Abstract
Neuronal pyroptosis is one of the crucial pathogenesis of neurodegenerative diseases, and the signaling pathway mediated by inflammasome is the main pathway of pyroptosis. Neuroinflammation is not only a common feature, but also an essential basis for the diagnosis of neurodegenerative diseases. Neuroinflammation is an important pathological feature of common nervous system diseases such as Alzheimer's disease (AD), and Parkinson's disease (PD). NLRP1 inflammasome plays an important role in activating pyroptosis and inducing inflammatory response, so the development of drugs targeting the regulation of NLRP1 inflammasome has become a reasonable research direction for the treatment of nervous system diseases. By analyzing the current research progress of the NLRP1 inflammasome which affects the process of neuronal pyroptosis and the development of related Chinese medicine, natural products were docked with NLRP1 targets to find higher activity. The natural products and NLRP1 target were docked by CB-Dock molecular docking platform. Molecular docking results showed that silibinin, crocin, and hyperoside had excellent binding affinity, and which were the most promising potential active compounds for the treatment of AD by regulating NLRP1. This paper discusses the feasibility and rationality of the NLRP1 inflammasome as an important target for Chinese medicine research on neurodegenerative diseases in the future.
Introduction
Neurodegenerative diseases are characterized by progressive and selective loss of related neuronal systems, such as Alzheimer's disease (AD), Parkinson's disease (PD), and hereditary ataxias (HA).1,2 Neurodegenerative diseases result from the abnormal death of neurons in the nervous system, accompanied by different degrees of inflammation and neuron loss. 3 Pyroptosis of nerve cells is an important mechanism leading to specific death of nerve cells, which can be caused by activation of inflammasome by various influencing factors. Pyroptosis is characterized by the expansion of cells until the cell membrane bursts, resulting in the release of cell contents that activates a strong inflammatory response that promotes neurodegenerative diseases by accelerating neuronal dysfunction and abnormal microglial activation. 4 In this process, the cell continues to expand until the cell membrane bursts, and the contents of the cell are released, triggering a strong inflammatory and immune response. 5 NLRP1, NLRP3, NLRP4 and other members of the inflammasome (NLR) family are related to the stability of the central nervous system (CNS). NLRP1 inflammasome is the first member of the NLR family, mainly exists in macrophages and microglia of the CNS, which plays an important role in the regulation of neuronal pyroptosis. 6 It is well-documented that natural products play an important role in alleviating a wide range of disorders, including obesity, diabetes, cancer, and cardiovascular diseases and neurological diseases. Additionally, several natural products have favorable effects on AD.
NLRP1 Inflammasome and Neuronal Pyroptosis
Inflammasome biology is one of the most exciting and fastest growing areas of immunology. The inflammasome plays an important role in host defense against invading pathogens and in the development of cancer, metabolic, and neurodegenerative diseases. 7 Pyroptosis, also known as inflammatory cell death, is a programed necrotic behavior of cells mediated by the Gasdermin family, which serves as an important non-specific defense mechanism for the body to actively resist the invasion of external pathogens and monitor internal danger signals. Pyroptosis in nerve cells is mainly characterized by the destruction of the integrity of nerve cell membrane, the formation of plasma membrane pores, and the release of cell contents. 8
The NLR protein family acts as innate immune sensors in humans. NLRP1 inflammasome belongs to the NLR protein family, which is composed of N-terminal PYD domain, a central NACHT domain, a leucine-rich LRR domain, a FIIND domain and a C-terminal CARD domain. 9 NLRP1 in the nervous system is activated in response to amyloid-β (Aβ) aggregates, forming an oligomeric complex with an adaptor protein ASC, which then activates the pro-inflammatory protease cysteine proteinase-1 (Caspase-1). 10 Activated Caspase-1 triggers a series of inflammatory responses in the nervous system by cutting inflammatory cytokines IL-1β and IL-18 into maturation, and inducing neuronal pyroptosis by cutting and activating porinforming protein Gasdermin-D. 11
NLRP1 Inflammasome Mediated Pyroptosis and Neurodegenerative Diseases
Neuroinflammation is an important pathological feature of common nervous system diseases such as AD, and PD. 12 NLRP1 inflammasome plays an important role in activating pyroptosis and inducing an inflammatory response. Therefore, the development of drugs targeting the regulation of the NLRP1 inflammasome has become a reasonable research direction for the treatment of nervous system diseases. 13
NLRP1 Inflammasome and AD
AD is a progressive, irreversible neurodegenerative disease that is the most common cause of late-stage cognitive impairment and dementia, and is pathologically manifested by the deposition of amyloid-β (Aβ) plaques, hyperphosphorylated tau tangles, and loss of neurons in specific brain regions, which when associated with neuronal and glial dysfunctions, eventually leads to dementia.14–16 Several recent studies have reported high performance of plasma phosphorylated tau (p-tau, such as p-tau217, pTau181) for Alzheimer's disease (AD) to detect and monitor AD pathology.17,18 In different stages of the disease, patients show different degrees of dysfunction and personality changes in memory and cognition, and a series of complications. 19
NLRP1 inflammasome in the CNS is primarily expressed by pyramidal neurons and oligodendrocytes. 20 NLRP1 is activated in response to Aβ aggregates, and its activation subsequently cleaves caspase-1 into its active subunits. The activated caspase-1 proteolytically processes interleukin-1β (IL-1β) and IL-18 into maturation while coordinately triggering caspase-6, which is responsible for apoptosis and axonal degeneration. Recent studies have found that the expression level of NLRP1 in the brain tissue of AD patients is significantly higher than that of normal human brain, indicating that NLRP1 target plays a key role in the development of AD.21,22 The latest study found that miR-181c-5p mimic attenuated Aβ1−42-induced neuronal pyroptosis, while up-regulation of NLRP1 aggravated neuronal pyroptosis in HT22 cells. In mice, miR-181c-5p agomir attenuated neuronal pyroptosis in both hippocampal and cortical tissues, and miR-181c-5p antagomir improved neuronal pyroptosis and cognitive impairment through NLRP1. 23
NLRP1 Inflammasome and PD
The concept of ‘idiopathic’ Parkinson's disease (PD) as a single entity has been challenged with the identification of several clinical subtypes, pathogenic genes and putative causative environmental agents. 24 The neurophysiological underpinnings of PD are characterized by a spectrum of motor and non-motor symptoms. 25 In addition to classic motor symptoms, non-motor manifestations (such as rapid eye movement sleep disorder, anosmia, constipation and depression) appear at prodromic/premotor stage and evolve, along with cognitive impairment and dysautonomia, as the disease progresses, usually dominating the advanced stages of the disease. 26 The key molecular pathogenic mechanisms include α-synuclein misfolding and aggregation, mitochondrial dysfunction, impairment of protein clearance (associated with deficient ubiquitin-proteasome and autophagy-lysosomal systems), neuroinflammation and oxidative stress.27,28 Overactivation of inflammasome and abnormal secretion of proinflammatory cytokines play an important role in neuroinflammation and neurodegeneration in Parkinson's disease (PD).
Activated Caspase-1 in nerve cells can act on α-synuclein to induce its shortening and aggregation behavior and increase the incidence probability of PD. Meanwhile, the NLRP1 inflammasome can initiate innate immunity by recruiting and activating Caspase-1. 29
Effect of Natural Products on NLRP1 Inflammasome in Neurodegenerative Diseases
Crocin
Crocin, a natural component derived from the flower of saffron (Crocus sativus L.), is recognized as a pharmacologically active compound that is particularly noted for its potent antioxidant and anti-inflammatory properties. 30 Crocin exhibits beneficial effects in the prevention and treatment of cardiovascular disease, cancer, diabetes, kidney disease, and neurological disorders. 31 Crocin exhibits numerous beneficial protective effects against neurodegenerative diseases, attributed to its anti-apoptotic, anti-inflammatory, and antioxidant properties. These effects are particularly relevant in the context of cerebral ischemia, ischemic stroke, traumatic brain injury, spinal cord injury, memory deficits, cognitive impairments, as well as conditions such as AD, PD, and schizophrenia. 32 Crocin mitigated spatial learning and memory deficits, as well as hippocampal neuronal injury induced by Aβ25−35. Crocin suppressed inflammation and activated the PI3 K/AKT signaling pathway in mice with Aβ25−35 treatment. 33 Crocin increased live cells in the hippocampus, enhancing pyramidal neurons in CA3 and granular cells in DG, which reversed arborization loss and reduced apoptotic cell numbers in the regions of AD rats, thereby improving synaptic loss and neuronal death by decreasing neuronal apoptosis. 34 Crocin enhanced memory, learning, and the expression of NGF, BDNF, and TrkB genes while significantly reducing tau gene expression in a trimethytin (TMT)-induced AD model. 35 Additionally, crocin has been found to enhance the expression of tyrosine hydroxylase (TH) and decrease the levels of IL-1β, IL-18, NLRP1, and AIM2 genes. These findings suggested that crocin mitigates neuroinflammation in PD by lowering IL-1β and caspase-1 levels through the inhibition of AIM2 and NLRP1 gene expression. 36
Ginsenoside Rg1
Ginsenoside Rg1, the ginsenoside found in ginseng roots, demonstrates notable therapeutic effects in anti-inflammatory, antioxidant, and neuroprotective activities, which is particularly beneficial in the treatment of brain disorders, including dementia and depression. 37 Ginsenoside Rg1 has been shown to ameliorate depression-like behaviors by inhibiting the hyperactivity of the hypothalamic-pituitary-adrenal (HPA) axis, regulating synaptic plasticity, and modulating gut microbiota. Furthermore, Ginsenoside Rg1 may alleviate AD by repairing dendrites and axons, and addressing inflammation related to microglia and astrocytes. 38 Ginsenoside Rg1 has the potential to restore mitophagy and improve memory deficits in cellular and mouse models of AD through the PINK1-Parkin pathway. 39 Ginsenoside Rg1 has been shown to inhibit calcium overload and mitigate the disruption of calcium homeostasis following oxygen-glucose deprivation/reperfusion (OGD/R) in HT22 cells, which plays a neuroprotective role in alleviating cerebral ischemia-reperfusion injury (CIRI) by reducing oxidative stress, preventing calcium overload, and attenuating neuroinflammation. 40 Ginsenoside Rg1 was found to enhance the expression of glucocorticosteroid receptors while simultaneously reducing the levels of NLRP1, ASC, caspase-1, IL-1β, and IL-18 in the hippocampus following neuroinflammatory damage induced by chronic dexamethasone. 41
Hyperoside
Hyperoside is a natural flavonol glycoside found in various plants, including Crataegus pinnatifida Bge, Forsythia suspensa, and Cuscuta chinensis Lam, which exhibits a wide range of biological activities, such as anticancer, anti-inflammatory, antibacterial, antiviral, and antidepressant. 42 Hyperoside significantly reduced plaque deposition in the hippocampus of APP/PS1 mice and mitigated neuronal cell damage in the CA1 and CA3 regions, which regulated cellular apoptosis through the endoplasmic reticulum-mitochondrial calcium (Ca2+) signaling pathway, leading to an increase in Bcl-2 levels while decreasing Bax and cytochrome c protein levels. Consequently, hyperoside ameliorated neuronal cell death, suggesting its potential as a therapeutic agent or preventive remedy for AD. 43 Hyperoside has the potential to reduce cell apoptosis and counteract the Aβ42-induced downregulation of the PI3 K/Akt/Nrf2/HO-1 signaling pathway. 44 Hyperoside enhanced spatial learning and memory, reduced amyloid plaque deposition and tau phosphorylation, lowered the activated microglia and astrocyte, and mitigate neuroinflammation and oxidative stress in APP/PS1 transgenic mice. 45 Hyperoside reduced depressive-like behaviors in mice subjected to chronic stress, likely by inhibiting the NLRP1 inflammasome via the CXCL1/CXCR2/BDNF signaling pathway. 46
Kaempferol
Kaempferol is a key flavonol found abundantly in teas, apples, grapes, strawberries, tomatoes, and onions. 47 A growing study suggested that kaempferol exhibited neuroprotective properties in various neurodegenerative disorders, including PD, ischemic stroke, epilepsy, and AD. 48 The administration of kaempferol to rat cortical neurons under conditions of H2O2 stress demonstrated a protective effect against Aβ25−35-induced toxicity through the inhibition of the caspase cascade. Additionally, kaempferol enhanced the survival rate of PC12 cells by regulating heme oxygenase-1 (HO-1) expression and mitogen-activated protein kinase (MAPK) pathways. 49 The administration of kaempferol significantly increased cell viability, reduced Ca2+ influx, attenuated intracellular ROS generation, and regulated the expression levels of Bcl-2, Bid, apoptosis-inducing factor (AIF), and MAPK in glutamate-treated HT22 cells. 50 Kaempferol effectively protected PC-12 cells from cell death and apoptosis induced by Aβ25−35, enhanced the expression of anti-apoptotic molecules while inhibiting pro-apoptotic expression. The protective effect of kaempferol against Aβ25−35-induced apoptosis in PC-12 cells was mediated through the ER/ERK/MAPK signaling pathway. 51 Kaempferol has the potential to inhibit the activation of caspase-8, caspase-3, and NLRP1/NLRP3 inflammasomes. Additionally, it may prevent the production of pro-inflammatory cytokines by inhibiting the NF-κB and JNK signaling pathways. 52
Luteolin
Luteolin is a flavonoid found in various vegetables, fruits, flowers, celery, broccoli, green pepper, oranges, dandelion, peppermint, and rosemary, which has multiple beneficial effects on the regulation of inflammation-related symptoms and diseases including anti-inflammatory, anti-cardiovascular, and anti-neurodegenerative properties. 53 Luteolin was studied most extensively in AD, and PD. 54 The potential mechanism of Luteolin has been partially clarified, involving NF-κB, TLR4, MAPK, Wnt/β-catenin, PI3 K/Akt, AMPK/mTOR, and Nrf-2. 55 Luteolin has been shown to significantly ameliorate cognitive deficits in 3×Tg-AD mice and to mitigate Aβ-induced oxidative stress, mitochondrial dysfunction, and neuronal apoptosis through a PPARγ-dependent mechanism for AD. 56 Luteolin has been identified as an effective agent for reducing the learning and memory deficits in 3×Tg-AD mice. This efficacy may be attributed to the ability of luteolin to inhibit ER stress, specifically through the modulation of GRP78 and IRE1α in astrocytes, which subsequently leads to a reduction in neuroinflammation characterized by decreased levels of TNF-α, IL-1β, IL-6, NO, COX-2, and iNOS proteins. 57 Treatment with luteolin significantly decreased the expression levels of NLRP1, NOX4, TXNIP, and NLRP3 proteins. Additionally, luteolin ameliorated the dysregulation of apoptotic protein expression. 58
Parthenolide
Parthenolide is a germacrane sesquiterpene lactone that is abundantly sourced from Tanacetum parthenium. 59 In vitro studies demonstrated that parthenolide significantly reduced the production of ROS and restored mitochondrial membrane potential in PC12 cells. In vivo, parthenolide improved deficits in spatial learning and memory in the 3×Tg-AD murine model through the modulation of the AMPK/GSK3β/Nrf2 signaling pathway, while also providing protection to neuronal cells against ROS-related apoptosis. 60 Parthenolide demonstrated significant improvements in cognitive and behavioral dysfunction, inhibited neuroinflammation, and exhibited anti-neuroinflammatory activity along with neuroprotective effects. The benefits were achieved through improving the MAPK/TRIM31/NLRP3 axis, contributing to the amelioration of cognitive disorders during the progression of AD. 61 Parthenolide dramatically alleviated LPS-induced inflammation in microglia, and OGD-mediated neuronal apoptosis and oxidative stress, repressed the STAT3/NF-κB and NLRP1, NLRP3 inflammasomes activation. 62
Silibinin
Silibinin is a flavonolignan compound from Silybum marianum (milk thistle plant) that possesses strong antioxidant, and anti-inflammatory activities. 63 Silibinin has the potential to enhance cognitive function through the reduction of neuroinflammation and oxidative stress, activation of the reactive reactive oxygen species- Brain-derived neurotrophic factor- Tropomyosin receptor kinase B (ROS-BDNF-TrkB) pathway within the hippocampus. 64 Silibinin significantly mitigated the cognitive deficits observed in formaldehyde-induced mice, promoted the nuclear translocation of NRF2 and enhanced HO-1 expression, while decreasing apoptosis rates in mature neuronal cells. 65 Silibinin demonstrated a neuroprotective effect in an STZ-induced sporadic AD model by downregulating ferroptotic damage, thereby mitigating downstream STING-mediated neuroinflammation. 66 Silibinin significantly reduced microglial activation and downregulated the levels of the pro-inflammatory cytokine IL-6, as well as the anti-inflammatory cytokine IL-4, along with iNOS and COX-2. 67 In vitro studies demonstrated that silibinin treatment of monocytes derived from preeclamptic women effectively reduced the activation of these cells through the inhibition of NLRP1/NLRP3 inflammasome activity and a decrease in NF-κB signaling. 68
Molecular Docking
CB-Dock was employed for blind docking, which provides the three-dimensional structure of both a protein and a ligand while predicting their binding affinity. The PDB format of NLRP1 (PDB: 7WGE) was obtained from the RCSB database. 69 Natural products in SDF format were selected as the ligand file and subsequently input into CB-Dock for molecular docking analysis. 70 Molecular docking results showed that silibinin, crocin, and hyperoside had good binding affinity, and which were the most promising potential active compounds for the treatment of AD by regulating NLRP1 (Table 1, and Figure 1). The binding affinity of silibinin to NLRP1 (−9.0) is significantly higher than that of its own ligand (−8.8). The studies reported that silibinin can be considered a therapeutic agent to reduce symptoms of cognitive impairment, acting through affecting inflammation, programed cell death, and oxidative stress. 71 Therefore, silibinin can be considered a lead compound inhibited the NLRP1.
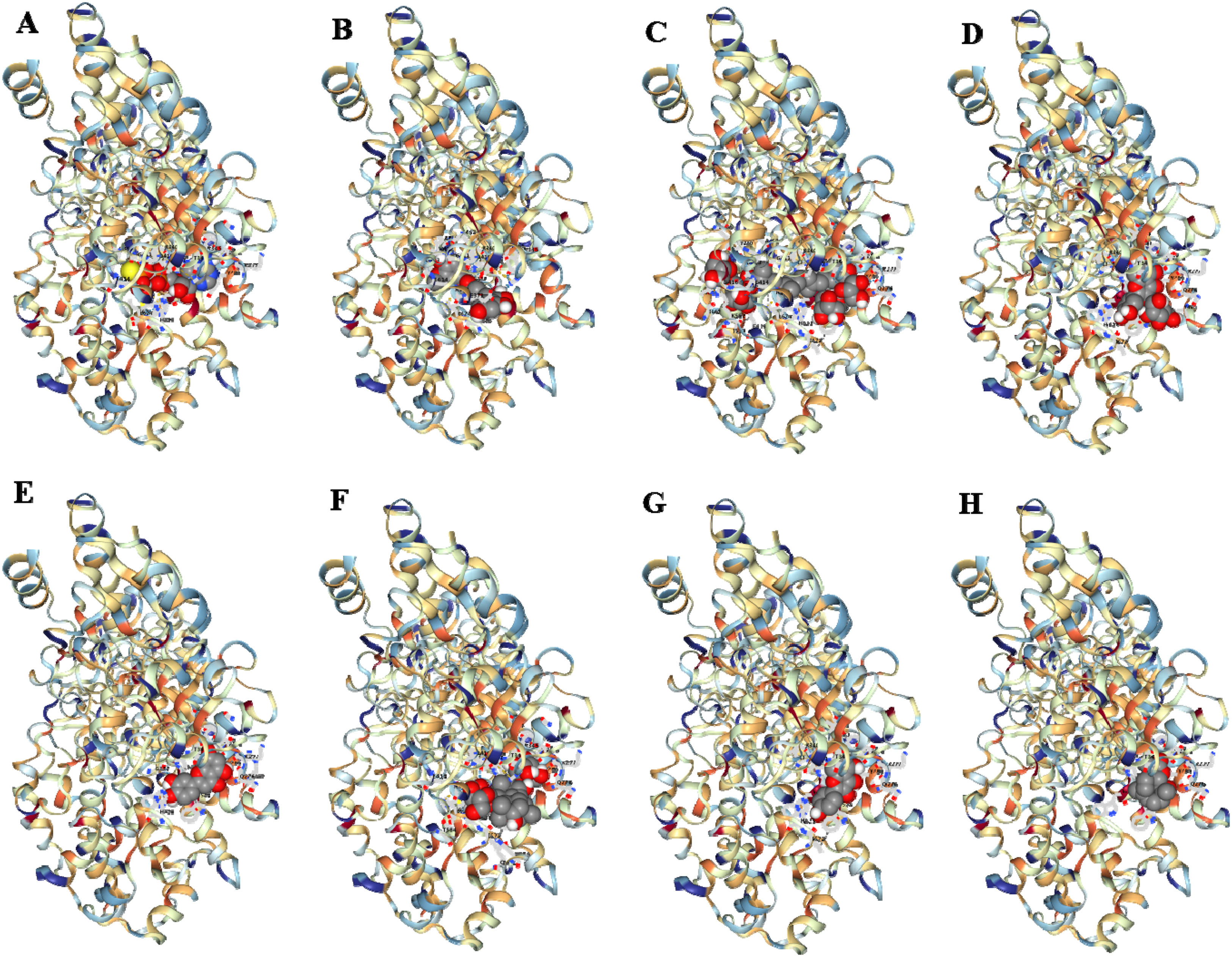
Molecular docking of natural products with NLRP1. Native ligand, (B) Silibinin, (C) Crocin, (D) Hyperoside, (E) Luteolin, (F) Ginsenoside Rg1, (G) Kaempferol, (H) Parthenolide.
Docking of Natural Products with NLRP1.

Conclusions
NLRP1 inflammasome is closely related to the development of neurodegenerative diseases. The natural products listed in this paper could binding to the NLRP1 and protect against the neurodegenerative diseases, such as silibinin, crocin, and hyperoside. Therefore, it is speculated that natural products may affect the process of neuronal pyroptosis by regulating NLRP1 inflammasome. In particular, silibinin can be used as lead compounds for the design of novel NLRP1 specific inhibitors. However, this paper only predicted the effect of natural products on NLRP1 inflammasome, which will be confirmed by in vivo and in vitro experiments in the following studies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
These works were financially supported by the Key Project Foundation of Natural Science Research in Universities of Anhui Province in China (KJ2021A0589).
