Abstract
Background and Objectives
Brown seaweed, Sargassum muticum, can grow up to a maximum length of 10 m. This study aimed to create novel bimetallic nanoparticles (NPs) by utilizing the entire plant extract. The bimetallic green-Ag-CuNPs was synthesized by using these substances extract of Sargassum muticum algae, copper oxide (CuO), silver (AgCl) by a reduction procedure and studied its toxic action against human colon cancer (HCT-116) cells.
Methods
SEM and TEM were used to measure the NPs’ size prior to the treatment of g-Ag-Cu NPs. The g-Ag-Cu nanoparticles had a circular shape and measured 46 ± 1 nm in size. MTT and NRU tests were used to assess the cytotoxicity of g-Ag-Cu NPs on HCT-116 cells.
Results
The results showed that the cytotoxicity of NPs increased in a concentration-dependent manner. The median inhibitory concentration (IC50) for HCT-116 cells at 24 h was determined to be 66.4 μg/ml based on the MTT result. At 50 µg/ml of g-Ag-Cu NPs, HCT-116 cells generated large amounts of intracellular ROS, induced caspase 3/7. Using Rhodamine123 labeling, the impact of NPs on mitochondrial membrane potential in HCT-116 cells was evaluated and it was highly compromised at 50 µg/ml of g-Ag-Cu NPs. Expression of apoptotic protein was assessed by using protein array methods and it was down and up regulated as per concentration of exposure of g-Ag-Cu NPs.
Conclusion
Our findings support the g-Ag-Cu NPs’ lethal effects on human colon cancer cells. We believe that because this nanoparticle increases cytotoxicity and triggers apoptosis, it may be useful as a supplement in the treatment of colon cancer.
Introduction
Nanoparticles have been utilized to make a wide range of items since nanotechnology first appeared and expanded quickly in the twenty-first century. As a result, nanomaterials have been incorporated into the industrial, agricultural, and biomedical sectors, among other fields of human endeavor. 1 The toxicity of nanomaterials is a controversial topic due to their varying sizes. Recently, however, there has been a greater focus on using bimetallic NPs because of their antimicrobial qualities and for cleaning contaminated groundwater and soil. 2 A common bimetallic alloy that is produced via chemical co-reduction from metal salts. Numerous studies have demonstrated the enhanced antibacterial activity of Ag-Cu NPs. Applications of bimetallic nanoparticles are numerous and include biological, environmental, chemical, and physical sectors. They also exhibit potent catalytic characteristics. 3 Bimetallic nanoparticles, such CuO-ZnO NPs, have been shown by certain studies to exhibit powerful cytotoxicity, antioxidant, and antibacterial properties, underscoring their potential for use in a range of therapeutic interventions. By employing it as a raw material for the synthesis of NPs and activated biochar as support, Sargassum spp. have significant potential to address environmental issues. 4 Taner et al 5 state that their antibacterial efficacy against Escherichia coli was enhanced when paired with Ag or Cu NPs. A synergistic effect was suggested as the cause of these multi-element antibacterial compounds increased antibacterial effectiveness. According to Tapiero et al, 6 Cu is an essential trace element required for human health and metabolism. There is a suggestion that nanoparticles are more harmful than materials in bulk. It is unclear how exactly nanoparticles cause toxicity, but some recent research has suggested that it could be due to a variety of factors, including increased surface area, permeability into cells, and accumulation within cells and organisms, 7 as well as changes in apoptosis, membrane damage, inflammation, and DNA damage. 8 Many potentially helpful substances, including steroids, flavonoids, fucoxanthin, sulphated polysaccharides, and terpenoids, have been discovered to be present in sargassum species. 9 Previously, extracts from the marine macroalgae Sargassum muticum were used to produce Ag NPs. 10 Researchers have used marine algae for the synthesis of nanoparticles in addition to its current uses in the production of human food, fertilizers, and cosmetic components.10,11
In this experiment, we thus investigated the cytotoxicity and apoptotic properties of g-Ag-CuNPs on HCT-116 cells over the course of 24 h. HCT116 cells are employed in numerous biomedical investigations concerning the growth of colon cancer and the inhibitors that accompany it. After breast cancer, colorectal cancer is the second most frequent cancer in Saudi Arabia. 12 NPs-mediated toxicity is caused by a number of routes, the most prominent of which is the excessive production of ROS in stressed living tissue. Apoptotic reactions, oxidative stress, and genotoxicity responses are the main toxicity mechanisms in nanoparticles. The purpose of this study was to evaluate the g-Ag-CuNPs cytotoxic effects on human colon cancer cells.
Materials and Methods
Substances and Reagents
Unless otherwise noted, every chemical utilized in this investigation was analytical grade and was acquired from Sigma-Aldrich (St. Louis, MO, USA), United States Biological (Salem, MA, USA), and Merck (Darmstadt, Germany). Kits and additional chemicals for the current experiment were purchased from Cayman Chemical in Ann Arbor, Michigan, USA, as well as local markets.
Collection and Preparation of S. muticum Extracts
Sargassum muticum, or brown seaweed, was collected from Dammam, Saudi Arabia's sea water locations. To get rid of the muddiness, it was rinsed in the lab with water. The cleaned algae were allowed to dry at room temperature for a week in the shade before being ground into a powder using a mortar and pestle. The dried form of S. muticum was pulverized into a 50 g powder using an electric mixer. For extraction of aqueous extract of S. muticum, we used 50 g powder of S. muticum and dissolved it in 200 ml of water in conical flask and kept it at 4 °C for overnight. Next day it was boiled at 100 °C for one hour. Then it was filtered through filter paper to obtain aqueous extract.
Green Synthesis of Copper and Silver Nanoparticle
Silver nitrate (AgNO3, 99%) and copper chloride (99%) were purchased from Sigma-Aldrich. In order to produce copper-silver nanoparticles (Cu-Ag NPs), roughly 10 ml of S. muticum aqueous extract (50 g powder S. muticum /200 ml of water) was combined with 20 ml of a solution containing 1 mM silver nitrate and 20 ml of copper chloride. For 60 min, the Cu-Ag NPs solution was incubated at room temperature. The conversion of AgNO3 and CuCl2 to Cu-Ag NPs was indicated by the emergence of brown color. The suspension is stored for further investigation in a dark environment.
Determination of g-Ag-CuNP Properties
The g-Ag-CuNPs were physically characterized using TEM and SEM (JEOL Inc., Tokyo, Japan). Using zeta sizer (Zetasizer Nano ZS, ZEN3600 and Malvern, UK) and dynamic light scattering instruments, we have measured the zeta potential and size of g-Ag-CuNPs in water.
Cell Culture
We acquired human colon cancer (HCT-116) cell lines from an American type culture collection located in Manassas, Virginia, USA. The cells were cultivated in Dulbecco's modified Eagle's medium (DMEM), enhanced with 10% fetal bovine serum (FBS) and 1% antibiotic. The cells were kept in an incubator with 5% CO2 at 37 °C.
Treatment of g-Ag-CuNPs
For a 24-h treatment, g-Ag-CuNPs were suspended in a cell culture medium and diluted to the proper concentrations (0, 5, 10, 25, 50, 100, and 150 µg/ml). A sonicator probe was then used to sonicate the suitable dilutions of g-Ag-CuNPs. To measure cytotoxicity, oxidative stress, apoptosis, and protein expression, the following treated cells were taken out. Every experiment used control cells, which were not exposed to NPs.
Determination of IC50 24 h of g-Ag-CuNPs
Using GraphPad Prism 8.0 software, the half maximum inhibitory concentration (IC50) of g-Ag-CuNPs on HCT-116 cells for 24 h was determined using the MTT assay after the experiment was repeated three times (Figure 2). IC50 is 66.4 µg/ml.
The three test concentrations of g-Ag-CuNPs, namely concentration I (1/10 th of LC50 = ∼6.64 µg/ml), concentration II (1/2 nd of LC50 = ∼33 µg/ml), and concentration III (3/fourth of LC50 = ∼50 µg/ml), were estimated based on the IC50-24 h value.
MTT Assay
According to Ali's 13 descriptions, the MTT assay was utilized to look into cytotoxicity HCT-116 cells after exposure of NPs for 24 h. HCT-116 cells were grown to 80% confluency by seeding 8 × 104 cells/well in a 96-well plate with 100 µl of the complete medium. Following an overnight incubation period, the medium was extracted, and cells were incubated for 24 h in 100 µL growth culture media that included a range of NP concentrations (0, 5, 10, 25, 50, 100, and 150 µg/ml). Subsequently, 100 µl of MTT solution (3 mg/ml) was added to each well as a replacement for the medium, and the mixture was incubated for 4 h at 37 °C in the dark.
After 3-4 h of incubation, the MTT solution was removed, and formazan crystals formed. The crystal was solubilized in (100 µl/well) DMSO and gently shacked for 15 min. The plates were read using a microplate reader (Synergy-H1; BioTek) at a wavelength of 570 nm. The assay was performed in triplicate with 10 replicates per sample. The survival rate of the cells was calculated using the following formula: The survival rate (%) = Optical Density (OD) values of the treated samples/OD value of control × 100.
NRU Assay
The g-Ag-CuNPs’ cytotoxicity was evaluated using the NRU test in accordance with Almutairi et al methodology. 1 A 96-well plate was used to seed HCT-116 cells at an initial density of 8 × 104 cells/well. Following a 24-h incubation period, the cells were treated with several concentrations of gCu-AgNPs (0, 5, 10, 25, 50, 100, and 150 µg/ml) until they reached a suitable confluence (∼70%-80%). Following a 24-h gCu-AgNP treatment, the medium was disposed of and 100 µl of NRU solution was combined with each well before being incubated for a further three hours. Following incubation, cells were cleaned using a 0.5% formaldehyde and 1% calcium chloride (CaCl2) solution.
The accumulated dye was extracted with 50% ethanol containing 1% (v/v) acetic acid. The absorbance was recorded at 540 nm using a microplate reader (Synergy-H1; BioTek). The cytotoxicity rate of the cells was calculated using the following formula: Cytotoxicity (%) = (OD in control cells − OD in treated cells)/(OD in control cells) × 100.
Evaluation of Intracellular Reactive Oxygen species
Reactive oxygen species (ROS) generation was assessed in HCT-116 cells after exposure to different concentrations of gCu-AgNPs by using 2,7-dichlorofluorescin diacetate (DCFH-DA) dye as a fluorescence agent based on the method. 1 ROS generation was studied by two methods: fluorometric analysis and microscopic fluorescence imaging. For fluorometric analysis, cells (7 × 104 per well) were seeded in 96-well black bottom culture plates and allowed to adhere them for 24 h in a CO2 incubator at 37C. After discarding the old medium, the HCT-116 cells were incubated in the medium containing various concentrations nanoparticles for 24 h. On the completion of respective exposure periods, cells were incubated with pre-prepared 1X DCFH-DA (10 mM) by adding µL of dye solution to 99 µL of DMEM in dark for 60 min at 37C. The reaction mixture was aspirated and replaced by 200 mL of PBS in each well. The plates were kept in a shaker for 10 min at room temperature in the dark. Fluorescence intensity was measured using a microplate reader at excitation wavelength 485 nm and at emission wavelength 535 nm and values were expressed as a percent of fluorescence intensity relative to control wells. A parallel set of cells (7 × 104 per well) was analyzed for intracellular fluorescence using an upright fluorescence microscope equipped with CCD cool camera (Nikon Eclipse 80i equipped with Nikon DS-Ri1 12.7-megapixel camera).
Determination of Mitochondrial Membrane Potential Assay
After NP treatment, the mitochondrial membrane potential (MMP) in HCT-116 cells was measured using Rhodamine 123 (Merck, USA). Because the mitochondria of living cells maintain their electron potential, rhodamine 123 tends to accumulate there and fluoresces more intensely than when it disperses throughout the cytoplasm of deceased cells, where the mitochondrial membrane has been destroyed.
For this purpose, HCT-116 cells were grown in black plate and eight chambered slide and after treatment cells was washed twice with PBS and incubated with 5 μg/mL of Rhodamine 123 (Merck, USA) for 30 min at 37 °C in the dark. Then the fluorophore in the cells was measured at 540 nm by using microplate reader and fluorescent images were taken by CRCL's LSM 780 NLO confocal microscope.
Measurement of Caspase-3/7 Enzyme
Following NP treatment, the caspase-3/7 enzyme in HCT-116 cells was assessed using a caspase-3/7 fluorescence assay kit (Item no. 10009135, www.caymanchem.com, USA). The enzyme caspase-3/7 is crucial to apoptosis. It catalyzes the particular cleavage of cellular proteins and activates death protease.
Determinations of Protein Expression
Apoptotic proteins have been identified applying a protein profiler array (RayBio, Human apoptosis Antibody Array C1 Kit) to investigate a possible mechanism of apoptosis induced by NPs in HCT-116 cells. NPs were applied to the cells for a whole day. Bradford's reagent was employed to assess the protein content of specimens. 14 After extracting proteins (250 μg) from three exposed and control cells, the human apoptosis antibody array was incubated with the samples for an entire night. Image Lab (6.0) software was used to quantify the apoptosis array data of pictures of the membranes using a Bio-Rad ChemiDoc XRS+ equipment. After examining the membrane results using the image lab software, the signal-fold expression levels of each sample were assessed in accordance with the manufacturer's guidelines.
Statistical Evaluation
The SPSS 26.0 program was used to carry out a one-way ANOVA test in order to identify any statistical differences between the exposed and control groups. In comparison to the control, the values of *P .05 and **P .01 were deemed statistically significant. For every experiment, a minimum of three separate experiments were conducted in duplicate.
Results
Characterization of g-Ag-Cu NPs
The synthesis of g-Ag-Cu NPs formation were confirmed by UV vis spectrum (Figure 1A), SEM-EDS data (Figure 1B, C). For instance, we measured Ag by 5.729 keV and Cu by 8.040 to confirm Ag-Cu nanoparticle production (Figure 1B). g-Ag-Cu NPs typically have a size of 46 ± 1 nm (Figure 1D). g-Ag-Cu NPs in suspension were measured for particle size and stability using DLS and zeta potentials. g-Ag-Cu NPs were measured to be 209.4 ± 2 nm in size. −0.840 mV was used to measure the net charges, or zeta potential, on the surface of the assessed NPs in aqueous solution.
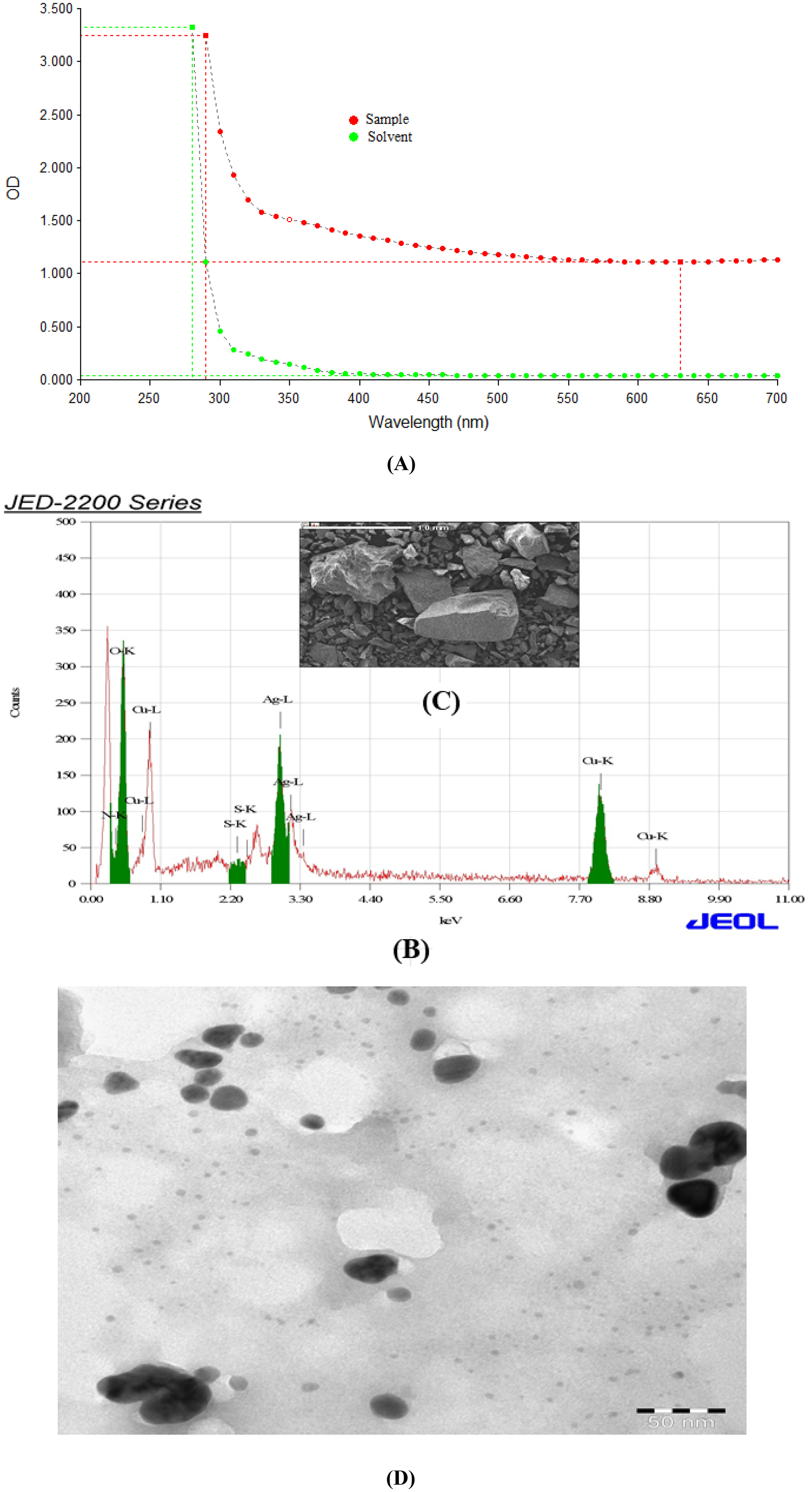
Characterization of gCu-Ag NPs (A) UVvis spectrum (B) EDX spectrum (C) image of gCu-Ag NPs by scanning electron microscope (D) image of g Cu-Ag NPs by transmission electron microscope.

Cytotoxicity gCu-Ag NPs in HCT cells for 24 h, based on the (A) MTT assay (B) NRU assay. Each value represents the mean ± SE of three experiments. n = 3, *P < .05, **P < .01, versus control.
IC50 Value
Based on the results of the MTT test, the dose-response curve graph produced by the program Origen Pro 8.5 was used to calculate the IC50 value for g-Ag-Cu NPs on HCT-116-116-116 cells at 24 h. We found that the g-Ag-Cu NPs’ IC50 value for HCT-116-116-116 cells was 66.4 μg/ml after 24 h.
Cytotoxicity
The number of viable cells, or metabolically active cells that change the tetrazolium salt (MTT) into a purple formazan crystal, determines the total cell viability. HCT-116 cells were exposed to several concentrations of g-Ag-Cu NPs (0, 5, 10, 25, 50, 100, and 150 µg/ml) for a duration of 24 h. The HCT-116 cells’ cell toxicity significantly increased, as shown by the results (1.6%, 5.44%, 11%, 16%, 18.58%, 41.3%, 59.6% and 72.7%) (Figure 2A). Moreover, a dose-dependent decline in cell viability was observed (Figure 2A).
The ability of live cells to ensnab and bind the neutral red dye in lysosomes is the basis for the neutral red intake experiment. The NRU assay results for HCT-116 cells demonstrate that there was a concentration-dependent decrease in cell viability due to g-Ag-Cu NPs (Figure 2B). The findings showed that the cell toxicity of HCT-116 cells increased by 1%, 2.04%, 6%, 9.03%, 13.58%, 15.3%, 39.6% and 50.7% (Figure 2B).
Reactive Oxygen species (ROS)
Using the 2′, 7-dichlorofluoresceindiacetate (DCFH-DA) dye as a fluorescence agent, the amount of ROS produced was quantified. Following a 24-h exposure of HCT-116 cell lines to varying concentrations of g-Ag-Cu NPs (6.64 µg/ml, 33 µg/ml, and 50 µg/ml), a ROS test was conducted. The outcome for HCT-116 cells indicates that the effect of reactive oxygen species is rising in treated cells as compared to control (109%, 119%, and 140%) (Figure 3A). When compared to other concentrations and the control, the rise in ROS at 2/3 of the IC50 concentration was noteworthy. ROS expression in HCT-116 cells was seen in Figure 3B.

(A) Percentage change in intracellular ROS generation in the HCT cells. (B) Induction of ROS levels in HCT cells after exposure with gCu-Ag NPs for 24 h. Each value represents the mean ± SE of three experiments. n = 3, (*P < .05, **P < .01) control.
Determination of MMP
The compromization of MMP in HCT-116 cells due to the action of g-Ag-Cu NPs was identified using the fluorescent agent Rhodamine 123. The results for HCT-116 cells show that, in comparison to control, treated cells are losing more MMP (Figure 4A, B). The drop in MMP at 2/3 of the IC50 concentration was greater than that of the other concentrations and the control.

(A) Images representing loss of MMP in HCT cells after exposure to different concentrations of g Cu-AgNPs for 24 h. (B) Induction of MMP in HCT cells. Each value represents the mean ± SE of three experiments. n = 3, (*P < .05, **P < .01) versus control.
Enzyme Caspase-3/7 Activity
The endo-protease family of enzymes known as caspases-3/7 plays a crucial role in the regulation of inflammation and apoptosis in cells. For a full day, the cells were exposed to varying concentrations of g-Ag-Cu NPs. Subsequently, the experiment was run, and the findings indicate that the amount of caspase-3/7 enzyme increased in a dose-dependent way, and that the activity of the enzyme was higher in treated HCT-116 cells than in control cells (Figure 5).

Caspase-3/7 activity in HCT cells after exposure of gCu-AgNPs. Each value represents the mean ± SE of three experiments *P < .05 and **P < .01 versus control.
Expression of Proteins
As seen in Figure 6A, B, apoptotic proteins were present in the cells following a 24-h exposure to NPs. In this case, we looked into the primary proteins involved in cell death using the human apoptosis protein array. A substantial change in the protein was discovered. Various proteins have been demonstrated to exhibit either up- or down-regulation based on their function within the inflammatory, apoptotic cascade. Additionally, it was shown that bcl-2 was downregulated, and some of these proteins, including bax, bcl2, bad, and caspase-3 and caspase-7, were irregularly upregulated (Figure 6A, B).

Equal quantity protein extracts from control and treated HCT-116 cells were analyzed using the antibody array. (a) The spots indicate the expression of proteins in HCT-116 cells after treatments of Cu-Ag NPs for 24 h. (b) The chemiluminescent intensities were quantified by densitometry. Representative bar graph of the apoptotic unregulated and downregulated protein. A Positive control was used normalize the results from different membranes.
Discussion
Because of its numerous scientific and technological uses, nanotechnology is one of the newest technologies in creative research. Humans are not aware of the increased risk of exposure to nanoparticles, which can enter biological systems through many routes, which is a significant and concurrent consequence of these growing nano-based applications. In instance, nanoparticles can morph in size over a range of mediums and offer a measurable increase in surface area in comparison to mass. Algae contain phytochemicals that bind metal nanoparticles and function as reducing agents, including various amino and hydroxyl group compounds. Green synthesized g-Ag-Cu NPs produced by S. muticum include Cu and Ag elements, according to EDX examination. The elements Cu and Ag exhibit spectra between 2.80 and 2.86 keV and 7.4 and 7.6 keV, respectively. According to Kannan et al, the synthesis of AgNPs from AgNO3 salts was facilitated by the presence of additional functional groups in U. rigida. It is believed that nanoparticles produced from naturally occurring materials with strong antioxidant activity are increasingly effective at reducing cell toxicity. According to Milledge et al (2016), seaweed has potential applications across a range of fields. Alpha and gamma tocopherol, fucoxanthin, a carotenoid pigment, and other carotenoids and phenolic compounds are common antioxidant components discovered in S. muticum.15–17 The cytotoxicity of nanoparticles is often assessed using the MTT, NRU, and blue tests. This result showed that even at the lowest concentration, both tests indicated that g-Ag-Cu NPs reduced the viability of HCT-116 cells. After 24 h of incubation, green-synthesized g-Ag-Cu NPs cause cell death in the HCT-116-116-116 cell line by inhibiting cell growth in a concentration-dependent manner (1-150 μg/ml). Using the MTT and NRU assays, a concentration-dependent reduction in cell viability was seen.
After a 24-h exposure, the median lethal concentration (IC50) of g-Ag-Cu NPs was determined to be 66.4 μg/ml in the current investigation. This result is largely consistent with previous research which employed Andrograohis paniculata's aqueous leaf extract to green synthesize ZnO NPs. 18 According to Alshahrani et al, 19 the increased production of ROS triggers the antioxidant defense system, boosting the activities of SOD and catalase, which catalyze the conversion of superoxide to hydrogen peroxides and hydrogen peroxide to oxygen and water, respectively. According to certain researchers, newly developed bimetallic nanoparticles were produced using green synthesis techniques and demonstrated cytotoxicity, apoptosis, and anti-cancer properties in malignant cells.20–22
ROS encourages growth and development in a healthy setting, but they can also have pathological effects, such as cell death, in a stressful environment. Too much ROS within the cell induces apoptosis. In our investigation, we found that g-Cu Ag NPs mitigated the growing impact of ROS in treated cells relative to control for HCT-116 cells. Additionally, the capacity of Cu-Ag-NPs to conformably increase intracellular ROS levels in human colon cancer cells was evaluated using the DCFHDA assay. Numerous cellular and molecular biological traits, including caspase cascade activation, cell shrinkage, and DNA breakage, are displayed by apoptotic cells. Caspases 3/7, which are essential for DNA fragmentation and apoptotic chromatin condensation in all examined cell types, are the hallmarks of apoptosis. Our study's findings showed that HCT-116 cells were experiencing more DNA fragmentation. Additionally, a protein array used to evaluate apoptosis in HCT-116 cell lines indicated a significant up- and down-regulated apoptotic protein at all exposure concentrations, suggesting that g-Cu-Ag NPs cause apoptosis to rise in a dose-dependent way.
Conclusion
This is likely the first study to highlight the substantial anticancer potential of Cu AgNPs synthesized by marine algae species, especially Cu-Ag-NPs made using S. muticum against malignant HCT-116-116-116 cells. To further elucidate their anticancer activity using animal models, however, in vivo investigations are necessary. Future research should also address issues such as genotoxicity, therapeutic window, safety profile, pharmacokinetics, and antibacterial resistance that are associated with biosynthesized Cu Ag-NPs.
Footnotes
Acknowledgments
This research was supported by Researchers Supporting Project number (RSP2024R27), King Saud University, Riyadh, Saudi Arabia.
Data Availability Statement
The original contributions presented in the study are included in the article; further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Researchers Supporting Project number (RSP2024R27), King Saud University, Riyadh, Saudi Arabia.
Statement of Informed Consent
Not applicable.
Statement of Human and Animal Rights
Not applicable.
