Abstract
Background
The leaves and rhizomes of Melicope pteleifolia are utilized for their therapeutic properties, being applied to treat various ailments such as scabies, wounds, ulcers, sore throats, rheumatism, eczema, dermatitis, bruises, and snake bites. The goal of this work is to investigate the anti-inflammatory and xanthine oxidase inhibitory activities as well as the isolation of bioactive compounds of extracts and fractions from Melicope ptelefolia.
Methods
The dried plants were extracted exhaustedly with 96% ethanol, the solvent was evaporated, and the resulting extracts (MPE) were partitioned successively with hexane, ethyl acetate, and methanol through flash chromatography to obtain three sub-fractions (MPH, MPEA, and MPM). The macrophage cell line RAW264.7 was used to assess the anti-inflammatory activity via inhibition of secretion of mediator (NO and TNF-α). The DPPH, ABTS, RP assay, and xanthine oxidase inhibition were applied to investigate the antioxidant and reducing uric acid in vitro, respectively. Structural chemicals were identified by NMR methods.
Results
All the extracts manifested anti-inflammatory and antioxidant activities. MPEA fraction showed a better antioxidant and NO inhibitory effects than that of MPE and MPH, while MPH fraction was stronger than those of MPE, MPEA, and MPM in the TNF-α inhibition experiment. From the most active fraction (MPEA), eight compounds (1-8) were isolated and identified by column chromatography. Of which, quercetin (3), quercitrin (7), kaempferol (2), and Luteolin 7 glucoside (8) were the potential compounds for anti-inflammation and suppressing activity of xanthine oxidase.
Conclusions
Melicope ptelefolia exhibited significant anti-inflammatory, antioxidant, and xanthine oxidase inhibitory properties, showing that it was a promising source for developing medications targeting inflammation and elevated uric acid levels. Its bioactive compounds are potentially used in pharmaceuticals to treat conditions like gout and other inflammatory illnesses.
Introduction
Gout is a disease caused by a disorder of purine metabolism, of which, the main characteristic is increased uric acid in the blood. When uric acid is saturated outside the cell membrane, it will cause the deposition of monosodium urate crystals (MSU) in the tissues. Depending on which tissue the urate microcrystals accumulate, the disease manifests itself by one or more clinical symptoms such as acute and/or chronic arthritis and periarticular inflammation, commonly known as gouty arthritis.1-4
Furthermore, MSU crystals interact with immune cells to induce the secretion of inflammatory mediators such as TNF-α, interleukin-1 (IL-1), IL-6, IL-8, and free radicals. resulting in tissue and cell damage in the body. 5 In addition, hyperuricemia causes oxidative stress, and oxidative stress exacerbates inflammation and disrupts endothelial function. 6 The disease is mainly related to excess nutrition. According to AnneKathrin Tausche (2009), at least 1% to 2% of adults in developed countries are affected, increasing in developing countries. 7 Suppose the disease is not diagnosed early and treated promptly. In that case, it will progress more and more severely, resulting in joint destruction, affecting the patient's quality of life, and being a burden to the family and society. 8
Currently, there have been many research results on gout treatment drugs, but most of them are chemical drugs. This can cause side effects related to long-term use of drugs. In particular, allopurinol and febuxostat are common drugs used in treatment but also cause many side effects such as abnormal liver function, diarrhea, nausea, headache, rash and edema. 9 On the other hand, the drugs only have the effect of preventing gout attacks but do not cure the disease. The main treatment method today is lifestyle changes combined with the use of drugs to reduce blood uric acid, treat concomitant diseases and control side effects of drugs. 9 Therefore, identifying new targeted drugs for treating gout is extremely important. Especially, plant bases-drugs with high treatment efficacy, easy to use and few side effects on patients, are increasingly interested, focusing on reducing uric acid levels in the blood, reducing inflammation symptoms and eliminating harmful free radicals.
Melicope pteleifolia (MP) is widely distributed across Vietnam, prevalent in northern and southern regions. This plant typically inhabits hilly terrains, secondary forests, sparse woodlands, and shrublands within delta areas. In traditional Vietnamese medicine, the leaves and rhizomes of Melicope pteleifolia are utilized for their therapeutic properties, being applied to treat various ailments such as scabies, wounds, ulcers, sore throats, rheumatism, eczema, dermatitis, bruises, and snake bites.10,11 Chemical investigations have demonstrated that the leaves of MP are abundant in flavonoid compounds, including formononetin, daidzein, oroxylin A, wogonin, 5,7-dihydroxy-3,4′-dimethoxyflavone, and kaempferol 3-O-β-D-glucopyranosyl (1→2)-α-D-xylopyranoside, as well as 3,7-dimethylkaempferol.12,13 Additionally, various alkaloids are found in the plant's stems.14,15 Biological studies have demonstrated that crude extracts of MP possessed a variety of bioactivities, including anti-inflammatory,16,17 antibacterial, 17 anti-tumor,18,19 and analgesic effects. 20 The broad spectrum of biological activities highlights the potential of MP as a valuable source for the development of therapeutic agents. This study proposes to assess the therapeutic potential of MP leaves in treating gout by focusing on three main areas: the inhibition of the xanthine oxidase enzyme, anti-inflammatory effects, and antioxidant properties. Given that there have been no prior studies evaluating the effectiveness of MP leaves in these areas, our research aims to establish a foundation for its use in reducing blood uric acid levels, which is crucial in managing gout. Through this evaluation, the study hopes to provide insights and guidelines for incorporating MP as a potential treatment option for individuals suffering from gout.
Results
Antioxidant Activity
Total Polyphenol and Flavonoid Contents
The early stages of gout are known to be significantly impacted by reactive oxygen species (ROS), which include free radicals like superoxide, hydroxyl radicals, and hydroperoxide molecules.21-24 Both the phagocytosis of monosodium urate (MSU) by macrophages and enzymatic reactions catalyzed by xanthine oxidase generate these ROS. 25 These free radicals not only make genetic material errors more likely, but they also interfere with DNA repair, changing the structure of chromosomes and raising the risk of a number of diseases.26,27 Using antioxidants could be a useful strategy to fight aging and reduce the risk of disease. The evaluation of antioxidant activity may be the starting point for upcoming biological screening studies. The MPE extract and its three fractions (MPH, MPEA, and MPM) were quantified for total flavonoids and polyphenols. As shown in Table 1, the ethyl acetate (MPEA) and methanol (MPM) fractions exhibited the highest TPC, at 115.76 ± 0.53 µg/g and 116.58 ± 3.16 µg/g, respectively. The ethanol extract showed a moderate average level of 68.52 ± 0.80 µg GAE/g DM, while the MPH fraction yielded the lowest total polyphenol content at 47.64 ± 1.32 µg/g. Due to the polarity of phenolic compounds, their solubility in n-hexane is limited. The superior TPC results in the MPM and MPEA fractions indicate that ethyl acetate and methanol are the most effective solvents for extracting phenolic compounds from MP leaves. As shown in Table 1, TFC values decreased in the following order: MPEA (79.58 ± 0.77 µg/g), MPM (59.14 ± 3.20 µg/g), MPE (37.42 ± 0.29 µg/g), and MPH (20.59 ± 0.25 µg/g). Both the TPC and TFC results indicated that the MPM and MPEA fractions had the highest flavonoid content, followed by the MPE fraction, with the MPH fraction exhibiting the lowest levels. Interestingly, while the TPC results for MPEA and MPM were comparable (P > .05), the TFC results demonstrated that the MPEA fraction had significantly higher flavonoid content than the MPM fraction (P < .05). These findings suggest that ethyl acetate is superior to methanol in terms of solubility and selectivity for flavonoid compounds within polyphenols.
Total Polyphenol (TPC) and Flavonoid (TFC) Content.

a,b,c,dFor a particular variable, means on the same column with different superscripts within the specified class variable are significantly (P < .05, Tukey test) different. Values indicate mean ± SE, n = 3.
Antioxidant Activity
All MP extracts and fractions demonstrated antioxidant effects based on the evaluated IC50 or OD0.5 values in standard assays. Antioxidant assays included DPPH, ABTS, reducing power (RP), conducted on MPE, MPH, MPEA, and MPM. The IC50 and OD0.5 values were used to compare antioxidant activity, as noted by Piaru 28 indicating that lower values reflect stronger activity. As shown in Table 2, the results highlight key findings of the study.
Antioxidant Activity of MP Extract and its Fractions via DPPH, ABTS, and Reducing Power.

For a particular variable, means on the same column with different superscripts within the specified class variable are significantly (P < .05, Tukey test) different. Values indicate mean ± SE, n = 3.
In three antioxidant assays, MPEA was the most active fraction, with the lowest IC50 value in each method compared to others (P < .05). Interestingly, in the ABTS method, the MPH showed the strongest inhibitory activity compared to other fractions (P < .05), although the TPC and TFC of MPH were the smallest. The higher concentration of coumarin-like substances in the hexane fraction of Melicope ptelefolia may explain this. 29 Coumarin cannot react with the Folin-Ciocalteu reagent because it lacks the necessary functional groups. This reagent primarily detects phenolic compounds, which typically have hydroxyl (-OH) groups that can undergo oxidation. Although coumarin has some phenolic characteristics, it does not possess enough free hydroxyl groups or the appropriate structure to enable the redox reaction for detection, resulting in no measurable change when treated with the reagent. However, coumarin can neutralize the ABTS radical by donating an electron or hydrogen atom.
Anti-Inflammatory Activity
The crude ethanol extract (MPE) together with n-hexane (MPH), ethyl acetate (MPEA), and methanol (MPM) fractions were assessed for the inhibition of nitric oxide and TNF-α secretion in LPS-activated RAW 264.7 cells. The pro-inflammatory mediators such as nitric oxide (NO) and TNF-α were used as biomarkers of inflammation. The suppression of NO and/or TNF-α production indicated anti-inflammatory activity. Therefore, we examined the effects of extracts on LPS-induced RAW264.7. First, the effect on cell viability was determined by MTT assay. The results are shown in Figure 1. The experimental results collectively indicated that the cells were safe at all extract concentrations below 100 µg/mL, as well as with Dexamethasone at a concentration of 100 nM. Therefore, we selected all extracts below 100 µg/mL and Dexamethasone at 100 nM for the subsequent in vitro nitric oxide and TNF-α secretion assay.

Effect of MP extract on RAW 264.7 macrophage cell viability. Cells were incubated for 24 h with the indicated concentrations (100, 75, 50, 25 μg/mL). Data are expressed as mean ± SD of three independent experiments. MPE: ethanol crude extract; MPH: n-hexane fraction; MPEA: ethyl acetate fraction; MPM: methanol fraction.
This paper investigated the anti-inflammatory effects of various extracts by measuring nitric oxide and TNF-α production in LPS-activated cells. RAW 264.7 macrophage cells were stimulated with LPS at a concentration of 1 µg/mL, resulting in a non-significant increase in cell viability compared to untreated cells. As a result, LPS at 1 µg/mL was employed to induce inflammation in the experiments. The macrophage cells were co-cultured with different known doses of extracts while inflammation was activated with LPS. As illustrated in Figure 2 and Figure 3 the results showed that nitric oxide production in LPS-activated cells was significantly reduced in a dose-dependent manner by the MPE, MPH, and MPEA fractions compared to LPS-stimulated cells (P < .05). Specifically, the MPEA fractions at a concentration of 100 µg/mL effectively inhibited nitric oxide production (Figure 2). Similar results were observed regarding the inhibitory effects of the extracts on TNF-α production. Compared to the LPS-stimulated RAW264.7, all extracts inhibited the production of the cytokine TNF-α in a dose-dependent manner. Among them, the MPH fraction exhibited the strongest inhibitory effect, followed by the MPEA, MPE, and MPM fractions (Figure 3). At a concentration of 100ug/ml, MPH and MPEA inhibited 74.45 and 63.34% of NO secretion, respectively, compared with the LPS-stimulated cell (P < .05). These findings indicate that the ethyl acetate and hexane fractions are promising candidates for inhibiting inflammation.
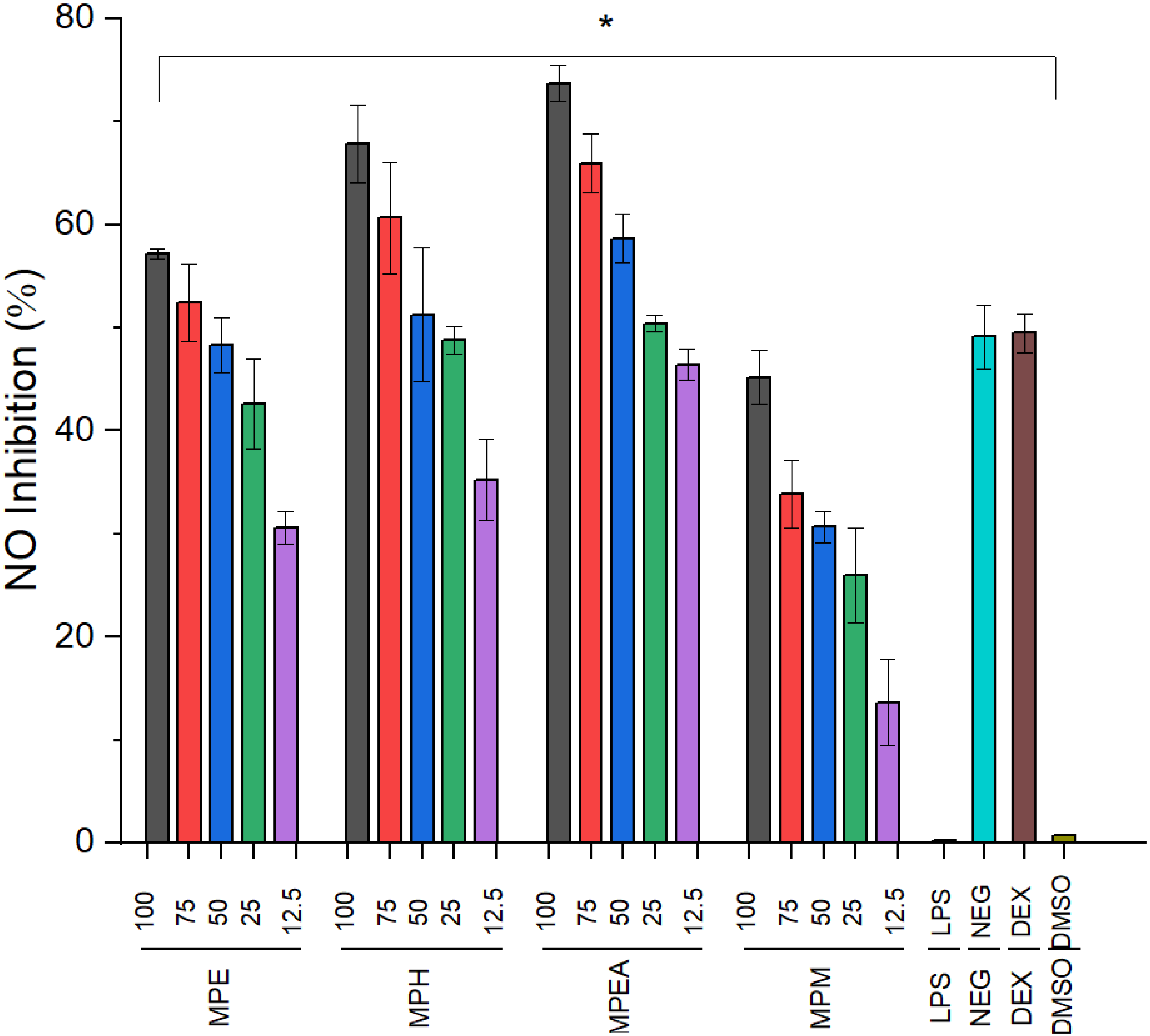
NO production in LPS-induced RAW264.7. NEG: untreated cell group; DEX: dexamethasone group; LPS: LPS-activated group. Data are expressed as mean ± SD of three independent experiments. * P < .05 compared to LPS. MPE: ethanol crude extract; MPH: n-hexane fraction; MPEA: ethyl acetate fraction; MPM: methanol fraction.

The secretion of TNF-α in LPS-induced RAW 264.7 macrophages. NEG: untreated cell group; DEX: dexamethasone group; LPS: LPS-activated group. Data are expressed as mean ± SD of three independent experiments. * P < .05 compared to LPS. MPE: ethanol crude extract; MPH: n-hexane fraction; MPEA: ethyl acetate fraction; MPM: methanol fraction.
Xanthine Oxidase Inhibition
When individuals suffer from gout, they experience oxidative stress and inflammation due to excessive uric acid production.25,30,31 This condition is primarily caused by the activity of xanthine oxidase. 31 Inhibiting this enzyme can help regulate uric acid levels in the blood, thereby improving symptoms associated with gout. 32
Figure 4 illustrates the inhibitory effects of MP extract on xanthine oxidase (XO), measured by their half-maximal inhibitory concentration (IC50) in comparison to allopurinol, the positive control. The results show that all MP extracts displayed a significant inhibitory effect on XO activity. The inhibition order is as follows: allopurinol < MPEA < MPM < MPE < MPH. This pattern resembled the TPC or TFC profiles.
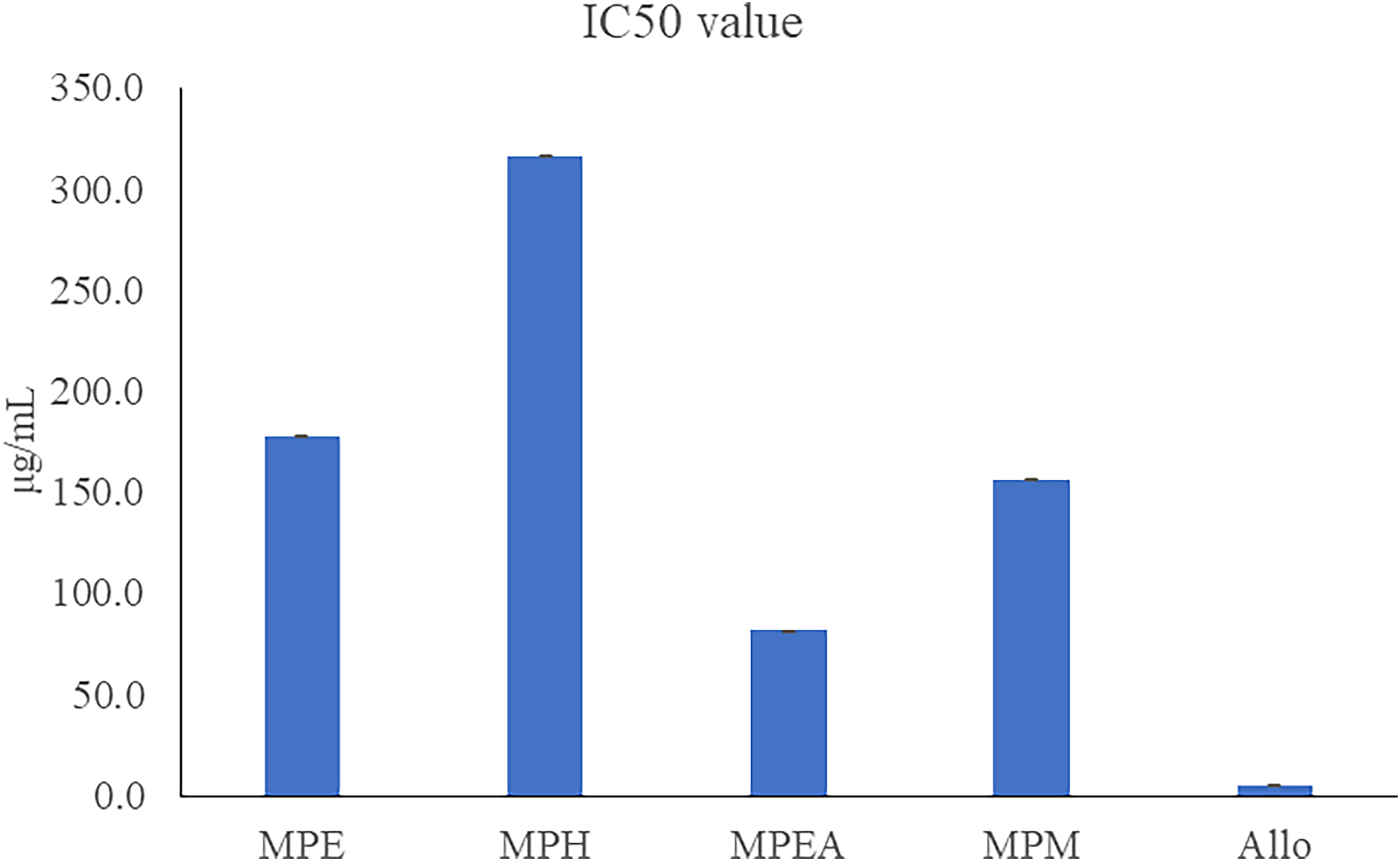
Xanthine oxidase inhibition. MPE: ethanol crude extract; MPH: n-hexane fraction; MPEA: ethyl acetate fraction; MPM: methanol fraction; Allo: Allopurinol.
Compounds that inhibit xanthine oxidase (XO) are widely used to treat gout by lowering plasma uric acid levels.31,33,34 The XO generates oxygen-derived free radicals, contributing to oxidative damage and various inflammatory conditions, including hepatitis, blood ischemia, and aging. 31 Currently, allopurinol is the only clinically approved XO inhibitor for gout treatment,35,36 but it has several side effects, such as hepatitis, kidney disease, and allergic reactions. 35 Therefore, there is an urgent need for new XO inhibitors that provide greater therapeutic benefits and fewer side effects for gout and other oxidative stress-related diseases that damage cells and tissues.37-39 Based on the xanthine oxidative assay of the extracts and their fractions, it is reasonable to assume that MPEA contains XO inhibitors. Consequently, the isolation was conducted on the MPEA fractions.
Isolation of XO Inhibitors
The ethyl acetate fraction showed the highest potential in biological activity evaluation, leading to the isolation of 8 compounds through column chromatography. NMR spectral analysis and published literature helped identify these substances as p-O-geranyl coumaric acid (1), 40 Kaempferol (2), 41 Quercetin (3), 41 Daidzein (4), 42 Daidzein-4′-O-Glucoside (5), 43 Formononetin-7-O-Glucoside (6), 44 Quercitrin (7), 45 and Luteolin 7-O- glucoside (8) 44 (Figure 5).

Chemical structure of isolated compounds.
The results of the antioxidant, anti-inflammatory, and XO inhibitory activities of the isolated compounds are summarized in Table 3. As shown in Table 3, quercetin (
Antioxidant, Anti-Inflammation and Anti-Xanthine Oxidase of Isolated Compounds.
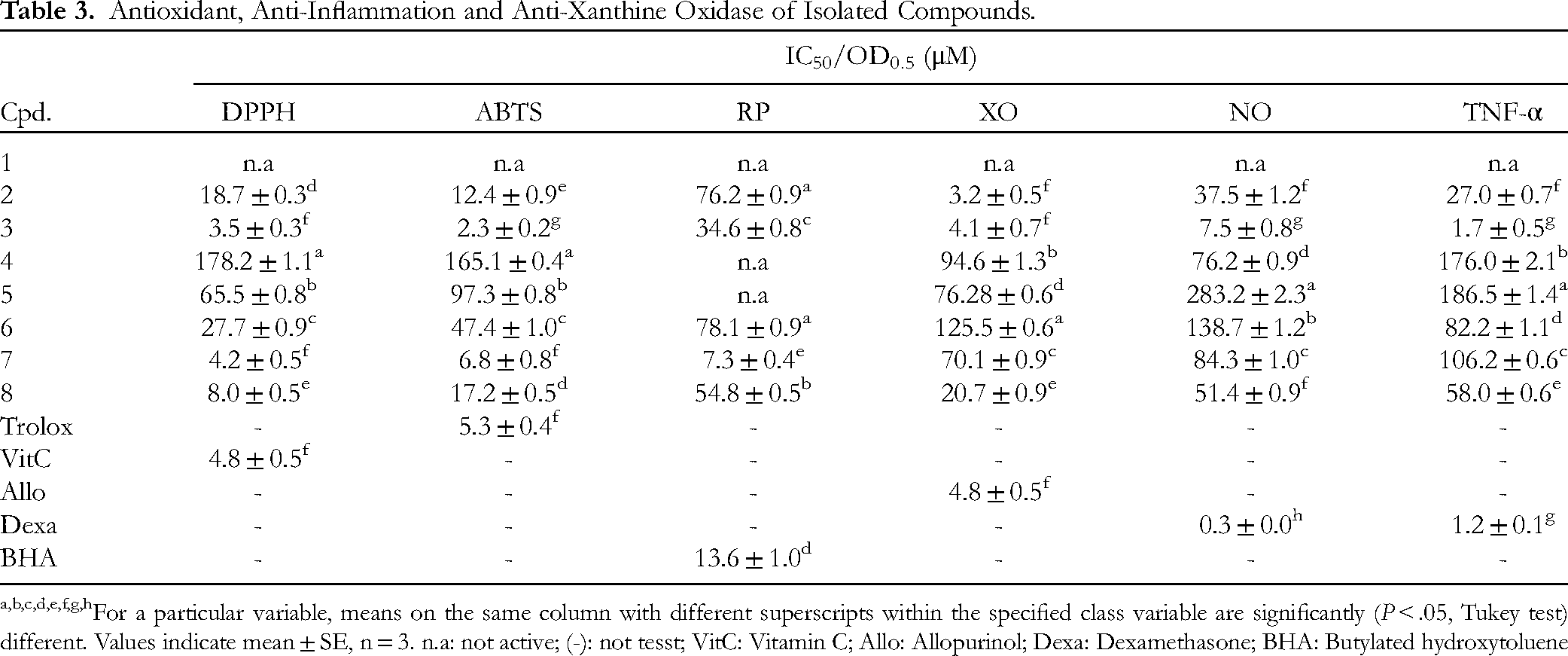
For a particular variable, means on the same column with different superscripts within the specified class variable are significantly (P < .05, Tukey test) different. Values indicate mean ± SE, n = 3. n.a: not active; (-): not tesst; VitC: Vitamin C; Allo: Allopurinol; Dexa: Dexamethasone; BHA: Butylated hydroxytoluene
Discussion
Existing anti-inflammatory and antimicrobial medications often come with numerous side effects. As a rich natural resource, plants offer significant advantages in searching and developing new, more effective drugs. Elevated blood uric acid levels lead to the formation of monosodium urate (MSU) crystals in the joints and surrounding tissues. The accumulation of MSU crystals activates the NLRP3 receptor, a member of the NOD-like receptor family, leading to the activation of the inflammasome. This process triggers the release of proinflammatory cytokines, including IL-8, IL-6, interferon-gamma (IFN-γ), and interleukin-1 beta (IL-1β).30,46
The presence of MSU crystals has several critical effects, primarily through the generation of reactive oxygen species (ROS) and the further activation of NLRP3.47,48 Specifically, MSU crystals stimulate the activities of NADPH oxidase, xanthine oxidase, and nitric oxide synthase, resulting in the production of significant amounts of ROS. These reactive species typically include hydrogen peroxide, superoxide anion, and nitric oxide (NO). Notably, nitric oxide can react with superoxide to form peroxynitrite (ONOO-), a reactive nitrogen species with a relatively long half-life. Peroxynitrite has been implicated in several detrimental effects, such as increasing apoptosis (programmed cell death), degenerating connective tissues, and contributing to joint damage.49,50 A previous study found that endogenous ROS are overproduced during acute gout attacks, suggesting that oxidative stress plays a significant role in the inflammatory processes associated with the presence of MSU crystals. 50
Therefore, studying antioxidants and anti-inflammation in gout is important because gout is a painful type of arthritis caused by high levels of uric acid in the blood. Antioxidants can help reduce oxidative stress in the body, which might lessen inflammation and pain. By focusing on these areas, researchers hope to find better ways to manage gout symptoms and improve patients’ quality of life.
Melicope pteleifolia demonstrated promising biological activities, particularly in inhibiting inflammatory mediators and xanthine oxidase, which are critical in managing hyperuricemia and gout. Several phytoconstituents from different plant species have been investigated for their ability to reduce uric acid levels and alleviate inflammation associated with gout. Among them, flavonoids, which were identified in M. pteleifolia, have been extensively studied for their xanthine oxidase inhibitory and anti-inflammatory properties. Quercetin and kaempferol, two key flavonoids found in this study, have previously been reported as potent inhibitors of xanthine oxidase and inflammatory mediators, in particular, has been shown to significantly reduce uric acid levels by inhibiting xanthine oxidase activity in hyperuricemic models,34,51,52 quercetin and kaempferol may modulate NLRP3 inflammasome activation, NF-κB signaling, and oxidative stress, key pathways in gout inflammation. Their ability to reduce IL-1β, IL-6, and TNF-α secretion suggests broader anti-inflammatory effects.49,53 Luteolin and its glycosides, also identified in M. pteleifolia, have demonstrated potent anti-inflammatory and antioxidant effects, which contribute to their potential in gout treatment.54,55 Other plants with known anti-gout activity include Terminalia bellerica, 56 Morus alba, 57 and Orthosiphon stamineus, 58 which have shown inhibitory effects on xanthine oxidase and inflammatory cytokines. In addition to flavonoids, alkaloids from plants have also shown promising anti-gout properties. Alkaloids such as berberine (from Berberis species) and colchicine (from Colchicum autumnale) have been investigated for their ability to regulate inflammation and uric acid metabolism.59-61 Coumarins, which are also present in M. pteleifolia have been linked to xanthine oxidase inhibition and anti-inflammatory pathways, suggesting their potential for further exploration in gout therapy.62,63 The findings from M. pteleifolia in this study alise previous reports, reinforcing the role of polyphenols and flavonoids as key bioactive compounds in managing hyperuricemia and inflammation. However, the therapeutic efficacy of M. pteleifolia compared to these other plant-based treatments remains to be systematically evaluated through direct comparative studies.
The xanthine oxidase inhibitory, anti-inflammatory, and antioxidant activities of the eight isolated compounds are summarized in Table 3, with allopurinol, dexamethasone, Vitamin C, BHA, and Trolox as positive controls. For XO inhibition, statistical analysis showed that kaempferol (IC50 of 3.2 µM), quercetin (IC50 of 4.1 µM), and allopurinol (IC50 of 4.8 µM) had no statistically significant differences (P > .05). This indicates that kaempferol and quercetin exhibited XO inhibitory activity comparable to allopurinol. In contrast, quercitrin (IC50 of 70.7 µM) and luteolin-7-glucoside (IC50 of 20.7 µM) exhibited significantly weaker XO inhibition compared to allopurinol (P < .05). p-O-geranyl coumaric acid did not exhibit measurable XO inhibition (IC50 > 500 µM) confirming its lack of activity. For NO inhibition, quercetin (IC50 = 7.5 µM) and kaempferol (IC50 = 37.5 µM) both exhibited significantly weaker inhibition than dexamethasone (IC50 = 0.3 µM, P < .05). However, quercetin showed significantly stronger NO inhibition than kaempferol (P < .05). Quercitrin and luteolin-7-glucoside exhibited moderate NO inhibition, while p-O-geranyl coumaric acid showed no significant inhibition compared to the positive control (P < .05). For TNF-α inhibition, quercetin (IC50 = 1.7 µM) showed no statistically significant difference from dexamethasone (IC50 = 1.20 µM, P > .05), indicating comparable potency. Kaempferol, quercitrin, and luteolin-7-glucoside exhibited significantly weaker TNF-α inhibition than dexamethasone (P < .05), while p-O-geranyl coumaric acid showed no activity. In DPPH radical scavenging activity, quercetin (IC50 = 3.5 µM), and quercitrin (IC50 = 4.2 µM) showed no significant difference compared to Vitamin C (IC50 = 4.8 µM) indicating comparable antioxidant activity. Two compounds Kaempferol (IC50 = 18.7 µM) and luteolin-7-glucoside (IC50 = 8.0 µM) exhibited significantly weaker activity than Vitamin C (P < .05). For ABTS radical scavenging activity and reducing power, quercetin (IC50 = 2.3; 34.6 µM, respectively) showed no significant difference from positive control (P > .05), indicating comparable antioxidant efficacy.
This study's results are consistent with previous publications on the anti-inflammatory and antioxidant effects of MP extracts or isolated compounds by inhibiting inflammatory mediators such as NO 64 or the pro-inflammatory cytokines such as TNF-α, 17 and scavenging free radicals such as H2O2, superoxide anion, hydroxyl free radicals, ABTS, and DPPH.65,66 This study provides additional evidence on the xanthine oxidase inhibitory and anti-inflammatory activities of quercetin, kaempferol, luteolin, and daidzein, which have been previously reported in the literature.
While this study focused on the individual activities of the isolated compounds, potential synergistic or antagonistic interactions remain an important consideration. Quercetin and kaempferol, which exhibited comparable xanthine oxidase inhibition, may act synergistically, enhancing enzyme inhibition through complementary binding. Similarly, their combined anti-inflammatory effects on NO and TNF-α suppression could be greater than individual activities, as previously reported for flavonoid mixtures.51,53,67 However, antagonistic interactions may also occur, particularly at high concentrations, where competition for enzyme binding sites or cellular transporters can reduce efficacy. Some flavonoids may also act as pro-oxidants at high doses, leading to unintended oxidative stress. 68 Future studies should investigate combinatorial effects using enzyme inhibition and cellular assays to determine whether these compounds exhibit synergy, additivity, or antagonism, providing a clearer picture of their therapeutic potential.
Limitations of the Current Study
Although this study provides valuable insights into the anti-inflammatory, antioxidant, and xanthine oxidase inhibitory effects of Melicope ptelefolia, there are several limitations that should be considered. First, the findings are based on in vitro experiments, which do not fully reflect how the compounds behave in a living system. Factors such as absorption, metabolism, and long-term effects remain unknown. To confirm the therapeutic potential of Melicope ptelefolia, further in vivo studies and clinical trials are needed. Second, while key compounds such as quercetin, quercitrin, kaempferol, and luteolin-7-glucoside were identified as bioactive components, their exact mechanisms of action have not been fully explored. More research is required to understand how these compounds interact with enzymes, inflammatory mediators, and uric acid metabolism at the molecular level such as inflammatory markers in gout are IL-6, IL-8, IFN-γ, NLRP3 receptor, IL-1β. Third, this study focused on isolating individual compounds, but natural plant extracts often work through synergistic effects, where multiple compounds interact to enhance or reduce overall activity. Future research should investigate whether the combined effects of different components are greater or different from those of individual compounds. Finally, safety and toxicity assessments were not conducted. Even though plant-based treatments are generally considered safe, some bioactive compounds can have unwanted side effects at high doses. Detailed toxicity studies will be essential to ensure the safe use of Melicope ptelefolia-derived treatments. In conclusion, while this study provides promising evidence for the potential of Melicope ptelefolia in managing inflammation and hyperuricemia, further in vivo studies, mechanism-based research, synergy investigations, and safety evaluations are necessary before clinical application can be considered.
Conclusion
This study confirms that Melicope ptelefolia extracts and isolated flavonoids, particularly kaempferol and quercetin, exhibit potent xanthine oxidase inhibitory, anti-inflammatory, and antioxidant activities, with kaempferol showing greater efficacy than allopurinol (IC50 of 3.2 µM vs 4.8 µM, P < .05). These compounds significantly reduced NO and TNF-α levels, suggesting their role in modulating gout-related inflammation beyond xanthine oxidase inhibition. While these findings highlight the therapeutic potential of M. ptelefolia, further mechanistic studies, in vivo validation, and toxicity assessments are essential to confirm its safety and efficacy. Future research should also compare its effects with clinically used drugs such as prednisolone and febuxostat, paving the way for its potential application as a natural alternative or complementary treatment for gout.
Material and Methods
General Procudure
NMR data were obtained on an NMR spectrometer (Bruker AM500, Germany); the internal standard is TMS, and the solvent is deuterated methanol and dimethyl sulfoxide. The ESI-MS spectrum was conducted on mass spectrometer equipment (MicroOTOF-Q, Brokers). Thin layer chromatography was deployed on an F254 silica gel plate (1.05554.0001, Merck) and a reversed-phase silica gel plate (1.15685.0001, Merck). The bands of the TLC plate were detected under ultraviolet illumination at 254 nm or sprayed with a solution of H2SO4 10% in ethanol. Column chromatography technique was accomplished using normal phase silica gel (Merck) and octadecyl silica gel (Merck). The size exclusion chromatography was performed on a glass column with Sephadex LH- 20 (GE Healthcare) as the carrier.
Chemicals
Solvents: distilled water, methanol, ethanol, n-hexane, ethyl acetate, chloroform (Chemsol, Vietnam); DPPH (2,2-diphenyl-1-picrylhydrazyl) (Wako, Japan); Gallic acid, Folin-Ciocalteu reagent, Na2CO3, Quercetin, NaNO2, AlCl3, NaOH, ABTS (diamonium-2,2′-azino-bis(3-ethylbenzthiazoline-6-sulphonate)), K2S2O8, Vitamin C, K3Fe(CN)6, CCl3COOH, FeCl3, acid acetic, NaH2PO4, Na2HPO4 (Merck, Germany), xanthine oxidase (Sigma), xanthine (Sigma), allopurinol (AK Sci), dimethyl sulfoxide (Merck). RPMI medium, 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide, Griess reagent, fetal bovine serum and lipopolysaccharide, Dexamethasone were from Sigma-Aldrich Co.
Plant Materials
The Melicope pteleifolia leaves were collected in October 2022 at Binh Chau Nature Reserve, Ba Ria Vung Tau province, VietNam. The sample was identified by Dr Dang Van Son, Institute of Tropical Biology (VAST). The specimen (Binh-10.2022) was stored at the Department of Biopharmaceutical Materials, Institute of Applied Materials Science (VAST).
Extraction
M. pteleifolia leaves (2 000 g) were extracted using 96% ethanol (3 × 10 L, for 24 h each). The combined extract was concentrated under reduced pressure using a Heidolph evaporator, yielding a semi-solid residue (MPE, 248.3 g). This residue was then adsorbed onto normal silica gel and loaded into a glass column, with silica gel serving as the adsorbent. The fractions were obtained by eluting with solvents of increasing polarity: n-hexane, ethyl acetate, and methanol. The obtained solvents were subsequently removed under reduced pressure, resulting in the corresponding fractions: MPH (hexane fraction), MPEA (ethyl acetate fraction), and MPM (methanol fraction). All were stored in the refrigerator at 4 °C until use.
Antioxidant Analysis
Total Polyphenol Content (TPC) and Total Flavonoid Content (TFC)
The overall concentration of polyphenols 69 and flavonoids 70 was evaluated utilizing established methodologies with minor adjustments.
DPPH Radical Scavenging Activity
2 mL of a 10−4 M DPPH solution in methanol was mixed with one-milliliter aliquots of each extract at four specified concentrations. The resulting mixtures were stored in darkness at room temperature for 30 min to allow for a complete reaction. Absorbance measurements were subsequently acquired at 517 nm using a UV-30 spectrophotometer. A methanol solution was utilized as the blank control for this experiment. 71
ABTS Radical Scavenging Activity
ABTS·+ cation radical was generated by combining 2 mL of a ABTS solution (7 mM) with 2 mL of a K2S2O8 solution (2.45 mM). The mixture was then incubated in the dark for approximately 16 h. Following incubation, the mixture was diluted with methanol to achieve an absorbance of 0.7000 at 734 nm. Subsequently, 100 μL of the extract was added to 5 mL of the diluted ABTS·+ solution, and the absorbance was measured at 734 nm after a 6-min reaction period. Methanol served as the blank control for this procedure. 72
Reducing Power (RP)
The reducing power assay was performed as follows: 1.0 mL of the sample (extracts/preference) at various concentrations in methanol was combined with 2.5 mL of phosphate buffer (pH 6.6) and 2.5 mL of 1% potassium ferricyanide. The mixture was then incubated at 50 °C in a water bath for 20 min. After cooling, 2.5 mL of 10% trichloroacetic acid was added, and the mixture was centrifuged at 3000 rpm for 10 min. Subsequently, 2.5 mL of the upper layer was mixed with 2.5 mL of distilled water and 0.5 mL of 0.1% ferric chloride solution. The absorbance of the resulting mixture was measured at 700 nm using a UV spectrometer. A blank was prepared without the addition of the extract. Ascorbic acid was used as the standard. 73
The concentrations of test compounds/extracts used in the antioxidant assays were shown in Table S1-S6.
Inflammatory Activity
Cell Culture
The mouse macrophage cell line RAW264.7 was obtained from ATCC. The cells were grown in RPMI containing 10% FBS in a humidified incubator with 5% CO2 at 37 °C.
Cell Viability
Determination of cell viability was measured using the MTT experiment.
NO and TNF-α Production Measurement
The cells were seeded into 96-well plates at a density of 2 × 104 cells/well/100 μL and incubated for 24 h. Then, treating the cells with the extracts in 1 h and activated by LPS (1 µg/mL). The NO level in the culture supernatant was determined using the Griess kit according to the manufacturer's instructions. The contents of TNF-α in cell culture supernatants were determined by ELISA kit according to the manufacturer's instructions as described previously. 74
Xanthine Oxidase Inhibition
The inhibition of xanthine oxidase was examined following the procedure detailed by Zhang et al. 75 In summary, the assay was performed using a 96-well plate, where each well was assigned a specific role, including a blank (phosphate buffer), a positive control (Allopurinol), and test sample wells (extracts/compounds). Each well contained 50 μl of phosphate buffer adjusted to pH 7.5. In the wells designated for the positive control, 50 μl of Allopurinol solution was added, whereas the sample wells received 50 μl of the corresponding extract/compound. Following this, 30 μl of enzyme solution (0.01 U/ml), prepared in the same phosphate buffer immediately before use, was added to each well. The reaction mixtures were incubated at 25 °C for 15 min to facilitate enzyme-substrate interactions. Subsequently, 60 μl of 150 μM xanthine was added to each well, and the mixtures were incubated again at 25 °C for an additional 30 min. The reaction was terminated by adding 25 μl of 1N hydrochloric acid (HCl) to each well.
Optical density was assessed at 290 nm using a microplate reader to determine the uric production of the reaction. All experiments were performed in triplicate for each concentration. The percentage of inhibition (I) of xanthine oxidase by the sample at the specified concentration was calculated utilizing the following formula:
As: the absorbance of the sample containing the inhibitor or extract.
Isolation of Pure Compounds
The MPEA fraction underwent normal phase silica gel column chromatography with an increasing polarity mixture of hexane and acetone (15:1, 10:1, 8:1, 5:1, 5:3, 1:1, v/v) and 100% acetone as eluent, yielding 10 fractions (MPEA.1-10). The MPEA3 sub-fraction was further processed with silica gel chromatography using a hexane-ethyl acetate-(1% acetic acid) methanol solvent system (10:1:0.1-8:1:0.1-6:1:0.1-5:1:0.2-5:1:0.5, v/v/v) to produce 6 fractions (MPEA3.1-6). Fraction MPEA3.2 was re-chromatographed on silica gel with a hexane-ethyl acetate-(1% acetic acid) methanol mixture (10:1:0.2-10:5:0.2, v/v/v) resulting in compounds 1 (82.3 mg) and 2 (12.6 mg). MPEA3.4 was further chromatographed on silica gel using hexane-ethyl acetate-(1% acetic acid) methanol (10:1:0.2-10:5:0.5, v/v/v), yielding 7 sub-fractions (MPEA3.4.1-7). Fraction MPEA3.4.4 was re-chromatographed on a silica gel column and eluted with a hexane-ethyl acetate-acetone mixture (10:1:0.5, v/v/v) to obtain compound
Fraction MPEA8 underwent chromatography on silica gel, eluted with a Chloroform-methanol-water mixture (20:1:0-8:2:0.2-20:6:1, v/v/v) to give 9 sub-fractions (MPEA8.1-9). MPEA8.5 was purified on a Sephadex LH20 column using MeOH-CHCl3 (3:1, v/v) as eluent to yield compound
Statistical Analysis
Data from distinct investigations were represented as mean ± SD and analyzed using the Student's t-test with ANOVA for multiple comparisons. Significant differences were defined as P-values < .05.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251328859 - Supplemental material for Anti-inflammatory and Xanthine Oxidase Inhibitor of Melicope Ptelefolia Leaves - A Promising Source of Gout Treatment
Supplemental material, sj-doc-1-npx-10.1177_1934578X251328859 for Anti-inflammatory and Xanthine Oxidase Inhibitor of Melicope Ptelefolia Leaves - A Promising Source of Gout Treatment by Le Xuan Binh, An Nguyen Minh Tran and Dung Tien Le in Natural Product Communications
Footnotes
Acknowledgments
This project is partially supported by the Vietnam Academy of Science and Technology (VAST) under grant number VAST04.02/23-24. The authors thank the Institute of Applied Materials Science, Vietnam Academy of Science and Technology for allowing the use of the equipment in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent
The authors have read the final manuscript and approved of the submission.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
