Abstract
Introduction
Chitin is considered the second most plentiful biopolymer in nature after cellulose. 1 It is a linear amino polysaccharide comprised of β-(1-4)-connected 2-deoxy-2-acetamido-D-glucose units. 2 The main source of chitin is crustacean shell derived from shrimp and crab. 3 Chitin is considered an important material because of its various characteristics, such as biodegradability, biocompatibility, non-toxicity, low immunogenicity, and thermal stability.4,5 Therefore, chitin and its derivatives are widely used in cosmetic, 6 food, 7 agricultural, 8 tissue engineering, 9 waste water treatment, 10 and packaging material applications. 11 It is reported that shrimp accounts for about 45% of processed seafood. 12 The shrimp processing industry produces massive amounts of by-products, such as shrimp shells. These shrimp by-products are usually applied for low-value animal feeds and biological fertilizers. 13 Therefore, it is particularly critical to convert by-products into high-value products. Shrimp shells are mainly composed of chitin, proteins, and minerals. 14 Chitin is extracted from shrimp shells by demineralization and deproteinization procedures.
Chitin is the second most abundant biopolymer after cellulose in the tissues of some aquatic animals. Chitosan has attracted consideration due to unique cationic nature, which is obtained after process called N- deacetylation of chitin. Chitin and chitosan are commercial attracted due to their physiological compatibility, non-toxicity, bio digestibility, and adsorption and chelating capacity. 15 The characteristics of chitosan with industrial and biotechnological applications such as food, medicine, agriculture, aquaculture and environmental engineering depend on the type and quality of chitin and chitosan from different sources and different concentrations of a wide range of acid and alkali used and the method of extracting chitin and chitosan from shell waste. Nguyen Van Toan 15 reported that chitin and chitosan produced with 3% HCl for demineralisation, 4% NaOH and 50% NaOH for deacetylation in good quality chitin and chitosan.16,17 The chemical method, this study was designed to compare quality of chitin and chitosan obtained from the shrimp wastes using reliable process.
Scientific Hypothesis
The quality of chitosan and chitin produced is related to the type of processing method and the initial quality of freshly caught shrimp.
Materials and Methods
This research work was carried out in 2023 at Fisheries Laboratory of University, Iran. The shrimp transferred to the Laboratory as fresh shrimp in crushed ice.
Chemicals
All chemicals were purchased from Behbahan Company in Iran with good grade quality.
Instruments
The charcoal, water, meter, scale, oven, electric furnace, Chinese container, Petri dish, distilled water, homogenizer, and pH meter were used in this research.
Description of Experiment
Sample Preparation
For this purpose, 8 kg of western shrimp L. vanamei (Vanami) (Image 1) was purchased from Behbahan fish market, which found 5.1 kg pure meat. Its waste was 710 g, including wet shell of shrimp, which after drying and grinding with an grinder, 265 g powder was obtained. Therefore, this type of shrimp had 58% of meat and 42% of wastes. The shell of shrimp was dried and ground into powder for extraction of chitin and chitosan.

Shrimp L. vanamei (Vanami).
Used Reagents
HCl and NaOH solutions in different concentrations used in this research.
Design of Experiment
The First Stage of Chitin Production from Shrimp Shell Powder
220 g Vanami shrimp shell powder was mixed in 1100 ml HCl 3 M (1 to 5 ratio) and kept at laboratory temperature (28°C) for 16 h. Then remaining acid was removed with frequent washing with distilled water until reaching pH 7. 15
The weight of dried chitin in the first stage was 112.6 g.
The Second Step of Chitin Extraction
To remove protein from shrimp shell powder, 90 g of raw chitin powder produced in initial stage was mixed in a NaOH 4 M solution at a ratio of one to five and then kept for 20 h at a temperature of 28°C. Then it was washed well with distilled water and filtered and dried. The 68 g dry chitin powder was obtained. After drying, a sample was taken to measure moisture, ash and pH. 15
The Production Stage of Chitosan from Produced Chitin
50 g produced chitin powder from second step was mixed in 50% NaOH solution at a ratio of 1 to 10 and kept in an oven for 16 h at a temperature of 60°C. Then it was washed well with distilled water, filtered and dried, and 37 g dried chitosan powder was obtained.
After drying, a sample was taken to measure moisture, ash and pH. 15
Physicochemical and Functional Properties of Produced Products
Calculation of Degree of Chitosan Deacetylation
For this purpose, acid and base titration method was performed. About 2 g of chitosan was dissolved in 60°C of 0.1 M HCl and mixed with 6 drops of methyl orange reagent at room temperature, then, this mixture was titrated with 0.1 M NaOH solution until solution turned orange. The acetylation degree was calculated using the following formula.
Macronutrient Composition Analysis
Moisture Content Determination
Moisture percentages of samples were determined on wet basis.
18
The samples were kept in an oven for 2 h at 103°C. The percentage moisture content was difference between weight wet and oven dried samples
19
and expressed as
Ash Content Determination
Chitin and chitosan ash content were calculated from each samples according to method. 20 Three samples of chitosan and three samples of chitin, each 2.0 g, were placed in cooled crucibles. The samples were heated in a muffle furnace at a temperature of 550°C for 7 h. The crucibles were cooled at temperature of less than 150°C and then placed in a desiccator. The desiccator were allowed to cool and then weighed. The amount of ash in each sample was calculated and average value was recorded. 18
Ash percentage was calculated as
pH Determination
The pH levels chitosan and chitin samples were measured using digital pH meter model PHS_550 device.
Percentage of Yield
The chitin and chitosan yield were calculated based on dry weight of chitin and chitosan powder relative to wet weight of dried shrimp shell.
21
The percentage yield of chitin and chitosan was determined by following formulas. (1) and (2):
Chitin extraction yield; % = Dried extracted chitin weight (g) / Dried shrimp shell weight (g) × 100 Chitosan extraction yield; % = Dried extracted chitosan weight (g) / Dried shrimp shell weight (g) × 100
Statistical Analysis
All experiments were carried out in triplicate, Average values and standard deviations were reported. Mean data were analyzed using ANOVA at level p < .05.
Results
Results of present study showed in Figures 1–3 and Table 1. The moisture, ash and pH values are three important factors to control the quality of produced chitin and chitosan respectively.
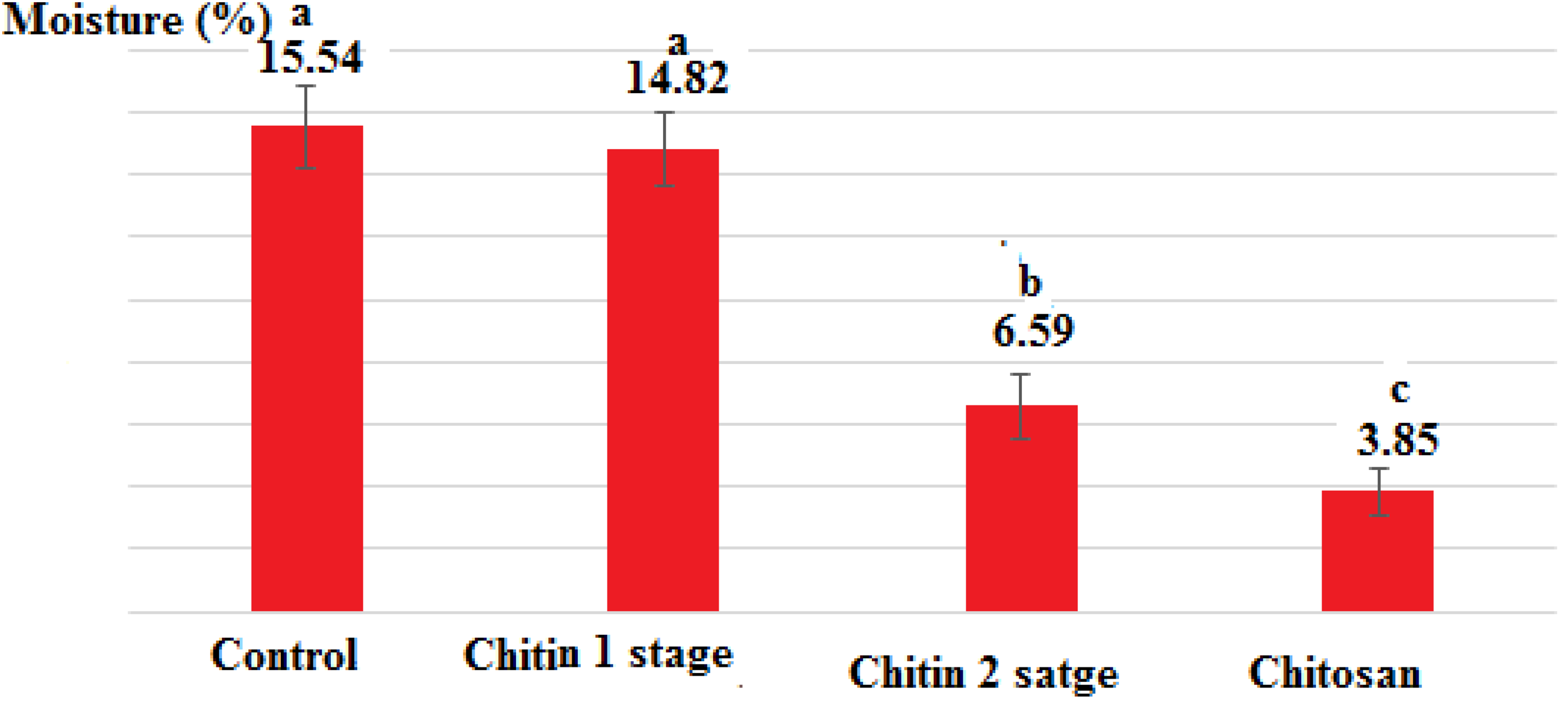
Moisture percentage of chitin and chitosan extracted from shrimp L. vanamei shells.

Ash percentage of chitin and chitosan extracted from shrimp L. vanamei shells.

pH values of chitin and chitosan extracted from shrimp L. vanamei shells.
Analysis of Proximate Compounds and pH Values of L.

ND: Not detected. Different letters in each column indicate significant differences.
In the first stage of chitin production, the percentage of moisture was lost by about 1% compared to the control, while in the second stage, more than 50% of the moisture was lost compared to the first stage and the control, which was agreed with results of another researcher. 15
In the second stage of chitin production, the percentage of ash lost by about 90% compared to the control which was agreed with results of another researcher. 15
Discussion
The composition of shell waste, chitin and chitosan obtained from L. vanamei shrimp source was given in in Table 1 and also Figures 1, 2 and 3. Hossain and Iqbal 20 reported moisture of shrimp shell waste 69.3%. The present study showed that moisture content in shrimp wastes was 15.54%. The variation in moisture content may be to the reason, Season, climate, weather and raw material condition. 21 In hot and humid climates, materials dry slower at room temperature and the percentage of moisture is higher. If the raw materials contain a high percentage of moisture, it loses moisture slowly. The lower result of moisture content of raw material was mainly due to collection procedure. The moisture content of chitin and chitosan obtained from shrimp were 6.59% and 3.85% respectively. These results showed moisture content in chitin was lower than results obtained from mussel shell (12.90%) by Abdulkarim et al 21 the reason may be due to drying conditions of chitin, where experimental chitin was dried in the hot air oven. According to results of Sukumaran et al, 22 commercial chitosan products should contain less than 10% moisture. The result of chitosan moisture content in present study was 3.85%. The results of study are evident to prove moisture content of chitosan which was within acceptable range.
The level of ash shows efficiency of demineralization (DM) step for removal of minerals. The ash content of fresh shells in present study was 4.67%. The mineral content of shrimp shell was high, which was caused by the type of species and its habitat and feeding conditions. It was reduced to 0.06%, after demineralization step. The mineral content of shrimp shells found high, because of hard structure due to strong bond between chitin and minerals. It elucidates that acid concentration and duration for demineralization was sufficient for shrimp shell waste. Chitosan with less than 1% ash content considered as a high quality grade. 23 In present study, yield of chitosan showed 54.41% compared to squilla (0.58%) and crab (0.76%) reported by some researchers. The residual ash content is considered as an important parameter affects solubility, consequently reducing viscosity. 24 Criteria to compare the quality of chitin and chitosan that were obtained from shrimp waste in present study included the control of ash, moisture content, pH value, yield percentage and degree of acetylation. Authors affirm that chitosan obtained from shrimp shell waste had a higher degree of deacetylation and good yield, due to used HCl and NaOH suitable percentages compared to other researchers.
Degree of Deacetylation
The chitin is usually obtained by strong alkaline hydrolysis at high temperatures. Concentrated NaOH (40%–50%) is commonly used.3,6 No and Lee 23 showed range of degree of deacetylation (DD) of chitosan from 56% to 99% with an average of 80% considered as high quality chitosan. The shrimp chitosan showed superior quality with DD value 49%. The lower DD values of chitosan obtained from shrimp wastes in present study was in accordance with findings of No and lee 23 and Toan 15 respectively. The yield of shell wastes was around 42% on wet weight basis, but chitin yield was 25.66% for shrimp wastes. These results were concurred with shrimp shell chitin estimated by Trung and Phuong. 24 The chitosan yield from shrimp wastes in present study showed better result after deacetylation process.
The FAO 25 recommended new approaches to fisheries management which included conservation, environmental, social, and economic considerations. The quantity of processing waste generated around 80% depends on species, processing stage, and technology used. The industries reject approximately up to 75% of total weight of raw material, these can create serious pollution and disposal problems. The shell wastes obtained from seafood industries have only a low economic value and they are used as either animal feed or organic manure. David et al 17 explained economic comparison of the yield of chitin and chitosan by experimental lab-scale and commercial process. The result showed yield for these two processes in present study found in this range.
The limitations include the high cost for obtained samples and the shortness of time the shrimp catch season.
Conclusions
Shrimp L. vanamei shell wastes were treated with chemical extraction procedure according to Toan method for production of chitin and chitosan. As per research output of present study, chitosan obtained from shrimp shell waste appeared to be highest degree of deacetylation along with good yield. Hence it can be concluded that shrimp chitosan appeared of superior quality. Utilization of shrimp shell waste for production of chitin and chitosan will give more economical and biological value along with reduction of environmental pollution. The results of the chitin and chitosan extraction method in this study found different compared to the results of Hamdi et al, 16 and Kumari et al, 7 which was caused by the type of species and the concentration of chemicals used in the method.
Footnotes
Acknowledgments
The authors would like to thank Behbahan Khatam Alanbia University of Technology.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding authors upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study did not involve human or animal testing.
The Ethical Declarations
The authors agreed that the paper submitted to be published is original, and confirm the following:
Confirmed that the manuscript has been submitted solely to this journal and is not published, in press, or submitted elsewhere Confirmed that all the research meets the ethical guidelines, including adherence to the legal requirements of the study country.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Behbahan Khatam Alanbia University of Technology, Behbahan, Iran.
Informed Consent/ Patient Consent
Not applicable.



