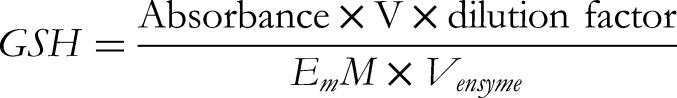Abstract
Background
Blumea lacera Burm. f. DC (Asteraceae) is an erect herb with antioxidant and α-amylase inhibitory activities, rendering it a potential agent for the evaluation of antidiabetic activity.
Methods
Experimental diabetes was induced in albino Wistar rats by a single intraperitoneally injection of streptozotocin (50 mg/kg b.w.) dissolved in cold citrate buffer at pH 4.5. Different fractions of methanol extract of aerial parts of B. lacera were subjected to acute antidiabetic study in streptozotocin-induced diabetic rats at a dose of 200 mg/kg tested up to 10 h. Methanol fraction (MFBL) at 100 and 200 mg/kg was administered to diabetic rats for sub-acute antidiabetic study. Serum biochemistry including lipid profile, insulin levels, and carbohydrate metabolizing enzymes as well as histopathology, were performed during the 30 days of interventional study. All the groups were compared with the standard drug Metformin. Gas chromatography-mass spectrometry (GC-MS) analysis of different fractions was conducted to identify the active compounds.
Results
MFBL showed a significant (P < 0.01) fall in blood glucose levels and hepatic enzymes in a dose-dependent manner in streptozotocin-induced diabetic rats. MFBL also modulated the activities of carbohydrate metabolizing enzymes, by significantly increasing the effects of hexokinase and glucose-6-phosphate dehydrogenase and significantly reducing the effects of glucose-6-phosphatase and fructose-1,6-bisphosphatase. The histopathology of liver and pancreas samples along with antioxidant study also supported biochemical observations. GC-MS analysis identified 17 major bioactive compounds including the antidiabetic agent, Myo-inositol, in different fractions.
Conclusion
The anti-diabetic effects of MFBL are attributed to its modulatory effects on carbohydrate metabolizing enzymes as well as the presence of antidiabetic phytochemical ‘Myo-inositol’ in MFBL. Further studies are warranted to identify multiple targets of active compounds present in B. lacera plant.
Introduction
Diabetes mellitus (DM) is a heterogeneous type of metabolic disorder characterized by hyperglycaemia ensuring either insufficient secretion or resistance to the action of insulin or both. Hyperglycaemia is a state of rise in blood glucose level which later develops into macro and microvascular complications. Diabetes is prevalent affecting around 366 million people in the year 2011 to grow to 552 million by the year 2030. There is an imbalance in the metabolism of carbohydrates, lipids, and protein in diabetes patients. Thus, the enzymes involved in the process of metabolism like gluconeogenesis, glycogenolysis, etc are distorted, leading to a chronic state of Diabetes. Diabetes is a multifactorial disorder having clinical manifestations like hyperglycemia, hypercholesterolemia, and hypertriglyceridemia that lead to impaired secretion of insulin and low insulin sensitivity to tissue. After a long time, a diabetic patient will have cardiovascular diseases, renal failure, nerve issues, and cataracts in the eye. 1
Although, there are lots of therapeutic medicines available in the market but their extended use results in serious side effects and hence, there is an urgent need to develop a phytochemical that controls hyperglycaemia and its complications. 2
Blumea lacera Burm.f. DC. (Asteraceae) commonly known as Kukursunga, has enormous pharmacological importance and is widely distributed in Bangladesh, India Australia, China, South Africa etc. B. lacera has α-amylase inhibitory activity as well as antioxidant effects. 3 This plant has an aromatic-scented strong herb with a length of 0.5 −1.0 m with purple flowers and toothed leaves. It has been used for fever, anti-inflammation, burns and cuts, blood diseases and bronchitis, etc. 4 This plant is used for anti-inflammatory, antioxidant, hypoglycaemic, wound healing activity, antipyretic, antimicrobial, antidiarrheal, etc. 5 Another species of the Blumea, B. eriantha has anti-angiogenic and antioxidant effects in its essential oil. 6 There is no such study conducted earlier by any researcher on this plant regarding its antidiabetic potency using animal models.
The present study focuses on the antidiabetic potential of a methanol fraction of aerial parts of B. lacera in streptozotocin (STZ) induced diabetic rats by monitoring the carbohydrate metabolizing enzymes, and hepatic key enzymes along with histopathological investigation.
Materials and Methods
Plant Materials
Aerial parts of B. lacera were aggregated in large quantities from Purohitpur village of Bhubaneswar, Odisha in the winter season, in the month of January. Plant material was authenticated by the Taxonomist, Regional Plant Research Centre from Bhubaneswar. A voucher specimen number (SPS/SOAU/08) was preserved in the Department of Pharmacology, School of Pharmaceutical Sciences, Bhubaneswar, Odisha. The plant materials were cleaned with water and shade-dried for 12-15 days. Then, they were coarsely powdered with the help of a mechanical grinder for the preparation of methanol extract using methanol as a solvent. 7
Fraction Preparation
Different fractions such as benzene, chloroform, ethyl acetate, and methanol were prepared from the best extract i.e., methanol extract by solvent-solvent separation technique in the order of their polarity index. They are stored in a desiccator for future use.8,9
Evaluation of Acute Oral Toxicity
An acute toxicity study was conducted as per the Organization for Economic Co-operation and Development (OECD 423) and Arrive guidelines for animal experimentation. Ethical permission was approved by the Institutional Animal Ethical Committee with letter number 57/SPS/IAEC/SOAU. The study protocol was passed by the University animal ethical committee (IAEC No. 1171/C/08/CPCSEA). Test fractions at levels starting from 250 mg/kg to a maximum of 2000 mg/kg were given to the test and control groups. Various signs of acute toxicity study such as diarrhea, lethargy, convulsion, salivation, changes in skin color, and sleep were observed critically for 4 h and followed up for 72 h. Mortality was observed for 14 days in all the treated mice. 10
Gas Chromatography and Mass Spectrometry Study of the Fractions of B. lacera
The gas chromatography-mass spectrometry analysis was conducted with the help of THERMO TRACE 1300 Gas Chromatograph and linked to a mass detector THERMO TSQ 8000 spectrometer with TG 5 MS (30 m × 0.25 mm, 0.25 μm) capillary column. The instrument has maintained an initial temperature of 70 °C for 3 min. The temperature was then increased by 10 °C every minute till a destination temperature of 300 °C and maintained for around 9 min. The temperature of the injection port was kept at 250 °C and the rate of helium flow at 1 μl/min. The ionization voltage was kept at 70 eV. The samples were taken injection in a 10:1 split manner. MS scanning range was set at 50 - 700 (m/z). The ion source temperature and interface temperature were maintained at 230 °C and 240 °C respectively. The start time of MS was 3 min, and the end time of MS was 35 min with 3 min of solvent cut time. Database of (NIST Version 2.0, 2005) library and Wiley spectral library were used to identify the compounds. 8
Experimental Animals and Induction of Diabetes
Male Wistar albino rats (180-250 mg/kg, aged about 3-4 months) were selected and acclimatized for 10 days under standard conditions (temperature 25 ± 2 οC, humidity at 50 ± 5%) with 12 h of light and dark cycle in a polycarbonate cage. They were fed with a standard pellet diet and drinking water ad libitum. The composition of the pellet diet includes wheat, barley, oats, soybean, wheat middlings, ground corn, soybean oil, animal fat, vegetable oil, and other essential nutrients and are procured from the National Institution of Nutrition, Indian Council of Medical Research, situated at Secunderabad, Telangana. Animals were used for experimentation with permission from the Institutional Animal Ethical Committee and guidelines were followed by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) and Arrive guidelines. The animals were fasted overnight (for about 12 h) on the last day of the experimental model before the induction of anesthesia or the collection of blood samples.
They were induced with a single dose intraperitoneal injection of 50 mg/kg b.w. of STZ dissolved in a freshly prepared citrate buffer (pH 4.5) in cold conditions. As the STZ is degraded within 15 to 20 min of preparation with citrate buffer, it is advisable to prepare immediately before the induction process. 11
Evaluation of Acute Antidiabetic Potency of the Fractions of B. lacera
Different fractions such as benzene, chloroform, ethyl acetate, and methanol at a dose level of 200 mg/kg were administered to the fasted diabetic rats, and blood sugar levels were recorded at 0, 1, 2, 3, 4, 6, 8 and 10 h. Analysis of Variance (ANOVA) followed by Tukey's test was used to compare the effect on the extent of fall of blood glucose measures between the treated groups.
Forty-two healthy Wistar male albino rats were divided into 7 groups of 6 animals each such as
Group 1: Negative control (NC) received distilled water and tween 80 orally in a volume of 10 mL/kg in normal rats. Group 2: Diabetic control (DC) received vehicle (tween 80 + distilled water) orally in a volume of 10 mL/kg in diabetic rats. Group 3: Standard group received Metformin (Glycomet 500, USV Ltd) in a dose of 250 mg/kg, p.o. in diabetic rats. Group 4: Test group received a benzene fraction of B. lacera (BFBL) at a dose of 200 mg/kg orally in diabetic rats. Group 5: Test group received a chloroform fraction of B. lacera (CFBL) at a dose of 200 mg/kg orally in diabetic rats. Group 6: Test group received a ethyl acetate fraction of B. lacera (EFBL) at a dose of 200 mg/kg orally in diabetic rats. Group 7: Test group received a methanol fraction of B. lacera (MFBL) at a dose of 200 mg/kg orally in diabetic rats.
Evaluation of Sub-acute Antidiabetic Potency of Methanol Fraction of B. lacera
After conducting an acute antidiabetic study by fractions in diabetic rats, a methanol fraction showed better potency in lowering blood glucose and this fraction was taken for further sub-acute antidiabetic study. Methanol fraction at a dose level of 100, 200 mg/kg was administered to the fasted diabetic rats daily for 30 days and blood glucose levels were determined with the help of a glucometer at 0, 5, 10, 15, 20, 25, and 30th days. Data are interpreted in GraphPad Prism v9.0 for statistical analysis.
Thirty healthy male albino rats were divided into 5 groups of 6 animals each such as
Group 1: NC received distilled water and tween 80 orally in a volume of 10 mL/kg in normal rats. Group 2: DC received vehicle (tween 80 + distilled water) orally in a volume of 10 mL/ kg in diabetic rats. Group 3: Standard group received Metformin (Glycomet 500, USV Ltd) in a dose of 250 mg/kg, p.o. in diabetic rats. Group 4: Test group received a methanol fraction (MFBL1) of B. lacera at a dose of 100 mg/kg orally in diabetic rats. Group 5: Test group received a methanol fraction (MFBL2) of B. lacera at a dose of 200 mg/kg orally in diabetic rats.
Evaluation of Body Weight, Serum Biochemical Parameters, Insulin and Carbohydrate Metabolizing Indices
The body weight of all the treated groups was recorded in terms of percentage loss in body weight at 0, 10, 20, and 30 days of the sub-acute study. Blood serum samples were collected at 0, 10, 20, and 30 days of sub-acute study under an aseptic condition with mild anaesthesia by heart puncture method and preserved in different tubes like 52 BD vacutainer sodium fluoride, BD vacutainer Buff. Sodium citrate. They are cold centrifuged (Remi C-24 BL) immediately at 10,000 rpm at 4 οC for 15 min to separate the serum and plasma. Then they are stored at −80 οC in a freezer for future analysis. The serum biochemical parameters such as Aspartate aminotransferase (AST), Alanine transaminase (ALT), total cholesterol (TC), triglyceride (TG), low-density lipoprotein (LDL), high-density lipoprotein (HDL), and very low-density lipoprotein (VLDL) were estimated at the end of the 30th day. Insulin, and carbohydrate metabolizing enzymes were estimated at 0, 10, 20, and 30 days of the study.12,13 Blood glucose was estimated daily before and after the administration of test and standard drugs (g/dl) by using a glucometer. At the end of the experimental period i.e. 30 th day, animals were sacrificed, blood serum samples were collected for the biochemical analysis. Immune-turbidimetric assay kit for estimation of HbA1c was obtained from Roche (Roche Diagnostics Ltd). The blood samples in K2EDTA tubes were centrifuged at 4000 rpm at 4 °C for 15 min to obtain the plasma samples which used for the assessment of glycated hemoglobin percentage (HbA1c%).
Effects of Methanol Fraction of B. lacera on Histopathology of Liver and Pancreas
Finally, on the 30th day, all the treated diabetic rats were sacrificed by decapitation method under the standard procedure, and major affecting organs like the liver, and pancreas were removed in a clean petri dish with a physiological solution. After cleaning, organs were fixed in a 10% formaldehyde solution and stored for histopathological slide preparation of the organs. Sections of organs were cut by using rotary microtome at about 4 - 5 µm thickness. Different stains such as Haematoxylin-eosin and Halami were used for better visibility of hepatic and beta cells of the pancreas respectively. 14 The photographs were taken with the help of a Magnus light microscope and Olympus Digital Single Lens Reflex (DSLR) camera.
Determination of Carbohydrate Metabolism Enzymes
Estimation of glucose-6-phosphatase was done by following the method of Hubscher and West. 1 mL of assay containing citrate buffer (pH 6.0), 14 mM NaF, 28 mM EDTA and 200 mM glucose-6-phosphatase and enzyme protein in appropriate amount were added. Incubation of tubes was done at 37 °C for 30 min and the reaction was ended by adding up 1 mL of 10% TCA. In the supernatant layer, the determination of inorganic phosphate (Pi) was done according to the method of Tausky and Shorr. Determination of Glucose-6-phosphate dehydrogenase (G-6-PDH), hexokinase, and fructose-1,6-bisphosphatase was done at the end of the sub-acute study and serum was separated by centrifugation as aforementioned. These tests were performed by Bio-RAD automated Microat-II analyzer affinity chromatography method.15,16
2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical Scavenging Activity
Free radical scavenging activity of DPPH by fractions was performed by adding 0.3 mM of total 1 mL of DPPH to the 2.5 mL of test sample/ standard of various concentrations and allowed to stand for reaction at room temperature in the dark. After thirty minutes, the absorbance of samples was taken at 517 nm and the percentage of antioxidant value was calculated by following formula below
2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging activity
It was prepared by 2.45 mM ammonium persulphate with the ABTS reagent 7 mM and allowed to stand in the dark at room temperature for 12 - 16 h.
Different test fractions of different concentrations were prepared and mixed with ABTS 0.9 mL of radical cation. Absorbance was recorded at 745 nm and all the experiments were conducted in a triplicate manner. 18
Determination of Reduced Glutathione (GSH)
The tissue homogenate (supernatant) and Trichloroacetic acid (TCA) 20% were added in the same quantity. The precipitation was centrifuged and supernatant of 0.01 mL, 0.5 M 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB), 2 mL of phosphate buffer and 0.4 mL double distilled water were mixed. Absorbance is recorded at 412 nm within 15 min. The concentration of reduced glutathione was denoted as U/mg protein.
19
E mM is Molar absorption coefficient = 13.60
Dilution factor is 30
V is Reaction volume
V Enzyme is the volume of sample (0.01)
U/mg protein = Units per milligram of protein
Determination of Superoxide Dismutase (SOD)
Here, the reaction mixture contained 0.5 mL of homogenate (supernatant), 0.4 mL 25 µM of Nitro blue tetrazolium (NBT), 1 mL of 50 mM sodium carbonate and 0.1 mM of 2 mL Ethylenediaminetetraacetic acid (EDTA). With the addition of 1 mM of 0.4 mL hydroxylamine- hydrochloride, the reaction was started. A change of absorbance was recorded at 560 nm. Without taking tissue homogenate, control was run simultaneously. The measure of antioxidant was expressed as the amount of enzyme required to inhibit the NBT reduction by 50% in mg/mL.
20
Determination of Catalase (CAT)
The supernatant (0.1 mL) was mixed in a cuvette which contained a phosphate buffer of 1.9 mL (PH 7.4, 50 mM). With the addition of 1 mL of freshly prepared 30 mM hydrogen peroxide, the reaction was started. The unit of catalase was expressed as nmole/min/l. The measure of antioxidant value lies in the rate of decomposition of hydrogen peroxide at 240 nm.
21
Statistical Analysis
All the data are represented by mean ± SEM. Data were interpreted and analyzed using GraphPad Prism v9.0. ANOVA test followed by Tukey's test was taken for interpretation of the effect of fall of blood glucose measures between all the treated models.
Results
Acute Oral Toxicity Study
Acute oral toxicity study of fractions of B. lacera is depicted in Table 1. We observed that all the animals were found to be healthy and safe to the maximum dose level of 2000 mg/kg and doses were fixed at 1/10th and 1/20th of the lethal dose ie 200 and 100 mg/kg b.w. following the OECD guidelines 423.
Acute Oral Toxicity Study of Fractions of B. lacera.

Identification of Phytochemicals from Different Fractions of B. lacera by Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
GC-MS analysis of different fractions of B. lacera showed the presence of various reported and unreported compounds having pharmacological importance (Figures 1–4). The major compounds identified from GC-MS are phenols, antioxidant, antidiabetic, lipid-lowering and α-glucosidase inhibitory in nature which gave clues for the blood glucose-lowering potential of this plant. The details of the identified phytochemicals from different fractions and their reported pharmacological activities are depicted in Table 2.
Identification of Phytochemicals and Their Reported Activities in Fractions of B. lacera by GC-MS Analysis.

Acute Effects of Fractions of B. lacera on Blood Glucose Levels in Diabetic Rats
Different fractions such as benzene, chloroform, ethyl acetate, and methanol at a dose level of 200 mg/kg were given to the diabetic rats, and blood glucose estimations were done at 1, 2, 3, 4, 6, 8, 10 h. Diabetic rats treated with BFBL registered a blood glucose 137.33 mg/dL (56.92%) at the end of the 10 h and significant (P < 0.01) lowering of blood glucose was found at 1 h and continued till the end of the study period. Similarly, rats treated with CFBL registered blood glucose at 131.00 mg/dL (57.08%) at the end of the experiment, and a significant (P < 0.01) lowering of blood glucose was seen at 1hr and sustained till the end of the study. Again, rats treated with EFBL showed blood glucose 121.67 mg/dL (59.31%) at the end of 10 h, and a significant (P < 0.01) lowering of blood glucose was seen at 1 h to the end of 10 h. Similarly, MFBL registered blood glucose of 99.17 mg/dL (68.61%) towards the normal range in diabetic rats and a significant lowering of blood glucose was noted from 1hr till the end of the study period (Figure 5).

GC-MS chromatogram of BFBL.

GC-MS chromatogram of CFBL.

GC-MS chromatogram of EFBL.

GC-MS chromatogram of MFBL.
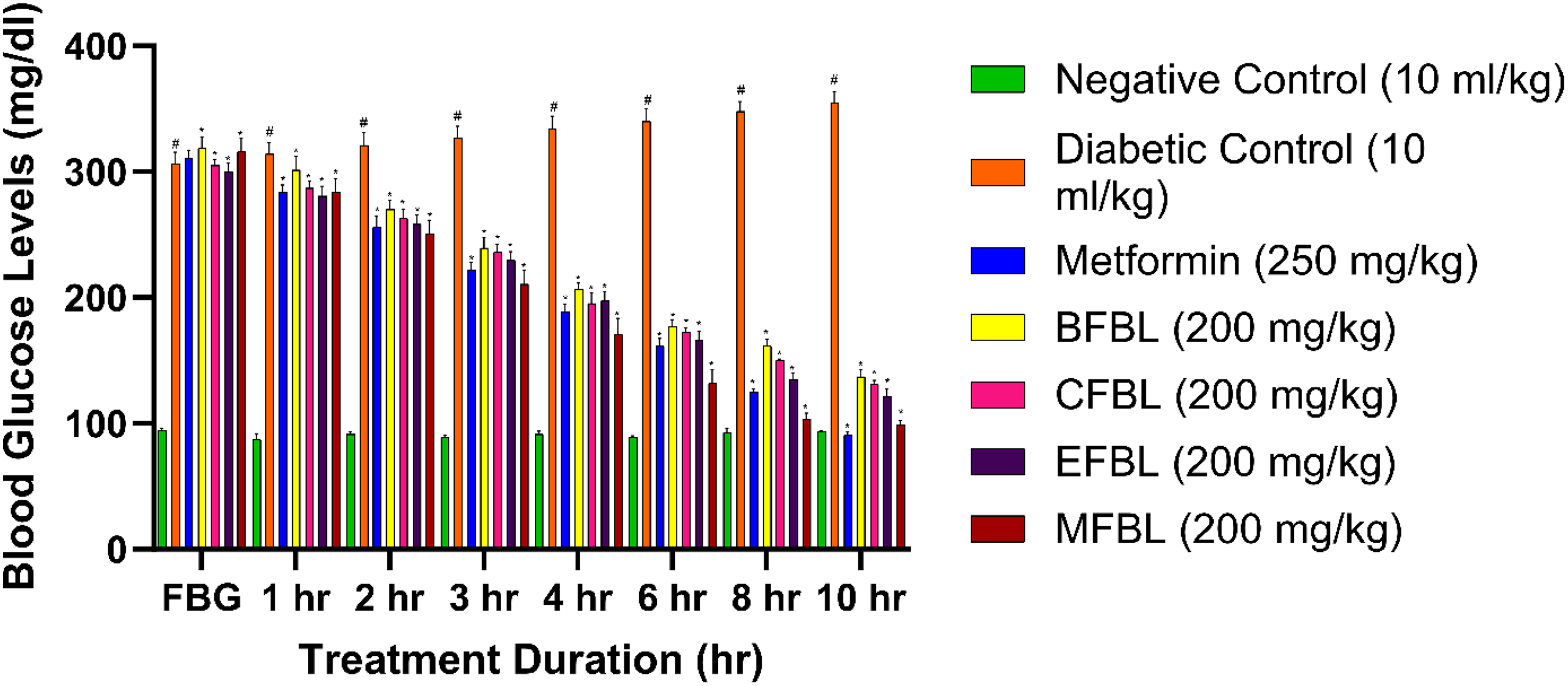
Acute antidiabetic effects of fractions of B. lacera on blood glucose levels. The values presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Moreover, all the fractions can reduce the blood glucose level significantly but, the MFBL of B. lacera at 200 mg/kg showed better potency among all. Thus, the researcher thought that MFBL might be a good choice for sub-acute challenges in diabetic rats.
Sub-Acute Effects of Methanol Fraction of B. lacera (MFBL) on Blood Glucose Levels in Diabetic Rats
The sub-acute effects of the MFBL of B. lacera on blood glucose levels in diabetic rats are presented in Figure 6. The methanol fraction of B. lacera ie MFBL at 100 mg/kg registered a blood glucose level of 115.67 mg/dL (62.15%) on the 30th day of the experimental period and a significant (P < 0.01) lowering of blood glucose was seen from fifth day onwards till the 30th day of the study. Similarly, MFBL at 200 mg/kg registered a blood glucose level of 102.00 mg/dL (67.96%) on the same day and a significant (P < 0.01) lowering of blood glucose was seen from the fifth day onwards till the 30th day of the study. While, Metformin standard drug at 250 mg/kg showed a blood glucose level of 92.83 mg/dL (70.14%) on the 30th day and a significant (P < 0.01) lowering of blood glucose was found from the fifth day onwards till the end of the study. The DC group showed a significant (P < 0.01) increase in blood glucose levels and registered the same 366.33 mg/dL at the end of the study period. The results revealed that there is a dose-dependent gradual decrease of blood glucose level significantly in the test fraction when compared to the DC group.

Sub-acute antidiabetic effects of methanol fraction of B. lacera (MFBL) on blood glucose levels. The values were presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Effects of MFBL on Body Weight of Treated Diabetic Rats
MFBL at 100 and 200 mg/kg registered 17.65, 11.67% as percentage loss in body weight of treated diabetic rats at the end of 30th day, when compared to the NC and DC group. On the other hand, diabetic rats treated with the standard drug Metformin at 250 mg/kg showed 8.34% (as % loss in body weight) on the same day. The lesser value of percentage loss in body weight implies improvement in the recovery of body weight in rats. There was a dose-dependent body weight recovery occurred in test treated groups as compared with the NC and DC group (Table 3).
Changes in Body Weight of MFBL-Treated Diabetic Rats.

Data represents Mean ± SEM, (N = 6) in individual groups; The values presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Effects of MFBL on serum Biochemical and Lipid Profile Estimation
The liver enzymes AST and ALT of MFBL at 100 mg/kg showed 43.10 and 42.37 µ/L respectively, while, MFBL at 200 mg/kg showed 33.87 and 36.77 µ/L respectively at the end of 30th day in diabetic rats. Moreover, Metformin-treated groups at 250 mg/kg registered the same 25.03, 22.77 µ/L on the same day. The DC group showed an increase in the level of hepatic enzymes due to disturbed metabolism in diabetes. The HbA1c level of MFBL at 100 and 200 mg/kg registered at 6.83 and 5.80% whereas, Metformin recorded the same 4.65% at the end of the experiment as compared to the NC and DC treated group (Figure 7a and 7b). In diabetes, there is a gradual rise in the amount of TC, TG, LDL, VLDL and a decrease in the amount of HDL due to disturbed lipid metabolism in a diabetic state. MFBL at a dose of 100 mg/kg registered TC, TG as 123.06 and 109.40 mg/dL whereas, MFBL at 200 mg/kg registered the same as 71.02, and 70.15 mg/dL respectively on the same day in treated rats. The Metformin standard group registered the same as 63.38 and 65.63 mg/dL respectively in diabetic rats. The LDL, VLDL, HDL, and PL of the test fraction MFBL at 100, 200 mg/kg registered 67.76, 33.29, 28.10, 132.03 mg/dL and 39.50, 25.05, 47.23 and 116.67 mg/dL respectively. Moreover, the Metformin-treated group showed 36.91, 19.43, 53.13, and 111.70 mg/dL similarly at the end of the 30th day. Results of the test fraction and Metformin showed a significant decrease in the level of LDL, VLDL, PL, and a increase in the level of HDL when compared to the DC group (Figure 8).

(a) Changes in serum biochemical parameters of MFBL-treated diabetic rats (b) Changes in serum biochemical parameters of MFBL-treated diabetic rats. The values are presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group. ALT- Alanine aminotransferase; AST- Aspartate aminotransferase; ALP- Alkaline Phosphatase; BT- Bilirubin total; BD- Bilirubin direct; TP- Total protein.

Changes in serum lipid profile of MFBL-treated diabetic rats. The values are presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0 .01 compared to the NC group. TC – Total cholesterol; TG – Triglyceride; HDL = High-density cholesterol; LDL = Low-density cholesterol; VLDL = Very low-density cholesterol; PL = Phospholipids.
Effects of MFBL on serum Insulin Levels
The results of the sub-acute effects of fractions of B. lacera on serum insulin levels in diabetic rats are presented in Figure 9. The test fractions, MFBL at 100 mg/kg enumerated 7.27, 8.31, 8.71, 9.09 µU/mL of insulin levels while, 200 mg/kg registered 7.16, 10.11, 11.64, 12.42 µU/mL at day 5, 10, 20 and 30 of sub-acute study respectively in diabetic rats. The standard Metformin groups registered insulin levels 6.98, 8.63, 10.13, and 12.77 µU/mL at day 5, 10, 20, and 30 of the sub-acute study respectively. However, the DC group registered the same as 5.71, 6.35, 7.73 and 8.49 µU/mL on days 5, 10, 20, and 30 of the experimental period respectively. In the case of NC group, there was an increase in the level of insulin gradually till the end of the study.

Changes in insulin level of MFBL-treated diabetic rats. The values are presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Sub-acute Effects of MFBL on Carbohydrate Metabolizing Indices
The results of the subacute effects of MFBL of B. lacera on carbohydrate metabolizing enzymes in diabetic rats are depicted in Table 4. The test fraction, MFBL at 100 mg/kg demonstrated enzyme activities of glucose-6-phosphatase, hexokinase, fructose-1,6-bis phosphatase, glucose-6-phosphate dehydrogenase as 3.75, 186.66, 0.15 and 10.67 Units/min/g respectively whereas, at 200 mg/kg enumerated the same as 3.10, 250, 24, 0.12, 13.92 Units/min/g respectively in a similar manner. The DC group enumerated the enzyme activity as 6.65, 139.58, 0.20, and 7.91 Units/min/g respectively at the end of the 30th day as compared to the NC-treated rats. Moreover, the Metformin-treated group at 250 mg/kg showed the same as 3.24, 258.95, 0.12, and 14.01 Units/min/g respectively at the end of the 30th day. It was revealed that the test fractions and Metformin group well-controlled over the carbohydrate metabolizing enzymes in the diabetic condition of rats in the subacute study when compared to the NC and DC group.
Changes in Carbohydrate Metabolizing Indices of MFBL-Treated Diabetic Rats.

The values are presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Effects of MFBL on Histopathology of Liver and Pancreas in Diabetic Rats
The histopathology study was conducted at the end of the sub-acute antidiabetic study (on the 30th day) to know the status of derangement in liver tissue and beta cell regeneration in islets of Langerhans of the pancreas. All the pancreas sections were stained in Halami stain for better visibility of beta cells and liver sections were stained with Haematoxylin & Eosin. The current interpretation of histopathology explored that the methanol fraction (MFBL 100 mg/kg b.w. and MFBL 200 mg/kg b.w.) and Metformin-treated groups were quite capable of combating the beta cell degeneration in the pancreas and derangements of hepatic cells due to STZ-induced diabetes. The pictures were captured by 40× and 100× magnification with the help of a Magnus light microscope and Olympus DSLR camera.
Effects of MFBL of B. lacera on Antioxidant Activity (In-Vitro)
The free radical scavenging effects of MFBL of B. lacera using DPPH and ABTS methods are presented in Figures 10 and 11 respectively. The IC50 values of the MFBL were 400 and 690 µg/mL in DPPH and ABTS models respectively.
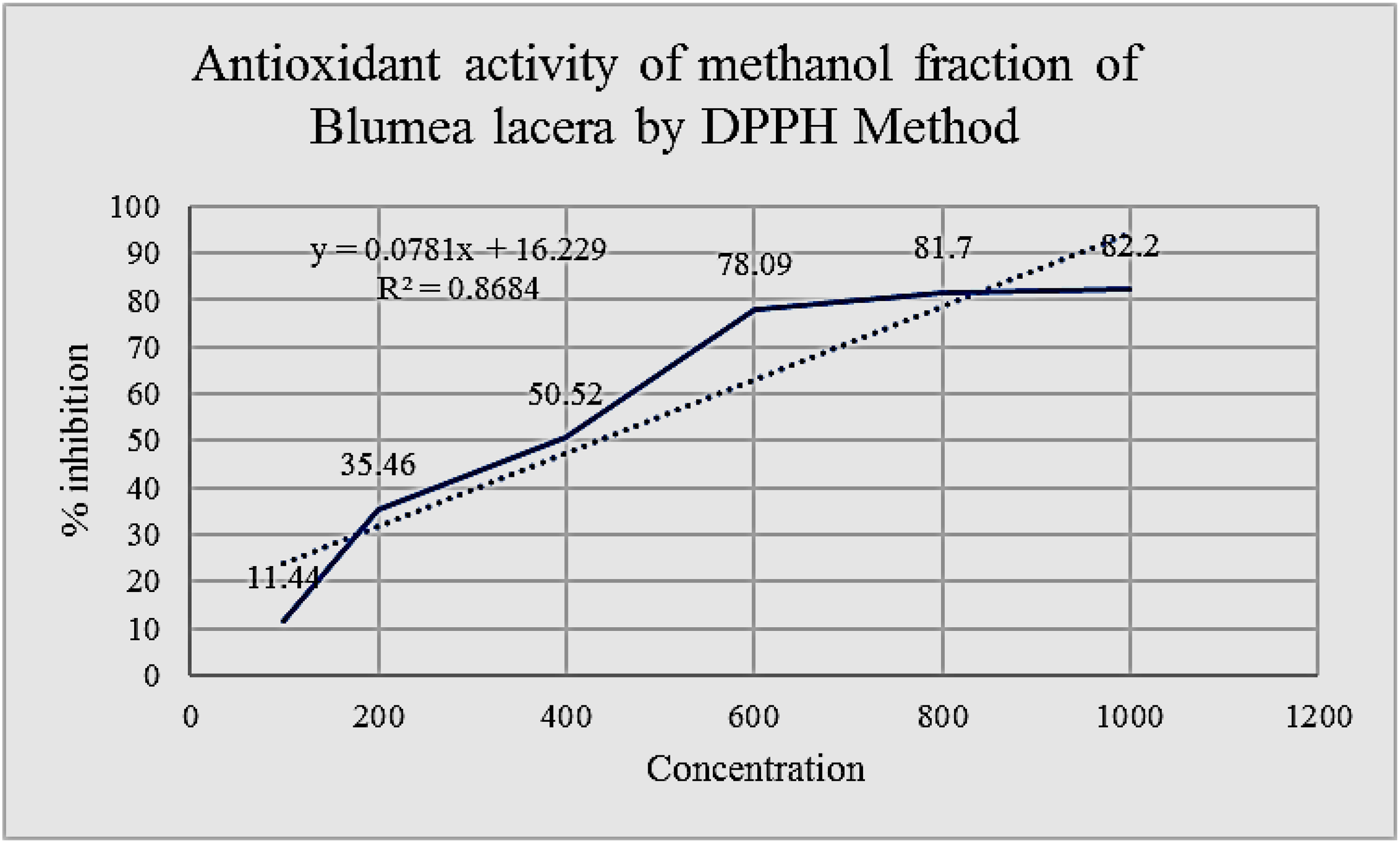
Effects of MFBL by DPPH radical scavenging assay.
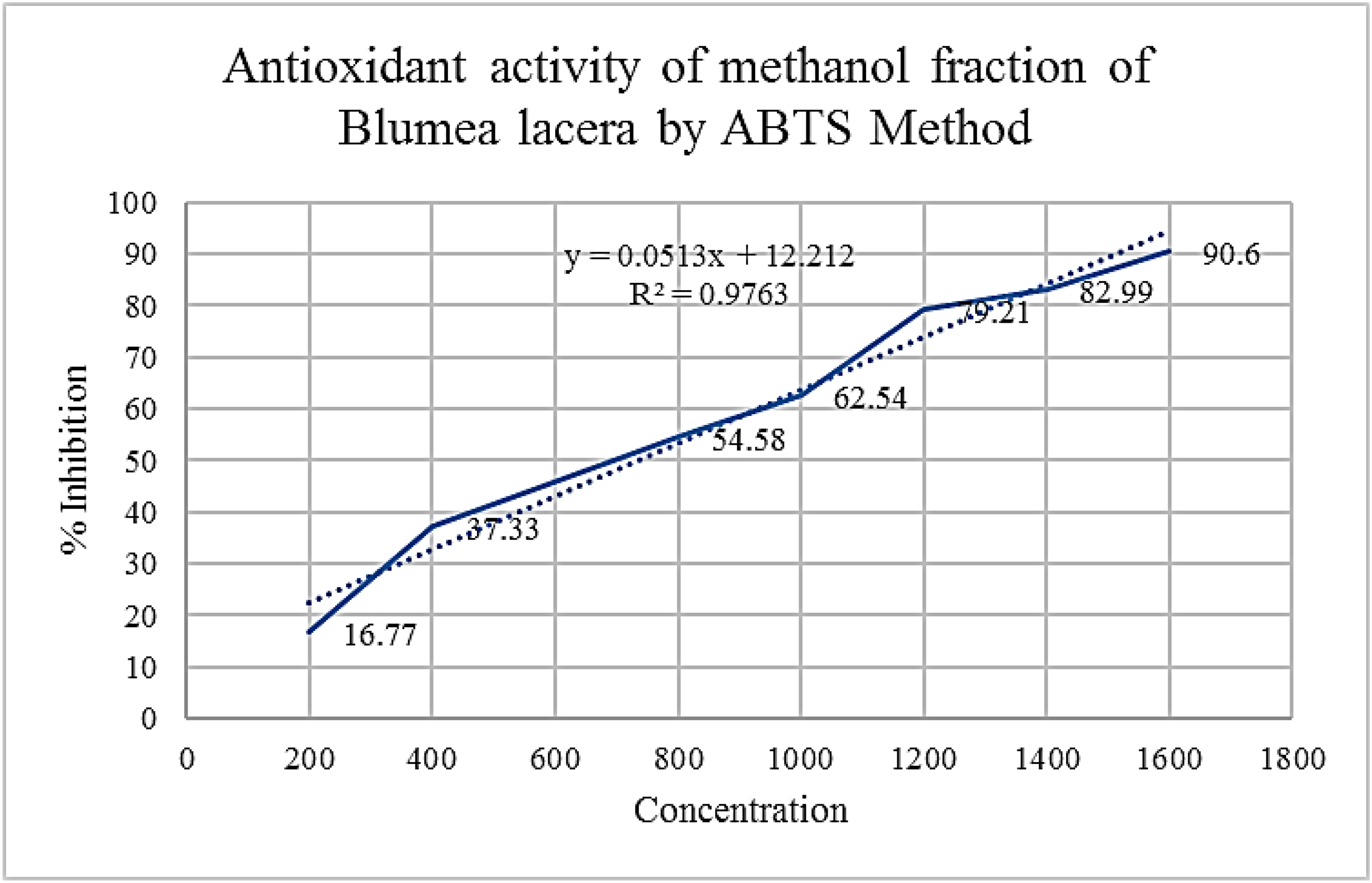
Effects of MFBL by ABTS radical scavenging assay.
Effects of MFBL on Antioxidant Enzyme Indices in Liver of Treated Diabetic Rats
MFBL at 100 mg/kg showed antioxidant enzymes levels such as SOD, CAT and GSH as 6.69, 14.77 U/mg protein, 5.34 µM/g of wet tissue while, at 200 mg/kg showed the same as 8.40, 22.05 U/mg protein, 6.77 µM/g of wet tissue respectively. At the same time, the Metformin standard group showed the same as 8.49, 18.58 U/mg protein, 7.97 µM/g of wet tissue respectively in treated diabetic rats. Likewise, the DC group showed the same as 5.05, 11.77 U/mg protein, 4.32 µM/g of wet tissue respectively in the diabetic rats.
Discussion
The unwanted side effects associated with current diabetes therapy including insulin have led researchers to investigate new lead compounds from natural resources having antidiabetic potential. From the literature, it has been suggested that MFBL of aerial parts of B. lacera did not exhibit any toxicity effects up to the dose level of 2000 mg/kg b.w. in the models which signifies the safeness of the fractionated drug. In the MFBL, Myoinositol (10.84%) is predominated followed by Desulfosinigrin (10.13%), Ribitol (5.40%), mannose (5.40%), and 1-mono linoleoyl glycerol trimethylsilyl ether (3.03%). Myoinositol, a six-carbon cyclitol has been classified as an insulin-sensitizing agent and has also been used in type 2 diabetes and metabolic syndrome. 22 It has also been used as a supplement to baby's milk powder. d-Mannose is a simple sugar used to treat Urinary tract infection (UTI). 23 The major identified constituents in the methanol fraction might be responsible for its anti-hyperglycemic potency. These new compounds have not been disclosed earlier for their potency against diabetes. It was also confirmed by the Duke's Library of phytochemicals and ethnobotanicals. 8 In the case of the acute antidiabetic study, the MFBL at 200 mg/kg b.w. dose registered a blood glucose level from 316.17 mg/dL to 99.17 mg/dL (68.61%) at the end of 10 h. The significant (P < 0.01) lowering of blood glucose has been seen in all the fractions starting from 1 h till the end of the 10 h. Again, MFBL was subjected to a long-term subacute antidiabetic study in diabetic rats to explore its persistent blood glucose-lowering potency. MFBL at 100 mg/kg treated diabetic rats registered blood glucose levels from 305.33 mg/dL on day 0 to 115.67 mg/dL on day 30 (62.15%). While, MFBL at 200 mg/kg showed a blood glucose level from 318.83 mg/dL on day 0 to 102.00 mg/dL on day 30 (67.96% decrease) as compared with the DC. Metformin-treated groups revealed similar type of significant (P < 0.01) blood glucose lowering effect (70.14% decrease) in the diabetic rats throughout the experiment. Whereas, the DC group showed a rise in the levels of blood glucose level in diabetic rats as they are diabetic. The mechanism underlying the antidiabetic potential may be due to the sensitization of beta cells to secret insulin or may be due to the enhanced effect on gluconeogenesis. The results of the biochemical parameters revealed that the liver enzymes such as AST and ALT were significantly decreased by the MFBL at a dose-dependent manner in treatment STZ groups as comparison with the control group having diabetes. At the same time, there was a significant decrease in the levels of TC, LDL, VLDL and an increase in the levels of HDL noted by the MFBL and Metformin-treated groups than the DC group. The blood glucose-lowering effects of B. lacera may be due to the potentiation of carbohydrate metabolism enzymes in diabetic rats.
The histology of the pancreas section of diabetic animals administered with MFBL at 200 mg/kg showed better restoration of beta cells and no necrosis or inflammation was found. All the pancreas sections were stained in Halami stain for better visibility of beta cells and liver sections were stained with H & E (Figures 12 and 13). Figure 12A depicts the microscopic liver sections of DC-treated groups representing an abnormal histology of the liver section due to STZ-induced diabetes in rats. Figure 12B presents the microscopic liver sections of Metformin-treated groups and normal histology of liver cells was found, hepatic cells were restored, and no inflammation or necrosis was noted. Figures 12C and D depict the microscopic picture of the liver section of diabetic rats treated with MFBL at 100 mg/kg and 200 mg/kg revealing restoration of hepatic cells and no necrosis was found. Figure 13A shows the microscopic picture of the pancreas section of untreated diabetic rats and shows islet atrophy and a decreased amount of beta cells in the islet, moderate to severe necrosis was found. Figure 13B showed a microscopic picture of diabetic rats treated with Metformin revealing restoration of beta cells and no necrosis or inflammation was found. Figure 13 (C and D) depicted about microscopic picture of the pancreas section of diabetic rats treated with MFBL at 100 mg/kg and 200 mg/kg revealed restoration of beta cells and no necrosis or inflammation was found.

Effects of MFBL on histopathology of liver section in diabetic-treated rats (Haematoxylin and Eosin stain; 40×). H & E photomicrograph of histopathology investigation of liver tissue on 30th day of sub-acute antidiabetic study at 40× magnification (A) DC 10 mL/kg b.w. (B) Metformin 250 mg/kg b.w. (C) MFBL 100 mg/kg b.w. (D) MFBL 200 mg/kg b.w.; black arrow indicates portal vein in hepatic cells.

Halami stained photomicrograph of histopathology investigation of pancreas tissue on 30th day at 100× magnification (A) DC 10 mL/kg b.w. (B) Metformin 250 mg/kg b.w. (C) MFBL 100 mg/kg b.w. (D) MFBL 200 mg/kg b.w. The yellow arrow indicates a decrease in the amount of beta cells in islet, islet atrophy, and moderate to severe necrosis were found. The black arrow indicates restoration of beta cells and no necrosis or inflammation was found.
As we know, during a chronic diabetic case, all the carbohydrate metabolizing enzymes are significantly altered. Glucose-6-phosphate dehydrogenase and hexokinase levels are declined in diabetic patients due to the shortage of insulin levels. MFBL restored these enzymes to normal levels in a dose-dependent manner by changing the insulin activity thereby, increased the glucose-utilization (Table 4).
There is an interrelationship between glucose toxicity, insulin biosynthesis and oxidative stress. Somehow, the pancreas is one such organ in our body that may be more sensitive towards enzymes of the antioxidant system. From the evidence, insulin resistance and beta cell sensitization play a key role in oxidative stress. The increase in the electron donors of Kreb's cycle which sensitizes the membrane of mitochondria results in the production of reactive oxygen species. 24
Free radical scavenging assay in-vitro methods like DPPH and ABTS are the two most popular methods. The IC50 value of MFBL by DPPH and ABTS methods are 400 and 690 µg/mL at 517 and 734 nm respectively (Figures 10 and 11). The results of in-vitro antioxidants revealed better antioxidant potency which are scavenged by the test methanol fraction well. GSH is one of the major thiol groups of nonprotein in living creatures. It has an important role in modulating the membrane function of secretion by the beta cells and intracellular state. Treatment of diabetic rats with B. lacera restored the levels of GSH to almost normal levels in the treated diabetic rats that reduced the oxidative stress. SOD is an antioxidant enzyme that catalyses the superoxide radical into hydrogen peroxide and CAT is another antioxidant heme protein that converts hydrogen peroxide into water and O2. In the present study, the SOD, and CAT in the liver were found to be decreased in DC group because of failure in the defence system of antioxidants to combat ROS-facilitated destruction. There is an increase in the in-vivo antioxidant enzymes such as SOD, CAT and GSH by the test fractions as compared with the DC group (Figure 14). Thus, the study on antidiabetic effects of B. lacera embodied in this study justifies the ethnomedicinal use of this plant scientifically.
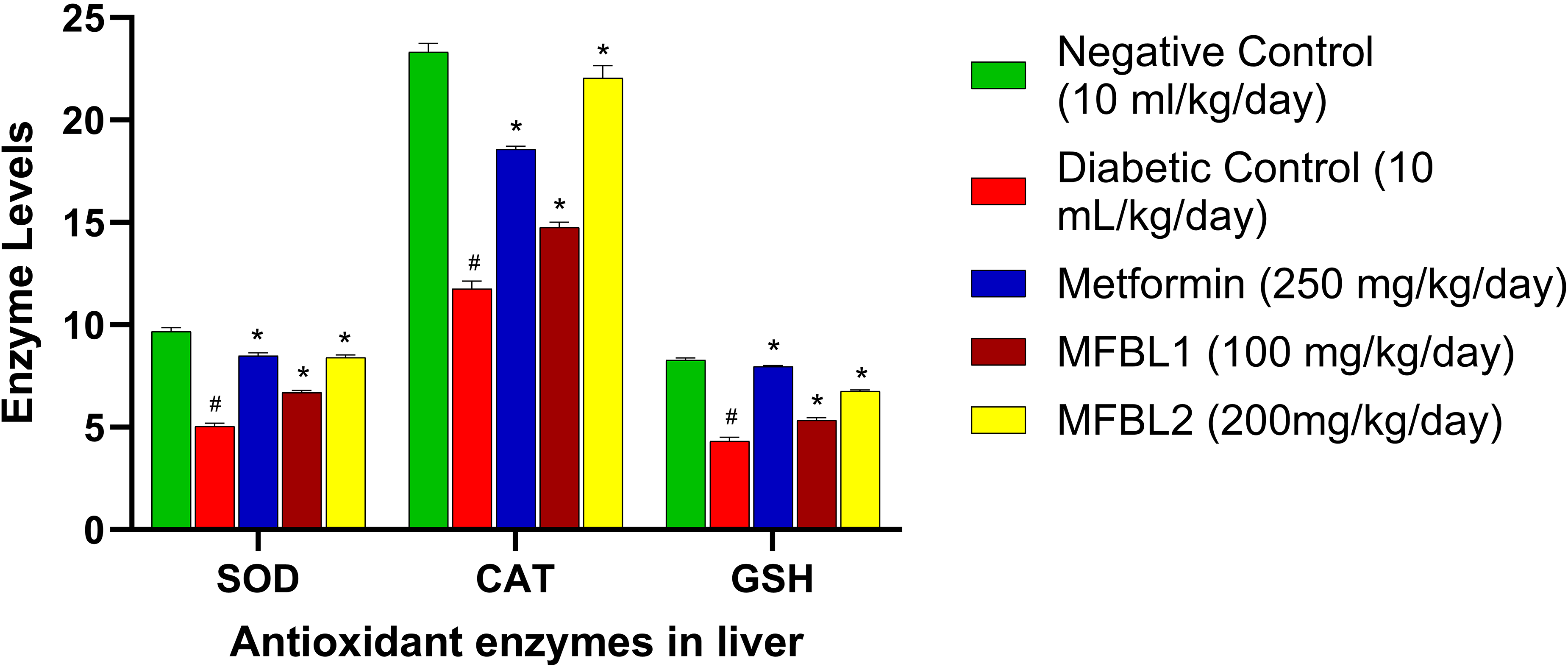
Effects of MFBL on antioxidant enzyme indices in liver of treated diabetic rats. The values are presented as mean ± SEM (N = 6). ANOVA with Tukey's multiple range test is used in statistical analysis (*P < 0.01 compared to the NC and DC group and #P < 0.01 compared to the NC group.
Conclusion
The current research work concluded that MFBL showed significant antidiabetic potential in STZ-induced diabetic rats as compared with the control (untreated diabetes) group with regards to the improvement in body weight, biochemical parameters, insulin levels, and carbohydrate metabolizing indices in a dose-dependent manner. It also revealed the improvement in the anatomy of damaged liver and pancreas sections after the treatment for 30 days at prescribed dose levels than the control one. The possible mechanism behind the antidiabetic potential of plant B. lacera may be due to the inhibition of carbohydrate metabolizing enzymes, and enhancement of insulin release from the pancreas. The improvement in the lipid profile signifies the protective effects against dyslipidemia. The promising phytochemicals like Myoinositol and Desulfosinigrin might be responsible for its antidiabetic potential due to their synergistic effects. This is the first report on the antidiabetic potential of B. lacera in a methanol fraction using the STZ diabetes model. The limitation of our study is that the molecular mechanism of action behind the anti-diabetic potency of the plant B. lacera has to be justified. As the study reveals that B. lacera possesses significant anti-diabetic properties, further analysis is required to know the exact molecular mechanism of action by using bioinformatics such as network pharmacology and molecular docking with experimental validation.
Footnotes
Acknowledgment
The authors are grateful to the Dean of the School of Pharmaceutical Sciences, Siksha ‘O’ Anusandhan Deemed to be University for providing lab facilities for carrying out research works.
Ethical Approval
In accordance to OECD guidelines, ethical permission was taken from Institutional Animal Ethical Committee before conducting in-vivo animal experiments with letter number 57/SPS/IAEC/SOAU. The study protocol was passed by the university animal ethical committee (IAEC No. 1171/C/08/CPCSEA).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the OECD guidelines, approved by Institutional Animal Ethical Committee with approval number IAEC No. 1171/C/08/CPCSEA.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Researchers Supporting Project Number (RSPD2025R770), King Saud University, Riyadh, Saudi Arabia.