Abstract
Background
Croton megalocarpus has been traditionally used for its medicinal properties, but its potential anticancer effects remain underexplored. This study evaluates the cytotoxicity of its solvent fractions against HeLa cells to identify its most potent anticancer fraction.
Methods
The dichloromethane (CMDE), ethyl acetate (CMEE), and methanol (CMME) fractions of C. megalocarpus were tested for cytotoxicity using MTT and clonogenic assays. Lipid peroxidation and glutathione depletion were assessed to evaluate oxidative stress. The surviving fraction was calculated based on plating efficiency, and statistical analyses were applied to validate findings.
Results
CMEE exhibited the most potent cytotoxic activity, showing a dose-dependent reduction in HeLa cell survival. At 320 µg/ml CMEE, the surviving fraction was reduced by 88% (p < 0.0001). DOX, used as a positive control, showed a comparable cytotoxic effect. Increased lipid peroxidation and glutathione depletion further indicated oxidative stress-mediated cytotoxicity.
Conclusion
The findings demonstrate that CMEE exerts significant anticancer activity through oxidative stress induction and inhibition of HeLa cell proliferation. These results support its potential as a source of novel anticancer agents, warranting further mechanistic and in vivo studies.
Introduction
Despite the plethora of modalities available for cancer treatment, the attainment of complete remission remains an elusive objective. This predicament is underscored by the persistent lack of significant reduction in cancer mortality rates observed in recent years, with over 50% of diagnosed patients succumbing to the disease. 1 In light of this daunting reality, the exploration of natural products emerges as a promising avenue for the discovery and development of novel anticancer agents.
The screening of natural products represents a potent strategy in the quest for compounds endowed with anticancer properties. Within the vast reservoir of secondary metabolites synthesized by diverse botanical sources lies the potential for unearthing molecules possessing remarkable efficacy against cancer.2,3 The exploitation of these natural reservoirs holds promise for the generation of innovative pharmaceutical compounds with the potential to profoundly impact the landscape of human healthcare.
Throughout the annals of drug discovery, natural products have emerged as indispensable reservoirs of bioactive compounds and structural scaffolds, underpinning the development of paradigm-shifting therapeutics. 4 From statins to vinca alkaloids, taxols, and antibiotics, these naturally occurring entities have served as cornerstones in the pharmacopeia, addressing a diverse spectrum of ailments, including neoplastic disorders, cardiovascular maladies, and inflammatory conditions.5-8 Thus, the sustained exploration of natural product reservoirs remains imperative in the relentless pursuit of innovative therapeutic modalities.
This renewed interest in natural products has led to the isolation and characterization of an expansive array of bioactive compounds, with over 210,272 natural products identified to date. 9 These compounds encompass diverse chemical classes, including terpenoid derivatives, alkaloids, glycosides, polyphenolics, and steroids, among others.5,6 Notably, many of the cornerstone chemotherapeutic drugs trace their origins to plants, underscoring the historical and ongoing significance of natural sources in drug discovery.1,2,7
While modern chemotherapeutic agents have demonstrated remarkable efficacy in combating various neoplastic diseases, their clinical utility is tempered by significant adverse effects. 10 Myelosuppression, gastrointestinal disturbances, reproductive toxicity, nephron damage, and hair follicle impairment represent prominent challenges associated with their use.11-13 Furthermore, the emergence of resistance phenomena poses a formidable obstacle, rendering tumors refractory to conventional treatment modalities.14,15 Thus, there persists an unmet clinical need for the discovery of novel biomolecules capable of selectively targeting cancer cells while sparing normal tissues, thereby minimizing toxicity and circumventing resistance mechanisms.
Croton megalocarpus stands as a notable botanical entity, characterized by its medium-sized stature, often reaching heights of up to 35 meters, and distinguished by a cylindrical, branchless bole extending up to 20 meters in height and 1 meter in diameter, adorned with a spreading and flat crown.16,17 Its ubiquity as a shade tree is underscored by its frequent cultivation for this purpose. Known by various vernacular names such as “mbali” in East Africa and “msuduzi” in Kenya, the plant holds cultural and medicinal significance across its range, recognized as a vital component of traditional medicine in Kenya. 18
A comprehensive understanding of C. megalocarpus’ therapeutic potential is further augmented by previous investigations into its phytochemical composition. Phytochemical screening of its leaves and stem bark has revealed the presence of an array of bioactive constituents including alkaloids, flavonoids, glycosides, saponins, sterols, tannins, and terpenoids.16,17,19
Croton megalocarpus was selected for this study based on its rich ethnomedicinal history and documented bioactive compounds with potential anticancer properties. Traditionally, C. megalocarpus has been used in East Africa for the treatment of bacterial, viral, and inflammatory conditions, suggesting the presence of pharmacologically active constituents. 20 Previous phytochemical analyses have revealed that C. megalocarpus contains alkaloids, flavonoids, terpenoids, tannins, and glycosides, many of which have demonstrated cytotoxic and anticancer properties in other plant species.16,17 Additionally, recent studies have reported its antiviral activity, particularly against HIV, and suggestive cytotoxic effects against MT-4 cell lines. 21
Given its rich ethnomedicinal legacy 20 and documented phytochemical profile, C. megalocarpus emerges as a compelling subject for further scientific inquiry. Building upon previous research indicating its antiviral activity, particularly against HIV 21 and suggestive evidence of cytotoxicity against MT-4 cell lines 17 this study aims to investigate its potential as an anticancer agent. By elucidating its pharmacological properties and mechanisms of action, this research endeavors to contribute to the expanding repertoire of natural products with therapeutic relevance in oncology.
Methods
Collection and Extraction of Plant Material
The stem bark of Croton megalocarpus were collected from botanical garden 1.2233° S, 36.8803° E in Nairobi, Kenya. Ethical approval was granted by KNH-UON ERC under approval number P992/12/2019, and a voucher specimen was deposited at the USIU-Africa herbarium.
Five hundred grams (500 g) of powdered stembark was extracted with 2.5 liters of dichloromethane and methanol (1:1 ratio) using cold maceration. The resulting crude extract was dried and suspended in distilled water, followed by sequential partitioning with solvents of increasing polarity: dichloromethane (CMDE), ethyl acetate (CMEE), and methanol (CMME). The obtained fractions were concentrated using a rotary evaporator and dried at 30 °C.
The dried stem bark extracts were dissolved in 0.5%–1% dimethyl sulfoxide (DMSO), diluted with RPMI 1640 medium, and prepared as a 4 mg/ml stock solution. The solution was then filtered through a 0.22 µm membrane filter and stored at 4 °C.
Preparation of Test Solutions
Prior to inoculation into cell culture, extracts of C. megalocarpus were prepared at concentrations of 10 mg/ml in dichloromethane, ethyl acetate, and methanol, and initially dissolved in dimethyl sulfoxide (DMSO). These extracts were then diluted in sterile minimum essential medium (MEM) as needed for the experiments, ensuring that the final concentration of DMSO did not exceed 0.01%. Additionally, doxorubicin (DOX) was directly dissolved in MEM. All solutions were freshly prepared immediately before use to maintain optimal efficacy.
Cell Culture
The human cervical adenocarcinoma cell line (HeLa, ATCC® CCL-2™) was maintained as monolayer cultures in MEM supplemented with 10% (v/v) heat-inactivated fetal calf serum (FCS) and 1% antibiotic-antimycotic solution containing 1000 U/ml penicillin, 10 mg/ml streptomycin sulfate, 5 mg/ml gentamicin, and 25 µg/ml amphotericin-B. The cultures were incubated at 37 °C in a CO2 incubator (Eppendorf, Chennai, India) under controlled conditions of 5% CO2, 95% air, and 90% relative humidity.
Experimental Procedures
MTT Assay
The cytotoxic effects of CMDE, CMEE, and CMME were assessed based on the methodologies outlined by Mosmann, 22 and Zhu & Paul. 23 HeLa cells (5 × 104 per well) were plated in 96-well microplates (Corning; Thermo Scientific, USA) with 100 µl of MEM and incubated for 24 h to allow proper attachment. 21 Following this, the culture medium was discarded, and the cells were washed twice with sterile phosphate-buffered saline (PBS) to remove residual media.
Subsequently, the cells were exposed to different concentrations of CMDE, CMEE, CMME (0, 20, 40, 80, 160, and 320 µg/ml), or the positive control DOX. The microplates were then incubated at 37 °C in a CO2-enriched atmosphere for 24 h. After the treatment period, the medium was replaced with 1.2 mM MTT solution prepared in serum-free MEM, followed by a further 3-h incubation to allow for formazan formation. 21 The resulting formazan crystals, indicating viable cells, were solubilized by adding DMSO, and the optical density (OD) was measured at 570 nm using a Tecan Infinite M 200 microplate reader (Männedorf, Germany).
The percentage of cell viability was calculated using the following formula:
Clonogenic Assay
Logarithmically growing HeLa cells (5 × 105) were seeded into culture flasks (Techno Plastic Products, Trasadingën, Switzerland) and cultured for 48 h to reach the log phase. The CMDE group of HeLa cells was treated with various concentrations (20, 40, 80, 160, or 320 µg/ml) of the dichloromethane extract of C. megalocarpus. The CMEE group of HeLa cells was exposed to different concentrations (20, 40, 80, 160, or 320 µg/ml) of the ethyl acetate extract of C. megalocarpus. The CMME group of HeLa cells was treated with various concentrations (20, 40, 80, 160, or 320 µg/ml) of the extract of C. megalocarpus. As a positive control, the DOX group of HeLa cells was treated with varying concentrations (4, 8, 12, 16, or 20 µg/ml) of doxorubicin. After treatment, the media were aspirated, and the cells were washed twice with sterile PBS, then treated with trypsin EDTA for cell harvesting.
For the clonogenic assay, 200 harvested HeLa cells were seeded into 60 mm petri dishes with 5 ml of MEM and incubated for 12 days to form colonies. At the end of this period, the dishes were stained with 1% crystal violet in methanol. Colonies with at least 50 cells were counted using a stereo zoom microscope (Ernst Wild M3, Wild Heerbrugg Ltd, Heerbrugg, Switzerland).24,25
The plating efficiency (PE) of the cells was determined using the formula:
The clonogenic assays revealed that the CMEE exerted the greatest cytotoxic effect and therefore following experiments were performed with CMEE only.
Biochemical Analyses
A separate experiment was conducted to examine the effects of different concentrations (20, 40, 80, 160, or 320 µg/ml) of CMEE on various biochemical parameters in cell homogenates (lipid peroxidation and glutathione) and in the medium (lactate dehydrogenase). Initially, 1 × 106 HeLa cells were seeded into multiple petri dishes and treated with CMEE at the specified concentrations, along with a positive control of 10 µg/ml DOX. The concentrations of glutathione (GSH) and lipid peroxidation (LOO) were measured in the cell homogenates, while the levels of lactate dehydrogenase (LDH) released into the medium were assessed at various time points after CMEE treatment (2, 4, 6, 8, 12, and 24 h). To ensure the reliability of the results, the experiments were repeated three times for validation.
Glutathione
To estimate glutathione levels, a method used by Mazzari et al (2022) was employed. 26 One milliliter of HeLa cell homogenate was mixed with 0.5 ml of ice-cold 10% 5-sulfosalicylic acid to precipitate the proteins. 26 The mixture was then placed on ice for 10 min and centrifuged at 15,000 rpm at 4 °C for 15 min. The resulting protein-free supernatant was collected and combined with 0.5 ml of NADPH (4 mg in 100 ml of 0.5% NaHCO3), 0.5 ml of glutathione reductase (6 units/ml, pH 7), and 1 ml of 0.6 M DTNB in 0.2 M phosphate buffer (pH 8).
The formation of TNB (5-thio-2-nitrobenzoic acid) was measured using a UV-Visible double-beam spectrophotometer (Shimadzu UV-260, Shimadzu Corporation, Tokyo, Japan) at 412 nm, with a blank sample (without GSH) used to determine the background rate of product formation. This background rate was subtracted from the sample values before quantifying GSH. The concentration of GSH was expressed as nM per 106 cells, and a standard curve was constructed using 1–10 nM GSH/ml.
Lipid Peroxidation
Lipid peroxidation in HeLa cells was evaluated by measuring thiobarbituric acid reactive substances (TBARS), which include lipid hydroperoxides, aldehydes, and malondialdehyde, using the method described by Ghanati & Bakhtiarian. 27 To perform the assessment, 1 ml of supernatant was mixed with 0.8% thiobarbituric acid, 20% acetic acid, and 0.1% SDS. The mixture was then incubated in a boiling water bath for 30 min to precipitate lipoproteins. 27 After cooling, the solution was extracted with N-butanol-pyridine, and the butanol layer was separated. The absorbance of this layer was measured at 532 nm using a UV-Visible double-beam spectrophotometer. Lipid peroxidation was quantified as nM TBARS per 106 cells by comparing the absorbance readings to a standard curve generated using 0–5 nM malondialdehyde (MDA).
Lactate Dehydrogenase
The release of lactate dehydrogenase (LDH) from HeLa cells into the media was evaluated at different time points (0, 0.5, 1, 2, 4, 8, and 12 h) post-treatment with CMEE or DOX, following a modified protocol by Decker & Lohmann-Matthes. 28 For each experimental group, the MEM from individual cell cultures was collected at specified intervals and subjected to centrifugation. Subsequently, 50 µl of the medium was combined with Tris-EDTA-NADH buffer and incubated at 37 °C for 10 min, followed by the addition of pyruvate solution. The absorbance was measured at 339 nm using a UV-Vis spectrophotometer. The resulting data were expressed in units per liter (U/L).
Statistical Analyses
Data analysis was carried out using SPSS software. The significance among all groups was assessed using one-way ANOVA, followed by Tukey's post-hoc test for multiple comparisons. Additionally, the Mann Whitney U test was employed to determine the statistical significance between treatments. All experiments utilized cells from the same stock simultaneously, and the experiments were replicated to validate the findings. The homogeneity test was applied to evaluate variance among each experiment, revealing no significant differences between them. Therefore, data from both experiments were pooled, and means were computed. Results are presented as mean ± standard error of the mean (SEM), with a p-value of <0.05 considered statistically significant.
Result
MTT Assay
The MTT assay revealed that treatment with DMSO did not significantly affect the spontaneous viability of HeLa cells, as shown in Figure 1. In contrast, exposure to CMDE, CMEE, and CMME led to a dose-dependent decline in cell viability, with the lowest viability observed at a concentration of 320 µg/ml. Among the three extracts, CMEE exhibited the strongest cytotoxic effect, demonstrating a three-fold higher potency compared to CMDE and CMME. These findings emphasize the varying cytotoxic potential of the extracts, with the ethyl acetate fraction displaying the highest efficacy. (Figure 1).

Dose-Dependent reduction in HeLa cell viability following treatment with Croton megalocarpus extracts and doxorubicin (MTT assay).
Figure 1 illustrates the cytotoxic effects of dichloromethane (CMDE), ethyl acetate (CMEE), and methanol (CMME) extracts of Croton megalocarpus on HeLa cell viability, assessed using the MTT assay. A dose-dependent decrease in cell viability was observed, with CMEE exhibiting the highest cytotoxicity, comparable to the positive control, doxorubicin (DOX).
Clonogenic Assay
The clonogenic assay demonstrated a dose-dependent reduction in the surviving fraction of HeLa cells following treatment with the dichloromethane (CMDE), ethyl acetate (CMEE), and methanol (CMME) extracts of C. megalocarpus. Cells treated with 20–320 μg/ml of these extracts exhibited a progressive increase in cytotoxicity, reaching its peak at 4 h post-treatment, after which the effect remained stable.
Comparatively, doxorubicin (DOX), a standard chemotherapeutic agent, also induced a significant reduction in cell survival, with the lowest survival fraction observed at 4 h post-treatment. At 80 μg/ml, CMDE, CMEE, and CMME significantly reduced the surviving fraction of HeLa cells, whereas at 12 μg/ml, DOX produced a comparable cytotoxic effect.
Among the extracts tested, CMEE exhibited the highest cytotoxic activity, resulting in an 88% reduction in colony formation at 320 μg/ml (p < 0.0001), demonstrating significantly greater cytotoxicity than CMDE and CMME (Table 1).
Clonogenic Survival of HeLa Cells Following Treatment with Croton megalocarpus Extracts.

Table 1 summarizes the plating efficiency and surviving fraction of HeLa cells following treatment with CMEE, CMDE, and CMME at different concentrations. A significant dose-dependent decline in colony formation was observed across all treatments.
Figure 2 depicts the dose-dependent decline in clonogenic survival of HeLa cells following treatment with CMEE and DOX, showing that CMEE exerts a potent cytotoxic effect, comparable to DOX at higher concentrations. The graph presents the surviving fraction of HeLa cells treated with CMEE (20-320 µg/ml) and DOX (4-20 µg/ml). Data are expressed as mean ± standard deviation from three independent experiments.

Dose-Dependent reduction in the surviving fraction of HeLa cells treated with ethyl acetate fraction of Croton megalocarpus (CMEE) and doxorubicin (DOX).
CMEE demonstrated a dose-dependent reduction in colony formation, with 320 µg/ml CMEE showing the greatest cytotoxic effect. DOX, as a positive control, also exhibited a strong inhibitory effect, further validating the results. The CMEE fraction was the most cytotoxic, justifying its selection for subsequent experiments.
These results highlight the concentration- and time-dependent cytotoxic effects of C. megalocarpus extracts on HeLa cells, with CMEE demonstrating the strongest activity. The findings suggest that these extracts, particularly CMEE, have promising anticancer potential and merit further exploration as potential therapeutic candidates for cervical cancer.
Biochemical Analyses
Biochemical analyses were conducted to assess the impact of CMEE treatment on various cellular parameters in HeLa cells. Interestingly, DMSO treatment, which served as a vehicle control, did not result in any significant alterations in the spontaneous levels of glutathione (GSH), TBARS, and lactate dehydrogenase (LDH) over the course of the assay time. This suggests that DMSO, at the concentrations used in this study, did not induce any notable changes in these cellular markers.
Glutathione
Exposure of HeLa cells to varying concentrations of CMEE resulted in a significant decrease in glutathione (GSH) levels across all post-treatment time points, with the most pronounced reduction observed at 320 µg/ml. Notably, while GSH levels exhibited a non-significant improvement over time in cells treated with different concentrations of CMEE, they reached their lowest levels at 8 h post-treatment. This decline in GSH concentration over time was consistent with the patterns observed in HeLa cells treated with doxorubicin (DOX) (Table 2). These findings suggest that CMEE treatment may lead to oxidative stress, as indicated by the depletion of this critical antioxidant.29,30
Time- and Dose-Dependent Reduction in Glutathione (GSH) Levels in HeLa Cells Treated with Croton megalocarpus Ethyl Acetate Fraction (CMEE) Over 24 Hours.
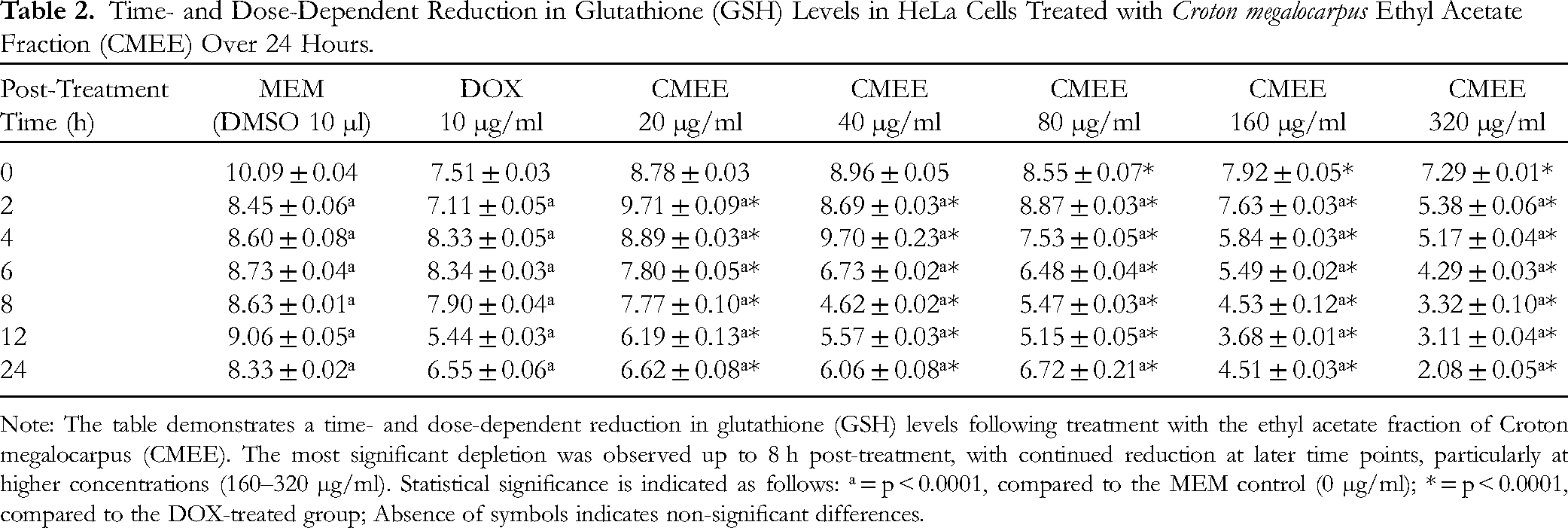
Note: The table demonstrates a time- and dose-dependent reduction in glutathione (GSH) levels following treatment with the ethyl acetate fraction of Croton megalocarpus (CMEE). The most significant depletion was observed up to 8 h post-treatment, with continued reduction at later time points, particularly at higher concentrations (160–320 µg/ml). Statistical significance is indicated as follows: ᵃ = p < 0.0001, compared to the MEM control (0 µg/ml); * = p < 0.0001, compared to the DOX-treated group; Absence of symbols indicates non-significant differences.
Lipid Peroxidation
In HeLa cells treated with varying concentrations of CMEE, lipid peroxidation levels, measured as TBARS, increased in a dose-dependent manner across all post-treatment time points. This increase was particularly significant at 160 and 320 µg/ml, where TBARS levels were markedly elevated. Additionally, a time-dependent rise in TBARS was observed, peaking at 6 h post-treatment. At 320 µg/ml CMEE, TBARS levels remained significantly higher than baseline at all time points, showing an approximately 1.5-fold increase compared to DMSO treatment and a 2-fold increase relative to DOX treatment (Figure 3).

Lipid peroxidation in HeLa cells following CMEE treatment.
Figure 3 illustrates the effect of the ethyl acetate fraction of Croton megalocarpus (CMEE) on lipid peroxidation in HeLa cells, measured as TBARS levels (nM per 106 cells) at different post-treatment time points. A dose- and time-dependent increase in TBARS levels was observed, with the highest lipid peroxidation occurring at 320 µg/ml CMEE at 6 h post-treatment. Statistical significance was determined using one-way ANOVA, and significance levels are indicated on the graph (ns = not significant, * = p ≤ 0.05, ** = p ≤ 0.01, *** = p ≤ 0.001). The results suggest that CMEE induces oxidative stress, potentially contributing to its cytotoxic effects in HeLa cells.
Lactate Dehydrogenase
Exposure of HeLa cells to various concentrations of C. megalocarpus ethyl acetate extract (CMEE) resulted in a dose-dependent elevation of lactate dehydrogenase (LDH) activity, with the most significant increase observed at 320 µg/ml (Table 3). Notably, LDH activity peaked at 2 h post-treatment, after which it gradually declined. It is important to note that the entire media was removed at each assay time point, which accounts for the lower LDH values recorded in subsequent assay periods.
Time- and Dose-Dependent Changes in LDH Release from HeLa Cells Following Treatment with Croton megalocarpus Ethyl Acetate Fraction (CMEE).
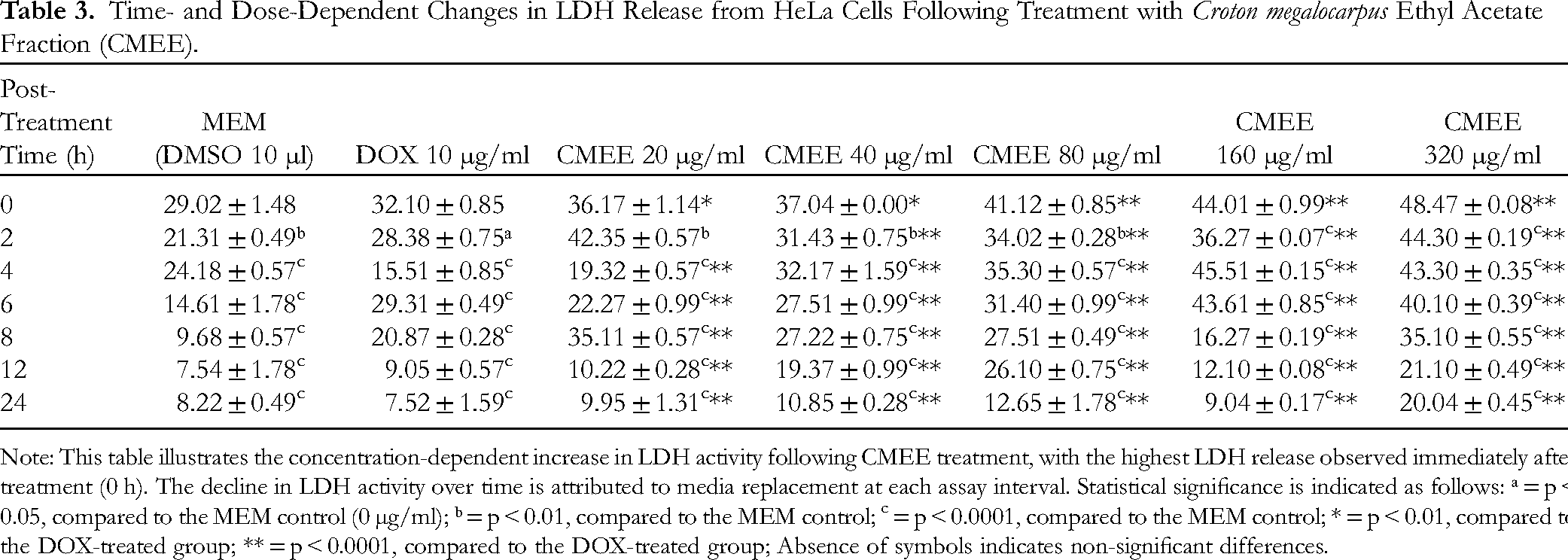
Note: This table illustrates the concentration-dependent increase in LDH activity following CMEE treatment, with the highest LDH release observed immediately after treatment (0 h). The decline in LDH activity over time is attributed to media replacement at each assay interval. Statistical significance is indicated as follows: ᵃ = p < 0.05, compared to the MEM control (0 µg/ml); ᵇ = p < 0.01, compared to the MEM control; c = p < 0.0001, compared to the MEM control; * = p < 0.01, compared to the DOX-treated group; ** = p < 0.0001, compared to the DOX-treated group; Absence of symbols indicates non-significant differences.
When comparing the effects of CMEE treatment to DMSO treatment, the release of LDH was found to be 1.3-fold higher after doxorubicin (DOX) treatment and 2-fold higher following treatment with 320 µg/ml CMEE at the 4-h mark (Table 3). These results indicate that CMEE treatment not only induces cell damage but also enhances LDH release, a marker of cellular cytotoxicity.
Discussion
The utilization of natural sources for deriving potential chemotherapeutic drugs remains a significant avenue for discovering novel molecules to combat cancer. Plants, in particular, have been a prolific source of secondary metabolites that have contributed to the development of numerous drugs for treating various human ailments. In fact, it is noteworthy that 75% of modern synthetic drugs have originated from plants. 31 Many anticancer drugs were initially obtained from plants before they underwent chemical synthesis.5,6
Key chemotherapeutic agents such as paclitaxel (derived from the Pacific yew tree, Taxus brevifolia), vincristine (derived from the Madagascar periwinkle, Catharanthus roseus), podophyllotoxins (derived from the mayapple, Podophyllum peltatum), and camptothecin (derived from the Chinese happy tree, Camptotheca acuminata), have been derived from natural sources. 4 Despite advancements in combinatorial chemistry and computer-based molecular modeling utilized by pharmaceutical industries for novel drug development, these methods may not completely replace the importance of natural products in the pursuit of new molecules for drug development.
Given the rich history of plant-derived chemotherapeutic agents and the potential of C. megalocarpus as a source of novel anticancer compounds, there is a compelling need to systematically explore the anticancer potential of C. megalocarpus stem bark extract in cultured HeLa cells. This investigation aims to contribute to the ongoing efforts in uncovering natural sources of effective anticancer agents that can be developed into new treatments for cervical cancer and potentially other types of cancer.
The dichloromethane, ethylacetate, and methanol fractions derived from C. megalocarpus stem bark were evaluated for their cytotoxic effects against HeLa cells. Our findings demonstrate that these fractions exhibited dose-dependent cytotoxicity, with the most pronounced effect observed at a concentration of 320 μg/ml after 4 h of exposure. A systematic analysis of their anticancer activity in vitro was conducted using the clonogenic assay, which assesses the ability of cells to form colonies, a hallmark of their proliferative capacity. These comprehensive in vitro studies provide valuable insights into the anticancer properties of the C. megalocarpus stem bark fractions and their potential as sources of novel therapeutic agents.
The CMEE extract exhibited a reduction in GSH concentration, along with an increase in lipid peroxidation and LDH levels in HeLa cells. Previous studies have reported similar effects with extracts of Colocasia gigantean and Helicia nilagirica, which also reduced GSH contents in HeLa cells.32,33 Elevated levels of glutathione in cancer cells are associated with therapy resistance, whereas decreased glutathione levels enhance cytotoxic effects. Conversely, increased lipid peroxidation induces oxidative stress and leads to cancer cell death.24,25 The cytotoxic effect observed with CMEE may be attributed to the elevated TBAR levels observed in our study. Furthermore, the assessment of LDH serves as an indirect measure of cytotoxicity, and CMEE treatment led to a concentration-dependent increase in LDH activity in our study. Similar increases in LDH activity have been reported following treatment with Tinospora cordifolia extract in HeLa cells.29,30
The cytotoxic effects of C. megalocarpus on HeLa cells may involve multiple potential mechanisms. One significant pathway is lipid peroxidation, a crucial event associated with cell death. Lipid peroxidation disrupts membrane function by increasing membrane permeability and oxidizing membrane proteins, ultimately leading to cell death through damage to cellular DNA. This is supported by the observed increase in TBARS and LDH levels following exposure to C. megalocarpus extract. Previous studies have directly linked cytotoxicity to elevated LDH activity.34,35 Additionally, glutathione plays a role in cellular processes such as proliferative response, cell cycle progression, and DNA damage.36,37
While our findings demonstrate the cytotoxic effects of the ethyl acetate fraction of Croton megalocarpus on HeLa cells, certain limitations should be acknowledged. The study does not explore specific mechanisms of cell death, such as apoptosis, DNA fragmentation, or caspase activation, which would provide deeper insights into the mode of action. Additionally, the results are based on in vitro experiments, and further validation in in vivo models is necessary to assess systemic effects and therapeutic relevance. The study also focuses on a single cancer cell line, and testing on other cancer types would help determine the broader applicability of the extract. Despite these limitations, the findings provide a strong foundation for further exploration of C. megalocarpus as a potential source of anticancer agents
Conclusion
This study provides evidence for the anticancer potential of the ethyl acetate fraction derived from Croton megalocarpus in HeLa cervical cancer cells. The cytotoxic effects of this fraction were demonstrated through a dose-dependent reduction in cell survival, as assessed by clonogenic assays. Furthermore, the ethyl acetate fraction was found to decrease glutathione levels, increase lipid peroxidation, and elevate lactate dehydrogenase release. The observed cytotoxicity and cellular damage may be attributed to the induction of DNA damage by the ethyl acetate fraction. This hypothesis is supported by the reduced glutathione levels, which suggest increased susceptibility to oxidative stress. Additionally, the increased lipid peroxidation and lactate dehydrogenase release point to compromised cell membrane integrity and cellular dysfunction, respectively.
These findings highlight the potential of Croton megalocarpus as a source of anticancer compounds and warrant further investigation into the specific phytochemicals responsible for the observed effects. Future studies should focus on the isolation and characterization of active compounds, as well as the elucidation of their mechanisms of action in various cancer cell lines and animal models. Ultimately, this research contributes to the growing body of knowledge on the medicinal properties of Croton megalocarpus and its potential applications in cancer therapy.
Footnotes
Acknowledgments
The authors acknowledge United States International University-Africa Internal Grant research fund for their support toward the successful completion of the research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the USIU-Africa Institutional ethics and research committee, Nairobi, Kenya.
Funding
This research was supported by an internal grant from United States International University-Africa, Nairobi, Kenya.
Statement of Human and Animal Rights
There are no human and animal subjects in this article and statement of Human and animal rights is not applicable. All of the experimental procedures were conducted in accordance with Good Laboratory Practice guideline for in vitro experiments.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable;



