 5 g of solid vitamin C powder was dissolved in 250 mL of warm deionized water. Added the vitamin C solution to the indophenol blue solution, stirred continuously until the emergence of white precipitation. Placed the mixed solution in a refrigerator set at 4°C for cooling. After cooling, the solution was subjected to centrifugation for precipitation, washed three times with deionized water, collected, lyophilized, and stored in a vacuum for subsequent use. Weighed 15 mg of solid reduced indophenol blue powder and 85 mg of solid indophenol blue powder, and dissolved them completely in 10 mL of ethylene glycol monomethyl ether to prepare the indophenol blue staining solution. Testing of free amino acids in the dentin matrix collagen: Prepared a standard glycine solution at a concentration of 50 μg/mL, and added 0, 0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 mL of the glycine solution to each centrifuge tube successively, filling each tube to 1 mL with distilled water to establish a glycine concentration gradient. Divided the cross-linked 2 mg of dentin powder into individual portions and placed them in the centrifuge tubes containing the standard.
Inhibition of Collagenase Activity
The inhibition of collagenase activity was assessed using the BioVisions Collagenase Activity Assay Kit (colorimetric method). The assays were conducted in accordance with the manufacturer's instructions on 96-well plates, with 3 replicates per group. The experiment consisted of seven groups: a negative control group, a positive control group, an EGCG group, an EGCG-EGCG group, an EC-EC group, and an EGCG-3 “Me” group. Each polyphenol solution had a concentration of 0.65% (w/v). After mixing, all samples were incubated at room temperature for 10 min. Subsequently, 60 µL per well was aspirated and mixed with 40 µL of collagenase substrate (FALGPA) in each set of reagent wells. Following completion of the reaction, absorbance was dynamically measured at a wavelength of 345 nm for 5-15 min using a microplate reader. Collagenase activity was calculated using the following equation.
In the equation, ΔA345 nm represents the difference between A345 nm at time point T2 and A345 nm at time point T1. The absorbance of A345 nm is measured within the linear range at both time points (T1 and T2). ΔT denotes the time interval between T2 and T1. The reaction solution has a volume of 0.2 mL. DF represents the dilution factor, which is set to 1 in this case. The FALGPA extinction coefficient is 0.53, while V indicates the volume of enzyme solution in mL.
Statistical Analysis
Data processing and analysis were performed using statistical analysis software SPSS 16.0. ANOVA was employed to compare overall differences among the five groups, while pairwise comparisons between groups were conducted using LSD-t test with a significance level of α=0.05.
Results
ATR Spectra of Polyphenols Treated Dentin Collagen
The corresponding ATR spectra of the blank group, EGCG, EGCG-3"Me, EGCG-EGCG, and EC-EC treated dentin slices are presented in Figure 2. The main spectral bands associated with type I collagen include the amide I band near 1640 cm−1, the amide II band near 1550 cm−1, the CH2 shear vibration near 1450 cm−1, and the amide III band near 1240 cm−1. As depicted in Figure 2, characteristic peaks representing dentin collagen remained after treatment in each group without any significant appearance of new waves. Comparing to the blank group, there were noticeable alterations observed in the ATR spectra of dentin collagen treated by EGCG, EGCG-3 “Me, and EGCG-EGCG. Specifically, an increase in wave width was observed within the amide I band along with a slight shift towards lower frequencies. In contrast to this, both position and width of each characteristic peak within EC-EC group were essentially unchanged when compared to those of the blank group.

(A) Mass spectrum of different phenol agents. (B) Chemical formula of different phenol agents.

Fourier-transform infrared (FTIR) spectra of dentin slices treated by Hepes, EGCG, EC-EC, EGCG-EGCG and EGCG-3"Me, Amides I, II, and III at 1640, 1550, and 1240 cm-1. Hepes treatment served as control.
The ratio between peak area at amide III band (A1240) and peak area at 1450 cm−1 (A1450) is considered as an important criterion for evaluating preservation of conformational integrity within collagen triple helix structure. Conversely, the ratio between peak area at amide II band (A1550) and peak area at 1450 cm−1 (A1450) was used to assess degree of collagen cross-linking, a decrease in A155/A145 indicated increased collagen cross-linking.
The Swelling Rate of Polyphenols Treated Dentin Collagen
The impact of each crosslinker treatment on the expansion rate of collagen in demineralized dentin matrix is illustrated in Figure 3. One-way ANOVA analysis revealed that the swelling rate of dentin collagen was significantly lower in all treatment groups compared to the control group (

The Swelling Rate of Matrix Collagen of Demineralized Dentin in Each Group After Cross-Linking. N = 5, **
Ultimate Tensile Strength
The ultimate tensile strength is another important characteristic of the mechanical properties of dentin collagen. The ultimate tensile strengths of five groups of demineralized dentin bars exhibit in Figure 4 were as follows: control group, 12.39 ± 5.11 MPa; EGCG group, 19.96 ± 7.85 MPa; EGCG-3"Me group, 18.15 ± 8.92 MPa; EGCG-EGCG group, 21.61 ± 7.59 MPa; EC-EC group, 15.48 ± 6 .68 Mpa. There were statistically significant differences among the five groups (

Ultimate Tensile Strength Test of Demineralized Dentin Beams Treated by Hepes, EGCG, EGCG-3"Me, EGCG-EGCG or EC-EC. n = 15, *
Dentin Collagen Thermal Stability Test
The thermal stability of dentin collagen was assessed by differential scanning calorimetry (DSC) to determine its denaturation temperature. The experimental results are depicted in Figure 5, with the peak on the curve indicating the denaturation temperature of collagen. The denaturation temperatures of dentin collagen in each group were as follows: blank 122.09°C; EGCG 125.31°C; EGCG-3 “Me 122.71°C; EGCG-EGCG 127.65°C; EC-EC 123.49°C. Compared to the blank group, all cross-linking agents led to higher denaturation temperatures of dentin collagen, with the most significant changes observed for EGCG-EGCG and EGCG.

A) The Thermal Stability of Collagen Treated by Hepes, EGCG, EGCG-3"Me, EGCG-EGCG or EC-EC. B) The Statistical Result of the Thermal Stability. n = 5, *
Effect of Polyphenols on Resistance to Bacterial Collagenase Digestion
The hydroxyproline content of demineralized dentin slices was measured after enzymatic hydrolysis in five different groups. The levels of hydroxyproline were significantly lower in the EGCG, EGCG-3 “Me, EGCG-EGCG, and EC-EC groups compared to the blank group (

Collagen Degradation of Dentin Slices Treated by hHepes, EGCG, EGCG-3"Me, EGCG-EGCG or EC-EC. n = 5, *
Ninhydrin Test
The absorbance Y of the measured glycine standard solution and its corresponding mass concentration X (μg/mL) were calculated, yielding the linear standard curve: Y = 0.022X-0.0288, with a correlation coefficient R2 = 0.9908, indicating a strong and significant correlation as demonstrated in the Figure 7.

The Standard Curve of Glycine for Cross-Linking Test.
The absorbance obtained from each experimental group were used to calculate the free amino group n (-NH2) (μmol) in dentin powder after crosslinking, based on the molecular weight of glycine (75.07), using a standard curve equation. As shown in the Figure 8, the blank group without crosslinking had a free amino group content of 0.133 ± 0.036 μmol, while the EGCG and EGCG-EGCG groups showed significantly decreased amounts of free amino groups compared to the blank group (
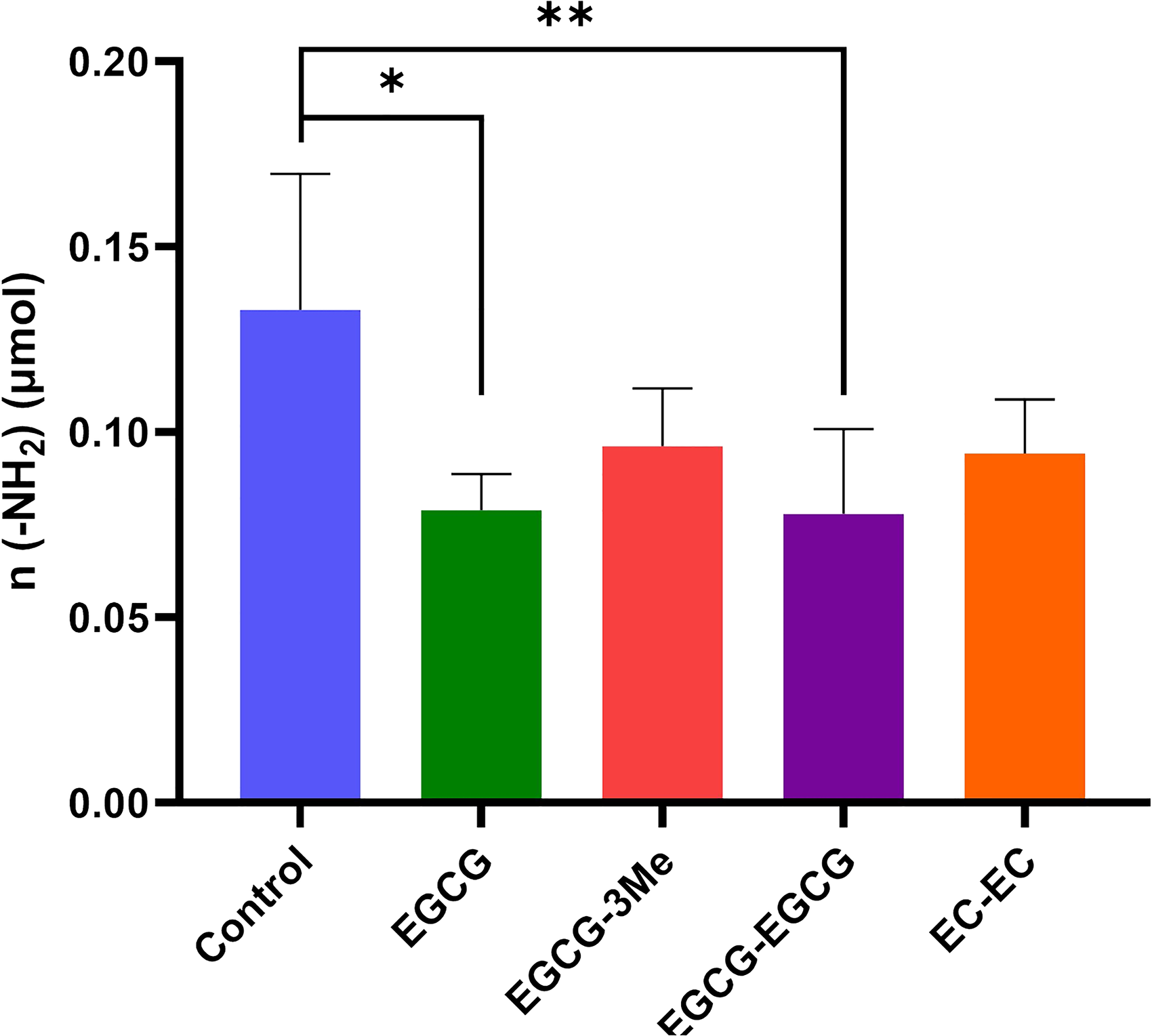
The Content of n (-NH2) in Demineralized Dentin Powder of Each Group After Cross-Linking. n = 5, *
Effect on Collagenase Activity
The effect on collagenase activity is illustrated in the Figure 9, demonstrating the inhibition caused by each compound. Compared to the negative control group, significant reduction in collagenase activity was observed in the positive control, EGCG-EGCG, and EC-EC groups (
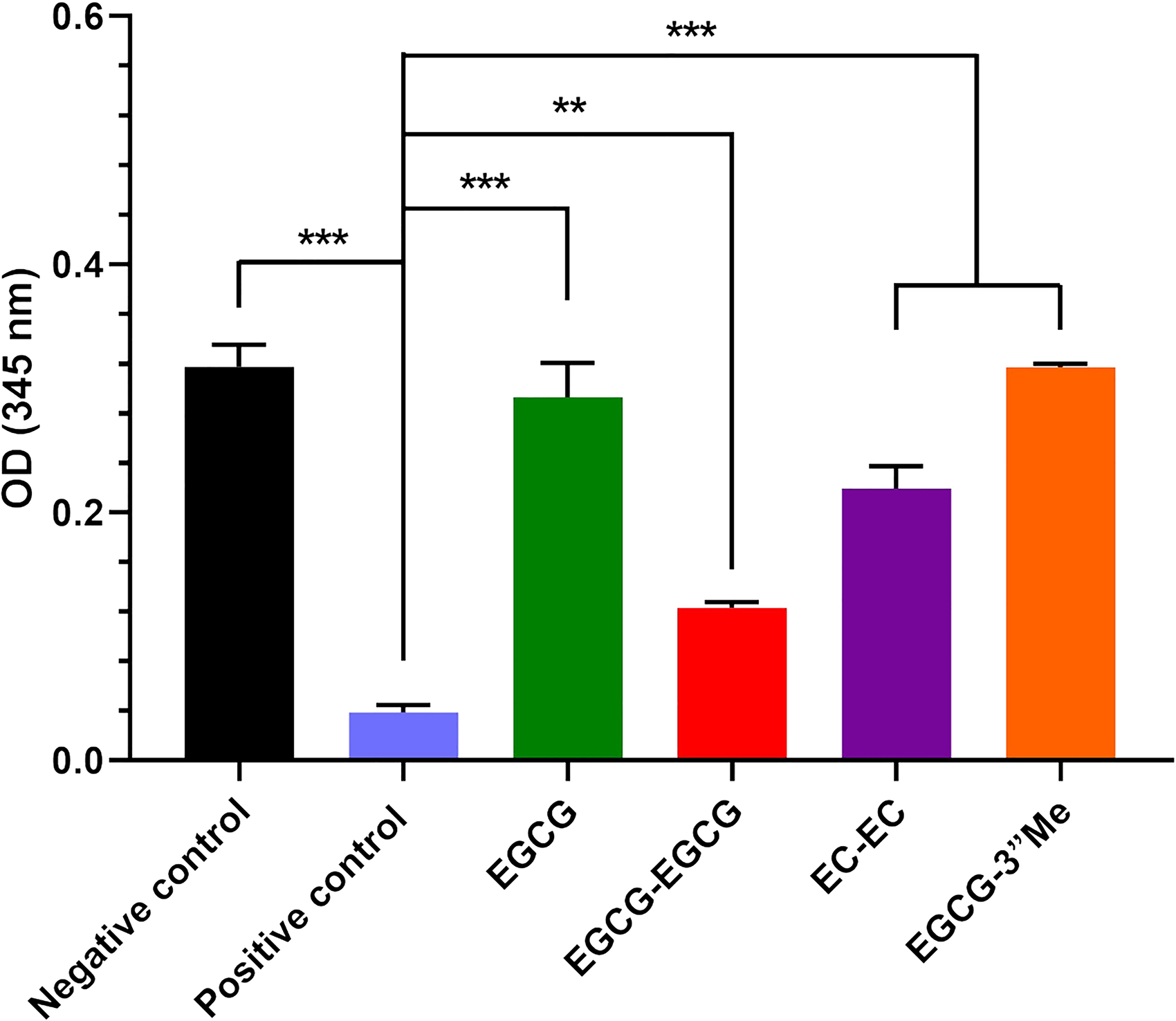
The Effect of EGCG, EGCG-3"Me, EGCG-EGCG or EC-EC on Collagenase Activity. n = 3, *
Discussion
Previous studies have demonstrated that PAs can enhance the cross-linking of dentin collagen, 16 inhibit enzymatic degradation of dentin collagen, 17 and maintain the stability of the dentin matrix. 18 Furthermore, subsequent investigations have revealed that different sources of PAs exhibit varying degrees of protective effects on dentin. 19 The proportion of PAs oligomers is often considered a crucial indicator for evaluating their impact. 20 The number of phenolic hydroxyl groups and the degree of polymerization are two pivotal factors influencing the cross-linking and stability of PAs on dentin collagen.
In this experiment, we u
The presence of phenolic hydroxyl groups in PAs greatly influences their cross-linking efficiency with collagen.
22
Notably, EGCG-EGCG displays a significantly higher count of these groups compared to EC-EC, EGCG, and EGCG-3"Me. Consequently, EGCG-EGCG demonstrates the most robust cross-linking action. The
The enhancement of cross-linking may be attributed to the increase in polymerization degree, as observed in EGCG-EGCG. However, solely prolonging the polymerization degree may not have a significant impact if we disregard the influence of phenolic hydroxyl groups, as exemplified by EC-EC's minimal effect on dentin collagen. It is possible that the variation exhibited by certain PAs with different degrees of polymerization is primarily due to an increase in the number of phenolic hydroxyl groups resulting from higher polymerization degrees. Interestingly, despite both EGCG and EC-EC containing the same number of phenolic hydroxyl groups, their cross-linking effects differ significantly, the EGCG exhibiting superior performance. The structural difference lies in the clustering of three hydroxyl groups on a benzene ring in EGCG versus two hydroxyl groups on the benzene ring in EC-EC. This suggests that the concentration of hydrogen bonding between collagen and phenolic hydroxyl groups gathered on one benzene ring enhances cross-linking. Therefore, collagen cross-linking with PAs is not only dependent on the number of phenolic hydroxyl groups but also closely related to the gallated moieties.
From the results of collagenase inhibition by polyphenols, both dimeric EGCG-EGCG and EC-EC exhibited superior inhibitory activity. The inhibitory effect of EGCG-EGCG was significantly greater than that of EC-EC, suggesting that substituted gallate groups also influence enzyme inhibition. However, neither monomers EGCG nor EGCG-3 “Me demonstrated significant inhibitory effects. This implies that while PAs may modify collagen, their inhibitory effect on collagenase is more closely associated with their degree of polymerization and hydroxyl groups.
PAs are non-specific inhibitors of MMPs, 23 current research suggests that PAs can induce multi-site cross-linking of amino acids within the catalytic region of enzymes, leading to irreversible alterations in the spatial structure or structural elasticity of this region. This interference disrupts enzyme specificity recognition and hampers the formation of enzyme-type I collagen complexes. 24 Moreover, also studies proposed that PAs can modulate conformational regulation in non-catalytic regions of proteases, thereby exerting inhibitory effects. 25 Sang Gu Kang et al, conducted a molecular simulation to demonstrate that (-)-epicatechin can directly blocks active residues as in reference compound EGCG while proanthocyanidin B2 inhibits the enzyme activity by occupying the substrate binding region in the protein structure. 26 This alteration irreversibly modifies the spatial arrangement of the protease, thereby impeding its specific recognition and formation of enzyme-collagen complexes. Additionally, PAs may impact allosteric regulation within the non-catalytic region of proteases, 27 exerting an inhibitory influence through this mechanism as well. Furthermore, PAs can directly occupy the substrate binding site within protein structures to function as enzymatic inhibitors. 28 Based on our current experimental findings, we hypothesize that dimeric PAs could form cross-links with enzymes, thus obstruct their binding sites on proteins and consequently exhibit enhanced inhibitory effects.
To enhance the stability and bioavailability of EGCG and confer novel biological functions, researchers have endeavored to perform various derivatizations of EGCG. Several modifications, such as esterified EGCG, 29 methylated EGCG, 30 acylated EGCG, 31 and glycosylated EGCG, 32 have been explored. It has been concluded that methylation of EGCG significantly improves its lipid solubility and stability, 33 thereby effectively increasing its bioavailability. However, in the present experiments, it was observed that compared to EGCG-3 “Me, unmodified EGCG exhibited higher cross-linking with dentin collagen and greater resistance to enzymatic degradation. This suggests that the reduction in hydroxyl groups due to methylation may diminish the biological activity of methylated EGCG on dentin matrix collagen. Henceforth, when modifying EGCG, striking an appropriate balance between enhancing stability and preserving biological activity is crucial depending on specific application requirements.
From the perspective of 3D structural diversity, PAs exhibit a significant degree of structural variation due to variations in monomer types, interflavonoid bonds (bond types and attachment positions), and degree of polymerization. 34 The diverse shapes and spatial arrangements also contribute to their distinct biological activities. Moreover, our current study revealed a close correlation between the cross-linking effect of PAs on dentin collagen and the position of the phenolic hydroxyl group within its structure. Regrettably, due to limitations in experimental design, we were unable to investigate this aspect comprehensively. In future experiments, we will explore the intricate relationship between PAs's three-dimensional structure and its impact on dentin collagen biology.
PAs has been extensively utilized in various fields due to their unique biological activities, particularly in the realm of oral health. They have demonstrated significant potential in the biomodification of dentin by interacting with type I collagen present within it. This interaction not only enhances the mechanical properties and biostability of dentin but also effectively reduces enzymatic degradation of collagen, thereby providing long-lasting protection for teeth. The bioactivity of proanthocyanidins is closely associated with their molecular weight, degree of polymerization, and monomer type. In this study, we extracted two catechin monomers and two proanthocyanidin dimers separately while systematically investigating the impact of different degrees of proanthocyanidins and their monomers on the physicochemical properties of dentin collagen. The results revealed that the effect exerted by different monomers on collagen primarily depended on the proportion of phenolic hydroxyl groups they possessed. Specifically, monomers with a higher proportion exhibited more efficient binding capabilities towards collagen, thereby enhancing its structural stability while reducing enzymatic degradation. Based on these findings, we claim that PAs can partially regulate the physical and chemical properties exhibited by dentin collagen. This will make an important contribution to innovation and development in the field of oral health.
Conclusions
In this study, different monomeric catechins and dimeric PAs were used to enhance the stability of dentin collagen through cross-linking and enzyme inhibition. PAs, abundant in phenolic hydroxyl groups, demonstrates the most potent effects, while EC dimer exhibits the least efficacy. The cross-linking process depends on the quantity and structural positioning of the phenolic hydroxyl groups. Dimeric PAs also exhibit more potent collagenase inhibition, suggesting that polymerization enhances enzyme inhibition. Methylation-like modifications can increase EGCG's stability but may potentially reduce its biological activity since it contains one fewer hydroxyl group than EGCG. Thus, this study highlights the role of natural polyphenols in enhancing collagen stability and contributing to dentin health. Future research should explore the structural subtleties of PAs for the development of optimized dental applications.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251332159 - Supplemental material for Stabilizing Impact of Monomeric Catechins and Dimeric Proanthocyanins on Dentin Collagen
Supplemental material, sj-docx-1-npx-10.1177_1934578X251332159 for Stabilizing Impact of Monomeric Catechins and Dimeric Proanthocyanins on Dentin Collagen by Xi Yang, Honglin Yang, Tongtong Han, Xinwei Zhu, Mei Zhao and Guanhu Bao in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by the Laboratory open project of Key Lab of Oral Diseases Research of Anhui Province.
Ethical Considerations
This study was approved by the Ethics Committee of Anhui Medical University Affiliated Stomatology Hospital Ethics Committee (Approval Number: T2024046).
Author Contributions/CRediT
Conceptualization, Honglin Yang.; methodology, Honglin Yang.; validation, Xi Yang.; formal analysis, Xi Yang. and Tongtong Han.; investigation, Xinwei Zhu. and Mei Zhao.; resources, Honglin Yang. and Guanhu Bao.; data curation, Xi Yang.; writing—original draft preparation, Xi Yang.; writing—review and editing, Honglin Yang.; supervision, Honglin Yang.; project administration, Honglin Yang. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Laboratory open project of Key Lab of Oral Diseases Research of Anhui Province (2020kqsy03), Scientific research project of colleges and universities in Anhui Province (2022AH050782). Laboratory open project of Key Lab of Oral Diseases Research of Anhui Province, Scientific research project of colleges and universities in Anhui Province, (grant number 2020kqsy03, 2022AH050782).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
